Effect of Amnioguard and Biomesh GTR Membranes with Novabone Putty in the Treatment of Periodontal Osseous Defects—A Communication
Abstract
1. Introduction
2. Materials and Methods
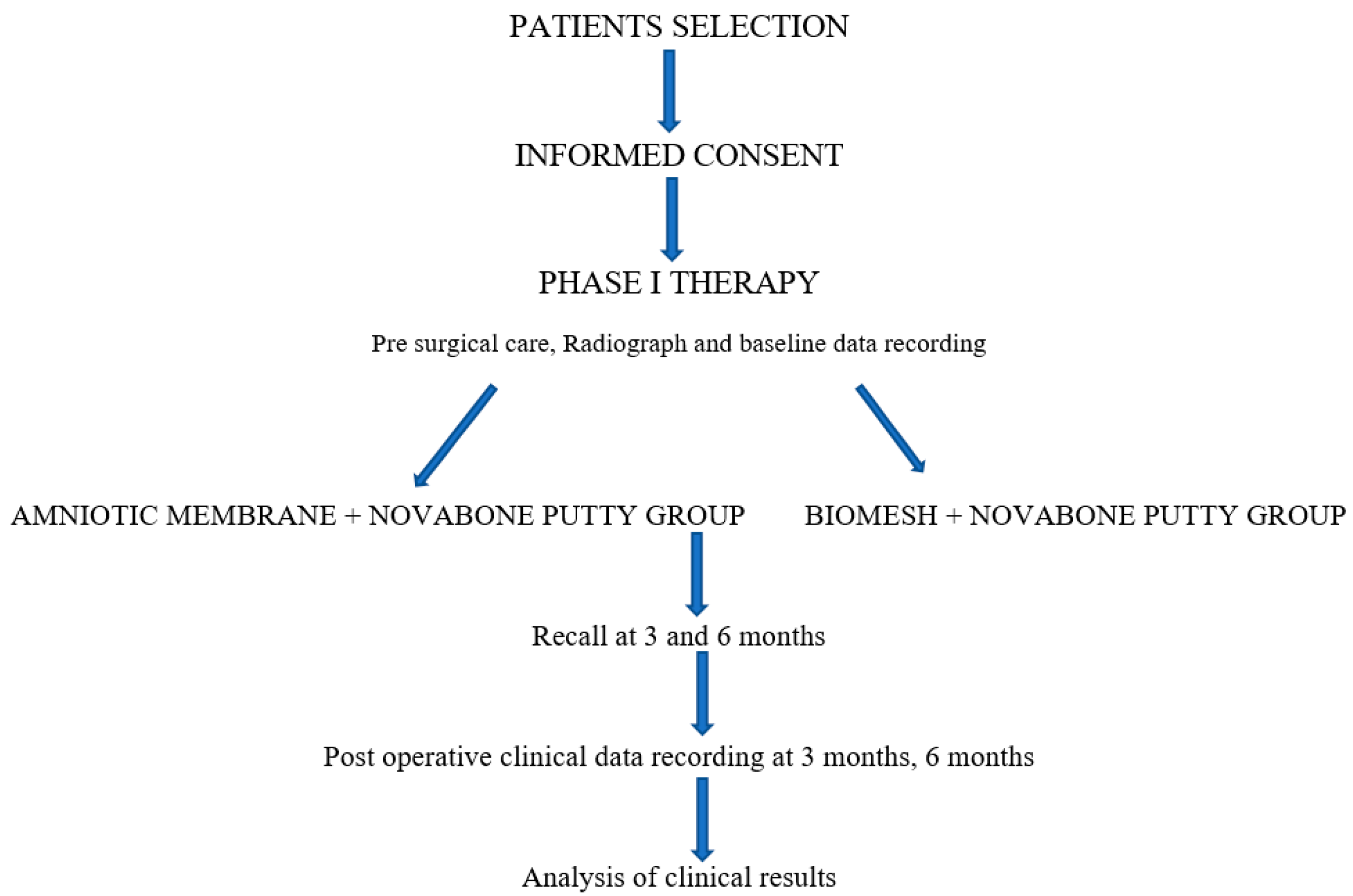
2.1. Study Design
2.2. Clinical Recordings
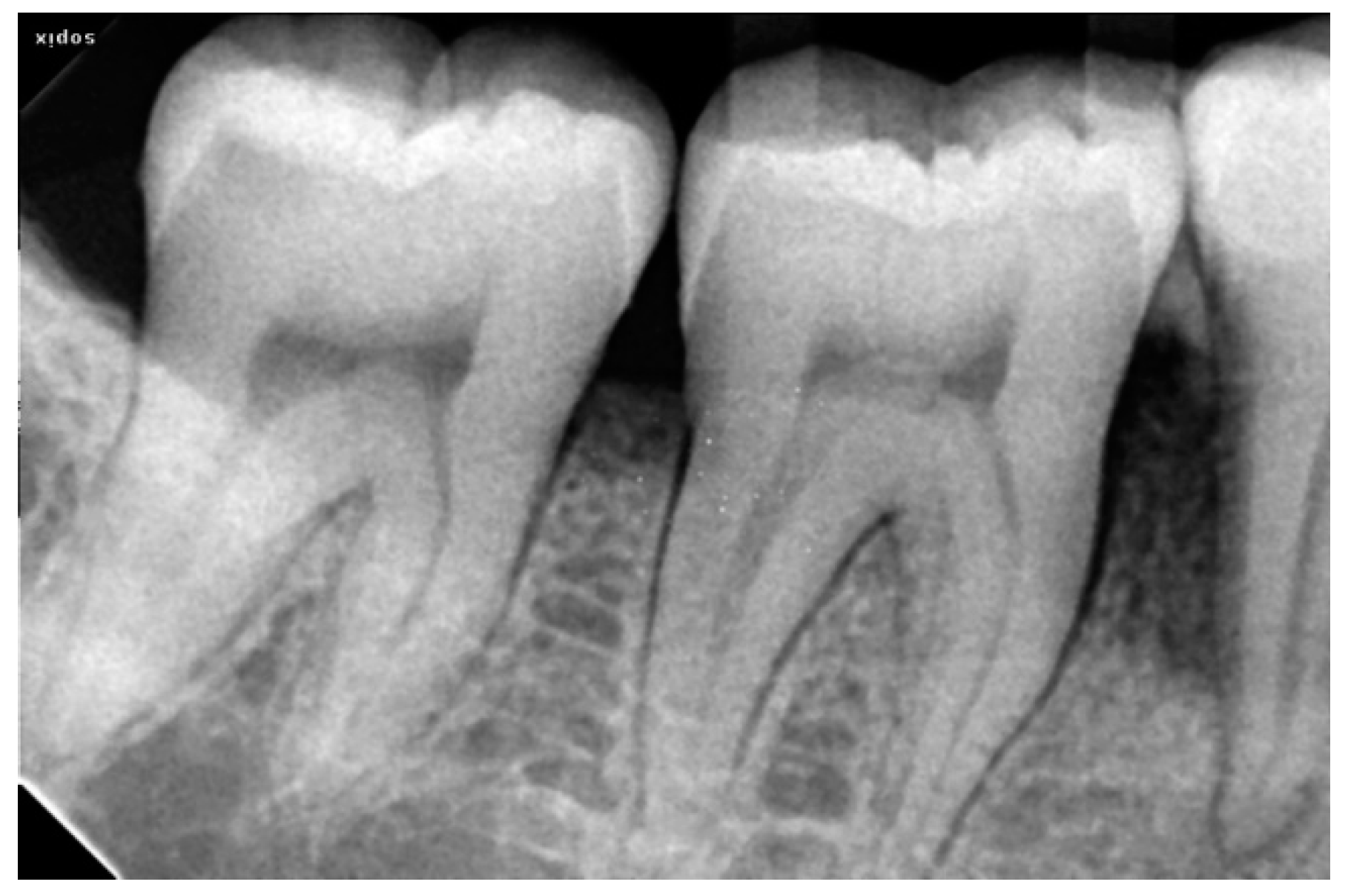
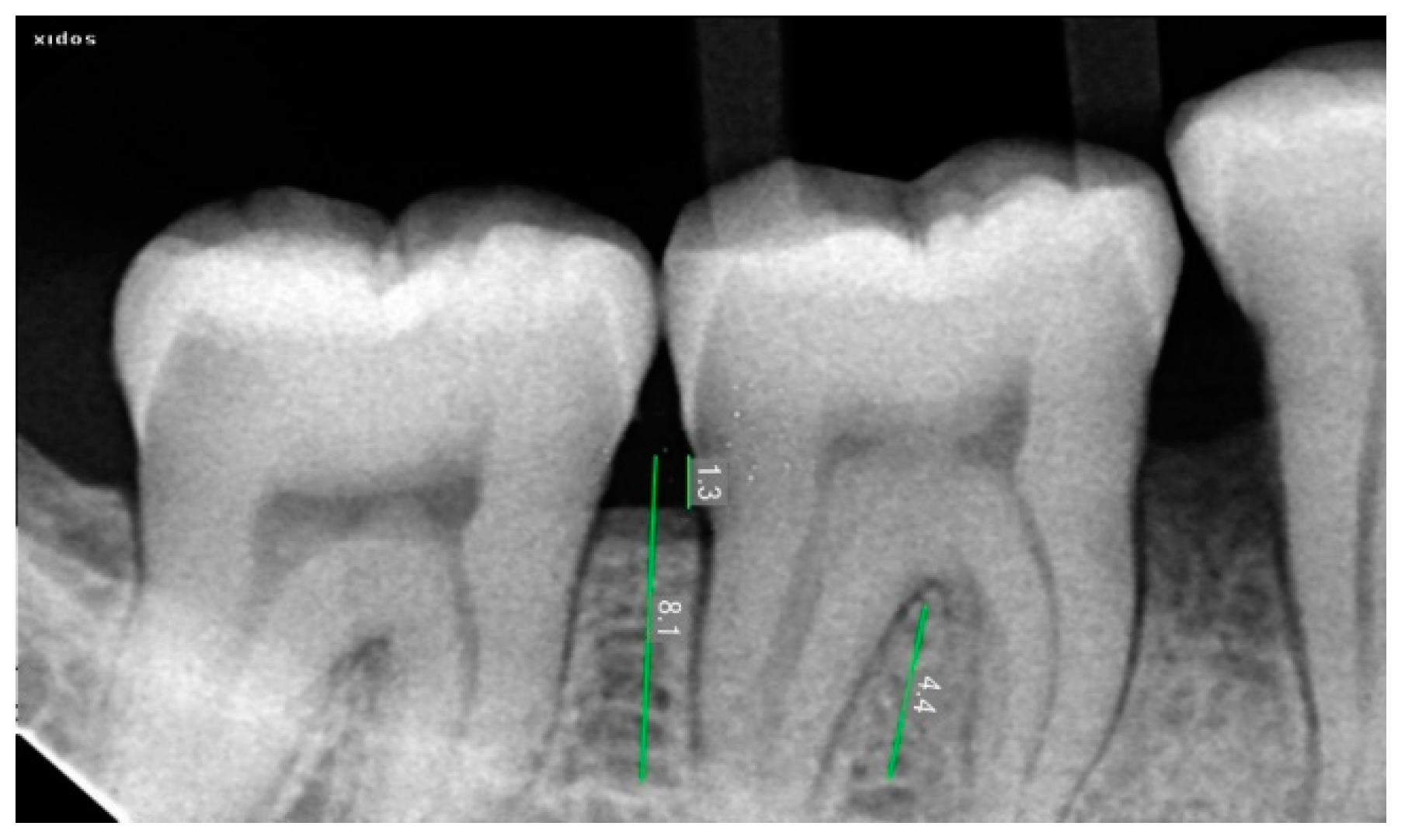
2.3. Radiographic Assessment
2.4. Surgical Procedure
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Melcher, A.H. On the Repair Potential of Periodontal Tissues. J. Periodontol. 1976, 47, 256–260. [Google Scholar] [CrossRef] [PubMed]
- Nyman, S.; Gottlow, J.; Karring, T.; Lindhe, J. The Regenerative Potential of the Periodontal Ligament. An Experimental Study in the Monkey. J. Clin. Periodontol. 1982, 9, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Gottlow, J.; Nyman, S.; Lindhe, J.; Karring, T.; Wennstrom, J. New Attachment Formation in the Human Periodontium by Guided Tissue Regeneration Case Reports. J. Clin. Periodontol. 1986, 13, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, M.; Sridhar, M.S.; Sangwan, V.S.; Rao, G.N. Amniotic Membrane Transplantation for Ocular Surface Reconstruction. Cornea 2005, 24, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Vandana, K.; Gupta, I. The Location of Cemento Enamel Junction for CAL Measurement: A Clinical Crisis. J. Indian Soc. Periodontol. 2009, 13, 12. [Google Scholar] [CrossRef] [PubMed]
- Hefti, A.F. Periodontal Probing. Crit. Rev. Oral Biol. Med. 1997, 8, 336–356. [Google Scholar] [CrossRef] [PubMed]
- Law, E.J.; Taib, H.; Berahim, Z. Amniotic Membrane: An Approach to Periodontal Regeneration. Cureus 2022, 14, e27832. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, N.; Lavu, V.; Balaji, S.K. Clinical Efficacy of Amniotic Membrane with Biphasic Calcium Phosphate in Guided Tissue Regeneration of Intrabony Defects- a Randomized Controlled Clinical Trial. Biomater. Res. 2021, 25, 15. [Google Scholar] [CrossRef] [PubMed]
- Elahi, A.; Taib, H.; Berahim, Z.; Ahmad, A.; Ab Hamid, S.S.; Mocktar, N.A. Amniotic Membrane as a Scaffold for Periodontal Tissue Engineering. J. Health Sci. Med. Res. 2021, 39, 169–180. [Google Scholar] [CrossRef]
- Suh, J.-J.; Chung, Y.-J.; Choi, B.-G.; Choi, S.-H.; Cho, K.-S. Clinical Study on Therapeutic Effects of Biodegradable Membrane Biomesh® and Autogenous Bone Grafts in Infrabony Defects. J. Korean Acad. Periodontol. 2000, 30, 779. [Google Scholar] [CrossRef]
- Srivastava, S.; Tandon, P.; Gupta, K.; Srivastava, A.; Kumar, V.; Shrivastava, T. A Comparative Clinico-Radiographic Study of Guided Tissue Regeneration with Bioresorbable Membrane and a Composite Synthetic Bone Graft for the Treatment of Periodontal Osseous Defects. J. Indian Soc. Periodontol. 2015, 19, 416. [Google Scholar] [CrossRef] [PubMed]
- Kiany, F.; Moloudi, F. Amnion Membrane as a Novel Barrier in the Treatment of Intrabony Defects: A Controlled Clinical Trial. Int. J. Oral Maxillofac. Implants 2015, 30, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Mopur, J.M.; Devi, T.R.; Ali, S.M.; Srinivasa, T.; Gopinath, V.; Salam, A.T. Clinical and Radiographic Evaluation of Regenerative Potential of GTR Membrane (Biomesh®) along with Alloplastic Bone Graft (Biograft®) in the Treatment of Periodontal Intrabony Defects. J. Contemp. Dent. Pract. 2013, 14, 434–439. [Google Scholar] [CrossRef] [PubMed]




| Pre Operative | Post Operative | ||
|---|---|---|---|
| Baseline | 3 months | ||
| (Mean ± SD) | (Mean ± SD) | ||
| AM + NB | 8.60 ± 1.17 mm | 6.90 ± 1.10 mm | AM + NB |
| BM + NB | 8.70 ± 0.95 mm | 6.10 ± 1.20 mm | BM + NB |
| p value | 0.853 | 0.165 | p value |
| Pre Operative | Post Operative | ||
|---|---|---|---|
| Baseline | 3 months | ||
| (Mean ± SD) | (Mean ± SD) | ||
| AM + NB | 13.70 ± 0.95 mm | 12.00 ± 0.81 mm | AM + NB |
| BM + NB | 14.10 ± 0.87 mm | 11.50 ± 1.35 mm | BM + NB |
| p value | 0.393 | 0.315 | p value |
| Defect Fill (Mean ± SD) | |
|---|---|
| AM + NB | 1.25 ± 0.38 mm |
| BM + NB | 2.64 ± 0.44 mm |
| p value | 0.0005 |
| AM + NB | 16% |
| BM + NB | 33% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chakraborthy, P.; Ravishankar, P.L.; Saravanan, A.V.; Alzahrani, K.J.; Halawan, I.F.; Alshammeri, S.; Alnfiai, M.M.; Baeshen, H.A.; Qattan, S.Y.M.; Almarghlani, A.; et al. Effect of Amnioguard and Biomesh GTR Membranes with Novabone Putty in the Treatment of Periodontal Osseous Defects—A Communication. Int. J. Environ. Res. Public Health 2023, 20, 816. https://doi.org/10.3390/ijerph20010816
Chakraborthy P, Ravishankar PL, Saravanan AV, Alzahrani KJ, Halawan IF, Alshammeri S, Alnfiai MM, Baeshen HA, Qattan SYM, Almarghlani A, et al. Effect of Amnioguard and Biomesh GTR Membranes with Novabone Putty in the Treatment of Periodontal Osseous Defects—A Communication. International Journal of Environmental Research and Public Health. 2023; 20(1):816. https://doi.org/10.3390/ijerph20010816
Chicago/Turabian StyleChakraborthy, Priyankar, Potluri Leela Ravishankar, Anbukumari Vadivelu Saravanan, Khalid J. Alzahrani, Ibrahim F. Halawan, Saleh Alshammeri, Mrim M. Alnfiai, Hosam Ali Baeshen, Sarah Yasser M. Qattan, Ammar Almarghlani, and et al. 2023. "Effect of Amnioguard and Biomesh GTR Membranes with Novabone Putty in the Treatment of Periodontal Osseous Defects—A Communication" International Journal of Environmental Research and Public Health 20, no. 1: 816. https://doi.org/10.3390/ijerph20010816
APA StyleChakraborthy, P., Ravishankar, P. L., Saravanan, A. V., Alzahrani, K. J., Halawan, I. F., Alshammeri, S., Alnfiai, M. M., Baeshen, H. A., Qattan, S. Y. M., Almarghlani, A., Karkala, S. R., Neelakandan, A., & Patil, S. (2023). Effect of Amnioguard and Biomesh GTR Membranes with Novabone Putty in the Treatment of Periodontal Osseous Defects—A Communication. International Journal of Environmental Research and Public Health, 20(1), 816. https://doi.org/10.3390/ijerph20010816







