Short-Term Effect of Fly Ash from Biomass Combustion on Spring Rape Plants Growth, Nutrient, and Trace Elements Accumulation, and Soil Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Pot Experiment Design
2.2. Analysis of Plant Samples
2.2.1. Relative Chlorophyll Content (CCl)
2.2.2. Analysis of the Mineral Composition of Seeds and Aboveground Parts of Rape Plants
2.3. Soil Properties Analysis
2.4. Soil Solution Samples Analysis
2.5. Statistical Analysis
3. Results
3.1. Physicochemical Properties of BAs
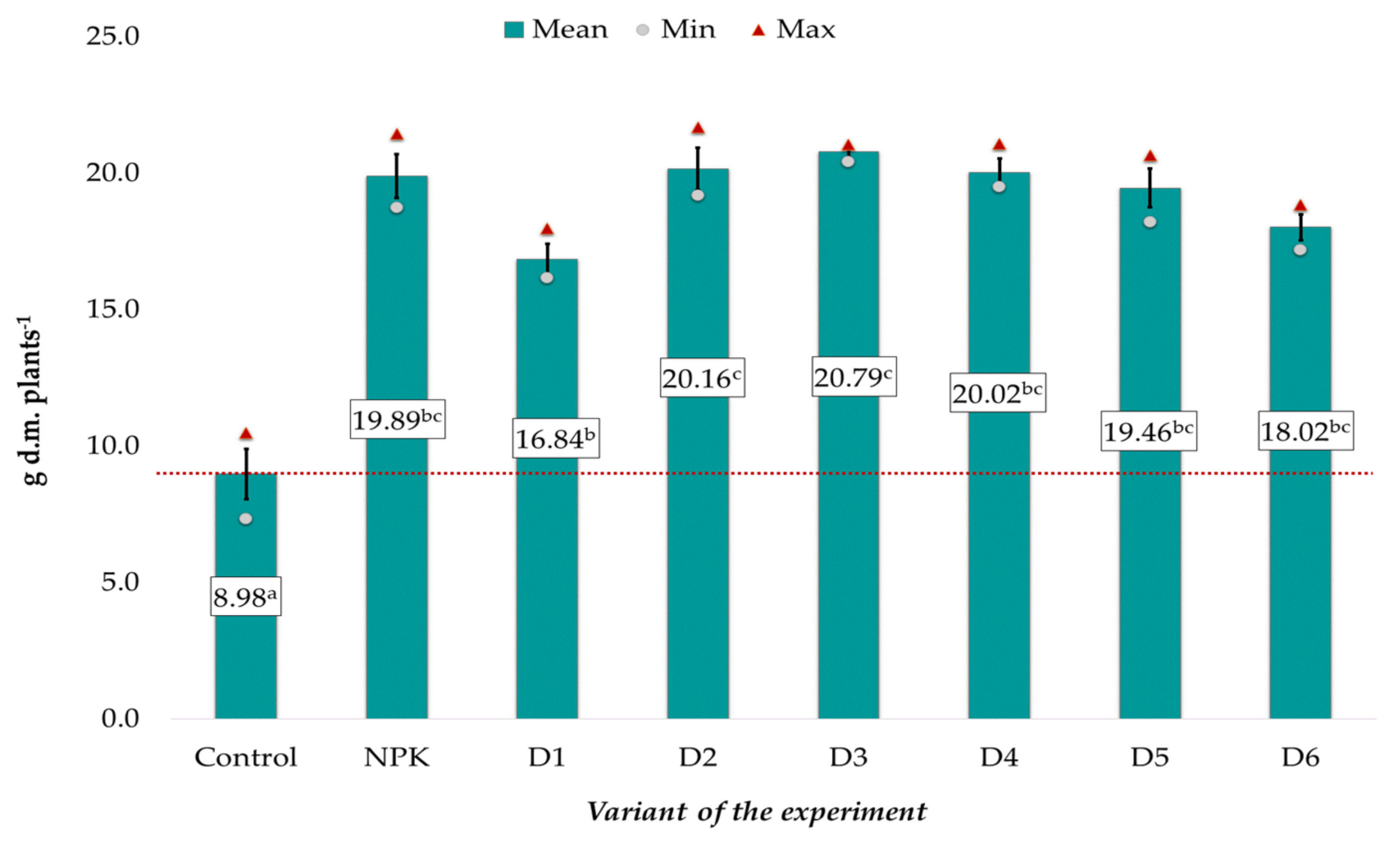
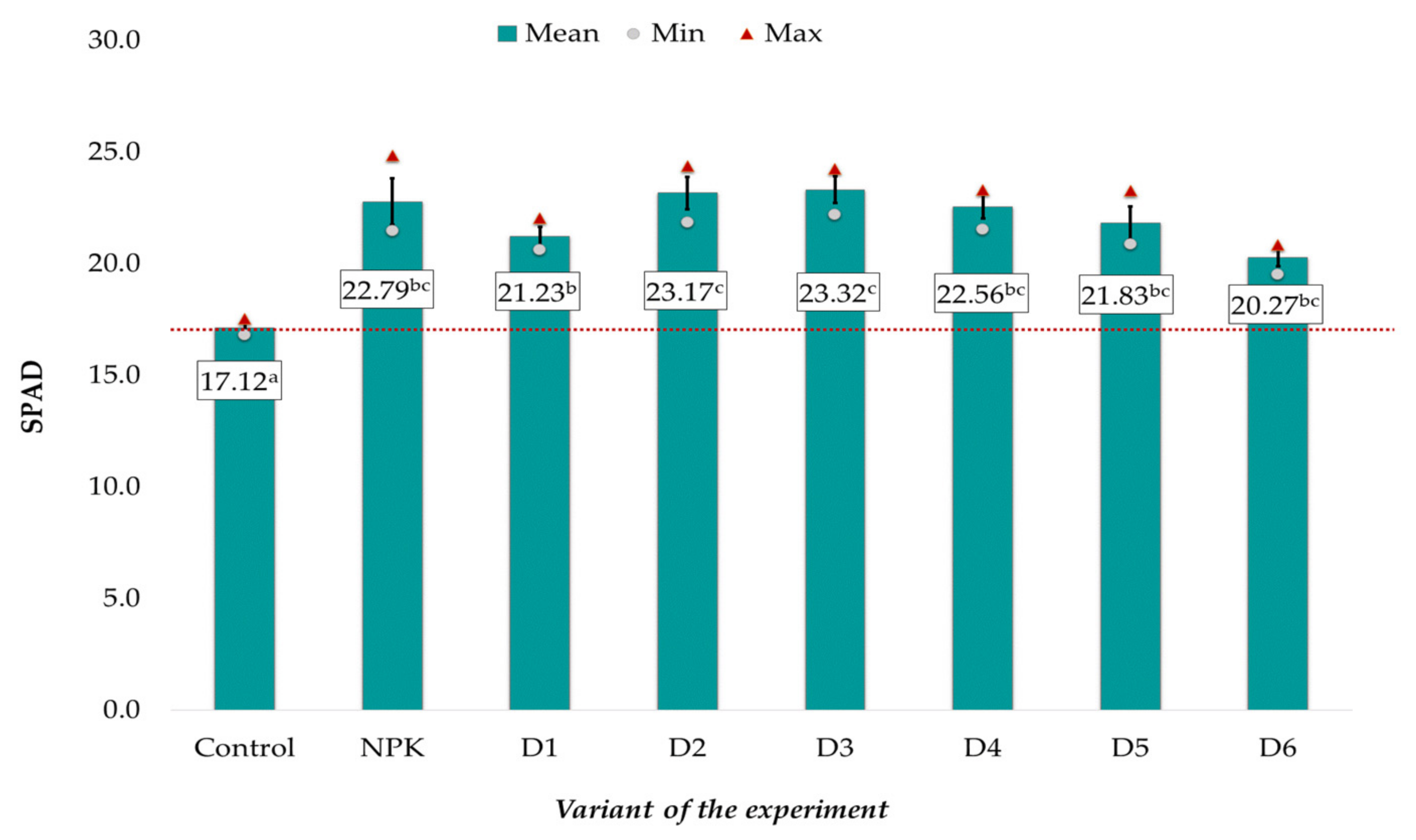
3.2. Effect of BAs on Plant Growth and Development and Grain Mineral Composition
3.3. Effect of BAs on Soil Properties
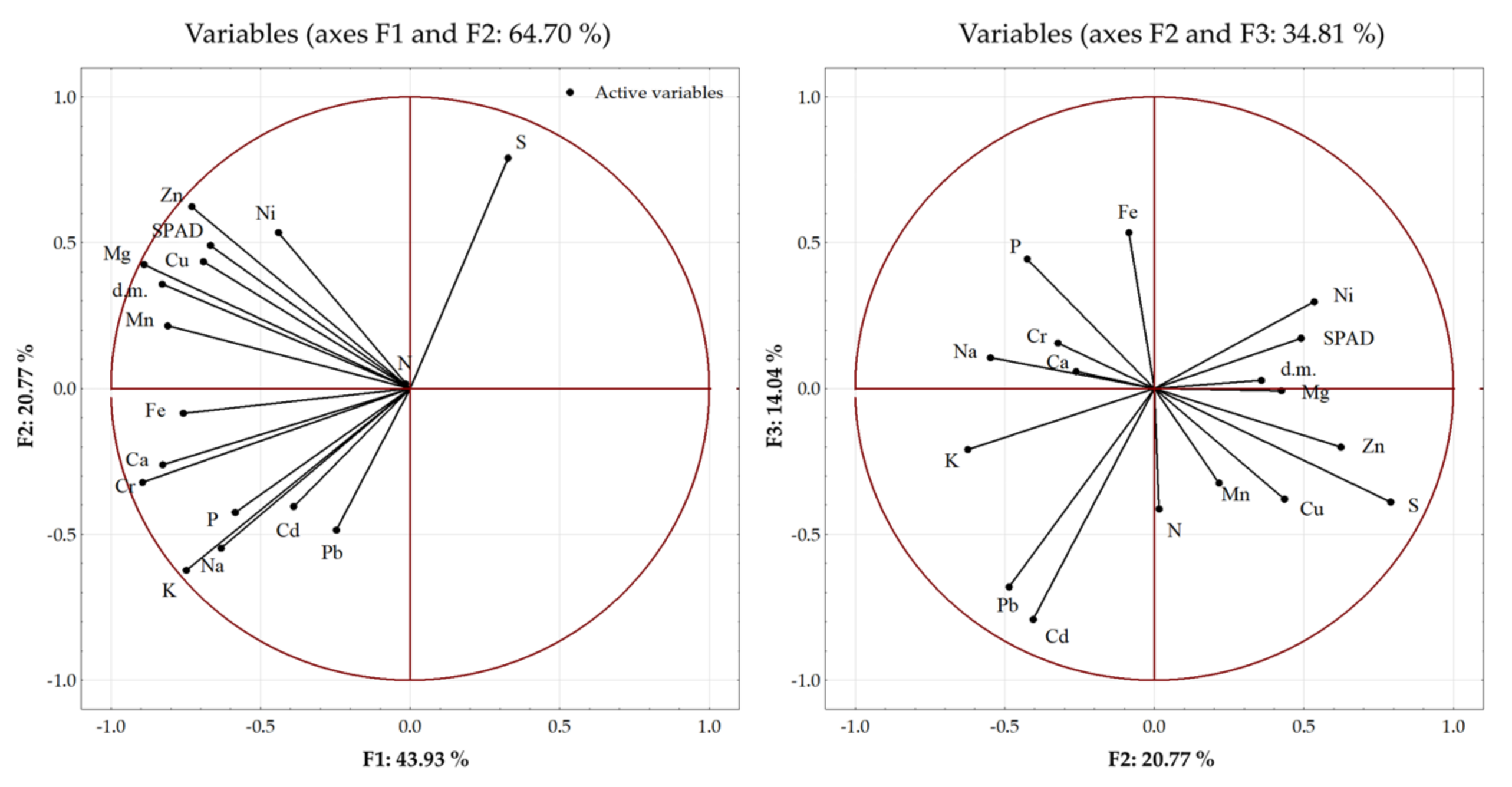
3.4. Relationship between Plants and Soil Parameters
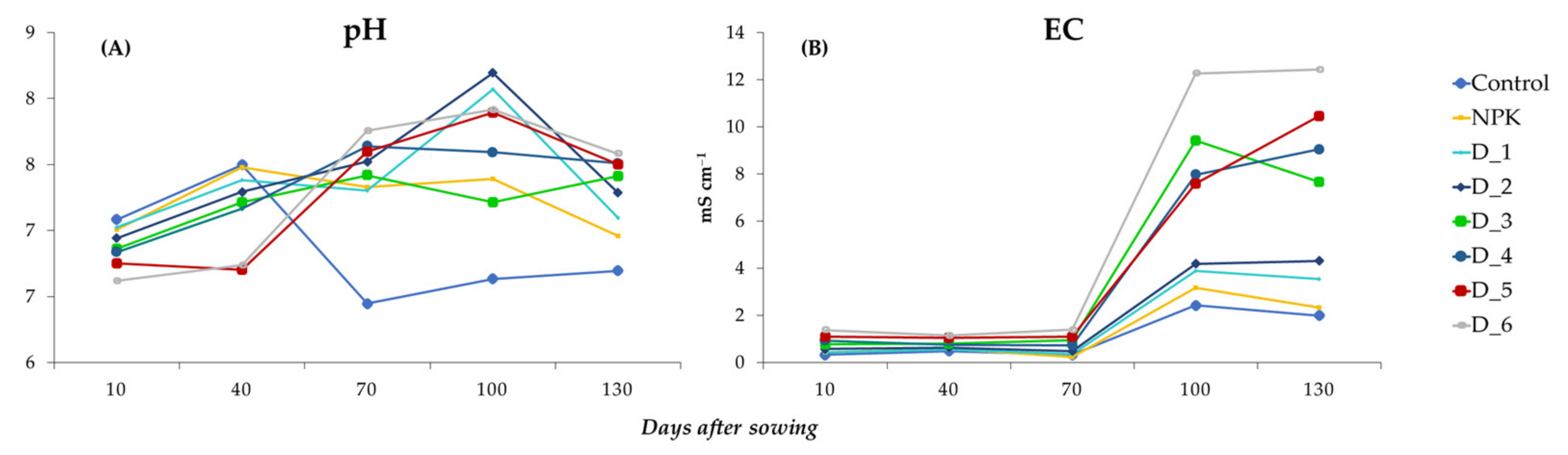
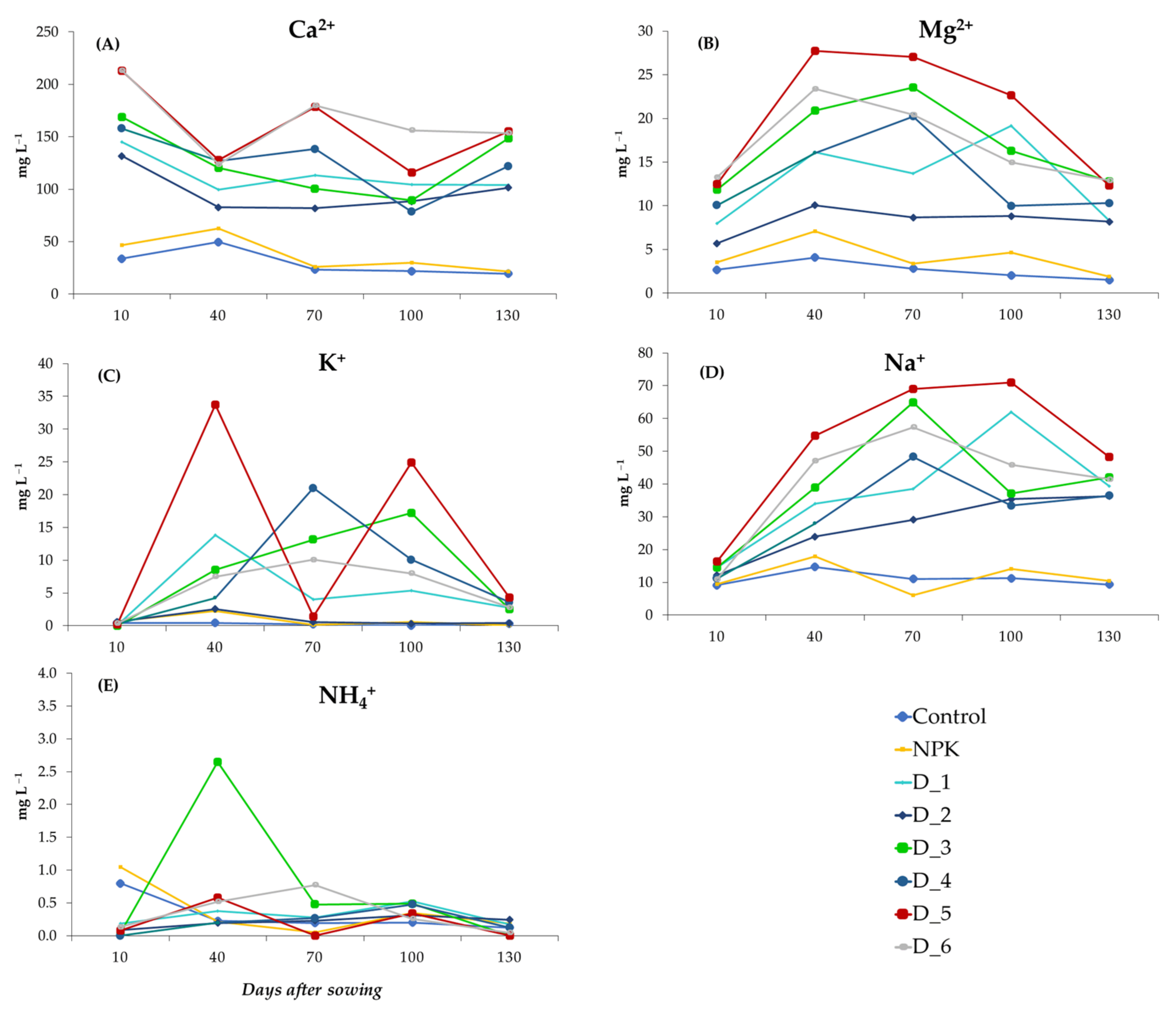
3.5. Effect of BAs on the Physicochemical Properties of Soil Solution Samples
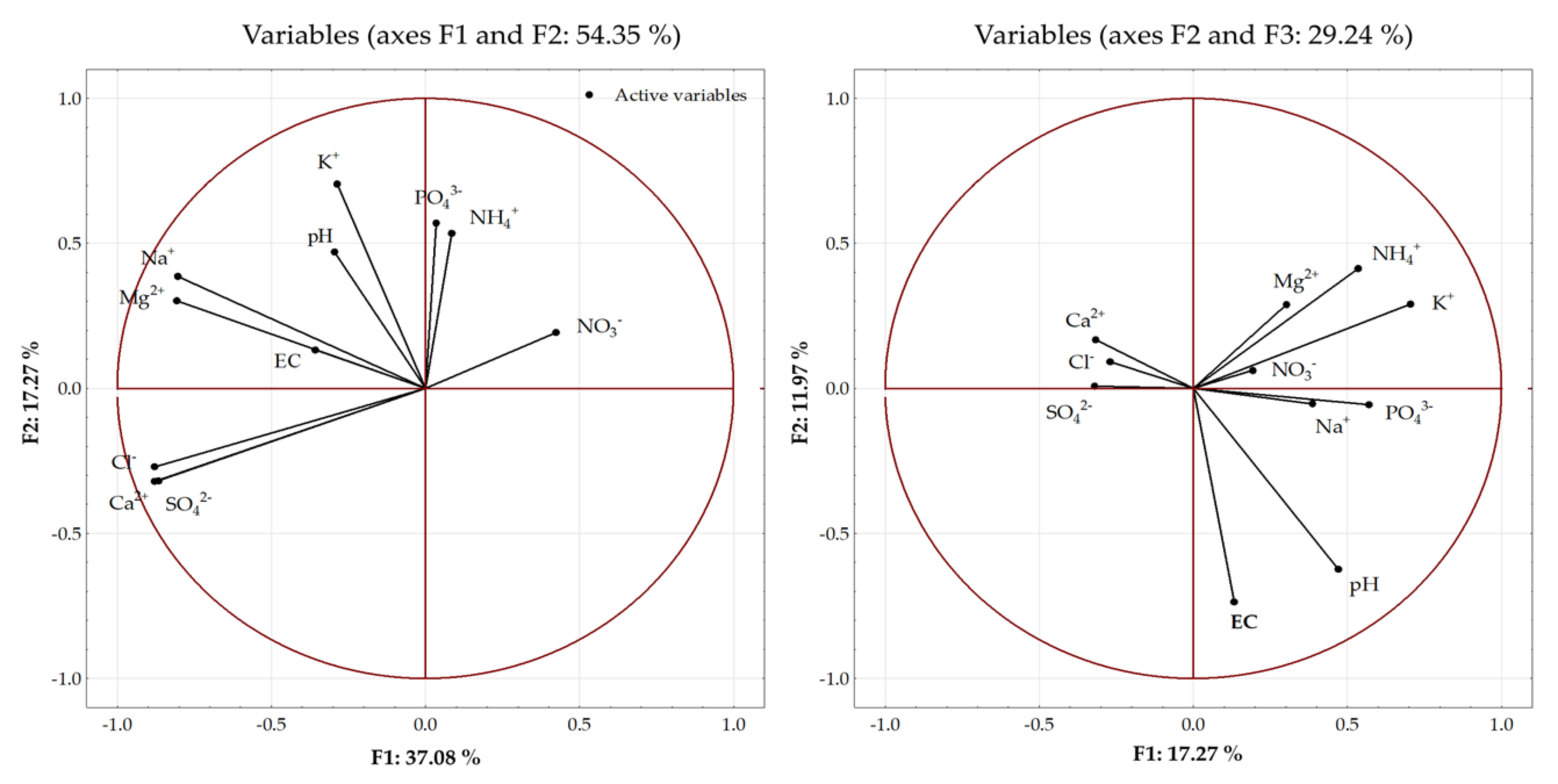
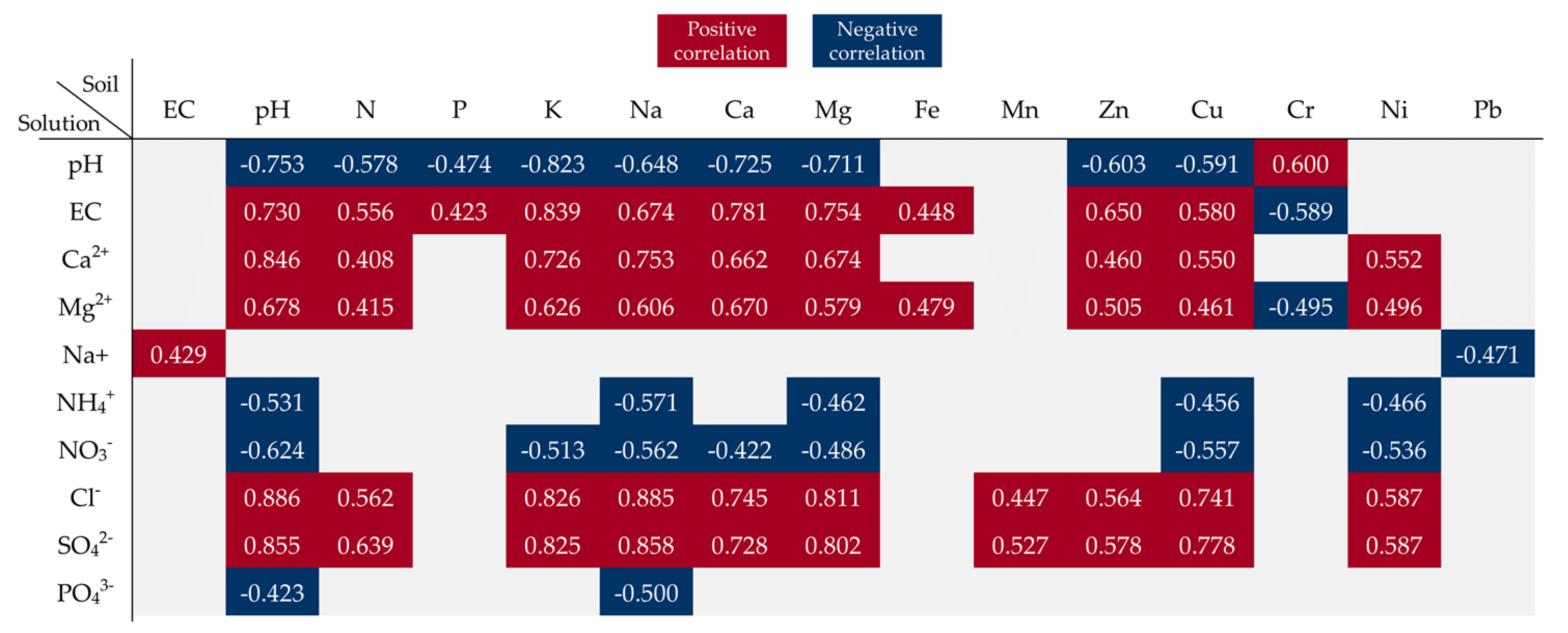
3.6. Relationship between Soil Parameters and Soil Solution Properties
4. Discussion
5. Conclusions
- The biomass ashes used in the experiment contained large amounts of macro- and micronutrients (except N), and starting from the dose of 1 Mg ha−1 (D1), the BAs application contributed to better plant growth and development compared to the control. Significant effectiveness of BAs in fertilization of spring rape plants, comparable to traditional NPK fertilization, was demonstrated. The utilization of nutrients contained in BAs is in line with the circular economy and fully justified.
- In addition to the supply of almost all essential nutrients to plants, short-term application of ashes positively changes the physicochemical and chemical properties of soils. Therefore, the application of this fertilizer in soil is consistent with the principles of sustainable development and may contribute to maintenance of the fertility and productivity of agricultural soils in many aspects.
- The application of BAs may increase the accumulation of alkaline substances in soil, especially those containing such elements as Ca, K, and Na, thereby causing excessive alkalization and salinization. There is also some risk that the BAs fertilization, especially in higher doses, may enhance the accumulation of trace elements in plants. These aspects should therefore be closely monitored, especially during a long-term use of these wastes, in order to avoid serious environmental problems.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhai, J.; Burke, I.T.; Stewart, D.I. Beneficial management of biomass combustion ashes. Renew. Sustain. Energy Rev. 2021, 151, 111555. [Google Scholar] [CrossRef]
- Thomas, B.; Yang, J.; Mo, K.; Abdalla, J.; Hawileh, R.; Ariyachandra, E. Biomass ashes from agricultural wastes as supplementary cementitious materials or aggregate replacement in cement/geopolymer concrete: A comprehensive review. J. Build. Eng. 2021, 40, 102332. [Google Scholar] [CrossRef]
- Mao, G.; Huang, N.; Chen, L.; Wang, H. Research on biomass energy and environment from the past to the future: A bibliometric analysis. Sci. Total Environ. 2021, 635, 1081–1090. [Google Scholar] [CrossRef] [PubMed]
- Waste Framework Directive (2008/98/EC) of the European Parliament and of the Council of 19 November 2008 on waste and repealing certain Directives. Available online: http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=CELEX:32008L0098:EN:NOT (accessed on 1 December 2022).
- Cruz, N.C.; Silva, F.C.; Tarelho, L.A.C.; Rodrigues, S.M. Critical review of key variables affecting potential recycling applications of ash produced at large-scale biomass combustion plants. Resour. Conserv. Recycl. 2019, 150, 104427. [Google Scholar] [CrossRef]
- Silva, F.C.; Cruz, N.C.; Tarelho, L.A.C.; Rodrigues, S.M. Use of biomass ash-based materials as soil fertilisers: Critical review of the existing regulatory framework. J. Clean. Prod. 2019, 214, 112–124. [Google Scholar] [CrossRef]
- Zając, G.; Szyszlak-Bargłowicz, J.; Gołębiowski, W.; Szczepanik, M. Chemical Characteristics of Biomass Ashes. Energies 2018, 11, 2885. [Google Scholar] [CrossRef]
- Vassilev, S.; Baxter, D.; Andersen, L.K.; Vassileva, C.G. An overview of the composition and application of biomass ash. Part 1. Phase–mineral and chemical composition and classification. Fuel 2013, 105, 40–76. [Google Scholar] [CrossRef]
- Trivedi, N.S.; Mandavgane, S.A.; Mehetre, S.; Kulkarni, B.D. Characterization and valorization of biomass ashes. Environ. Sci. Pollut. Res. 2016, 23, 20243–20256. [Google Scholar] [CrossRef]
- Kohli, S.J.; Goyal, D. Effect of fly ash application on some soil physical properties and microbial activities. Acta Agrophysica 2010, 16, 327–335. [Google Scholar]
- Huotari, N.; Tillman-Sutela, E.; Moilanen, M.; Laiho, R. Recycling of ash—For the good of the environment? For. Ecol. Manag. 2015, 348, 226–240. [Google Scholar] [CrossRef]
- Saletnik, B.; Zagula, G.; Bajcar, M.; Czernicka, M.; Puchalski, C. Biochar and Biomass Ash as a Soil Ameliorant: The Effect on Selected Soil Properties and Yield of Giant Miscanthus (Miscanthus × giganteus). Energies 2018, 11, 2535. [Google Scholar] [CrossRef]
- Shi, R.; Li, J.; Jiang, J.; Mehmood, K.; Liu, Y.; Xu, R.; Qian, W. Characteristics of biomass ashes from different materials and their ameliorative effects on acid soils. J. Environ. Sci. 2017, 55, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Schönegger, D.; Gómez-Brandón, M.; Mazzier, T.; Insam, H.; Hermanns, R.; Leijenhorst, E.; Juárez, M.F.D. Phosphorus fertilising potential of fly ash and effects on soil microbiota and crop. Resour. Conserv. Recycl. 2018, 134, 262–270. [Google Scholar] [CrossRef]
- Stankowski, S.; Sobolewska, M.; Jaroszewska, A.; Gibczyńska, M. Influence of biomass ash, lime and gypsum fertilization on macro-and microelement contents in the soil and grains of spring wheat. Soil Sci. Annu. 2018, 69, 177–183. [Google Scholar] [CrossRef]
- Wierzbowska, J.; Sienkiewicz, S.; Żarczyński, P.; Krzebietke, S. Environmental Application of Ash from Incinerated Biomass. Agronomy 2020, 10, 482. [Google Scholar] [CrossRef]
- Kramar, V. Possible utilization directions of ash from biomass combustion. Biomass ash as fertilizer in agriculture. UABIO Bioenergy Assoc. Ukr. 2020, 27, 33–36. [Google Scholar]
- WBR. World Reference Base for Soil Resources; WBR: New York, NY, USA, 2014; Volume 43, pp. 187–189. [Google Scholar]
- Mehlich, A. Mehlich 3 soil test extractant: A modification of Mehlich 2 Extractant. Commun. Soil Sci. Plant Anal. 1984, 15, 14091416. [Google Scholar] [CrossRef]
- Qu, Z.; Fatehi, H.; Schmidt, F.M. Potassium Release from Biomass Particles during Combustion Real-Time In Situ TDLASDetection and Numerical Simulation. Appl. Sci. 2021, 11, 8887. [Google Scholar] [CrossRef]
- Bremner, J. Determination of nitrogen in soil by the Kjeldahl method. J. Agric. Sci. 1960, 55, 11–33. [Google Scholar] [CrossRef]
- Page, A.L.; Miller, R.H.; Keeney, D.R. Methods of Soil Analysis; Agron No 9, Part 2: Chemical and Microbiological Properties, 2nd ed.; American Society of Agronomy: Madison, WI, USA, 1982. [Google Scholar]
- Allison, L.E.; Brown, J.W.; Hayward, H.E.; Richards, A.; Bernstein, L.; Fireman, M.; Pearson, G.A.; Wilcox, L.; Bower, C.A.; Hatcher, J.T.; et al. Diagnostic and Improvement of Saline and Alkali Soils; Richards, L.A., Ed.; United States Department of Agriculture, Agriculture Handbook: Washington, DC, USA, 1954. Available online: https://www.ars.usda.gov/ARSUserFiles/20360500/hb60_pdf/hb60complete.pdf (accessed on 1 December 2022).
- Romdhane, L.; Ebinezer, L.B.; Panozzo, A.; Barion, G.; Dal Cortivo, C.; Radhouane, L.; Vamerali, T. Effects of Soil Amendment with Wood Ash on Transpiration, Growth, and Metal Uptake in Two Contrasting Maize (Zea mays L.) Hybrids to Drought Tolerance. Front. Plant Sci. 2021, 12, 661909. [Google Scholar] [CrossRef]
- Szpunar-Krok, E.; Szostek, M.; Pawlak, R.; Gorzelany, J.; Migut, D. Effect of Fertilisation with Ash from Biomass Combustion on the Mechanical Properties of Potato Tubers (Solanum tuberosum L.) Grown in Two Types of Soil. Agronomy 2022, 12, 379. [Google Scholar] [CrossRef]
- Kurzemana, F.R.; Fernández-Delgado Juárez, M.; Probst, M.; Gómez-Brandón, M.; Partl, C.; Insam, H. Effect of biomass fly ashes from fast pyrolysis bio-oil production on soil properties and plant yield. J. Environ. Manag. 2021, 298, 113479. [Google Scholar] [CrossRef] [PubMed]
- Ohno, T. Neutralization of soil acidity and relase of phosphorus and potassium by wood ash. J. Environ. Qual. 1992, 21, 433–438. [Google Scholar] [CrossRef]
- Huotari, N.; Tillman-Sutela, E.; Kubin, E. Ground vegetation has a major role in element dynamics in an ash-fertilized cut-away peatland. For. Ecol. Manag. 2011, 261, 2081–2088. [Google Scholar] [CrossRef]
- Hansen, M.; Bang-Andreasen, T.; Sørensen, H.; Ingerslev, M. Micro vertical changes in soil pH and base cations over time after application of wood ash on forest soil. For. Ecol. Manag. 2017, 406, 274–280. [Google Scholar] [CrossRef]
- Demeyer, A.; Voundi Nkana, J.C.; Verloo, M.G. Characteristics of wood ash and influence on soil properties and nutrient uptake: An overview. Bioresour. Technol. 2001, 77, 287–295. [Google Scholar] [CrossRef]
- Nkana, J.C.; Demayer, A.; Verloo, M.G. Effect of wood ash application on soil solution chemistry of tropical acid soils: Incubation study. Bioresour. Technol. 2002, 85, 323–325. [Google Scholar] [CrossRef]
- Mercl, F.; Tejnecký, V.; Száková, J.; Tlustoš, P. Nutrient Dynamics in Soil Solution and Wheat Response after Biomass Ash Amendments. Agron. J. 2016, 108, 2222–2234. [Google Scholar] [CrossRef]
- Gómez-Rey, M.; Coutinho, M. Wood ash effects on nutrient dynamics and soil propertiesunder Mediterranean climate. Ann. For. Sci. 2012, 69, 569–579. [Google Scholar] [CrossRef]
- Bielińska, E.J.; Baran, S.; Stankowski, S. Assessment concerning usability of fluidal ashes from hard coal for agricultural purposes. Inżynieria Rol. 2009, 6, 7–15. (In Polish) [Google Scholar]
- Hansen, M.; Kepfer-Rojas, S.; Bjerager, E.R.; Holm, P.E.; Skov, S.; Ingerslev, M. Effects of ash application on nutrient and heavy metal fluxes in the soil and soil solution in a Norway spruce plantation in Denmark. For. Ecol. Manag. 2018, 424, 494–504. [Google Scholar] [CrossRef]
- Zhan, G.; Erich, M.S.; Ohno, T. Release of trace elements from wood ash by nitric acid. Water Air Soil Pollut. 1996, 88, 297–311. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Tsadilas, C.D.; Rinklebe, J. A review of the distribution coefficients of trace elements in soils: Influence of sorption system, element characteristics, and soil colloidal properties. Adv. Colloid Interface Sci. 2013, 201–202, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowska, G.; Hrynkiewicz, K.; Mierek-Adamska, A.; Goc, A. The sensitivity of spring and winter varieties of oilseed rape to heavy metals and rhizobacteria. Oilseed Crops 2012, 33, 201–220. (In Polish) [Google Scholar]
- Nabeela, F.; Murad, W.; Khan, I.; Mian, I.A.; Rehman, H.; Adnan, M.; Azizullah, A. Effect of wood ash application on the morphological, physiological and biochemical parameters of Brassica napus L. Plant Physiol. Biochem. 2015, 95, 15–25. [Google Scholar] [CrossRef]



| pH | SOC | Nt | Available Forms of Nutrients * | Total Concentrations of Elements | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| P | K | Mg | Zn | Cu | Ni | Cr | Pb | Cd | |||
| H2O | % | mg kg−1 | |||||||||
| 6.78 ± 0.12 | 1.23 ± 0.16 | 0.09 ± 0.0 | 7.92 ± 0.7 | 26.4 ± 2.2 | 92.1 ± 6.8 | 43.2 ± 8.6 | 5.67 ± 0.9 | 12.2 ± 1.04 | 31.2 ± 5.01 | 16.4 ± 1.2 | 0.34 ± 0.1 |
| Parameter | Units | Total Concentration | Soluble in H2O | Soluble in 1 M HCl | |
|---|---|---|---|---|---|
| pH | - | 12.83 ± 0.37 | - | - | - |
| EC | mS cm−1 | 8.81 ± 0.24 | - | - | - |
| C | % | - | 2.99 ± 0.21 | - | - |
| N | mg kg−1 | - | 10 ± 0.52 | - | - |
| S | - | 4700 ± 50 | - | - | |
| P | - | 9244 ± 65 | - | - | |
| K | - | 165,617 ± 123 | - | - | |
| Mg | - | 13,512 ± 78 | - | - | |
| Ca | - | 145,081 ± 137 | - | - | |
| Na | - | 1452 ± 43 | - | - | |
| Fe | - | 4351 ± 16 | 0.06 ± 0.01 | 1239 ± 54 | |
| Mn | - | 1490 ± 21 | 0.08 ± 0.01 | 635 ± 27 | |
| Zn | - | 423 ± 11 | 0.02 ± 0.00 | 395 ± 19 | |
| Cu | - | 536 ± 28 | 0.07 ± 0.01 | 157 ± 16 | |
| Ni | - | 20.3 ± 1.1 | 0.01 ± 0.00 | 15.9 ± 1.8 | |
| Cr | - | 48.4 ± 2.9 | 0.09 ± 0.02 | 32.8 ± 2.7 | |
| Pb | - | 130 ± 3.7 | 0.01 ± 0.00 | 33.8 ± 1.6 | |
| Cd | - | 2.68 ± 0.81 | 0.01 ± 0.00 | 2.49 ± 0.9 |
| Variant | N | P | K | Mg | Ca | Na | S |
|---|---|---|---|---|---|---|---|
| g kg−1 | |||||||
| Control | 30.0 a,b ± 0.4 | 4.39 b ± 0.12 | 8.65 a ± 0.54 | 1.20 a ± 0.16 | 4.21 a,b ± 0.30 | 0.13 a ± 0.00 | 1.77 a ± 0.06 |
| NPK | 31.2 b ± 0.4 | 6.33 c ± 0.07 | 8.15 a ± 0.38 | 2.19 b ± 0.25 | 4.35 b ± 0.38 | 0.18 d ± 0.01 | 2.16 b ± 0.06 |
| D1 | 31.2 b ± 0.4 | 3.78 a ± 0.36 | 8.22 a ± 0.17 | 2.65 c,d ± 0.37 | 4.13 a ± 0.33 | 0.12 a ± 0.00 | 2.32 b ± 0.05 |
| D2 | 30.3 a,b ± 0.6 | 3.93 a ± 0.34 | 10.02 b ± 0.54 | 2.79 d ± 0.20 | 4.89 c ± 0.19 | 0.15 b,c ± 0.00 | 2.15 b ± 0.04 |
| D3 | 29.0 a ± 0.5 | 6.10 c ± 0.20 | 10.36 c ± 0.36 | 3.00 e ± 0.58 | 5.09 d ± 0.22 | 0.16 c,d ± 0.00 | 1.82 a ± 0.08 |
| D4 | 30.6 a,b ± 0.4 | 7.72 e ± 0.35 | 11.12 d ± 0.54 | 3.01 e ± 0.35 | 4.84 c ± 0.22 | 0.22 e ± 0.00 | 1.55 a ± 0.08 |
| D5 | 31.2 b ± 0.2 | 7.08 d ± 0.26 | 12.14 e ± 0.43 | 2.57 b,c ± 0.33 | 4.77 c ± 0.34 | 0.22 e ± 0.00 | 1.73 a ± 0.06 |
| D6 | 31.5 b ± 0.4 | 6.16 c ± 0.27 | 12.09 e ± 0.49 | 2.62 b,c ± 0.17 | 4.80 c ± 0.18 | 0.21 e ± 0.00 | 1.75 a ± 0.04 |
| Variant | Fe | Mn | Zn | Cu | Ni | Cr | Pb | Cd |
|---|---|---|---|---|---|---|---|---|
| mg kg−1 | ||||||||
| Control | 22.6 a ± 0.3 | 6.1 a ± 0.1 | 13.7 a ± 0.4 | 1.84 a ± 0.06 | 0.51 a,b ± 0.01 | 0.20 a ± 0.00 | 0.25 b,c ± 0.02 | 0.06 b ± 0.00 |
| NPK | 27.8 a ± 0.2 | 7.9 a ± 0.1 | 27.9 b,c ± 0.4 | 2.51 b ± 0.08 | 0.50 a,b ± 0.01 | 0.21 a ± 0.01 | 0.12 a ± 0.01 | 0.04 a ± 0.00 |
| D1 | 23.3 a ± 0.2 | 27.6 c ± 0.1 | 30.7 e ± 0.3 | 2.97 c ± 0.07 | 1.08 a ± 0.03 | 0.20 a ± 0.01 | 0.22 b ± 0.01 | 0.07 b ± 0.00 |
| D2 | 46.5 c ± 0.4 | 27.9 c ± 0.2 | 30.1 d,e ± 0.1 | 2.69 b ± 0.02 | 0.88 c ± 0.06 | 2.55 b ± 0.04 | 0.30 c,d ± 0.01 | 0.09 c ± 0.00 |
| D3 | 48.5 c ± 0.5 | 27.9 c ± 0.4 | 28.9 c,d ± 0.3 | 2.46 b ± 0.05 | 1.05 d ± 0.04 | 2.87 d ± 0.04 | 0.33 d,e ± 0.01 | 0.07 b ± 0.00 |
| D4 | 82.1 d ± 1.7 | 28.3 c ± 0.2 | 28.3 c ± 0.2 | 2.78 b,c ± 0.03 | 1.21 e ± 0.01 | 3.87 e ± 0.08 | 0.13 a ± 0.01 | 0.06 b ± 0.00 |
| D5 | 39.8 b,c ± 1.0 | 25.4 b ± 0.4 | 27.0 b ± 0.2 | 2.73 b,c ± 0.02 | 0.43 a ± 0.03 | 2.75 c,d ± 0.04 | 0.39 f ± 0.01 | 0.10 c ± 0.00 |
| D6 | 35.4 b ± 1.9 | 24.8 b ± 0.9 | 26.9 b ± 0.1 | 2.79 b,c ± 0.03 | 0.61 b ± 0.03 | 2.56 b,c ± 0.05 | 0.36 e,f ± 0.01 | 0.11 c ± 0.00 |
| Variant | pH | EC | Total Macronutrients (mg kg−1) | |||||
|---|---|---|---|---|---|---|---|---|
| [H2O] | [mS cm−1] | N | P | K | Mg | Ca | Na | |
| Control | 6.48 a ± 0.06 | 0.10 a ± 0.00 | 1927 a ± 22 | 979 a ± 27 | 1196 b ± 19 | 1294 a,b ± 4 | 1558 a,b ± 21 | 138 a ± 2 |
| NPK | 6.59 a ± 0.04 | 0.13 a ± 0.00 | 2058 b,c ± 20 | 1891 c ± 17 | 1110 a ± 21 | 1247 a ± 17 | 1481 a ± 21 | 136 a ± 4 |
| D1 | 6.92 b ± 0.08 | 0.49 e ± 0.01 | 1991 a,b ± 11 | 1845 c ± 19 | 1237 b ± 22 | 1350 b,c ± 20 | 1639 c,d ± 13 | 157 b,c ± 3 |
| D2 | 7.09 d ± 0.02 | 0.16 b,c ± 0.00 | 2122 c ± 18 | 1725 d ± 23 | 1321 c ± 14 | 1364 b,c ± 14 | 1521 a,b ± 15 | 164 c ± 4 |
| D3 | 6.86 c ± 0.07 | 0.17 c,d ± 0.00 | 2081 c ± 17 | 1398 b ± 19 | 1195 b ± 11 | 1294 a,b ± 12 | 1581 b,c ± 23 | 147 a,b ± 3 |
| D4 | 6.83 c ± 0.06 | 0.16 b,c ± 0.00 | 2302 d ± 13 | 1481 b ± 19 | 1337 c ± 15 | 1448 d,e ± 20 | 1842 e ± 22 | 166 c ± 3 |
| D5 | 7.09 d ± 0.02 | 0.19 d ± 0.00 | 1988 a,b ± 14 | 1489 b ± 10 | 1385 c ± 13 | 1391 c,d ± 15 | 1711 d ± 14 | 164 c ± 4 |
| D6 | 7.29 e ± 0.04 | 0.13 a ± 0.00 | 2230 d ± 12 | 2326 e ± 33 | 1580 d ± 18 | 1491 e ± 14 | 1865 e ± 28 | 171 c ± 4 |
| Variants | Fe | Mn | Zn | Cu | Ni | Cr | Pb | Cd |
|---|---|---|---|---|---|---|---|---|
| mg kg−1 | ||||||||
| Control | 2421 b ± 31 | 127 a ± 1 | 41.1 a,b ± 0.7 | 4.71 a ± 0.16 | 14.6 a ± 0.3 | 29.3 b,c ± 0.5 | 15.6 c ± 0.1 | 0.25 a ± 0.01 |
| NPK | 2436 b ± 24 | 129 a ± 2 | 40.7 a ± 0.3 | 4.54 a ± 0.25 | 14.9 a ± 0.1 | 28.7 b,c ± 0.3 | 15.3 b,c ± 0.1 | 0.25 a ± 0.01 |
| D1 | 2870 d ± 19 | 163 c ± 2 | 42.0 a,b ± 0.4 | 4.88 a ± 0.37 | 17.8 c ± 0.1 | 30.7 c,d ± 0.4 | 14.9 a,b ± 0.2 | 0.32 b,c ± 0.01 |
| D2 | 1983 a ± 9 | 157 b,c ± 6 | 40.4 a ± 0.4 | 6.14 c ± 0.20 | 16.3 b ± 0.2 | 31.9 d ± 0.3 | 15.2 b,c ± 0.1 | 0.22 a ± 0.01 |
| D3 | 2701 c ± 11 | 120 a ± 1 | 43.1 b ± 0.3 | 5.43 b ± 0.58 | 16.4 b ± 0.6 | 27.4 a,b ± 0.6 | 15.5 b,c ± 0.2 | 0.25 a ± 0.01 |
| D4 | 3481 e ± 15 | 204 d ± 4 | 49.5 c ± 0.5 | 5.96 c ± 0.35 | 19.0 d ± 0.2 | 30.4 c,d ± 0.5 | 16.7 d ± 0.1 | 0.34 c ± 0.01 |
| D5 | 2630 c ± 26 | 164 c ± 3 | 41.4 a,b ± 0.5 | 4.99 a,b ± 0.33 | 16.3 b ± 0.3 | 26.3 a ± 0.4 | 14.3 a ± 0.2 | 0.24 a ± 0.01 |
| D6 | 2916 d ± 28 | 144 b ± 3 | 48.2 c ± 0.3 | 6.27 c ± 0.17 | 16.5 b ± 0.4 | 26.6 a ± 0.3 | 15.7 c ± 0.1 | 0.30 b ± 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szostek, M.; Szpunar-Krok, E.; Jańczak-Pieniążek, M.; Ilek, A. Short-Term Effect of Fly Ash from Biomass Combustion on Spring Rape Plants Growth, Nutrient, and Trace Elements Accumulation, and Soil Properties. Int. J. Environ. Res. Public Health 2023, 20, 455. https://doi.org/10.3390/ijerph20010455
Szostek M, Szpunar-Krok E, Jańczak-Pieniążek M, Ilek A. Short-Term Effect of Fly Ash from Biomass Combustion on Spring Rape Plants Growth, Nutrient, and Trace Elements Accumulation, and Soil Properties. International Journal of Environmental Research and Public Health. 2023; 20(1):455. https://doi.org/10.3390/ijerph20010455
Chicago/Turabian StyleSzostek, Małgorzata, Ewa Szpunar-Krok, Marta Jańczak-Pieniążek, and Anna Ilek. 2023. "Short-Term Effect of Fly Ash from Biomass Combustion on Spring Rape Plants Growth, Nutrient, and Trace Elements Accumulation, and Soil Properties" International Journal of Environmental Research and Public Health 20, no. 1: 455. https://doi.org/10.3390/ijerph20010455
APA StyleSzostek, M., Szpunar-Krok, E., Jańczak-Pieniążek, M., & Ilek, A. (2023). Short-Term Effect of Fly Ash from Biomass Combustion on Spring Rape Plants Growth, Nutrient, and Trace Elements Accumulation, and Soil Properties. International Journal of Environmental Research and Public Health, 20(1), 455. https://doi.org/10.3390/ijerph20010455








