Antibiotics in Dentistry: A Narrative Review of the Evidence beyond the Myth
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Eligibility Criteria
- Antibiotic OR Antibiotics
- Stewardship OR Administration OR Prescription OR Use OR Adherence OR Compliance OR Resistance
- Bacterial resistance OR Antimicrobial resistance
- Dentist OR Dentistry.
2.2. Data Extraction and Collection
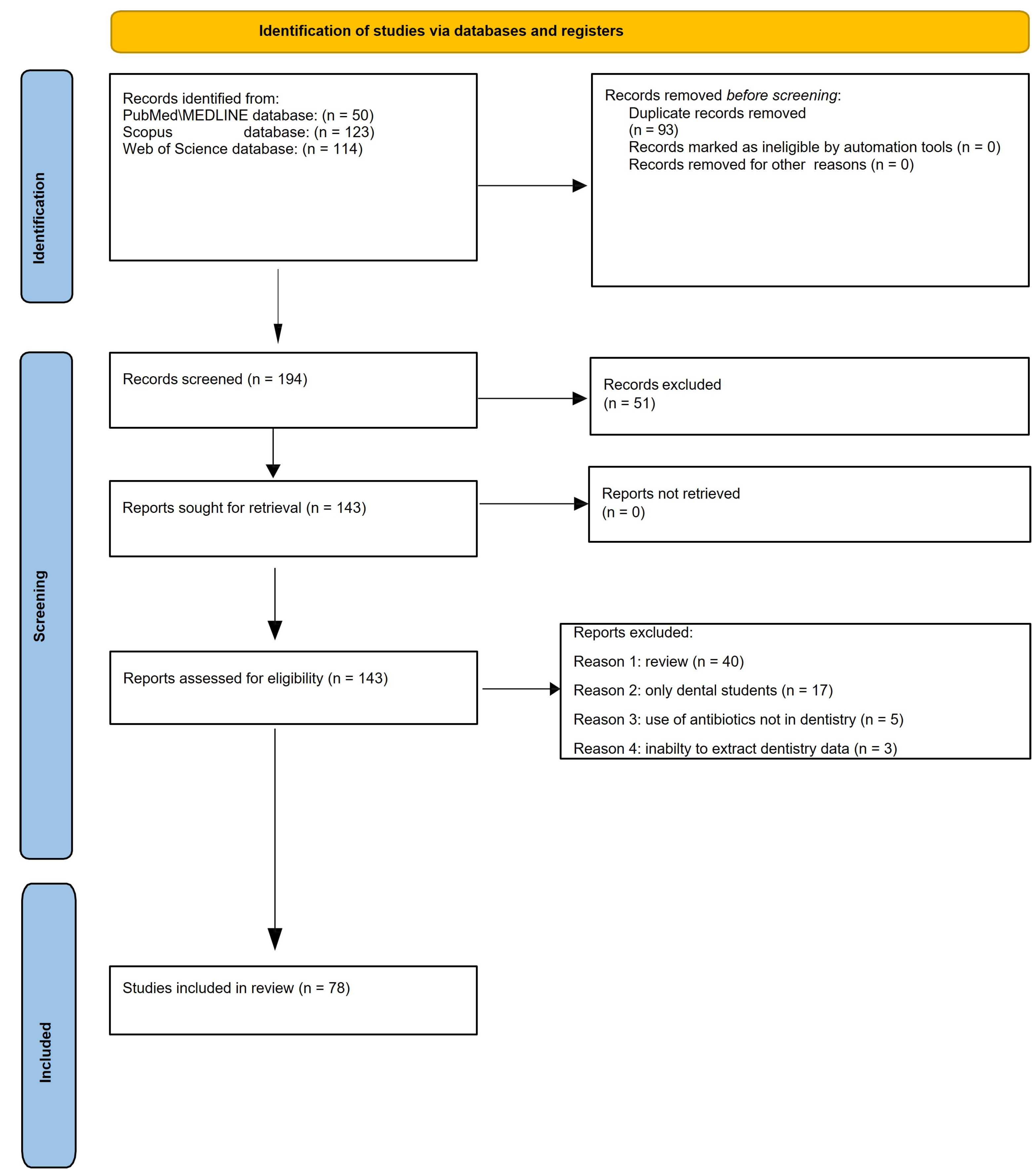
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Studies Reporting Epidemiology of Antibiotic Consumption and Prescription in Dentistry
3.4. Studies Reporting Antibiotic Therapy in Dentistry
3.5. Studies Reporting Antibiotic Prophylaxis in Dentistry
3.6. Studies Reporting Dental Patients’ Adherence to Antibiotic Prescription
3.7. Studies Reporting Antimicrobial Resistance in Dentistry
4. Discussion
4.1. Epidemiology of Antibiotic Consumption and Prescription in Dentistry
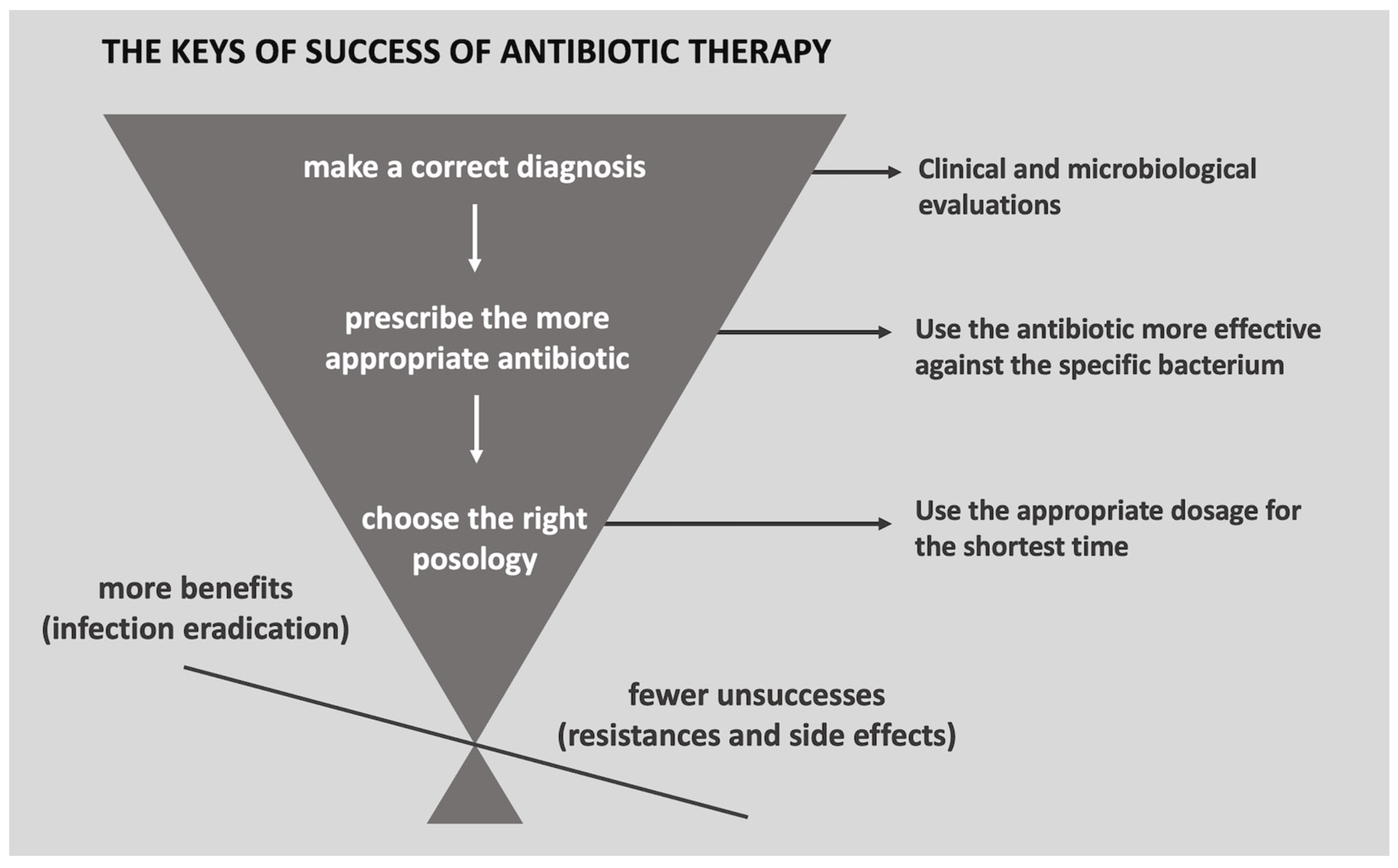
4.2. Antibiotic Therapy in Dentistry: Current Measures
Antibiotic Therapy in Dentistry: Guidelines for More Judicious Use of Antibiotic Therapy in Dentistry
4.3. Antibiotic Prophylaxis in Dentistry
Antibiotic Prophylaxis Prescription in Dentistry
4.4. Dental Patients’ Adherence to Antibiotic Prescription
- Direct methods are generally not applicable to dental patients and measuring drug levels in body fluids, such as blood or urine, or measuring biomarkers.
4.5. Antimicrobial Resistance in Dentistry
4.5.1. Antimicrobial Resistance in Biofilm
4.5.2. Antimicrobial Resistance in Major Periodontal Pathogens
4.5.3. Chlorhexidine and Antimicrobial Resistance
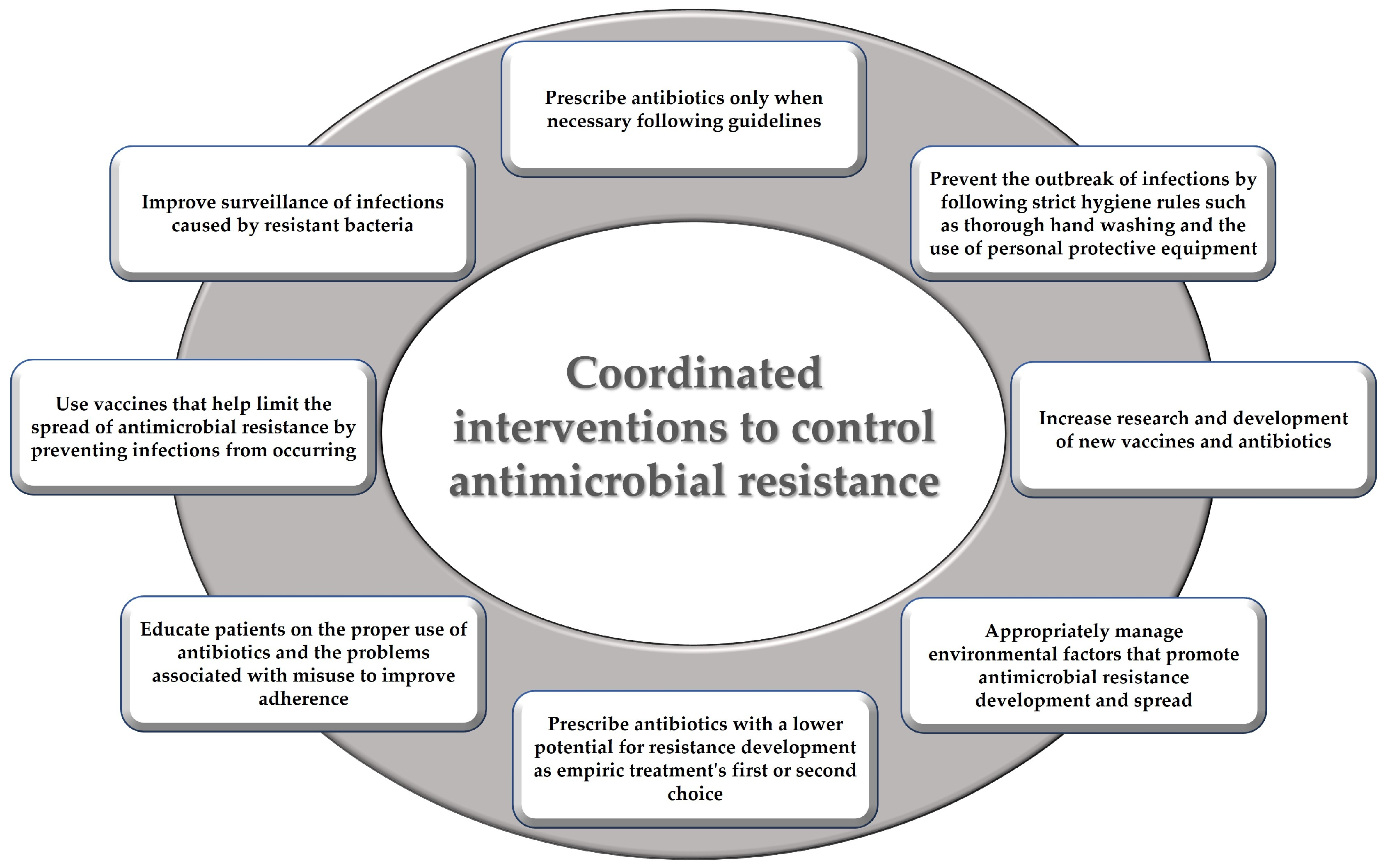
4.5.4. Measures to Counteract Antimicrobial Resistance in Dentistry: Antibiotic Stewardship
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ribeiro da Cunha, B.; Fonseca, L.P.; Calado, C.R.C. Antibiotic Discovery: Where Have We Come from, Where Do We Go? Antibiotics 2019, 8, 45. [Google Scholar] [CrossRef] [PubMed]
- Uddin, T.M.; Chakraborty, A.J.; Khusro, A.; Zidan, B.R.M.; Mitra, S.; Bin Emran, T.; Dhama, K.; Ripon, M.K.H.; Gajdács, M.; Sahibzada, M.U.K.; et al. Antibiotic Resistance in Microbes: History, Mechanisms, Therapeutic Strategies and Future Prospects. J. Infect. Public. Health 2021, 14, 1750–1766. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.L. Changing Patterns of Infectious Disease. Nature 2000, 406, 762–767. [Google Scholar] [CrossRef] [PubMed]
- Guerrini, L.; Monaco, A.; Pietropaoli, D.; Ortu, E.; Giannoni, M.; Marci, M.C. Antibiotics in Dentistry: A Narrative Review of Literature and Guidelines Considering Antibiotic Resistance. Open Dent. J. 2019, 13, 383–398. [Google Scholar] [CrossRef]
- D’Ambrosio, F.; Di Spirito, F.; Amato, A.; Caggiano, M.; Lo Giudice, R.; Martina, S. Attitudes towards Antibiotic Prescription and Antimicrobial Resistance Awareness among Italian Dentists: What Are the Milestones? Healthcare 2022, 10, 1585. [Google Scholar] [CrossRef]
- D’Ambrosio, F.; Di Spirito, F.; De Caro, F.; Lanza, A.; Passarella, D.; Sbordone, L. Adherence to Antibiotic Prescription of Dental Patients: The Other Side of the Antimicrobial Resistance. Healthcare 2022, 10, 1636. [Google Scholar] [CrossRef]
- Durand, G.A.; Raoult, D.; Dubourg, G. Antibiotic Discovery: History, Methods and Perspectives. Int. J. Antimicrob. Agents 2019, 53, 371–382. [Google Scholar] [CrossRef]
- Waxman, D.J.; Strominger, J.L. Penicillin-binding proteins and the mechanism of action of beta-lactam antibiotics. Annu. Rev. Biochem. 1983, 52, 825–869. [Google Scholar] [CrossRef]
- Bush, K.; Bradford, P.A. β-Lactams and β-Lactamase Inhibitors: An Overview. Cold Spring Harb. Perspect. Med. 2016, 6, a025247. [Google Scholar] [CrossRef]
- Gaynor, M.; Mankin, A. Macrolide Antibiotics: Binding Site, Mechanism of Action, Resistance. Curr. Top. Med. Chem. 2003, 3, 949–960. [Google Scholar] [CrossRef]
- Chopra, I.; Roberts, M. Tetracycline Antibiotics: Mode of Action, Applications, Molecular Biology, and Epidemiology of Bacterial Resistance. Microbiol. Mol. Biol. Rev. 2001, 65, 232–260. [Google Scholar] [CrossRef] [PubMed]
- Spížek, J.; Řezanka, T. Lincosamides: Chemical Structure, Biosynthesis, Mechanism of Action, Resistance, and Applications. Biochem. Pharmacol. 2017, 133, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Oberoi, S.S.; Dhingra, C.; Sharma, G.; Sardana, D. Antibiotics in Dental Practice: How Justified Are We. Int. Dent. J. 2015, 65, 4–10. [Google Scholar] [CrossRef]
- Thompson, W.; Williams, D.; Pulcini, C.; Sanderson, S.; Calfon, P.; Verma, M. Tackling Antibiotic Resistance: Why Dentistry Matters. Int. Dent. J. 2021, 71, 450–453. [Google Scholar] [CrossRef]
- Barone, A.; Chatelain, S.; Derchi, G.; Di Spirito, F.; Martuscelli, R.; Porzio, M.; Sbordone, L. Antibiotic’s Effectiveness after Erupted Tooth Extractions: A Retrospective Study. Oral. Dis. 2020, 26, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Amato, M.; Di Spirito, F.; D’Ambrosio, F.; Boccia, G.; Moccia, G.; De Caro, F. Probiotics in Periodontal and Peri-Implant Health Management: Biofilm Control, Dysbiosis Reversal, and Host Modulation. Microorganisms 2022, 10, 2289. [Google Scholar] [CrossRef]
- Ahsan, S.; Hydrie, M.Z.I.; Hyder Naqvi, S.M.Z.; Shaikh, M.A.; Shah, M.Z.; Jafry, S.I.A. Antibiotic Prescription Patterns for Treating Dental Infections in Children among General and Pediatric Dentists in Teaching Institutions of Karachi, Pakistan. PLoS ONE 2020, 15, e0235671. [Google Scholar] [CrossRef]
- Al-Harthi, S.E.; Khan, L.M.; Abed, H.H.; Alkreathy, H.M.; Ali, A.S. Appraisal of Antimicrobial Prescribing Practices of Governmental and Non-Governmental Dentists for Hospitals in the Western Region of Saudi Arabia. Saudi Med. J. 2013, 34, 1262–1269. [Google Scholar] [PubMed]
- Al-Harthi, S.E.; Khan, L.M.; Osman, A.-M.M.; Alim, M.A.; Saadah, O.I.; Almohammadi, A.A.; Khan, F.M.; Kamel, F.O. Perceptions and Knowledge Regarding Antimicrobial Stewardship among Clinicians in Jeddah, Saudi Arabia. Saudi Med. J. 2015, 36, 813–820. [Google Scholar] [CrossRef]
- Al-Johani, K.; Reddy, S.; Al Mushayt, A.; El-Housseiny, A. Pattern of Prescription of Antibiotics among Dental Practitioners in Jeddah, KSA: A Cross-Sectional Survey. Niger. J. Clin. Pract. 2017, 20, 804–810. [Google Scholar] [CrossRef]
- Al-Taani, G.M.; Al-Azzam, S.; Karasneh, R.A.; Ababneh, M.; Al-Batayneh, O.B.; Khader, Y.S.; Conway, B.R.; Aldeyab, M.A. Antibiotic Use and Resistance: Information Sources and Application by Dentists in Jordan. J. Infect. Dev. Ctries. 2022, 16, 1607–1613. [Google Scholar] [CrossRef] [PubMed]
- Baskaradoss, J.K.; Alrumaih, A.; Alshebel, A.; Alfaqih, A.; Aleesa, M.; Alkhashan, S.; Altuwaijri, M. Pattern of Antibiotic Prescription among Dentists in Riyadh, Saudi Arabia. J. Investig. Clin. Dent. 2018, 9, e12339. [Google Scholar] [CrossRef]
- Bhuvaraghan, A.; King, R.; Larvin, H.; Aggarwal, V.R. Antibiotic Use and Misuse in Dentistry in India—A Systematic Review. Antibiotics 2021, 10, 1459. [Google Scholar] [CrossRef] [PubMed]
- Bird, L.; Landes, D.; Robson, T.; Sturrock, A.; Ling, J. Higher Antibiotic Prescribing Propensity of Dentists in Deprived Areas and Those with Greater Access to Care in the North East and Cumbria, UK. Br. Dent. J. 2018, 225, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Cope, A.L.; Chestnutt, I.G.; Wood, F.; Francis, N.A. Dental Consultations in UK General Practice and Antibiotic Prescribing Rates: A Retrospective Cohort Study. Br. J. Gen. Pract. 2016, 66, e329–e336. [Google Scholar] [CrossRef]
- Cope, A.L.; Francis, N.A.; Wood, F.; Chestnutt, I.G. Antibiotic Prescribing in UK General Dental Practice: A Cross-Sectional Study. Community Dent. Oral. Epidemiol. 2016, 44, 145–153. [Google Scholar] [CrossRef]
- Duncan, E.M.; Goulao, B.; Clarkson, J.; Young, L.; Ramsay, C.R. ‘You Had to Do Something’: Prescribing Antibiotics in Scotland during the COVID-19 Pandemic Restrictions and Remobilisation. Br. Dent. J. 2021, 1–6. [Google Scholar] [CrossRef]
- Durkin, M.J.; Feng, Q.; Suda, K.J.; Lockhart, P.B.; Thornhill, M.H.; Warren, K.; Munshi, K.D.; Henderson, R.R.; Fraser, V.J. Longitudinal Antibiotic Prescribing Trends among US Dental Specialists within a Pharmacy Benefits Manager, 2013 through 2015. J. Am. Dent. Assoc. 2019, 150, 846–853.e5. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.B.; Chong, S.; Le, N.D. A Survey of Antibiotic Use in Dentistry. J. Am. Dent. Assoc. 2000, 131, 1600–1609. [Google Scholar] [CrossRef]
- Farkaš, M.; Ivančić Jokić, N.; Mavrinac, M.; Tambić Andrašević, A. Antibiotic Prescribing Habits and Antimicrobial Resistance Awareness of Dental Practitioners in Primorsko-Goranska County, Croatia. Microb. Drug. Resist. 2021, 27, 1482–1488. [Google Scholar] [CrossRef]
- Garg, A.K.; Agrawal, N.; Tewari, R.K.; Kumar, A.; Chandra, A. Antibiotic Prescription Pattern among Indian Oral Healthcare Providers: A Cross-Sectional Survey. J. Antimicrob. Chemother. 2014, 69, 526–528. [Google Scholar] [CrossRef] [PubMed]
- George, A.; Narayan, V.; Joseph, B.; Balram, B.; Anil, S. Antimicrobial Prescription Patterns among Oral Implantologists of Kerala, India: A Cross-Sectional Survey. J. Pharm. Bioallied Sci. 2022, 14, 712. [Google Scholar] [CrossRef] [PubMed]
- Gowri, S.; Mehta, D.; Kannan, S. Antibiotic Use in Dentistry: A Cross-Sectional Survey from a Developing Country. J. Orofac. Sci. 2015, 7, 90. [Google Scholar] [CrossRef]
- Goulao, B.; Scott, C.; Black, I.; Clarkson, J.; McArthur, L.; Ramsay, C.; Young, L.; Duncan, E. Audit and Feedback with or without Training In-Practice Targeting Antibiotic Prescribing (TiPTAP): A Study Protocol of a Cluster Randomised Trial in Dental Primary Care. Implement. Sci. 2021, 16, 32. [Google Scholar] [CrossRef]
- Jones, E.; Cope, A. Knowledge and Attitudes of Recently Qualified Dentists Working in Wales towards Antimicrobial Prescribing and Resistance. Eur. J. Dent. Educ. 2018, 22, e730–e736. [Google Scholar] [CrossRef]
- Kusumoto, J.; Uda, A.; Kimura, T.; Furudoi, S.; Yoshii, R.; Matsumura, M.; Miyara, T.; Akashi, M. Effect of Educational Intervention on the Appropriate Use of Oral Antimicrobials in Oral and Maxillofacial Surgery: A Retrospective Secondary Data Analysis. BMC Oral Health 2021, 21, 20. [Google Scholar] [CrossRef] [PubMed]
- Licata, F.; Di Gennaro, G.; Cautela, V.; Nobile, C.G.A.; Bianco, A. Endodontic Infections and the Extent of Antibiotic Overprescription among Italian Dental Practitioners. Antimicrob. Agents Chemother. 2021, 65, e00914-21. [Google Scholar] [CrossRef] [PubMed]
- Lokhasudhan, G.; Nasim, I. Knowledge, Attitude, and Practice Survey on Usage of Antibiotics among Dental Practitioners in Southern Region of India. J. Adv. Pharm. Educ. Res. 2017, 7, 160–162. [Google Scholar]
- Mansour, H.; Feghali, M.; Saleh, N.; Zeitouny, M. Knowledge, Practice and Attitudes Regarding Antibiotics Use among Lebanese Dentists. Pharm. Pract. 2019, 16, 1272. [Google Scholar] [CrossRef]
- McKay, J.; Begley, E.; Kerlin, P.; O’Carolan, D.; Cleary, G.; McKenna, G.; Burnett, K. An Analysis of the Clinical Appropriateness of Out-of-Hours Emergency Dental Prescribing of Antibiotics in Northern Ireland. Br. Dent. J. 2020, 228, 598–603. [Google Scholar] [CrossRef]
- Mengari, L.; Mandorah, A.; Badahdah, R. Knowledge and Practice of Antibiotic Prescription Among Dentists for Endodontic Emergencies. J. Res. Med. Dent. Sci. 2020, 8, 6–16. [Google Scholar]
- Mustafa, L.; Islami, H.; Sutej, I. Administration of Systemic Antibiotics for Dental Treatment in Kosovo Major Dental Clinics: A National Survey. Eur. J. Dent. 2022, 16, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Nourah, D.; Aldahlawi, S. Prescribing Practice of Systemic Antibiotics by Periodontists in Saudi Arabia. Int. J. Med. Dent. 2022, 25, 533–543. [Google Scholar]
- Ogunbodede, E.O.; Fatusi, O.A.; Folayan, M.O.; Olayiwola, G. Retrospective Survey of Antibiotic Prescriptions in Dentistry. J. Contemp. Dent. Pract. 2005, 6, 64–71. [Google Scholar]
- Ono, A.; Ishikane, M.; Kusama, Y.; Tanaka, C.; Ono, S.; Tsuzuki, S.; Muraki, Y.; Yamasaki, D.; Tanabe, M.; Ohmagari, N. The First National Survey of Antimicrobial Use among Dentists in Japan from 2015 to 2017 Based on the National Database of Health Insurance Claims and Specific Health Checkups of Japan. PLoS ONE 2020, 15, e0244521. [Google Scholar] [CrossRef] [PubMed]
- Osailan, S.M.; Alenazi, A.; Alburaih, J.A.; Almuqbil, A.F.; Almansour, N.N.; Barnawi, N.I.; Alkahtani, A.K.; Almutairi, A.B.; Alzahrani, K.T. Knowledge and Attitude towards Antibiotics Prescription and Antimicrobial Resistance among Dental Surgeons in Saudi Arabia. J. Pharm. Res. Int. 2021, 33, 96–104. [Google Scholar] [CrossRef]
- Palmer, N.O.; Martin, M.V.; Pealing, R.; Ireland, R.S.; Roy, K.; Smith, A.; Bagg, J. Antibiotic Prescribing Knowledge of National Health Service General Dental Practitioners in England and Scotland. J. Antimicrob. Chemother. 2001, 47, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Palmer, N.; Clover, H. A Pilot Study to Investigate Antibiotic Prescribing in Private Dental Practice in the UK. Prim. Dent. J. 2019, 8, 24–29. [Google Scholar] [CrossRef]
- Pisarnturakit, P.P.; Sooampon, S.; Sooampon, S. Managing Knowledge for Health Care Quality: An Investigation of Rational Antibiotic Use among Thai Dentists. Int. J. Health Plann Manag. 2020, 35, 606–613. [Google Scholar] [CrossRef]
- Rodriguez-Núñez, A.; Cisneros-Cabello, R.; Velasco-Ortega, E.; Llamas-Carreras, J.M.; Tórres-Lagares, D.; Segura-Egea, J.J. Antibiotic Use by Members of the Spanish Endodontic Society. J. Endod. 2009, 35, 1198–1203. [Google Scholar] [CrossRef]
- Rubanenko, M.; Blumer, S.; Maalof, K.; Elbahary, S.; Katz, L.; Kharouba, J. Assessment of the Knowledge and Approach of General Dentists Who Treat Children and Pediatric Dentists Regarding the Proper Use of Antibiotics for Children. Antibiotics 2021, 10, 1181. [Google Scholar] [CrossRef] [PubMed]
- Salako, N.O.; Rotimi, V.O.; Adib, S.M.; Al-Mutawa, S. Pattern of Antibiotic Prescription in the Management of Oral Diseases among Dentists in Kuwait. J. Dent. 2004, 32, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, J.; Kunderova, M.; Pilbauerova, N.; Kapitan, M. A Review of Evidence-Based Recommendations for Pericoronitis Management and a Systematic Review of Antibiotic Prescribing for Pericoronitis among Dentists: Inappropriate Pericoronitis Treatment Is a Critical Factor of Antibiotic Overuse in Dentistry. Int. J. Environ. Res. Public. Health 2021, 18, 6796. [Google Scholar] [CrossRef] [PubMed]
- Sefah, I.A.; Sneddon, J.; Essah, D.O.; Kurdi, A.; Fadare, J.; Jairoun, A.A.; Godman, B. Evaluation of Antibiotic Prescribing for Ambulatory Patients Seeking Primary Dental Care Services in a Public Hospital in Ghana: A Clinical Audit Study. JAC Antimicrob. Resist. 2022, 4, dlac079. [Google Scholar] [CrossRef] [PubMed]
- Segura-Egea, J.J.; Velasco-Ortega, E.; Torres-Lagares, D.; Velasco-Ponferrada, M.C.; Monsalve-Guil, L.; Llamas-Carreras, J.M. Pattern of Antibiotic Prescription in the Management of Endodontic Infections amongst Spanish Oral Surgeons. Int. Endod. J. 2010, 43, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Shalini, K.; Vidushi, S.; Nisha, Y. Knowledge and attitude of antibiotic prescription among implantologists: An observational study. Indian Drugs 2022, 59, 53–57. [Google Scholar] [CrossRef]
- Shemesh, A.; Batashvili, G.; Shuster, A.; Slutzky, H.; Moshonov, J.; Buchkovskii, O.; Lvovsky, A.; Azizi, H.; Levin, A.; Itzhak, J.B.; et al. International Questionnaire Study on Systemic Antibiotics in Endodontics. Part 1. Prescribing Practices for Endodontic Diagnoses and Clinical Scenarios. Clin. Oral Investig. 2022, 26, 2921–2926. [Google Scholar] [CrossRef]
- Sneddon, J.; Thompson, W.; Kpobi, L.N.A.; Ade, D.A.; Sefah, I.A.; Afriyie, D.; Goldthorpe, J.; Turner, R.; Nawaz, S.; Wilson, S.; et al. Exploring the Use of Antibiotics for Dental Patients in a Middle-Income Country: Interviews with Clinicians in Two Ghanaian Hospitals. Antibiotics 2022, 11, 1081. [Google Scholar] [CrossRef]
- Teoh, L.; Sloan, A.J.; McCullough, M.J.; Thompson, W. Measuring Antibiotic Stewardship Programmes and Initiatives: An Umbrella Review in Primary Care Medicine and a Systematic Review of Dentistry. Antibiotics 2020, 9, 607. [Google Scholar] [CrossRef]
- Teoh, L.; Marino, R.J.; Stewart, K.; McCullough, M.J. A Survey of Prescribing Practices by General Dentists in Australia. BMC Oral Health 2019, 19, 193. [Google Scholar] [CrossRef]
- Thompson, W.; Tonkin-Crine, S.; Pavitt, S.H.; McEachan, R.R.C.; Douglas, G.V.A.; Aggarwal, V.R.; Sandoe, J.A.T. Factors Associated with Antibiotic Prescribing for Adults with Acute Conditions: An Umbrella Review across Primary Care and a Systematic Review Focusing on Primary Dental Care. J. Antimicrob. Chemother. 2019, 74, 2139–2152. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.K.; Suresan, V.; Das, S.; Dash, B.; Banka, A.; Suresh, A. Antibiotic Prescribing Practices amongst the Dental Practitioners of Bhubaneswar City: A Cross-Sectional Study. World J. Dent. 2022, 13, 520–526. [Google Scholar] [CrossRef]
- Cope, A.L.; Wood, F.; Francis, N.A.; Chestnutt, I.G. General Practitioners’ Attitudes towards the Management of Dental Conditions and Use of Antibiotics in These Consultations: A Qualitative Study. BMJ Open 2015, 5, e008551. [Google Scholar] [CrossRef] [PubMed]
- Froum, S.; Weinberg, M. An Evaluation of Antibiotic Use in Periodontal and Implant Practices. Int. J. Periodontics Restor. Dent. 2015, 35, 481–487. [Google Scholar] [CrossRef] [PubMed]
- Kaul, R.; Sandhu, H.; Talwar, B.; Chengappa, D.M.; Bali, A.; Koul, R. Oral Pain and Infection Control Strategies for Treating Children and Adolescents in India. J. Family Med. Prim. Care 2021, 10, 1987. [Google Scholar] [CrossRef]
- Khattri, S.; Kumbargere Nagraj, S.; Arora, A.; Eachempati, P.; Kusum, C.K.; Bhat, K.G.; Johnson, T.M.; Lodi, G. Adjunctive Systemic Antimicrobials for the Non-Surgical Treatment of Periodontitis. Cochrane Database Syst. Rev. 2020, 11, CD012568. [Google Scholar] [CrossRef]
- Silva, M.; Paulo, M.; Cardoso, M.; Martins, M.; Noites, R. The Use of Systemic Antibiotics in Endodontics: A Cross-Sectional Study. Rev. Port. Estomatol. Med. Dentária Cir. Maxilofac. 2017, 58, 205–211. [Google Scholar] [CrossRef]
- Thompson, W.; Teoh, L.; Pulcini, C.; Williams, D.; Pitkeathley, C.; Carter, V.; Sanderson, S.; Torres, G.; Walsh, T. Dental Antibiotic Stewardship: Study Protocol for Developing International Consensus on a Core Outcome Set. Trials 2022, 23, 116. [Google Scholar] [CrossRef]
- Bianco, A.; Cautela, V.; Napolitano, F.; Licata, F.; Pavia, M. Appropriateness of Antibiotic Prescription for Prophylactic Purposes among Italian Dental Practitioners: Results from a Cross-Sectional Study. Antibiotics 2021, 10, 547. [Google Scholar] [CrossRef]
- Ireland, R.S.; Palmer, N.O.; Lindenmeyer, A.; Mills, N. An Investigation of Antibiotic Prophylaxis in Implant Practice in the UK. Br. Dent. J. 2012, 213, E14. [Google Scholar] [CrossRef]
- Kirnbauer, B.; Jakse, N.; Truschnegg, A.; Dzidic, I.; Mukaddam, K.; Payer, M. Is Perioperative Antibiotic Prophylaxis in the Case of Routine Surgical Removal of the Third Molar Still Justified? A Randomized, Double-Blind, Placebo-Controlled Clinical Trial with a Split-Mouth Design. Clin. Oral Investig. 2022, 26, 6409–6421. [Google Scholar] [CrossRef]
- Lollobrigida, M.; Pingitore, G.; Lamazza, L.; Mazzucchi, G.; Serafini, G.; De Biase, A. Antibiotics to Prevent Surgical Site Infection (SSI) in Oral Surgery: Survey among Italian Dentists. Antibiotics 2021, 10, 949. [Google Scholar] [CrossRef]
- Salgado-Peralvo, A.-O.; Peña-Cardelles, J.-F.; Kewalramani, N.; Mateos-Moreno, M.-V.; Jiménez-Guerra, Á.; Velasco-Ortega, E.; Uribarri, A.; Moreno-Muñoz, J.; Ortiz-García, I.; Núñez-Márquez, E.; et al. Preventive Antibiotic Therapy in the Placement of Immediate Implants: A Systematic Review. Antibiotics 2021, 11, 5. [Google Scholar] [CrossRef] [PubMed]
- Salgado-Peralvo, A.-O.; Peña-Cardelles, J.-F.; Kewalramani, N.; Garcia-Sanchez, A.; Mateos-Moreno, M.-V.; Velasco-Ortega, E.; Ortiz-García, I.; Jiménez-Guerra, Á.; Végh, D.; Pedrinaci, I.; et al. Is Antibiotic Prophylaxis Necessary before Dental Implant Procedures in Patients with Orthopaedic Prostheses? A Systematic Review. Antibiotics 2022, 11, 93. [Google Scholar] [CrossRef] [PubMed]
- Salgado-Peralvo, A.-O.; Mateos-Moreno, M.-V.; Velasco-Ortega, E.; Peña-Cardelles, J.-F.; Kewalramani, N. Preventive Antibiotic Therapy in Bone Augmentation Procedures in Oral Implantology: A Systematic Review. J. Stomatol. Oral Maxillofac. Surg. 2022, 123, 74–80. [Google Scholar] [CrossRef]
- Sato, M.; Yamana, H.; Ono, S.; Ishimaru, M.; Matsui, H.; Yasunaga, H. Amoxicillin vs Third-generation Cephalosporin for Infection Prophylaxis after Third Molar Extraction. Oral Dis. 2022, 1–9. [Google Scholar] [CrossRef]
- Sato, M.; Yamana, H.; Ono, S.; Ishimaru, M.; Matsui, H.; Yasunaga, H. Trends in Prophylactic Antibiotic Use for Tooth Extraction from 2015 to 2018 in Japan: An Analysis Using a Health Insurance Claims Database. J. Infect. Chemother. 2022, 28, 504–509. [Google Scholar] [CrossRef] [PubMed]
- Sologova, D.; Diachkova, E.; Gor, I.; Sologova, S.; Grigorevskikh, E.; Arazashvili, L.; Petruk, P.; Tarasenko, S. Antibiotics Efficiency in the Infection Complications Prevention after Third Molar Extraction: A Systematic Review. Dent. J. 2022, 10, 72. [Google Scholar] [CrossRef]
- Singh Gill, A.; Morrissey, H.; Rahman, A. A Systematic Review and Meta-Analysis Evaluating Antibiotic Prophylaxis in Dental Implants and Extraction Procedures. Medicina (B Aires) 2018, 54, 95. [Google Scholar] [CrossRef]
- Williams, R.G.M. Antibiotic Prophylaxis during Dental Implant Placement in the UK. Br. Dent. J. 2020, 229, 787–792. [Google Scholar] [CrossRef]
- Abe, F.C.; Kodaira, K.; de Cássia Bergamaschi Motta, C.; Barberato-Filho, S.; Silva, M.T.; Guimarães, C.C.; Martins, C.C.; Lopes, L.C. Antimicrobial Resistance of Microorganisms Present in Periodontal Diseases: A Systematic Review and Meta-Analysis. Front. Microbiol. 2022, 13, 3712. [Google Scholar] [CrossRef]
- Abe, F.C.; de Cássia Bergamaschi Motta, C.; Figueiró, M.F.; Lopes, L.C. Resistance Profile to Antimicrobial Agents in the Main Circulating Bacteria Isolated from Acute Periodontal and Endodontic Infections in Latin America (MICROBE- DENT). Medicine 2018, 97, e13158. [Google Scholar] [CrossRef]
- Almeida, V.d.S.M.; Azevedo, J.; Leal, H.F.; de Queiroz, A.T.L.; da Silva Filho, H.P.; Reis, J.N. Bacterial Diversity and Prevalence of Antibiotic Resistance Genes in the Oral Microbiome. PLoS ONE 2020, 15, e0239664. [Google Scholar] [CrossRef]
- Alzahrani, A.A.H.; Alzahrani, M.S.A.; Aldannish, B.H.; Alghamdi, H.S.; Albanghali, M.A.; Almalki, S.S.R. Inappropriate Dental Antibiotic Prescriptions: Potential Driver of the Antimicrobial Resistance in Albaha Region, Saudi Arabia. Risk Manag. Healthc. Policy 2020, 13, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Groppo, F.C.; Castro, F.M.; Pacheco, A.B.; Motta, R.H.; Filho, T.R.; Ramacciato, J.C.; Florio, F.M.; Meechan, J.G. Antimicrobial Resistance of Staphylococcus Aureus and Oral Streptococci Strains from High-Risk Endocarditis Patients. Gen. Dent. 2005, 53, 410–413. [Google Scholar]
- Irshad, M.; Alam, M.K.; Alawneh, A.; Alhadi, M.A.; Alhadi, A.A.; Almunajem, Y.S.; Alanezi, F.F.; Al Sagoor, S.A.; Bajawi, A.M.; Alfawzan, A.A.; et al. Characterization and Antimicrobial Susceptibility of Pathogens Associated with Periodontal Abscess. Antibiotics 2020, 9, 654. [Google Scholar] [CrossRef] [PubMed]
- Kiros, A.; Saravanan, M.; Niguse, S.; Gebregziabher, D.; Kahsay, G.; Dhandapani, R.; Paramasivam, R.; Araya, T.; Asmelash, T. Bacterial Profile, Antimicrobial Susceptibility Pattern, and Associated Factors among Dental Caries-Suspected Patients Attending the Ayder Comprehensive Specialized Hospital and Private Dental Clinic in Mekelle, Northern Ethiopia. BioMed Res. Int. 2022, 2022, 3463472. [Google Scholar] [CrossRef] [PubMed]
- Lang, P.M.; Jacinto, R.C.; Dal Pizzol, T.S.; Ferreira, M.B.C.; Montagner, F. Resistance Profiles to Antimicrobial Agents in Bacteria Isolated from Acute Endodontic Infections: Systematic Review and Meta-Analysis. Int. J. Antimicrob. Agents 2016, 48, 467–474. [Google Scholar] [CrossRef] [PubMed]
- Laumen, J.G.E.; Van Dijck, C.; Manoharan-Basil, S.S.; Abdellati, S.; De Baetselier, I.; Cuylaerts, V.; De Block, T.; Van den Bossche, D.; Xavier, B.B.; Malhotra-Kumar, S.; et al. Sub-Inhibitory Concentrations of Chlorhexidine Induce Resistance to Chlorhexidine and Decrease Antibiotic Susceptibility in Neisseria Gonorrhoeae. Front. Microbiol. 2021, 12, 3521. [Google Scholar] [CrossRef]
- Rodrigues, R.M.J.; Goncalves, C.; Souto, R.; Feres-Filho, E.J.; Uzeda, M.; Colombo, A.P.V. Antibiotic Resistance Profile of the Subgingival Microbiota Following Systemic or Local Tetracycline Therapy. J. Clin. Periodontol. 2004, 31, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.A.; Bastos, E.M.A.; Rodrigues, P.H.; de Uzeda, M.; de Carvalho, M.A.R.; de Macedo Farias, L.; Andrade Moreira, E.S. Susceptibility of Prevotella Intermedia/Prevotella Nigrescens (and Porphyromonas Gingivalis) to Propolis (Bee Glue) and Other Antimicrobial Agents. Anaerobe 2002, 8, 9–15. [Google Scholar] [CrossRef] [PubMed]
- van Winkelhoff, A.J.; Gonzales, D.H.; Winkel, E.G.; Dellemijn-Kippuw, N.; Vandenbroucke-Grauls, C.M.J.E.; Sanz, M. Antimicrobial Resistance in the Subgingival Microflora in Patients with Adult Periodontitis. J. Clin. Periodontol. 2000, 27, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Priyadharsini, V.J.; SmilineGirija, A.S.; Paramasivam, A. Enterococcus Faecalis an Emerging Microbial Menace in Dentistry-An Insight into the In-Silico Detection of Drug Resistant Genes and Its Protein Diversity. J. Clin. Diagn. Res. 2018, 12, GC06–GC10. [Google Scholar] [CrossRef]
- Klein, E.Y.; Van Boeckel, T.P.; Martinez, E.M.; Pant, S.; Gandra, S.; Levin, S.A.; Goossens, H.; Laxminarayan, R. Global Increase and Geographic Convergence in Antibiotic Consumption between 2000 and 2015. Proc. Natl. Acad. Sci. USA 2018, 115, E3463–E3470. [Google Scholar] [CrossRef]
- Hicks, L.A.; Bartoces, M.G.; Roberts, R.M.; Suda, K.J.; Hunkler, R.J.; Taylor, T.H.; Schrag, S.J. US Outpatient Antibiotic Prescribing Variation According to Geography, Patient Population, and Provider Specialty in 2011. Clin. Infect. Dis. 2015, 60, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Page, A.T.; Kruger, E.; Tennant, M. Dispensing Patterns of Medicines Prescribed by Australian Dentists From 2006 to 2018—A Pharmacoepidemiological Study. Int. Dent. J. 2021, 71, 106–112. [Google Scholar] [CrossRef]
- Cope, A.L.; Francis, N.; Wood, F.; Chestnutt, I.G. Systemic Antibiotics for Symptomatic Apical Periodontitis and Acute Apical Abscess in Adults. Cochrane Database Syst. Rev. 2018, 26, CD010136. [Google Scholar] [CrossRef]
- Suda, K.J.; Calip, G.S.; Zhou, J.; Rowan, S.; Gross, A.E.; Hershow, R.C.; Perez, R.I.; McGregor, J.C.; Evans, C.T. Assessment of the Appropriateness of Antibiotic Prescriptions for Infection Prophylaxis Before Dental Procedures, 2011 to 2015. JAMA Netw. Open 2019, 2, e193909. [Google Scholar] [CrossRef]
- Langford, B.J.; So, M.; Raybardhan, S.; Leung, V.; Soucy, J.-P.R.; Westwood, D.; Daneman, N.; MacFadden, D.R. Antibiotic Prescribing in Patients with COVID-19: Rapid Review and Meta-Analysis. Clin. Microbiol. Infect. 2021, 27, 520–531. [Google Scholar] [CrossRef]
- Lee, D.S.; Lee, S.-J.; Choe, H.-S. Community-Acquired Urinary Tract Infection by Escherichia coli in the Era of Antibiotic Resistance. BioMed Res. Int. 2018, 2018, 7656752. [Google Scholar] [CrossRef]
- Martins, J.R.; Chagas, O.L.; Velasques, B.D.; Bobrowski, Â.N.; Correa, M.B.; Torriani, M.A. The Use of Antibiotics in Odontogenic Infections: What Is the Best Choice? A Systematic Review. J. Oral Maxillofac. Surg. 2017, 75, 2606.e1–2606.e11. [Google Scholar] [CrossRef] [PubMed]
- di Stasio, D.; Lauritano, D.; Minervini, G.; Paparella, R.S.; Petruzzi, M.; Romano, A.; Candotto, V.; Lucchese, A. Management of Denture Stomatitis: A Narrative Review. J. Biol. Regul. Homeost. Agents 2018, 32, 113–116. [Google Scholar]
- Bertossi, D.; Barone, A.; Iurlaro, A.; Marconcini, S.; De Santis, D.; Finotti, M.; Procacci, P. Odontogenic Orofacial Infections. J. Craniofacial Surg. 2017, 28, 197–202. [Google Scholar] [CrossRef]
- Contaldo, M.; Romano, A.; Mascitti, M.; Fiori, F.; Della Vella, F.; Serpico, R.; Santarelli, A. Association between Denture Stomatitis, Candida Species and Diabetic Status. J. Biol. Regul. Homeost. Agents 2019, 33 (Suppl. S1), 35–41. [Google Scholar]
- Sukumar, S.; Martin, F.; Hughes, T.; Adler, C. Think before You Prescribe: How Dentistry Contributes to Antibiotic Resistance. Aust. Dent. J. 2020, 65, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, F.; Pelella, S.; Argentino, S.; Sisalli, L.; Sbordone, L. Oral Manifestations and the Role of the Oral Healthcare Workers in COVID-19. Oral Dis. 2022, 28, 1003–1004. [Google Scholar] [CrossRef]
- di Spirito, F.; Amato, A.; di Palo, M.P.; Contaldo, M.; D’Ambrosio, F.; lo Giudice, R.; Amato, M. Oral Lesions Following Anti-SARS-CoV-2 Vaccination: A Systematic Review. Int. J. Environ. Res. Public. Health 2022, 19, 10228. [Google Scholar] [CrossRef]
- Di Spirito, F.; Iandolo, A.; Amato, A.; Caggiano, M.; Raimondo, A.; Lembo, S.; Martina, S. Prevalence, Features and Degree of Association of Oral Lesions in COVID-19: A Systematic Review of Systematic Reviews. Int. J. Environ. Res. Public. Health 2022, 19, 7486. [Google Scholar] [CrossRef] [PubMed]
- NICE. Antimicrobial Stewardship: Systems and Processes for Effective Antimicrobial Medicine Use. 2015. Available online: https://www.nice.org.uk/guidance/ng15/evidence/full-guideline-pdf-252320799 (accessed on 28 September 2022).
- Dar-Odeh, N.S.; Abu-Hammad, O.A.; Al-Omiri, M.K.; Khraisat, A.S.; Shehabi, A.A. Antibiotic Prescribing Practices by Dentists: A Review. Ther. Clin. Risk Manag. 2010, 6, 301. [Google Scholar] [CrossRef] [PubMed]
- Milic, T.; Raidoo, P.; Gebauer, D. Antibiotic Prophylaxis in Oral and Maxillofacial Surgery: A Systematic Review. Br. J. Oral Maxillofac. Surg. 2021, 59, 633–642. [Google Scholar] [CrossRef]
- Wilson, W.; Taubert, K.A.; Gewitz, M.; Lockhart, P.B.; Baddour, L.M.; Levison, M.; Bolger, A.; Cabell, C.H.; Takahashi, M.; Baltimore, R.S.; et al. Prevention of Infective Endocarditis. Circulation 2007, 116, 1736–1754. [Google Scholar] [CrossRef]
- Gould, F.K. Guidelines for the Prevention of Endocarditis: Report of the Working Party of the British Society for Antimicrobial Chemotherapy. J. Antimicrob. Chemother. 2006, 57, 1035–1042. [Google Scholar] [CrossRef]
- Thornhill, M.H.; Chambers, J.B.; Dayer, M.; Shanson, D. A Change in the NICE Guidelines on Antibiotic Prophylaxis for Dental Procedures. Br. J. Gen. Pract. 2016, 66, 460–461. [Google Scholar] [CrossRef]
- Caggiano, M.; Di Spirito, F.; Acerra, A.; Galdi, M.; Sisalli, L. Multiple-Drugs-Related Osteonecrosis of the Jaw in a Patient Affected by Multiple Myeloma: A Case Report. Dent. J. 2023, 11, 104. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, F.; Argentino, S.; Martuscelli, R.; Sbordone, L. MRONJ Incidence after Multiple Teeth Extractions in Patients Taking Oral without “Drug. Holiday”: A Retrospective Chart Review. Oral Implantol. 2019, 12, 105–110. [Google Scholar]
- Buonavoglia, A.; Leone, P.; Solimando, A.G.; Fasano, R.; Malerba, E.; Prete, M.; Corrente, M.; Prati, C.; Vacca, A.; Racanelli, V. Antibiotics or No Antibiotics, That Is the Question: An Update on Efficient and Effective Use of Antibiotics in Dental Practice. Antibiotics 2021, 10, 550. [Google Scholar] [CrossRef] [PubMed]
- Vila, P.M.; Zenga, J.; Fowler, S.; Jackson, R.S. Antibiotic Prophylaxis in Clean-Contaminated Head and Neck Surgery: A Systematic Review and Meta-analysis. Otolaryngol.-Head Neck Surg. 2017, 157, 580–588. [Google Scholar] [CrossRef] [PubMed]
- Dammling, C.; Abramowicz, S.; Kinard, B. Current Concepts in Prophylactic Antibiotics in Oral and Maxillofacial Surgery. Oral Maxillofac. Surg. Clin. N. Am. 2022, 34, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Ramos, E.; Santamaría, J.; Santamaría, G.; Barbier, L.; Arteagoitia, I. Do Systemic Antibiotics Prevent Dry Socket and Infection after Third Molar Extraction? A Systematic Review and Meta-Analysis. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2016, 122, 403–425. [Google Scholar] [CrossRef]
- Arteagoitia, M.; Barbier, L.; Santamaria, J.; Santamaria, G.; Ramos, E. Efficacy of Amoxicillin and Amoxicillin/Clavulanic Acid in the Prevention of Infection and Dry Socket after Third Molar Extraction. A Systematic Review and Meta-Analysis. Med. Oral Patol. Oral Cir. Bucal 2016, 21, e494. [Google Scholar] [CrossRef]
- Rodrigues, W.C.; Okamoto, R.; Pellizzer, E.P.; dos Carrijo, A.C.N.; de Almeida, R.S.; de Melo, W.M. Antibiotic Prophylaxis for Third Molar Extraction in Healthy Patients: Current Scientific Evidence. Quintessence Int. 2015, 46, 149–161. [Google Scholar] [CrossRef]
- Lodi, G.; Azzi, L.; Varoni, E.M.; Pentenero, M.; Del Fabbro, M.; Carrassi, A.; Sardella, A.; Manfredi, M. Antibiotics to Prevent Complications Following Tooth Extractions. Cochrane Database Syst. Rev. 2021, 2, CD003811. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Grusovin, M.G.; Worthington, H. V Interventions for Replacing Missing Teeth: Antibiotics at Dental Implant Placement to Prevent Complications. Cochrane Database Syst. Rev. 2013, 7, CD004152. [Google Scholar] [CrossRef] [PubMed]
- El-Kholey, K.E. Efficacy of Two Antibiotic Regimens in the Reduction of Early Dental Implant Failure: A Pilot Study. Int. J. Oral Maxillofac. Surg. 2014, 43, 487–490. [Google Scholar] [CrossRef]
- Cahill, T.J.; Harrison, J.L.; Jewell, P.; Onakpoya, I.; Chambers, J.B.; Dayer, M.; Lockhart, P.; Roberts, N.; Shanson, D.; Thornhill, M.; et al. Antibiotic prophylaxis for infective endocarditis: A systematic review and meta-analysis. Heart 2017, 103, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Cannon, R.B.; Houlton, J.J.; Mendez, E.; Futran, N.D. Methods to Reduce Postoperative Surgical Site Infections after Head and Neck Oncology Surgery. Lancet Oncol. 2017, 18, e405–e413. [Google Scholar] [CrossRef]
- Fernandes, M.; Leite, A.; Basto, M.; Nobre, M.A.; Vieira, N.; Fernandes, R.; Nogueira, P.; Jorge, P. Non-Adherence to Antibiotic Therapy in Patients Visiting Community Pharmacies. Int. J. Clin. Pharm. 2014, 36, 86–91. [Google Scholar] [CrossRef]
- Haskard Zolnierek, K.B.; DiMatteo, M.R. Physician Communication and Patient Adherence to Treatment. Med. Care 2009, 47, 826–834. [Google Scholar] [CrossRef]
- Demonceau, J.; Ruppar, T.; Kristanto, P.; Hughes, D.A.; Fargher, E.; Kardas, P.; De Geest, S.; Dobbels, F.; Lewek, P.; Urquhart, J.; et al. Identification and Assessment of Adherence-Enhancing Interventions in Studies Assessing Medication Adherence Through Electronically Compiled Drug Dosing Histories: A Systematic Literature Review and Meta-Analysis. Drugs 2013, 73, 545–562. [Google Scholar] [CrossRef]
- Claxton, A.J.; Cramer, J.; Pierce, C. A Systematic Review of the Associations between Dose Regimens and Medication Compliance. Clin. Ther. 2001, 23, 1296–1310. [Google Scholar] [CrossRef]
- Robin DiMatteo, M.; Giordani, P.J.; Lepper, H.S.; Croghan, T.W. Patient Adherence and Medical Treatment Outcomes. Med. Care 2002, 40, 794–811. [Google Scholar] [CrossRef]
- Lam, W.Y.; Fresco, P. Medication Adherence Measures: An Overview. BioMed Res. Int. 2015, 2015, 217047. [Google Scholar] [CrossRef]
- Munita, J.M.; Arias, C.A. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016, 4, 481–511. [Google Scholar] [CrossRef] [PubMed]
- Marcinkiewicz, J.; Strus, M.; Pasich, E. Antibiotic Resistance: A “Dark Side” of Biofilm-associated Chronic Infections. Pol. Arch. Med. Wewn. 2013, 123, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Rath, S.; Bal, S.C.B.; Dubey, D. Oral Biofilm: Development Mechanism, Multidrug Resistance, and Their Effective Management with Novel Techniques. Rambam Maimonides Med. J. 2021, 12, e0004. [Google Scholar] [CrossRef]
- Stewart, P.S.; William Costerton, J. Antibiotic Resistance of Bacteria in Biofilms. Lancet 2001, 358, 135–138. [Google Scholar] [CrossRef] [PubMed]
- Sbordone, C.; Toti, P.; Brevi, B.; Martuscelli, R.; Sbordone, L.; Di Spirito, F. Computed Tomography-Aided Descriptive Analysis of Maxillary and Mandibular Atrophies. J. Stomatol. Oral Maxillofac. Surg. 2019, 120, 99–105. [Google Scholar] [CrossRef]
- Di Spirito, F.; Toti, P.; Brevi, B.; Martuscelli, R.; Sbordone, L.; Sbordone, C. Computed Tomography Evaluation of Jaw Atrophies before and after Surgical Bone Augmentation. Int. J. Clin. Dent. 2019, 12, 259–270. [Google Scholar]
- Ramaglia, L.; Di Spirito, F.; Sirignano, M.; La Rocca, M.; Esposito, U.; Sbordone, L. A 5-Year Longitudinal Cohort Study on Crown to Implant Ratio Effect on Marginal Bone Level in Single Implants. Clin. Implant. Dent. Relat. Res. 2019, 21, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, F.; Iacono, V.J.; Alfredo, I.; Alessandra, A.; Sbordone, L.; Lanza, A. Evidence-Based Recommendations on Periodontal Practice and the Management of Periodontal Patients During and After the COVID-19 Era: Challenging Infectious Diseases Spread by Airborne Transmission. Open Dent. J. 2021, 15, 325–336. [Google Scholar] [CrossRef]
- Di Spirito, F. Oral-Systemic Health and Disorders: Latest Prospects on Oral Antisepsis. Appl. Sci. 2022, 12, 8185. [Google Scholar] [CrossRef]
- Di Spirito, F. Oral and Systemic Health in the Elderly. Appl. Sci. 2022, 12, 11718. [Google Scholar] [CrossRef]
- Khan, S.A.; Kong, E.F.; Meiller, T.F.; Jabra-Rizk, M.A. Periodontal Diseases: Bug Induced, Host Promoted. PLoS Pathog. 2015, 11, e1004952. [Google Scholar] [CrossRef]
- Bui, F.Q.; Almeida-da-Silva, C.L.C.; Huynh, B.; Trinh, A.; Liu, J.; Woodward, J.; Asadi, H.; Ojcius, D.M. Association between Periodontal Pathogens and Systemic Disease. Biomed. J. 2019, 42, 27–35. [Google Scholar] [CrossRef]
- Contaldo, M.; Itro, A.; Lajolo, C.; Gioco, G.; Inchingolo, F.; Serpico, R. Overview on Osteoporosis, Periodontitis and Oral Dysbiosis: The Emerging Role of Oral Microbiota. Appl. Sci. 2020, 10, 6000. [Google Scholar] [CrossRef]
- Cieplik, F.; Jakubovics, N.S.; Buchalla, W.; Maisch, T.; Hellwig, E.; Al-Ahmad, A. Resistance Toward Chlorhexidine in Oral Bacteria—Is There Cause for Concern? Front. Microbiol. 2019, 10, 587. [Google Scholar] [CrossRef] [PubMed]
- Di Spirito, F.; D’Ambrosio, F.; Di Palo, M.P.; Giordano, F.; Coppola, N.; Contaldo, M. COVID-19 and Related Vaccinations in Children: Pathogenic Aspects of Oral Lesions. Children 2023, 10, 809. [Google Scholar] [CrossRef]
- Di Spirito, F.; Caggiano, M.; Di Palo, M.P.; Contaldo, M.; D’Ambrosio, F.; Martina, S.; Amato, A. Oral Lesions in Pediatric Subjects: SARS-CoV-2 Infection and COVID-19 Vaccination. Appl. Sci. 2022, 12, 8995. [Google Scholar] [CrossRef]
- Di Spirito, F.; Pantaleo, G.; Di Palo, M.P.; Amato, A.; Raimondo, A.; Amato, M. Oral Human Papillomavirus Benign Lesions and HPV-Related Cancer in Healthy Children: A Systematic Review. Cancers 2023, 15, 1096. [Google Scholar] [CrossRef] [PubMed]
- Brookes, Z.L.S.; Belfield, L.A.; Ashworth, A.; Casas-Agustench, P.; Raja, M.; Pollard, A.J.; Bescos, R. Effects of Chlorhexidine Mouthwash on the Oral Microbiome. J. Dent. 2021, 113, 103768. [Google Scholar] [CrossRef] [PubMed]
- Tribble, G.D.; Angelov, N.; Weltman, R.; Wang, B.-Y.; Eswaran, S.V.; Gay, I.C.; Parthasarathy, K.; Dao, D.-H.V.; Richardson, K.N.; Ismail, N.M.; et al. Frequency of Tongue Cleaning Impacts the Human Tongue Microbiome Composition and Enterosalivary Circulation of Nitrate. Front. Cell. Infect. Microbiol. 2019, 9, 39. [Google Scholar] [CrossRef]
- Di Spirito, F.; Amato, A.; Di Palo, M.P.; Cannatà, D.; Giordano, F.; D’Ambrosio, F.; Martina, S. Periodontal Management in Periodontally Healthy Orthodontic Patients with Fixed Appliances: An Umbrella Review of Self-Care Instructions and Evidence-Based Recommendations. Dent. J. 2023, 11, 35. [Google Scholar] [CrossRef] [PubMed]
- Kampf, G. Biocidal Agents Used for Disinfection Can Enhance Antibiotic Resistance in Gram-Negative Species. Antibiotics 2018, 7, 110. [Google Scholar] [CrossRef]
- Saleem, H.G.M.; Seers, C.A.; Sabri, A.N.; Reynolds, E.C. Dental Plaque Bacteria with Reduced Susceptibility to Chlorhexidine Are Multidrug Resistant. BMC Microbiol. 2016, 16, 214. [Google Scholar] [CrossRef] [PubMed]
- Paoletti, I.; Fusco, A.; Grimaldi, E.; Perillo, L.; Coretti, L.; Di Domenico, M.; Cozza, V.; Lucchese, A.; Contaldo, M.; Serpico, R.; et al. Assessment of Host Defence Mechanisms Induced by Candida Species. Int. J. Immunopathol. Pharmacol. 2013, 26, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Boccia, G.; Di Spirito, F.; D’Ambrosio, F.; Di Palo, M.P.; Giordano, F.; Amato, M. Local and Systemic Antibiotics in Peri-Implantitis Management: An Umbrella Review. Antibiotics 2023, 12, 114. [Google Scholar] [CrossRef]
- Appaneal, H.J.; Caffrey, A.R.; Lopes, V.; Dosa, D.; LaPlante, K.L. Antibiotic Prescribing in Outpatient Settings: Rural Patients Are More Likely to Receive Fluoroquinolones and Longer Antibiotic Courses. Antibiotics 2023, 12, 224. [Google Scholar] [CrossRef]
- Guarch-Ibáñez, B.; Fernández-Polo, A.; Hernández, S.; Velasco-Arnaiz, E.; Giménez, M.; Sala-Castellvi, P.; Pineda, V.; Melendo, S. Assessment of the Plans to Optimize Antimicrobial Use in the Pediatric Population in Catalan Hospitals: The VINCat Pediatric PROA SHARP Survey. Antibiotics 2023, 12, 250. [Google Scholar] [CrossRef]
- Perrella, A.; Fortinguerra, F.; Pierantozzi, A.; Capoluongo, N.; Carannante, N.; Lo Vecchio, A.; Bernardi, F.F.; Trotta, F.; Cangini, A. Hospital Antibiotic Use during COVID-19 Pandemic in Italy. Antibiotics 2023, 12, 168. [Google Scholar] [CrossRef] [PubMed]
- Virhia, J.; Gilmour, M.; Russell, C.; Mutua, E.; Nasuwa, F.; Mmbaga, B.T.; Mshana, S.E.; Dunlea, T.; Shirima, G.; Seni, J.; et al. “If You Do Not Take the Medicine and Complete the Dose…It Could Cause You More Trouble”: Bringing Awareness, Local Knowledge and Experience into Antimicrobial Stewardship in Tanzania. Antibiotics 2023, 12, 243. [Google Scholar] [CrossRef]
- 2021 AWaRe Classification. Available online: https://www.who.int/publications/i/item/2021-aware-classification (accessed on 23 January 2023).
- Mind Me the Antimicrobial Creed. Available online: https://www.safetyandquality.gov.au/sites/default/files/migrated/SAQ113_Antibiotic_Awareness_Week_Card_FILM_MINDME_FINAL.pdf (accessed on 23 January 2023).
- Evans, R.P. Surgical Site Infection Prevention and Control: An Emerging Paradigm. J. Bone Jt. Surg. 2009, 91, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Klugman, K.P.; Black, S. Impact of Existing Vaccines in Reducing Antibiotic Resistance: Primary and Secondary Effects. Proc. Natl. Acad. Sci. USA 2018, 115, 12896–12901. [Google Scholar] [CrossRef] [PubMed]
- Löffler, C.; Böhmer, F. The Effect of Interventions Aiming to Optimise the Prescription of Antibiotics in Dental Care—A Systematic Review. PLoS ONE 2017, 12, e0188061. [Google Scholar] [CrossRef] [PubMed]
| Epidemiology of Antibiotic Consumption and Prescription in Dentistry | ||
|---|---|---|
| Study Characteristics | Title | Conclusion(s) |
| Ahsan, 2020 PLoS One [17] Cross-sectional | “Antibiotic prescription patterns for treating dental infections in children among general and pediatric dentists in teaching institutions of Karachi, Pakistan” | Most dentists, especially with a high volume of pediatric patients, have not shown adherence to professional guidelines in prescribing antibiotics to treat dental infections in children. |
| Al-Harthi, 2013 Saudi Med J [18] Cross-sectional | “Appraisal of antimicrobial prescribing practices of governmental and non-governmental dentists for hospitals in the western region of Saudi Arabia” | Professional guidelines for prescribing antibiotics were not considered attractive to all respondents. The questionnaire showed divergence in antibiotic prescribing among different specialists. |
| Al-Harthi, 2015 Saudi Med J [19] Cross-sectional | “Perceptions and knowledge regarding antimicrobial stewardship among clinicians in Jeddah, Saudi Arabia” | The questionnaire showed divergence in antibiotic prescribing among different specialists, with greater adherence by primary care physicians than other specialists, including dentists. More knowledge was demonstrated on the topic of antimicrobial resistance. |
| Al-Johani, 2017 Niger J Clin Pract [20] Cross-sectional | “Pattern of prescription of antibiotics among dental practitioners in Jeddah, KSA: A cross-sectional survey” | The questionnaire showed a lack of adherence to antibiotic prescribing guidelines among dentists in Jeddah. The antibiotic most commonly used by dentists for most orofacial infections was amoxicillin (73.8%). |
| Al-Taani, 2022 J Infect Dev Ctries [21] Cross-sectional | “Antibiotic use and resistance: Information sources and application by dentists in Jordan” | The questionnaire showed that only 35.5% of dentists surveyed refer to published guidelines for proper antibiotic prescribing in dentistry. Only 9.3% of dentists were aware of national action plans on antibiotic resistance. More than half of the respondents expressed a desire to receive more information on the proper use of antibiotics. |
| Baskaradoss, 2018 J Investing Clin Dent [22] Cross-sectional | “Pattern of antibiotic prescription among dentists in Riyadh, Saudi Arabia” | The most frequently prescribed antibiotic was amoxicillin. Most dentists (more than 70%) prescribed antibiotics to heart patients. Dentists with higher educational qualifications followed more appropriate prescribing patterns than their other colleagues. Antibiotic prescribing patterns were inappropriate among Riyadh dentists. |
| Bhuvaraghan, 2021 Antibiotics (Basel) [23] Systematic Review | “Antibiotic Use and Misuse in Dentistry in India-A Systematic Review” | The study showed significant abuse or misuse in prescribing antibiotics for prophylactic and therapeutic purposes in dentistry, particularly for the use of broad-spectrum antibiotics. Antibiotic self-medication for dental problems by the general population was found to be widespread. |
| Bird, 2018 Br Dent J [24] Cross-sectional | “Higher antibiotic prescribing propensity of dentists in deprived areas and those with greater access to care in the North East and Cumbria” | The rate of antibiotic prescriptions in dentistry is recorded to be higher in deprived areas of the North East and Cumbria. Areas with similar deprivation have shown that the prescribing rate depends on the bias or preferences of the practitioner. |
| Cope, 2016 Br J Gen Pract [25] Retrospective | “Dental consultations in UK general practice and antibiotic prescribing rates: A retrospective cohort study” | In the United Kingdom, consultation rates for dental reasons in general practice are low, but more than half hesitate to prescribe antibiotics. This raises concerns about antibiotic resistance and patient morbidity. |
| Cope, 2016 Community Dent Oral Epidemiol [26] Cross-sectional | “Antibiotic prescribing in UK general dental practice: A cross-sectional study” | The study showed a high rate of inappropriate antibiotic prescriptions among general dental practitioners. In the healthcare setting, numerous antibiotic prescriptions in the absence of infection haves been associated with clinical temporal pressures and patient characteristics, such as expectations for treatment with antibiotics and refusal of surgical treatment. |
| D’Ambrosio, 2022 Healthcare (Basel) [5] Cross-sectional | “Attitudes towards Antibiotic Prescription and Antimicrobial Resistance Awareness among Italian Dentists: What Are the Milestones?” | The main reasons for prescribing antibiotics in dentistry were abscesses, extractions, and pulpits. In case of allergy to penicillins, most dentists have prescribed macrolides, but only a small fraction of them have consulted the guidelines for an antibiotic prescription. In Italy, a high prevalence of misuse and overuse of antibiotics was recorded, similar to other countries. |
| Duncan, 2021 Br Dent J [27] Cross-sectional | ““You had to do something”: prescribing antibiotics in Scotland during the COVID-19 pandemic restrictions and remobilization” | Antibiotic prescriptions increased by 49% after the suspension of routine dental care due to the COVID-19 pandemic. The data showed that following remobilization, antibiotic prescribing remained about 28% higher than in the pre-pandemic period. The survey showed that dentists were concerned about the increased use of antibiotics. |
| Durkin, 2019 J Am Dent Assoc [28] Cross-sectional | “Knowledge and attitudes of recently qualified dentists working in Wales towards antimicrobial prescribing and resistance” | The study found that recently qualified dentists working in Wales were influenced by the use of guidelines and teachings received from students for antibiotic prescriptions. However, most were not confident in treating acute dental conditions. Antibiotic prescribing was also influenced by pressures induced by patients. |
| Epstein, 2000 J Am Dent Assoc [29] Cross-sectional | “A survey of antibiotic use in dentistry” | The questionnaire showed that about 85% of respondents appropriately prescribed antibiotics in dentistry for therapeutic use for dosage and duration. More than 80% of dentists said they follow the American Heart Association’s guidelines for antibiotic prophylaxis, which has been prescribed more frequently for patients with a history of rheumatoid fever, joint replacements, and heart murmur. |
| Farkaš, 2021 Microb Drug Resist [30] Cross-sectional | “Antibiotic Prescribing Habits and Antimicrobial Resistance Awareness of Dental Practitioners in Primorsko-Goranska County, Croatia” | The study showed the overuse of antibiotics among dentists in Croatia, particularly in cases where surgical treatment was the indication of first choice. Broad-spectrum antibiotics were the most prescribed drugs. |
| Garg, 2014 J Antimicrob Chemother [31] Cross-sectional | “Antibiotic prescription pattern among Indian oral healthcare providers: A cross-sectional survey” | The study showed that antibiotics were overprescribed in India, particularly in irreversible pulpitis, necrotic pulp, and acute apical periodontitis. Amoxicillin, with or without clavulanic acid, was the antibiotic of first choice by dentists for nonallergic patients. |
| George, 2022 J Pharm Bioallied Sci [32] Cross-sectional | “Antimicrobial prescription patterns among oral implantologists of Kerala, India: A cross-sectional survey” | Most systemic antibiotic prescriptions in implant surgery were not in accordance with current evidence. Many implantologists prescribed systemic antibiotics for the prevention of infection following simple implant insertions. |
| Gowri, 2015 J Orofacial Sci [33] Cross-sectional | “Antibiotic use in dentistry: A cross-sectional survey from a developing country” | The study showed the lack of knowledge, attitude, and practice of antibiotic use among dentists at a university and hospital institution. |
| Goulao, 2021 Implement Sci [34] Randomised Control Trial | “Audit and feedback with or without training in-practice targeting antibiotic prescribing (TiPTAP): a study protocol of a cluster randomised trial in dental primary care” | Training courses to improve the appropriateness of antibiotic prescriptions in primary dental care have shown good results. |
| Jones, 2018 Eur J Dent Educ [35] Cross-sectional | “Knowledge and attitudes of recently qualified dentists working in Wales towards antimicrobial prescribing and resistance” | Recently qualified dentists in Wales reported that the guidelines and teachings received in the course of the study were the main factors influencing their choices for prescribing antibiotics. However, some participants still did not feel confident in prescribing antibiotics for some acute dental conditions. |
| Kusumoto, 2021 BMC Oral Health [36] Retrospective | “Effect of educational intervention on the appropriate use of oral antimicrobials in oral and maxillofacial surgery: a retrospective secondary data analysis” | The study showed that educational intervention helped dentists prescribe antibiotics more appropriately than in the past. |
| Licata, 2021 Antimicrob Agents Chemother [37] Cross-sectional | “Endodontic Infections and the Extent of Antibiotic Overprescription among Italian Dental Practitioners” | Acute abscesses without systemic involvement and acute apical periodontitis were most frequently associated with antibiotic overprescription among Italian dentists. |
| Lokhasudhan, 2017 J Adv Pharm Educ Res [38] Cross-sectional | “Knowledge, attitude, and practice survey on usage of antibiotics among dental practitioners in southern region of India” | The study showed the over-prescription of antibiotics as an intracanal medicine for prophylaxis before endodontic treatment and for the management of patients with systemic diseases among endodontists and neoendodontists. |
| Mansour, 2018 Pharm Pract (Granada) [39] Cross-sectional | “Knowledge, practice and attitudes regarding antibiotics use among Lebanese dentists” | A lack of uniformity with the guidelines was found in the prophylactic and therapeutic prescriptions of Lebanese dentists. The latter showed greater knowledge of the problems related to antibiotic resistance. |
| McKay, 2020 Br Dent J [40] Cross-sectional | “An analysis of the clinical appropriateness of out-of-hours emergency dental prescribing of antibiotics in Northern Ireland” | The study reported that a high number of antibiotic prescriptions made in out-of-hours emergency dental clinics in Northern Ireland did not follow guidelines. |
| Mengari, 2020 JRMDS [41] Cross-sectional | “Knowledge and Practice of Antibiotic Prescription Among Dentists for Endodontic Emergencies” | The study showed that antibiotic prescriptions for endodontic emergencies were different among dentists working in governmental or private sectors or in educational institutions. General dentists prescribed antibiotics more frequently, even when it was not necessary, than endodontists. |
| Mustafa, 2022 Eur J Dent [42] Cross-sectional | “Administration of Systemic Antibiotics for Dental Treatment in Kosovo Major Dental Clinics: A National Survey” | Prescriptions of amoxicillin with or without clavulanic acid increased dramatically from 2015 to 2019 in dental clinics in Kosovo. |
| Nourah, 2021 Int J Med Dent [43] Cross-sectional | “Prescribing practice of systemic antibiotics by periodontists in Saudi Arabia” | Patterns of systemic antibiotic prescriptions were heterogeneous among periodontists in Saudi Arabia. |
| Ogunbodede, 2005 J Contemp Dent Pract [44] Retrospective | “Retrospective survey of antibiotic prescriptions in dentistry” | The total number of drugs prescribed for dental reasons ranged from one to seven, and penicillins were the most prescribed antibiotic. The dose, frequency, and duration were wrong in some prescriptions. Indications on the best time to take the antibiotic in relation to meals had not been specified in any prescription. |
| Ono, 2020 PLoS One [45] Cross-sectional | “The first national survey of antimicrobial use among dentists in Japan from 2015 to 2017 based on the national database of health insurance claims and specific health checkups of Japan” | Cephalosporins were the antibiotic most prescribed by Japanese dentists between 2015 and 2017. At the same time, around 99% of outpatients had been prescribed an antibiotic. |
| Osailan, 2021 J Pharm Policy Pract [46] Cross-sectional | “Knowledge and Attitude towards Antibiotics Prescription and Antimicrobial Resistance among Dental Surgeons in Saudi Arabia” | The study showed that antibiotic prescriptions among Saudi Arabian oral surgeons were inappropriate in most cases. Young age, male gender, higher level of studies, and poor aptitude were factors related to inappropriate antibiotic use. |
| Palmer, 2001 J Antimicrob Chemother [47] Cross-sectional | “Antibiotic prescribing knowledge of National Health Service general dental practitioners in England and Scotland” | The study showed that dentists who had taken at least one postgraduate course on antibiotic use had significantly greater knowledge. Significant differences were also found in the appropriateness of antibiotic prescriptions of dentists in English Health Authorities compared to dentists in Scottish Health Boards. |
| Palmer, 2019 Prim Dent J [48] Cross-sectional | “A Pilot Study to Investigate Antibiotic Prescribing in Private Dental Practice in the UK” | The study showed that UK dentists working in private structures prescribed antibiotics less and more appropriately than NHS dentists. |
| Pisarnturakit, 2020 Int J Health Plann Manage [49] Cross-sectional | “Managing knowledge for health care quality: An investigation of rational antibiotic use among Thai dentists” | Thai dentists have been largely shown to use antibiotics appropriately. Mobile applications were reported as the preferred means of filling the remaining gaps in knowledge of antibiotic use. |
| Rodriguez-Núñez, 2009 J Endod [50] Cross-sectional | “Antibiotic Use by Members of the Spanish Endodontic Society” | Most members of the Spanish Endodontic Society had indicated the appropriate antibiotic therapy for several orofacial infections. However, some inappropriate choices were made in some cases of minimal or no infections. |
| Rubanenko, 2021 Antibiotics (Basel) [51] Cross-sectional | “Assessment of the knowledge and approach of general dentists who treat children and pediatric dentists regarding the proper use of antibiotics for children” | The level of knowledge about when antibiotics should or should not be used in children is poor among both general and pediatric dentists. Antibiotic prescriptions, in a few cases, have been in accordance with European and American Pediatric Dentistry guidelines. |
| Salako, 2004 J Dent [52] Cross-sectional | “Pattern of antibiotic prescription in the management of oral diseases among dentists in Kuwait” | The study showed a lack of uniformity in prescribing antibiotics for oral diseases among Kuwaiti dentists. Uncertainty in diagnosis, patient expectations, and lack of time for immediate treatment were the main factors influencing antibiotic prescriptions. |
| Schmidt, 2021 Int J Environ Res Public Health [53] Systematic Review | “A Review of Evidence-Based Recommendations for Pericoronitis Management and a Systematic Review of Antibiotic Prescribing for Pericoronitis among Dentists: Inappropriate Pericoronitis Treatment Is a Critical Factor of Antibiotic Overuse in Dentistry” | Pericoronitis was the second leading cause of antibiotic use in dentistry. Antibiotics, particularly amoxicillin or metronidazole, were given to more than half of the subjects with pericoronitis. Antibiotic prescriptions were inappropriate and noncompliant with guidelines in most cases. |
| Sefah, 2022 JAC-antimicrobial resistance [54] Retrospective | “Evaluation of antibiotic prescribing for ambulatory patients seeking primary dental care services in a public hospital in Ghana: a clinical audit study” | Antibiotics were prescribed to more than 90 percent of patients who required primary dental care services in Ghana’s public hospitals. In almost all cases, the antibiotic prescription was not in accordance with the Ghana Standard Treatment Guidelines. |
| Segura-Egea, 2010 Int Endod J [55] Cross-sectional | “Pattern of antibiotic prescription in the management of endodontic infections amongst Spanish oral surgeons” | Most members of the Spanish Oral Surgery Society had selected the appropriate antibiotic for endodontic infections; however, still, many prescribed the antibiotics inappropriately. Odontologists have more frequently prescribed antibiotics than stomatologists for necrotic teeth with sinus tract and chronic apical periodontitis. |
| Shalini, 2022 Indian Drugs [56] Cross-sectional | “Knowledge and attitude of antibiotic prescription among implantologists: an observational study” | The study showed a lack of congruence among implantologists about the recommended protocols for the use of antibiotics for the prophylaxis or management of implant complications. |
| Shemesh, 2022 Clin Oral Investig [57] Cross-sectional | “International questionnaire study on systemic antibiotics in endodontics. Part 1. Prescribing practices for endodontic diagnoses and clinical scenarios” | The study showed a lack of congruence between the recommended protocols for the use of systemic antibiotics for endodontic treatment and the clinical prescriptions of Israeli and Soviet dentists. |
| Sneddon, 2022 Antibiotics [58] Cross-sectional | “Exploring the Use of Antibiotics for Dental Patients in a Middle-Income Country: Interviews with Clinicians in Two Ghanaian Hospitals” | The rate of antibiotic prescriptions in dentistry had been influenced by the work environment, clinical issues such as lack of available sterile instrumentation, and patient preferences or needs. |
| Teoh, 2020 Antibiotics (Basel) [59] Systematic Review | “Measuring Antibiotic Stewardship Programmes and Initiatives: An Umbrella Review in Primary Care Medicine and a Systematic Review of Dentistry” | Most of the antibiotic prescriptions (80%) take place in dentistry, but the increase in prescriptions also includes primary medical care. |
| Teoh, 2019 BMC Oral Health [60] Cross-sectional | “A survey of prescribing practices by general dentists in Australia” | Dentists with less than five years since graduation had a lower rate of antibiotic prescription abuse than their colleagues. Years of practice were the demographic factor that most influenced the antibiotic prescription rate. |
| Thompson, 2019 J Antimicrob Chemother [61] Systematic Review | “Factors associated with antibiotic prescribing for adults with acute conditions: an umbrella review across primary care and a systematic review focusing on primary dental care” | Factors potentially influencing antibiotic prescriptions were clinician-related, clinical context-related, patient-related, and social–political context-related. |
| Verma, 2022 World J Dentistry [62] Cross-sectional | “Antibiotic Prescribing Practices amongst the Dental Practitioners of Bhubaneswar City: A Cross-sectional Study” | The study showed gaps in knowledge of antibiotic prescription guidelines among dentists in the city of Bhubaneswar. |
| Antibiotic Therapy in Dentistry | ||
|---|---|---|
| Study Characteristics | Title | Conclusion(s) |
| Cope, 2015 BMJ Open [63] Qualitative Study | “General practitioners’ attitudes towards the management of dental conditions and use of antibiotics in these consultations: a qualitative study” | Antibiotics were considered the first-line treatment for many primary care physicians for the immediate management of acute dental problems. Often the patients themselves required the administration of the antibiotic. General practitioners who prescribed antibiotics rarely wanted to encourage patients to visit dentistry. |
| Froum, 2015 Int J Periodontics Restorative Dent [64] Cross-sectional | “An evaluation of antibiotic use in periodontal and implant practices” | The prescription, duration, and initiation of antibiotics were very heterogeneous in the ten therapeutic treatments in periodontal and implant practice, such as the treatment of acute and chronic periodontitis, sinus lifts or crest augmentation surgery, and immediate or delayed implant placement. |
| Kaul, 2021 J Family Med Prim Care [65] Cross-sectional | “Oral pain and infection control strategies for treating children and adolescents in India” | The study recorded the overuse of antibiotics and analgesics, especially in the postoperative phase and by general dentists, compared to specialists, for pain control and treatment of infections in children and adolescents. |
| Khattri, 2020 Cochrane Database Syst Rev [66] Systematic Review with Meta-analysis | “Adjunctive systemic antimicrobials for the non-surgical treatment of periodontitis” | No statistically significant evidence was found on the long-term efficacy of systemic antibiotics used in addition to non-surgical periodontitis treatments. The superiority of one antibiotic over the others has not been defined either. |
| Silva, 2017 SPMED [67] Cross-sectional | “The use of systemic antibiotics in endodontics: a cross-sectional study” | A considerable part of Portuguese dentists had inappropriately prescribed antibiotics as a therapeutic treatment for inflammatory endodontic diseases. |
| Thompson, 2022 Trials [68] Cross-sectional | “Dental antibiotic stewardship: study protocol for developing international consensus on a core outcome set” | The study showed that current international guidelines for antibiotic therapy in dentistry are very heterogeneous. |
| Antibiotic Prophylaxis in Dentistry | ||
|---|---|---|
| Study Characteristics | Title | Conclusion(s) |
| Bianco, 2021 Antibiotics (Basel) [69] Cross-sectional | “Appropriateness of Antibiotic Prescription for Prophylactic Purposes among Italian Dental Practitioners: Results from a Cross-Sectional Study” | The prescription of antibiotics by Italian dentists for prophylactic reasons was found to be not in accordance with the guidelines in 70.9% of cases. |
| Ireland, 2012 Br Dent J [70] Cross-sectional | “An investigation of antibiotic prophylaxis in implant practice in the UK” | Pre- and post-operative prescription regimens of antibiotics in dental implant practice in the United Kingdom are highly variable. In most cases, antibiotics were prescribed to prevent infection of the surgical site or to reduce bacteremia. |
| Kirnbauer, 2022 Clin Oral Investig [71] Randomized Control Trial | “Is perioperative antibiotic prophylaxis in the case of routine surgical removal of the third molar still justified? A randomized, double-blind, placebo-controlled clinical trial with a split-mouth design” | Oral antibiotics administered in the perioperative phase of routine surgical extractions of wisdom teeth in the absence of inflammation did not show a reduction in post-operative complications or greater benefit in patient-related outcome measures. |
| Lollobrigida, 2021 Antibiotics (Basel) [72] Cross-sectional | “Antibiotics to Prevent Surgical Site Infection (SSI) in Oral Surgery: Survey among Italian Dentists” | The choices reported by the dentists about antibiotic prescriptions for the prevention of infections in post-extraction surgical sites were appropriate for deciduous tooth extractions and simple extractions in healthy adult subjects. However, responses were more heterogeneous in adult extractions with comorbidities, complex or multiple extractions, drainage abscesses, and implant placement. The dosage to be used was also found to be very heterogeneous. |
| Salgado-Peralvo, 2021 Antibiotics (Basel) [73] Systematic Review | “Preventive Antibiotic Therapy in the Placement of Immediate Implants: A Systematic Review” | The study showed the efficacy of prophylactic treatment with 2–3 g of amoxicillin one hour before immediate implant placement and after for 5–7 days at a dosage of 500 mg every 8 h to reduce the rate of early failure. In subjects with penicillin allergy, azithromycin, clarithromycin, or metronidazole were recommended, but not clindamycin, if possible. |
| Salgado-Peralvo, 2022 Antibiotics (Basel) [74] Systematic Review | “Is Antibiotic Prophylaxis Necessary before Dental Implant Procedures in Patients with Orthopaedic Prostheses? A Systematic Review” | There is no evidence showing a relationship between implant placement and an increased risk of orthopedic prosthesis infection. Therefore, the authors concluded that antibiotic prophylaxis is not justified in these cases. |
| Salgado-Peralvo, 2022 J Stomatol Oral Maxillofac Surg [75] Systematic Review | “Preventive antibiotic therapy in bone augmentation procedures in oral implantology: A systematic review” | The study showed that the administration of 2/3 g of amoxicillin one hour before bone augmentation procedures in oral implantology allowed a reduction in the rate of early implant failure and the infection risk of the grafted bone particles. |
| Sato, 2022 Oral Dis [76] Retrospective | “Amoxicillin vs. third-generation cephalosporin for infection prophylaxis after third molar extraction” | Antibiotic prophylaxis with amoxicillin prior to extraction of impacted third molars showed a lower incidence of surgical site infections than using third-generation cephalosporins. |
| Sato, 2022 J Infect Chemother [77] Retrospective | “Trends in prophylactic antibiotic use for tooth extraction from 2015 to 2018 in Japan: An analysis using a health insurance claims database” | The trend in antibiotic prophylaxis for the extraction of third molars changed from 2015 to 2018 in Japan after the National Action Plan, with an increase in the use of amoxicillin and a decrease in third-generation cephalosporins. |
| Sologova, 2022 Dent J (Basel) [78] Systematic Review | “Antibiotics Efficiency in the Infection Complications Prevention after Third Molar Extraction: A Systematic Review” | The study showed that amoxicillin, with or without clavulanic acid, is the most widely used antibiotic at different dosages and durations to prevent infectious complications following the extraction of third molars. |
| Sing Gill, 2018 Medicina (Kaunas) [79] Systematic Review with Meta-Analysis | “A Systematic Review and Meta-Analysis Evaluating Antibiotic Prophylaxis in Dental Implants and Extraction Procedures” | The use of antibiotics prophylactically for third-molar extractions in healthy patients was supported by little evidence. In contrast, no significant evidence showed a higher incidence of adverse events to antibiotics compared with placebo. |
| Williams, 2020 Br Dent J [80] Cross-sectional | “Antibiotic prophylaxis during dental implant placement in the UK” | The study showed heterogeneous choices to prescribe antibiotics prophylactically for implant placement. Almost half of the dentists did not prescribe antibiotics routinely. In other cases, antibiotics were prescribed for complex procedures and by more qualified dentists. |
| Dental Patients’ Adherence to Antibiotic Prescription | |
|---|---|
| Total | 0 studies |
| Antimicrobial Resistance in Dentistry | ||
|---|---|---|
| Study Characteristics | Title | Conclusion(s) |
| Abe, 2022 Front Microbiol [81] Systematic Review with Meta-Analysis | “Antimicrobial resistance of microorganisms present in periodontal diseases: A systematic review and meta-analysis” | No evidence was found for the presence of specific antibiotic resistance profiles in microorganisms implicated in periodontal disease. The highest antibiotic resistance recorded was for ampicillin, while the lowest was for ciprofloxacin. |
| Abe, 2018 Medicine [82] Systematic Review | “Resistance profile to antimicrobial agents in the main circulating bacteria isolated from acute periodontal and endodontic infections in Latin America (MICROBE- DENT) A systematic review protocol” | Further studies are needed to assess the prevalence of antimicrobial resistance in endodontics and periodontics in Latin America. |
| Almeida, 2020 PLoS One [83] Cross-sectional | “Bacterial diversity and prevalence of antibiotic resistance genes in the oral microbiome” | No significant differences were found between the taxonomies of healthy or diseased oral microbiomes. However, healthy subjects showed a more diverse microbiological community. At least one antibiotic resistance gene was found in 72.7% of the samples. |
| Alzahrani, 2020 Risk Manag Healthc Policy [84] Retrospective | “Inappropriate Dental Antibiotic Prescriptions: Potential Driver of the Antimicrobial Resistance in Albaha Region, Saudi Arabia” | Misuse and abuse of antibiotic prescriptions have been found among dentists in Saudi Arabia. The inappropriate use of antibiotics could lead to the development of antibiotic resistance phenomena. |
| Groppo, 2005 Gen Dent [85] Clinical Trial | “Antimicrobial resistance of Staphylococcus aureus and oral streptococci strains from high-risk endocarditis patients” | Microorganisms causing bacterial endocarditis have shown high rates of antimicrobial resistance to antibiotics commonly used for prophylaxis in dentistry. |
| Irshad, 2020 Antibiotics (Basel) [86] Clinical Trial | “Characterization and Antimicrobial Susceptibility of Pathogens Associated with Periodontal Abscess” | The study showed that several bacterial species isolated from periodontal abscesses had high rates of antimicrobial resistance to amoxicillin, tetracyclines, and metronidazole, while azithromycin was not associated with antimicrobial resistance in these cases. |
| Kiros, 2022 Biomed Res Int [87] Cross-sectional | “Bacterial Profile, Antimicrobial Susceptibility Pattern, and Associated Factors among Dental Caries-Suspected Patients Attending the Ayder Comprehensive Specialized Hospital and Private Dental Clinic in Mekelle, Northern Ethiopia” | The study recorded multidrug resistance of 40.4% of microorganisms associated with dental caries. The highest resistance rate was found for penicillin and tetracyclines. |
| Lang, 2016 Int J Antimicrob Agents [88] Systematic Review with Meta-Analysis | “Resistance profiles to antimicrobial agents in bacteria isolated from acute endodontic infections: systematic review and meta-analysis” | The study showed that the antibiotic resistance profiles of bacteria causing acute endodontic infections were lower for amoxicillin. In addition, resistance rates increased when multiple cycles of antibiotics were given. |
| Laumen, 2021 Front Microbiol [89] Clinical Trial | “Sub-Inhibitory Concentrations of Chlorhexidine Induce Resistance to Chlorhexidine and Decrease Antibiotic Susceptibility in Neisseria gonorrhoeae” | The frequent use of chlorhexidine led to an increased rate of resistance of Neisseria gonorrhoeae to chlorhexidine itself and other antibiotics. |
| Rodrigues, 2004 J Clin Periodontol [90] Clinical Trial | “Antibiotic resistance profile of the subgingival microbiota following systemic or local tetracycline therapy” | Local or systemic administration of tetracycline in subjects with chronic periodontitis resulted in the transient selection of subgingival microorganisms intrinsically resistant to tetracycline itself. |
| Santos, 2002 Anaerobe [91] Clinical Trial | “Susceptibility of Prevotella intermedia/Prevotella nigrescens (and Porphyromonas gingivalis) to propolis (bee glue) and other antimicrobial agents” | Prevotella intermedia and Prevotella nigrescens were susceptible to penicillins, meropenem, erythromycin, and metronidazole, while a smaller percentage were susceptible to tetracyclines and a considerable number were resistant to clindamycin. Propolis was an effective alternative to periodontal pathogens. |
| van Winkelhoff, 2000 J Clin Periodontol [92] Comparative Study | “Antimicrobial resistance in the subgingival microflora in patients with adult periodontitis. A comparison between The Netherlands and Spain” | The increased use of antibiotics in Spain has led to a higher rate of antibiotic resistance of subgingival microorganisms in adult subjects with periodontitis. The resistance rate was found to be much lower among Dutch patients. |
| Vijayashree, 2018 J Clin Diagnostic Res [93] Retrospective | “Enterococcus faecalis an Emerging Microbial Menace in Dentistry-An Insight into the In silico Detection of Drug Resistant Genes and Its Protein Diversity” | Enterococcus Faecalis has been associated with endodontic and periodontal infections. The study showed that the bacterial genome hosts one or more genes encoding resistance to the most common antibiotics used in dentistry. |
| General Factors | Mechanisms |
|---|---|
| Food chain | Vegetables and meats from crops and farms using massive doses of antibiotics lead to the selection of super-resistant bacteria that can be transmitted to the final consumer (the human) when not adequately sanitized |
| Iatrogenic |
|
| Additional factors in dentistry | |
| Bacterial plaque and biofilm | A mature and thick dental plaque creates a progressive lower penetration of the antibiotic molecules through the biofilm layers, from the outer to the inner ones, thus invalidating the effectiveness of topical antibiotics and making systemic ones useless |
| Local inflammation | Local inflammation, as those occurring in periodontitis, is responsible for changes in pH that affect the antibiotic activities |
| Chlorhexidine |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Contaldo, M.; D’Ambrosio, F.; Ferraro, G.A.; Di Stasio, D.; Di Palo, M.P.; Serpico, R.; Simeone, M. Antibiotics in Dentistry: A Narrative Review of the Evidence beyond the Myth. Int. J. Environ. Res. Public Health 2023, 20, 6025. https://doi.org/10.3390/ijerph20116025
Contaldo M, D’Ambrosio F, Ferraro GA, Di Stasio D, Di Palo MP, Serpico R, Simeone M. Antibiotics in Dentistry: A Narrative Review of the Evidence beyond the Myth. International Journal of Environmental Research and Public Health. 2023; 20(11):6025. https://doi.org/10.3390/ijerph20116025
Chicago/Turabian StyleContaldo, Maria, Francesco D’Ambrosio, Giuseppe A. Ferraro, Dario Di Stasio, Maria Pia Di Palo, Rosario Serpico, and Michele Simeone. 2023. "Antibiotics in Dentistry: A Narrative Review of the Evidence beyond the Myth" International Journal of Environmental Research and Public Health 20, no. 11: 6025. https://doi.org/10.3390/ijerph20116025
APA StyleContaldo, M., D’Ambrosio, F., Ferraro, G. A., Di Stasio, D., Di Palo, M. P., Serpico, R., & Simeone, M. (2023). Antibiotics in Dentistry: A Narrative Review of the Evidence beyond the Myth. International Journal of Environmental Research and Public Health, 20(11), 6025. https://doi.org/10.3390/ijerph20116025










