Self-Selected Walking Cadence after 16-Week Light-Intensity Physical Activity Intervention for Older Cancer Survivors
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Participants
2.2. Instrumentation
2.3. Intervention
2.4. Other Data Collection
2.5. Outcome Measures
2.6. Statistical Analyses
3. Results
3.1. Quantity of Ambulatory Behavior
3.2. Quality of Ambulatory Behavior
3.2.1. Purposeful Stepping and Medium-Intensity Walking
3.2.2. Distribution of Steps across Cadence Bands
3.2.3. Peak Cadence
3.2.4. Average Cadence
3.3. Post Hoc Analyses
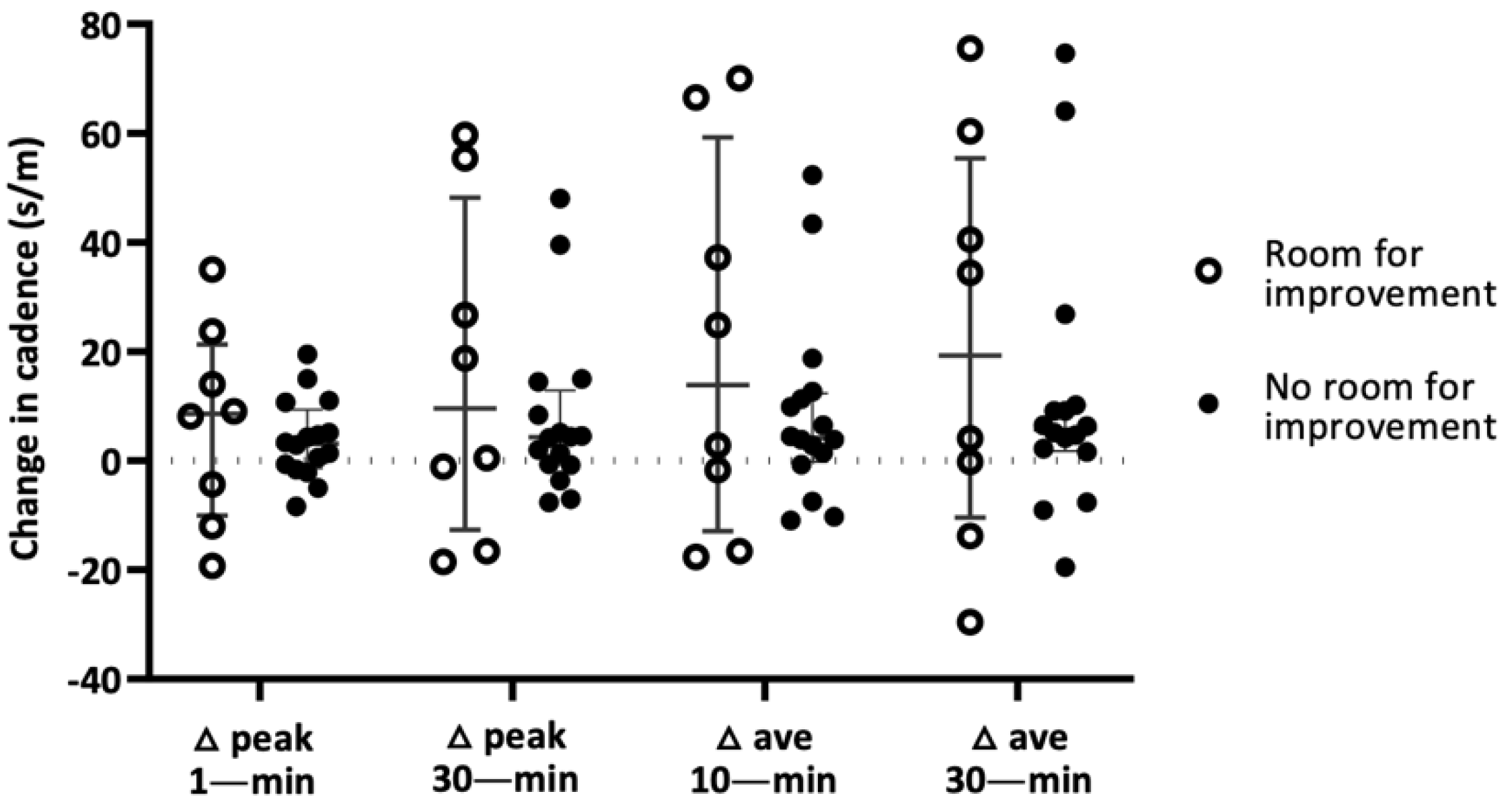
3.3.1. Room for Improvement—Potential Ceiling Effect of Baseline Physical Performance
3.3.2. Influence of Baseline and Change in Average Daily Steps on Change in Peak and Average Cadence
4. Discussion
4.1. Influence of Baseline Physical Function on Cadence Outcomes
4.2. Influence of Baseline and Change in Physical Activity on Cadence Outcomes
4.3. Limitations
4.4. Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Trial Registration
References
- Bluethmann, S.M.; Mariotto, A.B.; Rowland, J.H. Anticipating the “Silver Tsunami”: Prevalence Trajectories and Comorbidity Burden among Older Cancer Survivors in the United States. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1029–1036. [Google Scholar] [CrossRef]
- Weaver, K.E.; Leach, C.R.; Leng, X.; Danhauer, S.C.; Klepin, H.D.; Vaughan, L.; Naughton, M.; Chlebowski, R.T.; Vitolins, M.Z.; Paskett, E. Physical Functioning among Women 80 Years of Age and Older With and Without a Cancer History. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2016, 71 (Suppl. S1), S23–S30. [Google Scholar] [CrossRef]
- Deimling, G.T.; Arendt, J.A.; Kypriotakis, G.; Bowman, K.F. Functioning of older, long-term cancer survivors: The role of cancer and comorbidities. J. Am. Geriatr. Soc. 2009, 57 (Suppl. S2), S289–S292. [Google Scholar] [CrossRef]
- Sogaard, M.; Thomsen, R.W.; Bossen, K.S.; Sorensen, H.T.; Norgaard, M. The impact of comorbidity on cancer survival: A review. Clin. Epidemiol. 2013, 5 (Suppl. S1), 3–29. [Google Scholar] [CrossRef]
- den Ouden, M.E.; Schuurmans, M.J.; Arts, I.E.; van der Schouw, Y.T. Physical performance characteristics related to disability in older persons: A systematic review. Maturitas 2011, 69, 208–219. [Google Scholar] [CrossRef]
- Onder, G.; Penninx, B.W.; Ferrucci, L.; Fried, L.P.; Guralnik, J.M.; Pahor, M. Measures of physical performance and risk for progressive and catastrophic disability: Results from the Women’s Health and Aging Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2005, 60, 74–79. [Google Scholar] [CrossRef]
- Sweeney, C.; Schmitz, K.H.; Lazovich, D.; Virnig, B.A.; Wallace, R.B.; Folsom, A.R. Functional limitations in elderly female cancer survivors. J. Natl. Cancer Inst. 2006, 98, 521–529. [Google Scholar] [CrossRef]
- Dunlop, D.D.; Song, J.; Arnston, E.K.; Semanik, P.A.; Lee, J.; Chang, R.W.; Hootman, J.M. Sedentary time in US older adults associated with disability in activities of daily living independent of physical activity. J. Phys. Act. Health 2015, 12, 93–101. [Google Scholar] [CrossRef]
- Fielding, R.A.; Guralnik, J.M.; King, A.C.; Pahor, M.; McDermott, M.M.; Tudor-Locke, C.; Manini, T.M.; Glynn, N.; Marsh, A.P.; Axtell, R.S.; et al. Dose of physical activity, physical functioning and disability risk in mobility-limited older adults: Results from the LIFE study randomized trial. PLoS ONE 2017, 12, e0182155. [Google Scholar] [CrossRef]
- Rejeski, W.J.; Fielding, R.A.; Blair, S.N.; Guralnik, J.M.; Gill, T.M.; Hadley, E.C.; King, A.C.; Kritchevsky, S.B.; Miller, M.E.; Newman, A.B.; et al. The lifestyle interventions and independence for elders (LIFE) pilot study: Design and methods. Contemp. Clin. Trials. 2005, 26, 141–154. [Google Scholar] [CrossRef]
- Daum, C.W.; Cochrane, S.K.; Fitzgerald, J.D.; Johnson, L.; Buford, T.W. Exercise Interventions for Preserving Physical Function Among Cancer Survivors in Middle to Late Life. J. Frailty Aging 2016, 5, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Tudor-Locke, C.; Swift, D.L.; Schuna, J.M., Jr.; Dragg, A.T.; Davis, A.B.; Martin, C.K.; Johnson, W.D.; Church, T.S. WalkMore: A randomized controlled trial of pedometer-based interventions differing on intensity messages. BMC Public Health 2014, 14, 168. [Google Scholar] [CrossRef] [PubMed]
- Committee PAGA. Physical Activity Guidelines Advisory Committee Report, 2008; 2008:A1-H14; US Department of Health and Human Services: Washington, DC, USA, 2008.
- Beauchamp, M.R.; Harden, S.M.; Wolf, S.A.; Rhodes, R.E.; Liu, Y.; Dunlop, W.L.; Schmader, T.; Sheel, A.W.; Zumbo, B.D.; Estabrooks, P.A. GrOup based physical Activity for oLder adults (GOAL) randomized controlled trial: Study protocol. BMC Public Health 2015, 15, 592. [Google Scholar] [CrossRef]
- Kruger, J.; Buchner, D.M.; Prohaska, T.R. The prescribed amount of physical activity in randomized clinical trials in older adults. Gerontologist 2009, 49 (Suppl. S1), S100–S107. [Google Scholar] [CrossRef]
- Rejeski, W.J.; Axtell, R.; Fielding, R.; Katula, J.; King, A.C.; Manini, T.M.; Miller, M.E. Promoting physical activity for elders with compromised function: The lifestyle interventions and independence for elders (LIFE) study physical activity intervention. Clin. Interv. Aging 2013, 8, 1119–1131. [Google Scholar] [CrossRef]
- Santanasto, A.; Glynn, N.W.; Lovato, L.C.; Blair, S.N.; Fielding, R.A.; Gill, T.M.; Guralnik, J.M.; Hsu, F.; King, A.C.; Strotmeyer, E.S.; et al. Effect of Physical Activity versus Health Education on Physical Function, Grip Strength and Mobility. J. Am. Geriatr. Soc. 2017, 65, 1427–1433. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Schuna, J.M., Jr.; Han, H.O.; Aguiar, E.J.; Green, M.A.; Busa, M.A.; Johnson, W.D. Step-Based Physical Activity Metrics and Cardiometabolic Risk: NHANES 2005–2006. Med. Sci. Sports Exerc. 2017, 49, 283–291. [Google Scholar] [CrossRef]
- Dohrn, I.M.; Hagstromer, M.; Hellenius, M.L.; Stahle, A. Gait Speed, Quality of Life, and Sedentary Time are Associated with Steps per Day in Community-Dwelling Older Adults with Osteoporosis. J. Aging Phys. Act. 2016, 24, 22–31. [Google Scholar] [CrossRef]
- Withall, J.; Stathi, A.; Davis, M.; Coulson, J.; Thompson, J.L.; Fox, K.R. Objective indicators of physical activity and sedentary time and associations with subjective well-being in adults aged 70 and over. Int. J. Environ. Res. Public Health 2014, 11, 643–656. [Google Scholar] [CrossRef]
- Rowlands, A.V.; Schuna, J.M., Jr.; Stiles, V.H.; Tudor-Locke, C. Cadence, peak vertical acceleration, and peak loading rate during ambulatory activities: Implications for activity prescription for bone health. J. Phys. Act. Health 2014, 11, 1291–1294. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Brashear, M.M.; Katzmarzyk, P.T.; Johnson, W.D. Peak stepping cadence in free-living adults: 2005–2006 NHANES. J. Phys. Act. Health 2012, 9, 1125–1129. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Craig, C.L.; Brown, W.J.; Clemes, S.A.; De Cocker, K.; Giles-Corti, B.; Hatano, Y.; Inoue, S.; Matsudo, S.M.; Mutrie, N.; et al. How many steps/day are enough? For adults. Int. J. Behav. Nutr. Phys. Act. 2011, 8, 79. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Craig, C.L.; Thyfault, J.P.; Spence, J.C. A step-defined sedentary lifestyle index: <5000 steps/day. Appl. Physiol. Nutr. Metab. 2013, 38, 100–114. [Google Scholar] [CrossRef]
- Ayabe, M.; Yahiro, T.; Yoshioka, M.; Higuchi, H.; Higaki, Y.; Tanaka, H. Objectively measured age-related changes in the intensity distribution of daily physical activity in adults. J. Phys. Act. Health 2009, 6, 419–425. [Google Scholar] [CrossRef]
- Cavanaugh, J.T.; Coleman, K.L.; Gaines, J.M.; Laing, L.; Morey, M.C. Using step activity monitoring to characterize ambulatory activity in community-dwelling older adults. J. Am. Geriatr. Soc. 2007, 55, 120–124. [Google Scholar] [CrossRef]
- Barreira, T.V.; Harrington, D.M.; Schuna, J.M., Jr.; Tudor-Locke, C.; Katzmarzyk, P.T. Pattern changes in step count accumulation and peak cadence due to a physical activity intervention. J. Sci. Med. Sport. 2016, 19, 227–231. [Google Scholar] [CrossRef]
- Tucker, J.M.; Welk, G.J.; Beyler, N.K. Physical activity in U.S.: Adults compliance with the Physical Activity Guidelines for Americans. Am. J. Prev. Med. 2011, 40, 454–461. [Google Scholar] [CrossRef]
- Talbot, L.A.; Gaines, J.M.; Huynh, T.N.; Metter, E.J. A home-based pedometer-driven walking program to increase physical activity in older adults with osteoarthritis of the knee: A preliminary study. J. Am. Geriatr. Soc. 2003, 51, 387–392. [Google Scholar] [CrossRef]
- Maruya, K.; Asakawa, Y.; Ishibashi, H.; Fujita, H.; Arai, T.; Yamaguchi, H. Effect of a simple and adherent home exercise program on the physical function of community dwelling adults sixty years of age and older with pre-sarcopenia or sarcopenia. J. Phys. Ther. Sci. 2016, 28, 3183–3188. [Google Scholar] [CrossRef]
- Pahor, M.; Guralnik, J.M.; Ambrosius, W.T.; Blair, S.; Bonds, D.E.; Church, T.S.; LIFE Study Investigators. Effect of structured physical activity on prevention of major mobility disability in older adults: The LIFE study randomized clinical trial. JAMA 2014, 311, 2387–2396. [Google Scholar] [CrossRef]
- Blair, C.K.; Harding, E.; Wiggins, C.; Kang, H.; Schwartz, M.; Tarnower, A.; Kinney, A.Y. A Home-Based Mobile Health Intervention to Replace Sedentary Time With Light Physical Activity in Older Cancer Survivors: Randomized Controlled Pilot Trial. JMIR Cancer 2021, 7, e18819. [Google Scholar] [CrossRef] [PubMed]
- Grant, P.M.; Ryan, C.G.; Tigbe, W.W.; Granat, M.H. The validation of a novel activity monitor in the measurement of posture and motion during everyday activities. Br. J. Sports Med. 2006, 40, 992–997. [Google Scholar] [CrossRef] [PubMed]
- Chastin, S.F.M.; Dontje, M.L.; Skelton, D.A.; Čukić, I.; Shaw, R.J.; Gill, J.M.R.; Greig, C.A.; Gale, C.R.; Deary, I.J.; Der Deary, G.; et al. Systematic comparative validation of self-report measures of sedentary time against an objective measure of postural sitting (activPAL). Int. J. Behav. Nutr. Phys. Act. 2018, 15, 21. [Google Scholar] [CrossRef] [PubMed]
- Tudor-Locke, C.; Camhi, S.M.; Leonardi, C.; Johnson, W.D.; Katzmarzyk, P.T.; Earnest, C.P.; Church, T.S. Patterns of adult stepping cadence in the 2005–2006 NHANES. Prev. Med. 2011, 53, 178–181. [Google Scholar] [CrossRef]
- Gardner, A.W.; Montgomery, P.S.; Scott, K.J.; Afaq, A.; Blevins, S.M. Patterns of ambulatory activity in subjects with and without intermittent claudication. J. Vasc. Surg. 2007, 46, 1208–1214. [Google Scholar] [CrossRef]
- Sangha, O.; Stucki, G.; Liang, M.H.; Fossel, A.H.; Katz, J.N. The Self-Administered Comorbidity Questionnaire: A new method to assess comorbidity for clinical and health services research. Arthritis Rheum. 2003, 49, 156–163. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Ferrucci, L.; Pieper, C.F.; Leveille, S.G.; Markides, K.S.; Ostir, G.V.; Studenski, S.; Berkman, L.F.; Wallace, R.B. Lower extremity function and subsequent disability: Consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2000, 55, M221–M231. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Ferrucci, L.; Simonsick, E.M.; Salive, M.E.; Wallace, R.B. Lower-extremity function in persons over the age of 70 years as a predictor of subsequent disability. N. Engl. J. Med. 1995, 332, 556–561. [Google Scholar] [CrossRef]
- Pavasini, R.; Guralnik, J.; Brown, J.C.; Di Bari, M.; Cesari, M.; Landi, F.; Vaes, B.; Legrand, D.; Verghese, J.; Wang, C.; et al. Short Physical Performance Battery and all-cause mortality: Systematic review and meta-analysis. BMC Med. 2016, 14, 215. [Google Scholar] [CrossRef]
- Vasunilashorn, S.; Coppin, A.K.; Patel, K.V.; Lauretani, F.; Ferrucci, L.; Bandinelli, S.; Guralnik, J.M. Use of the Short Physical Performance Battery Score to predict loss of ability to walk 400 meters: Analysis from the InCHIANTI study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2009, 64, 223–229. [Google Scholar] [CrossRef]
- Tudor-Locke, C.; Rowe, D.A. Using cadence to study free-living ambulatory behaviour. Sports Med. 2012, 42, 381–398. [Google Scholar] [CrossRef] [PubMed]
- Tudor-Locke, C.; Han, H.; Aguiar, E.J.; Barreira, T.V.; Schuna, J.M., Jr.; Kang, M.; Rowe, D.A. How fast is fast enough? Walking cadence (steps/min) as a practical estimate of intensity in adults: A narrative review. Br. J. Sports Med. 2018, 52, 776–788. [Google Scholar] [CrossRef] [PubMed]
- Schuna, J.M., Jr.; Brouillette, R.M.; Foil, H.C.; Fontenot, S.L.; Keller, J.N.; Tudor-Locke, C. Steps per day, peak cadence, body mass index, and age in community-dwelling older adults. Med. Sci. Sports Exerc. 2013, 45, 914–919. [Google Scholar] [CrossRef] [PubMed]
- Slaght, J.; Senechal, M.; Hrubeniuk, T.J.; Mayo, A.; Bouchard, D.R. Walking Cadence to Exercise at Moderate Intensity for Adults: A Systematic Review. J. Sports Med. 2017, 2017, 4641203. [Google Scholar] [CrossRef] [PubMed]
- Gardner, A.W.; Parker, D.E.; Montgomery, P.S.; Scott, K.J.; Blevins, S.M. Efficacy of Quantified Home-Based Exercise and Supervised Exercise in Patients with Intermittent Claudication: A Randomized Controlled Trial. Circulation 2011, 123, 491–498. [Google Scholar] [CrossRef]
- Glazer, N.L.; Lyass, A.; Esliger, D.W.; Blease, S.J.; Freedson, P.S.; Massaro, J.M.; Murabito, J.M.; Vasan, R.S. Sustained and shorter bouts of physical activity are related to cardiovascular health. Med. Sci. Sports Exerc. 2013, 45, 109–115. [Google Scholar] [CrossRef]

| Intervention | Control | |
|---|---|---|
| Characteristics | n = 24 | n = 17 |
| Female; n(%) | 14 (58.3) | 9 (52.9) |
| Age (years): mean ± SD | 69.6 (3.4) | 70.8 (5.4) |
| Height (cm): mean ± SD | 164.9 (11.4) | 168.3 (11.0) |
| Weight (kg): mean ± SD | 79.0 (16.2) | 83.6 (16.8) |
| BMI (kg/m2): mean ± SD | 29.0 (4.7) | 29.5 (5.3) |
| Cancer type | ||
| Breast; n(%) | 10 (41.7) | 4 (23.5) |
| Prostate; n(%) | 6 (25.0) | 6 (35.3) |
| Other; n(%) | 8 (33.3) | 7 (41.2) |
| Age at diagnosis (years): mean ± SD | 65.2 (3.7) | 66.2 (5.1) |
| Health status (self-report) | ||
| Very good/excellent; n(%) | 13 (54.2) | 9 (52.9) |
| Good; n(%) | 9 (37.5) | 7 (41.2) |
| Fair/poor; n(%) | 2 (8.3) | 1 (5.9) |
| Chronic conditions (comorbidities) ≥ 3 | 8 (33.3) | 7 (41.2) |
| Race/ethnicity | ||
| White (Non-Hispanic); n(%) | 19 (79.2) | 13 (76.5) |
| Hispanic; n(%) | 5 (20.8) | 4 (23.5) |
| Marital Status | ||
| Married or living in marriage-like relationship; n(%) | 17 (70.8) | 14 (82.4) |
| Not Married (single, divorced, widowed); n(%) | 7 (29.2) | 3 (17.7) |
| Education | ||
| Less than high school or high school graduate; n(%) | 3 (12.5) | 3 (17.6) |
| Post high-school training or some college; n(%) | 6 (25.0) | 5 (29.4) |
| College degree or higher; n(%) | 15 (62.5) | 9 (53.0) |
| Annual household income | ||
| ≥50,000/year; n(%) | 12 (50.0) | 13 (76.5) |
| <50,000/year; n(%) | 10 (41.7) | 4 (23.5) |
| Declined response; n(%) | 2 (8.3) | 0 |
| Average Steps/day | |||
|---|---|---|---|
| Intervention (n = 24) | Control (n = 17) | a p | |
| Pre | 7378 (4368–8586) | 7732 (5748–10,022) | |
| Post | 8423 (6001–11,063) | 7890 (6159–9627) | |
| Δ | 976 (−388–3532) | 354 (−658–1300) | a Pic = 0.19 |
| a p | a Pi = 0.02 * | a Pc = 0.61 | |
| Variable | Intervention (n = 24) | Control (n = 17) | Pic | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Pre | Post | Δ | Pi | Pre | Post | Δ | Pc | ||
| Steps ≥40 s/m | 6860 (4128–8183) | 8104 (5669–10,647) | 954 (−325–3356) | 0.01 * | 7141 (5440–9437) | 7411 (5861–9217) | 327 (−558–1230) | 0.71 | 0.15 |
| Steps ≥80 s/m | 4125 (2815–6461) | 6123 (3480–8117) | 679 (−333–2767) | <0.01 * | 4651 (3512–6581) | 4706 (4204–7079) | 59 (−368–764) | 0.89 | 0.08 |
| Time spent ≥40 s/m (min) | 90.2 (54.9–113.0) | 102.3 (69.7–131.1) | 8.4 (−6.0–27.8) | 0.04 * | 97.4 (72.6–121.2) | 99.3 (76.1–117.9) | 6.4 (−10.3–17.2) | 0.60 | 0.35 |
| Time spent ≥80 s/m (min) | 60.5 (37.8–77.8) | 74.3 (50.8–100.3) | 7.8 (−4.0–24.6) | 0.02 * | 66.5 (49.2–89.5) | 67.1 (54.5–90.4) | 1.2 (−7.2–10.7) | 0.70 | 0.15 |
| Sporadic Movement | Purposeful Steps | Slow Walking | Medium Walking | Brisk Walking | Fast Locomotion | ||
|---|---|---|---|---|---|---|---|
| Steps/min | 20–39 | 40–59 | 60–79 | 80–99 | 100–119 * | ≥120 | |
| I | Pre | 327 | 677 | 1030 | 1826 | 1488 | 103 |
| (267–517) | (541–981) | (791–1364) | (1448–2342) | (828–2912) | (45–782) | ||
| Post | 346 | 711 | 1021 | 1925 | 3013 | 245 | |
| (231–498) | (480–1103) | (776–1601) | (1479–3201) | (1424–4190) | (46–1018) | ||
| Δ | −12 | −8 | 4 | −29 | 478 | 15 | |
| (−53–31) | (−100–77) | (−161–238) | (−270–470) | (−121–1844) | (−20–362) | ||
| C | Pre | 351 | 771 | 1185 | 2286 | 2088 | 106 |
| (293–585) | (657–1140) | (996–1580) | (2089–3099) | (1658–2968) | (71–194) | ||
| Post | 425 | 915 | 1249 | 2462 | 1996 | 121 | |
| (347–557) | (747–1023) | (1039–1630) | (1950–3759) | (1165–2317) | (79–205) | ||
| Δ | 40 | 66 | 208 | 271 | −92 | 25 | |
| (−100–113) | (−149–226) | (−176–305) | (−139–868) | (−510–181) | (−27–79) | ||
| Variable | Intervention (n = 24) | Control (n = 17) | Pic | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Pre | Post | Δ | Pi | Pre | Post | Δ | Pc | ||
| Peak 1-min (s/m) | 100.8 (90.3–113.0) | 108.2 (100.5–115.7) | 3.8 (−1.9–10.9) | 0.06 | 103.0 (89.7–110.7) | 99.7 (96.7–110.8) | −0.9 (−5.8–7.7) | 0.86 | 0.18 |
| Peak 30-min (s/m) | 61.7 (54.2–85.2) | 78.5 (61.9–103.2) | 4.3 (−0.96–16.8) | 0.03 * | 63.7 (59.3–87.9) | 65.6 (61.3–83.9) | 1.9 (−4.4–3.9) | 0.82 | 0.03 * |
| Average 30-min (s/m) | 30.6 (25.2–62.3) | 60.9 (31.4–93.7) | 5.7 (0.8–30.7) | 0.01 * | 41.9 (30.5–74.6) | 41.7 (29.7–69.7) | −0.8 (−6.9–5.0) | 0.61 | 0.03 * |
| Average 10-min (s/m) | 50.3 (42.5–81.7) | 73.7 (50.6–106.0) | 4.1 (−1.3–21.8) | 0.02 * | 61.9 (51.0–87.1) | 57.9 (48.0–82.6) | −6.6 (−10.0–5.4) | 0.38 | 0.04 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Harding, E.M.; Gibson, A.L.; Kang, H.; Zuhl, M.N.; Sharma, H.; Blair, C.K. Self-Selected Walking Cadence after 16-Week Light-Intensity Physical Activity Intervention for Older Cancer Survivors. Int. J. Environ. Res. Public Health 2022, 19, 4768. https://doi.org/10.3390/ijerph19084768
Harding EM, Gibson AL, Kang H, Zuhl MN, Sharma H, Blair CK. Self-Selected Walking Cadence after 16-Week Light-Intensity Physical Activity Intervention for Older Cancer Survivors. International Journal of Environmental Research and Public Health. 2022; 19(8):4768. https://doi.org/10.3390/ijerph19084768
Chicago/Turabian StyleHarding, Elizabeth M., Ann L. Gibson, Huining Kang, Micah N. Zuhl, Harsh Sharma, and Cindy K. Blair. 2022. "Self-Selected Walking Cadence after 16-Week Light-Intensity Physical Activity Intervention for Older Cancer Survivors" International Journal of Environmental Research and Public Health 19, no. 8: 4768. https://doi.org/10.3390/ijerph19084768
APA StyleHarding, E. M., Gibson, A. L., Kang, H., Zuhl, M. N., Sharma, H., & Blair, C. K. (2022). Self-Selected Walking Cadence after 16-Week Light-Intensity Physical Activity Intervention for Older Cancer Survivors. International Journal of Environmental Research and Public Health, 19(8), 4768. https://doi.org/10.3390/ijerph19084768







