Association between PhA and Physical Performance Variables in Cancer Patients
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Procedure
2.3. Measurements
2.4. Statistical Analysis
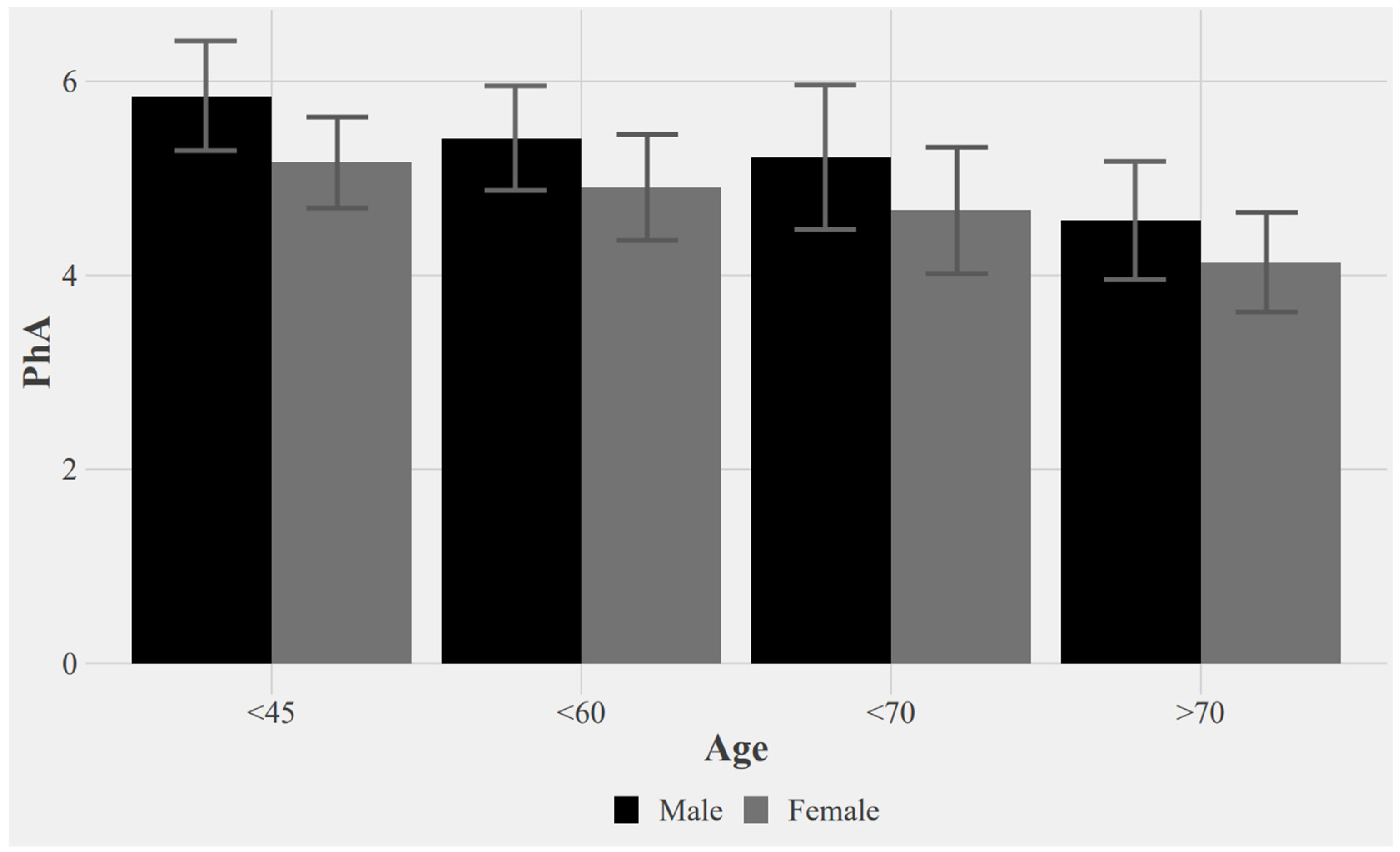
3. Results
4. Discussion
5. Conclusions
6. Future Research Lines
7. Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Norman, K.; Stobäus, N.; Pirlich, M.; Bosy-Westphal, A. Bioelectrical phase angle and impedance vector analysis–Clinical relevance and applicability of impedance parameters. Clin. Nutr. 2012, 31, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Micheli, M.L.; Pagani, L.; Marella, M.; Gulisano, M.; Piccoli, A.; Angelini, F.; Burtscher, M.; Gatterer, H. Bioimpedance and impedance vector patterns as predictors of league level in male soccer players. Int. J. Sport Physiol. Perform. 2014, 9, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Nabuco, H.C.G.; Silva, A.M.; Sardinha, L.B.; Rodrigues, F.B.; Tomeleri, C.M.; Ravagnani, F.C.P.; Cyrino, E.S.; Ravagnani, C.F.C. Phase Angle is Moderately Associated with Short-term Maximal Intensity Efforts in Soccer Players. Int. J. Sport. Med. 2019, 40, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Lukaski, H.; Raymond-Pope, C.J. New Frontiers of Body Composition in Sport. Int. J. Sport Med. 2021, 42, 588–601. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Courneya, K.S.; Matthews, C.; Demark-Wahnefried, W.; Galvão, D.A.; Pinto, B.M.; Irwin, M.L.; Wolin, K.Y.; Segal, R.J.; Lucia, A.; et al. American college of sports medicine roundtable on exercise guidelines for cancer survivors. Med. Sci. Sport Exerc. 2010, 42, 1409–1426. [Google Scholar] [CrossRef]
- Wolin, K.Y.; Schwartz, A.L.; Matthews, C.E.; Courneya, K.S.; Schmitz, K.H. Implementing the exercise guidelines for cancer survivors. J. Support. Oncol. 2012, 10, 171–177. [Google Scholar] [CrossRef]
- PROMIS. Dynamic Tools to Measure Health Outcomes from the Patient Perspective. Available online: https://www.healthmeasures.net/ (accessed on 17 August 2022).
- Di Vincenzo, O.; Marra, M.; Di Gregorio, A.; Pasanisi, F.; Scalfi, L. Bioelectrical impedance analysis (BIA) -derived phase angle in sarcopenia: A systematic review. Clin. Nutr. 2021, 40, 3052–3061. [Google Scholar] [CrossRef]
- Streb, A.R.; Hansen, F.; Gabiatti, M.P.; Tozetto, W.R.; Del Duca, G.F. Phase angle associated with different indicators of health-related physical fitness in adults with obesity. Physiol. Behav. 2020, 225, 113104. [Google Scholar] [CrossRef]
- Uemura, K.; Yamada, M.; Okamoto, H. Association of bioimpedance phase angle and prospective falls in older adults. Geriatr. Gerontol. Int. 2019, 19, 503–507. [Google Scholar] [CrossRef]
- Dittmar, M.; Reber, H.; Kahaly, G.J. Bioimpedance phase angle indicates catabolism in Type 2 diabetes. Diabet. Med. 2015, 32, 1177–1185. [Google Scholar] [CrossRef]
- Arietaleanizbeaskoa, M.S.; Gil Rey, E.; Mendizabal Gallastegui, N.; García-Álvarez, A.; De La Fuente, I.; Domínguez-Martinez, S.; Pablo, S.; Coca, A.; Gutiérrez Santamaría, B.; Grandes, G. Implementing Exercise in Standard Cancer Care (Bizi Orain Hybrid Exercise Program): Protocol for a Randomized Controlled Trial. JMIR Res. Protoc. 2021, 10, e24835. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.V.; Friedenreich, C.M.; Moore, S.C.; Hayes, S.C.; Silver, J.K.; Campbell, K.L.; Winters-Stone, K.; Gerber, L.H.; George, S.M.; Fulton, J.E.; et al. American College of Sports Medicine Roundtable Report on Physical Activity, Sedentary Behavior, and Cancer Prevention and Control. Med. Sci. Sports Exerc. 2019, 51, 2391–2402. [Google Scholar] [CrossRef]
- Cormie, P.; Lamb, S.; Newton, R.U.; Valentine, L.; McKiernan, S.; Spry, N.; Joseph, D.; Taaffe, D.R.; Doran, C.M.; Galvão, D.A. Implementing exercise in cancer care: Study protocol to evaluate a community-based exercise program for people with cancer. BMC Cancer 2017, 17, 103. [Google Scholar] [CrossRef] [PubMed]
- Walter-Kroker, A.; Kroker, A.; Mattiucci-Guehlke, M.; Glaab, T. A practical guide to bioelectrical impedance analysis using the example of chronic obstructive pulmonary disease. Nutr. J. 2011, 10, 35. [Google Scholar] [CrossRef] [PubMed]
- Więch, P.; Wołoszyn, F.; Trojnar, P.; Skórka, M.; Bazaliński, D. Does Body Position Influence Bioelectrical Impedance? An Observational Pilot Study. Int. J. Environ. Res. Public Health 2022, 19, 9908. [Google Scholar] [CrossRef]
- Lange-Maia, B.S.; Newman, A.B.; Strotmeyer, E.S.; Harris, T.B.; Caserotti, P.; Glynn, N.W. Performance on fast- and usual-paced 400-m walk tests in older adults: Are they comparable? Aging Clin. Exp. Res. 2015, 27, 309–314. [Google Scholar] [CrossRef]
- Gil-Rey, E.; Quevedo-Jerez, K.; Maldonado-Martin, S.; Herrero-Roman, F. Exercise Intensity Guidelines for Cancer Survivors: A Comparison with Reference Values. Int. J. Sports Med. 2014, 35, e1–e9. [Google Scholar] [CrossRef]
- Mishra, S.I.; Scherer, R.W.; Geigle, P.M.; Berlanstein, D.R.; Topaloglu, O.; Gotay, C.C.; Snyder, C. Exercise interventions on health-related quality of life for cancer survivors. Cochrane Database Syst. Rev. 2012, 2012, CD007566. [Google Scholar] [CrossRef]
- Blair, S.N.; Kampert, J.B.; Kohl, H.W., 3rd; Barlow, C.E.; Macera, C.A.; Paffenbarger, R.S.J.; Gibbons, L.W. Influences of cardiorespiratory fitness and other precursors on cardiovascular disease and all-cause mortality in men and women. JAMA 1996, 276, 205–210. [Google Scholar] [CrossRef]
- Simonsick, E.M.; Fan, E.; Fleg, J.L. Estimating cardiorespiratory fitness in well-functioning older adults: Treadmill validation of the long distance corridor walk. J. Am. Geriatr. Soc. 2006, 54, 127–132. [Google Scholar] [CrossRef]
- Kraemer, W.J.; Fleck, S.J. Strength Training for Young Athletes; Human Kinetics: Champaign, IL, USA, 2005. [Google Scholar]
- Hui, D.; Moore, J.; Park, M.; Liu, D.; Bruera, E. Phase Angle and the Diagnosis of Impending Death in Patients with Advanced Cancer: Preliminary Findings. Oncologist 2019, 24, e365–e373. [Google Scholar] [CrossRef] [PubMed]
- Bosy-Westphal, A.; Danielzik, S.; Dörhöfer, R.-P.; Later, W.; Wiese, S.; Müller, M.J. Phase angle from bioelectrical impedance analysis: Population reference values by age, sex, and body mass index. JPEN J. Parenter. Enteral Nutr. 2006, 30, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Barbosa-Silva, M.C.G.; Barros, A.J.D.; Wang, J.; Heymsfield, S.B.; Pierson, R.N.J. Bioelectrical impedance analysis: Population reference values for phase angle by age and sex. Am. J. Clin. Nutr. 2005, 82, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.D.; Oliveira, R.; Brito, J.P.; Costa, T.; Ramalho, F.; Pimenta, N.; Santos-Rocha, R. Phase angle cutoff value as a marker of the health status and functional capacity in breast cancer survivors. Physiol. Behav. 2021, 235, 113400. [Google Scholar] [CrossRef]
- Schmidt, K.; Vogt, L.; Thiel, C.; Jäger, E.; Banzer, W. Validity of the six-minute walk test in cancer patients. Int. J. Sport Med. 2013, 34, 631–636. [Google Scholar] [CrossRef] [PubMed]
- Sieber, C.C. Malnutrition and sarcopenia. Aging Clin. Exp. Res. 2019, 31, 793–798. [Google Scholar] [CrossRef]
- Hui, D.; Bansal, S.; Morgado, M.; Dev, R.; Chisholm, G.; Bruera, E. Phase angle for prognostication of survival in patients with advanced cancer: Preliminary findings. Cancer 2014, 120, 2207–2214. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.; Birdsell, L.; Macdonald, N.; Reiman, T.; Clandinin, M.T.; McCargar, L.J.; Murphy, R.; Ghosh, S.; Sawyer, M.B.; Baracos, V.E. Cancer cachexia in the age of obesity: Skeletal muscle depletion is a powerful prognostic factor, independent of body mass index. J. Clin. Oncol. 2013, 31, 1539–1547. [Google Scholar] [CrossRef]
- Paiva, S.I.; Borges, L.R.; Halpern-Silveira, D.; Assunção, M.C.F.; Barros, A.J.D.; Gonzalez, M.C. Standardized phase angle from bioelectrical impedance analysis as prognostic factor for survival in patients with cancer. Support. Care Cancer 2010, 19, 187–192. [Google Scholar] [CrossRef]
- Stegel, P.; Kozjek, N.R.; Brumen, B.A.; Strojan, P. Bioelectrical impedance phase angle as indicator and predictor of cachexia in head and neck cancer patients treated with (chemo)radiotherapy. Eur. J. Clin. Nutr. 2016, 70, 602–606. [Google Scholar] [CrossRef]
- Baracos, V.E.; Reiman, T.; Mourtzakis, M.; Gioulbasanis, I.; Antoun, S. Body composition in patients with non-small cell lung cancer: A contemporary view of cancer cachexia with the use of computed tomography image analysis. Am. J. Clin. Nutr. 2010, 91, 1133S–1137S. [Google Scholar] [CrossRef] [PubMed]
- Janssen, I.; Baumgartner, R.N.; Ross, R.; Rosenberg, I.H.; Roubenoff, R. Skeletal muscle cutpoints associated with elevated physical disability risk in older men and women. Am. J. Epidemiol. 2004, 159, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Ravasco, P. Nutrition in Cancer Patients. J. Clin. Med. 2019, 8, 1211. [Google Scholar] [CrossRef] [PubMed]
- Koeppel, M.; Mathis, K.; Schmitz, K.H.; Wiskemann, J. Muscle hypertrophy in cancer patients and survivors via strength training. A meta-analysis and meta-regression. Crit. Rev. Oncol. Hematol. 2021, 163, 103371. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Saito, K.; Tanaka, S.; Maki, M.; Yachi, Y.; Asumi, M.; Sugawara, A.; Totsuka, K.; Shimano, H.; Ohashi, Y.; et al. Cardiorespiratory fitness as a quantitative predictor of all-cause mortality and cardiovascular events in healthy men and women: A meta-analysis. JAMA 2009, 301, 2024–2035. [Google Scholar] [CrossRef]
- O’Neill, J.O.; Young, J.B.; Pothier, C.E.; Lauer, M.S. Peak oxygen consumption as a predictor of death in patients with heart failure receiving beta-blockers. Circulation 2005, 111, 2313–2318. [Google Scholar] [CrossRef]
- Burnett, D.; Kluding, P.; Porter, C.; Fabian, C.; Klemp, J. Cardiorespiratory fitness in breast cancer survivors. Springerplus 2013, 2, 68. [Google Scholar] [CrossRef]
- Jones, L.W.; Courneya, K.S.; Mackey, J.R.; Muss, H.B.; Pituskin, E.N.; Scott, J.M.; Hornsby, W.E.; Coan, A.D.; Herndon, J.E., 2nd; Douglas, P.S.; et al. Cardiopulmonary function and age-related decline across the breast cancer survivorship continuum. J. Clin. Oncol. 2012, 30, 2530–2537. [Google Scholar] [CrossRef]
- Lakoski, S.G.; Barlow, C.E.; Koelwyn, G.J.; Hornsby, W.E.; Hernandez, J.; Defina, L.F.; Radford, N.B.; Thomas, S.M.; Herndon, J.E., 2nd; Peppercorn, J.; et al. The influence of adjuvant therapy on cardiorespiratory fitness in early-stage breast cancer seven years after diagnosis: The Cooper Center Longitudinal Study. Breast Cancer Res. Treat. 2013, 138, 909–916. [Google Scholar] [CrossRef]
- Peel, A.B.; Thomas, S.M.; Dittus, K.; Jones, L.W.; Lakoski, S.G. Cardiorespiratory fitness in breast cancer patients: A call for normative values. J. Am. Heart Assoc. 2014, 3, e000432. [Google Scholar] [CrossRef]
- Steins Bisschop, C.N.; Velthuis, M.J.; Wittink, H.; Kuiper, K.; Takken, T.; van der Meulen, W.J.T.M.; Lindeman, E.; Peeters, P.H.M.; May, A.M. Cardiopulmonary exercise testing in cancer rehabilitation: A systematic review. Sports Med. 2012, 42, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Lundby, C.; Montero, D.; Joyner, M. Biology of VO(2) max: Looking under the physiology lamp. Acta Physiol. 2017, 220, 218–228. [Google Scholar] [CrossRef] [PubMed]
- Seiler, K.S.; Kjerland, G.Ø. Quantifying training intensity distribution in elite endurance athletes: Is there evidence for an “optimal” distribution? Scand. J. Med. Sci. Sports 2006, 16, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Schneider, J.; Schlüter, K.; Sprave, T.; Wiskemann, J.; Rosenberger, F. Exercise intensity prescription in cancer survivors: Ventilatory and lactate thresholds are useful submaximal alternatives to VO2peak. Support. Care Cancer 2020, 28, 5521–5528. [Google Scholar] [CrossRef]
- Lindemann, U.; Krumpoch, S.; Becker, C.; Sieber, C.C.; Freiberger, E. The course of gait speed during a 400m walk test of mobility limitations in community-dwelling older adults. Z. Gerontol. Geriatr. 2021, 54, 768–774. [Google Scholar] [CrossRef] [PubMed]
- Rolland, Y.M.; Cesari, M.; Miller, M.E.; Penninx, B.W.; Atkinson, H.H.; Pahor, M. Reliability of the 400-m usual-pace walk test as an assessment of mobility limitation in older adults. J. Am. Geriatr. Soc. 2004, 52, 972–976. [Google Scholar] [CrossRef]

| Variable | n (%) | Mean ± SD |
|---|---|---|
| Age (years) | 311 (100) | 55.54 ± 10.98 |
| Sex | ||
| Female Male | 225 (712.3) 86 (27.7) | |
| Height (cm) | 164.7 ± 8.7 | |
| Weight (kg) | 70.93 ± 14.52 | |
| BMI (kg/m2) | 26.1 ± 4.9 | |
| PhA (°) | 4.88 ± 0.68 | |
| 400 mWT (seg.) | 278.73 ± 53.37 | |
| VO2peak (mL/kg/min) | 15.31 ± 4 | |
| VT1 | 54.09 ± 16.81 | |
| VT2 | 80.97 ± 23.17 | |
| UBS | 61.70 ± 23.17 | |
| LBS | 33.26 ± 13.37 | |
| Surgery | 164 (52.9) | |
| Metastatic | 50 (19.6) | |
| Cancer stage | ||
| I II III IV | 48 (15.4) 80 (25.7) 62 (19.9) 121 (38.9) |
| Variable | Mixed Linear Regression Estimate [95% CI] | Mixed Linear Regression p-Value | Pearson Correlation [95% CI] | p-Value |
|---|---|---|---|---|
| 400 mWT (sec) | −22.57 [−27.58; −17.53] | <0.001 | −0.36 [−0.42; −0.3] | <0.001 |
| UBS (kg) | 6.3 [4.95; 7.65] | <0.001 | 0.54 [0.48; 0.59] | <0.001 |
| LBS (kg) | 13.25 [10.13; 16.35] | <0.001 | 0.47 [0.42; 0.53] | <0.001 |
| VO2peak (mL/kg/min) | 1.55 [0.98; 2.12] | <0.001 | 0.25 [0.18; 0.32] | <0.001 |
| VT1 (W) | 6.53 [3.83; 9.20] | <0.001 | 0.35 [0.28; 0.42] | <0.001 |
| VT2 (W) | 10.36 [7.02; 13.64] | <0.001 | 0.42 [0.36; 0.49] | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutiérrez-Santamaría, B.; Martinez Aguirre-Betolaza, A.; García-Álvarez, A.; Arietaleanizbeaskoa, M.S.; Mendizabal-Gallastegui, N.; Grandes, G.; Castañeda-Babarro, A.; Coca, A. Association between PhA and Physical Performance Variables in Cancer Patients. Int. J. Environ. Res. Public Health 2023, 20, 1145. https://doi.org/10.3390/ijerph20021145
Gutiérrez-Santamaría B, Martinez Aguirre-Betolaza A, García-Álvarez A, Arietaleanizbeaskoa MS, Mendizabal-Gallastegui N, Grandes G, Castañeda-Babarro A, Coca A. Association between PhA and Physical Performance Variables in Cancer Patients. International Journal of Environmental Research and Public Health. 2023; 20(2):1145. https://doi.org/10.3390/ijerph20021145
Chicago/Turabian StyleGutiérrez-Santamaría, Borja, Aitor Martinez Aguirre-Betolaza, Arturo García-Álvarez, Maria Soledad Arietaleanizbeaskoa, Nere Mendizabal-Gallastegui, Gonzalo Grandes, Arkaitz Castañeda-Babarro, and Aitor Coca. 2023. "Association between PhA and Physical Performance Variables in Cancer Patients" International Journal of Environmental Research and Public Health 20, no. 2: 1145. https://doi.org/10.3390/ijerph20021145
APA StyleGutiérrez-Santamaría, B., Martinez Aguirre-Betolaza, A., García-Álvarez, A., Arietaleanizbeaskoa, M. S., Mendizabal-Gallastegui, N., Grandes, G., Castañeda-Babarro, A., & Coca, A. (2023). Association between PhA and Physical Performance Variables in Cancer Patients. International Journal of Environmental Research and Public Health, 20(2), 1145. https://doi.org/10.3390/ijerph20021145








