The Impact of Maternal Prenatal Stress Related to the COVID-19 Pandemic during the First 1000 Days: A Historical Perspective
Abstract
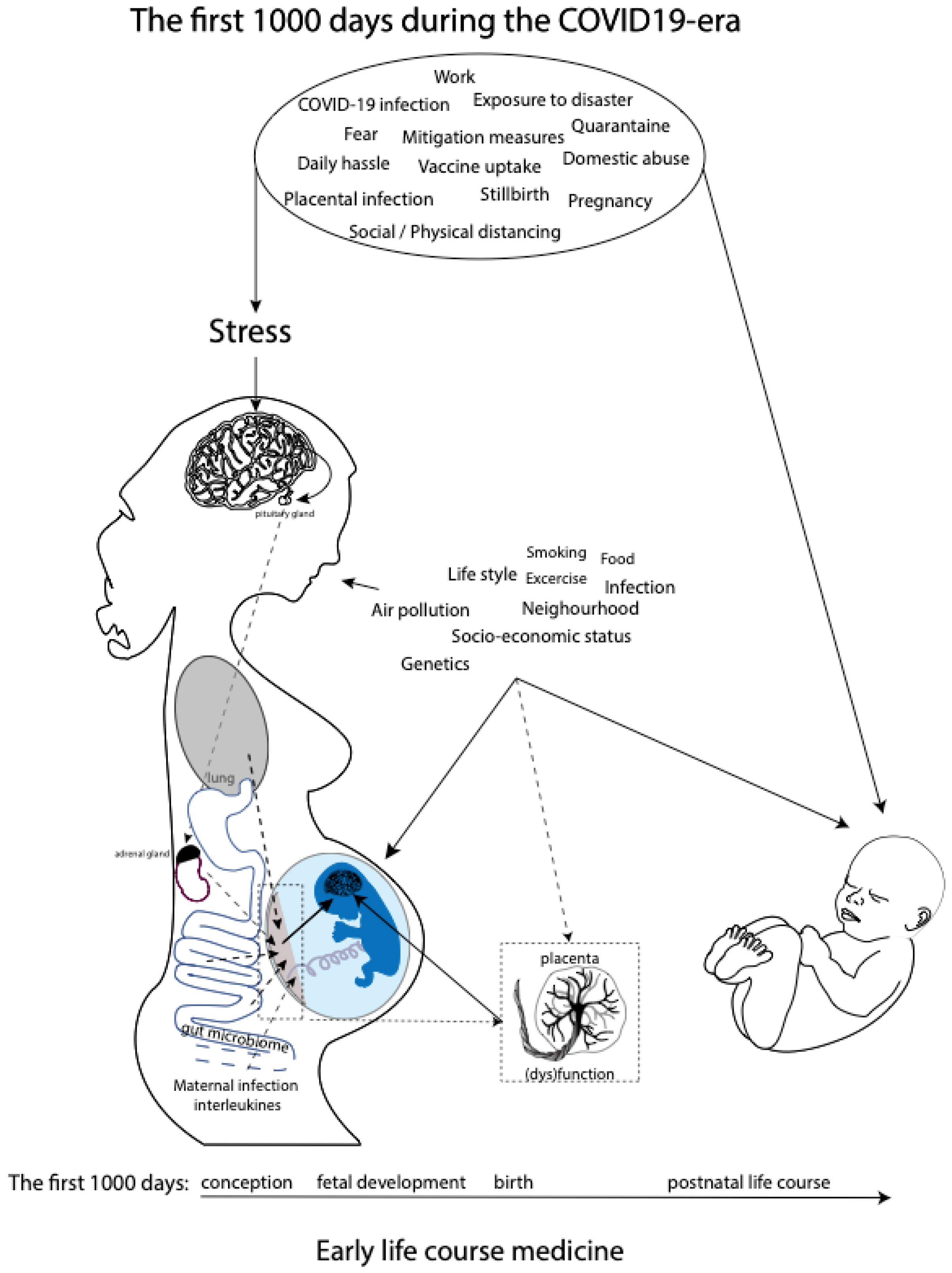
:1. Introduction
2. Materials and Methods
3. Results
3.1. Natural Disasters
3.1.1. Biological
The 1918 Influenza Pandemic (1918/1919)
3.1.2. Geological
Tangshan Earthquake, China (1976)
3.1.3. Hydrometerological
Quebec Ice Storm (1998), Hurricane Katrina, Louisiana (2005), Superstorm Sandy (2012)
3.1.4. Floods
Iowa Flood (2008) and The QF2011 Queensland Flood (Brisbane) (2011)
3.1.5. Manmade Disasters
Famines
The 9/11 Terrorist Attacks (2001)
Tailings Dam Breakage, Brazil (2015)
4. Discussion
4.1. Prenatal Maternal Stress and Developmental Origins of Health and Disease (DOHaD)
4.2. COVID-19 Pandemic, Maternal Prenatal Stress, and the First 1000 Days
| Disaster | Duration Disaster | Type | Offspring Effects during the Life Course | Sample Size | Outcome Variables | Confounders/Limitations |
|---|---|---|---|---|---|---|
| 1918 Influenza Pandemic (1918/1919) | 1–2 years | Natural | Exposure during mid- and late gestation is associated with multiple morbidities later in life, such as depression, diabetes, renal disease, ischemic heart disease, higher mortality, and lower socioeconomic status (SES) [32,33,34,35]. | I. Wisconsin Longitudinal Survey (1957, second-generation USA) [32] II. Swedish population database (1968–2012) [33] III. Taiwan population cohort (1916–1926) n = 870.468 [34] IV. National Health Interview Surveys (NHIS, USA) n = 101,068 [35] | I. First generation: flu indicators (male/female/either sex), controls (year of birth, male/female), dependent variables (male/female years of schooling, job prestige, and family SES). Second generation: controls (female indicator, birth year and order), dependent variables (years of schooling, family income, net worth, self-reported health, height, and BMI). Third generation: controls (birth year, female indicator, birth order, dependent variable (years of schooling). II. Occupational status 1970 (men), income (men), hospitalization (duration; men/women), mortality cause (men/women), birth year, and exposure trimester 1–3. III. Anthropometric outcome elementary school/middle school, educational attainment and outcome, health attainment and outcome, height in childhood and adolescents, and maternal mortality IV. Health outcome (medically important condition, e.g., diabetes and cardiovascular disease). | I. The 1918 pandemic co-occurred with a world war. II. Not fully able to distinguish between socioeconomic and biological factors. III. Possible positive selection due to high infant mortality rates (18%). IV. Underestimated effect expected higher mortality bore age 60, self-reported data. |
| The Dutch Famine, (“Dutch Hunger Winter”) (1944/1945) | ±5 months | Natural and manmade | Life-long effects on health; effects depending on the timing of exposure in gestation and the course of the catch-up period [74]. Exposure during early gestation is related to coronary heart disease, raised serum lipids, and more obesity, whereas mid-gestation exposure is associated with obstructive airways disease and microalbuminuria during the life course [78]. Twice more likely to develop schizophrenia [75,76,77]. | I. Review of the literature [74] II. n = 100,543 [78] III. n > 40.000 [75] IV. n > 40.000 [76] V. n= 2414 [77] | I. Review of the literature. II. Ecological measurement of nutritional status and date of conception not measured, and preterm labor is risk factor, too. III. Selective conception, selective survival. IV. The ascertainment of the population at risk, the ascertainment of cases [2], the reliability of diagnosis [3], the degree of food deprivation [4], the control of coincident factors [5], the use of group vs. individual data [6], and the control of social class [7]. V. Pinpointing the exact timing of famine exposure during gestation and associated outcomes in later life, due to the relatively small sample size and partial overlap between the three famine exposed groups on the other. | |
| Great Leap Forward Famine, China (1959–1961) | ±3 years | Natural and manmade | Prenatal exposure increases risk for schizophrenia later in life [81]. | 1987 Chinese National Disability Sample Survey n = 1,579,316 | Chinese National Disability Sample Survey (CNDSS), with focus on disability. Two step: interview + questionnaire. On-site diagnosis professional psychiatrists. | Large-scale sample survey, accuracy on diagnosis might not as a psychiatric clinic, selected cohorts were adults 22–32 years, and researchers not able to look at cohort differences in schizophrenia rate beyond this age. |
| Biafra Famine, Nigeria (1967–1970) | ±3 years | Manmade | Exposure during fetal and infant period is associated with increased risk of hypertension and glucose tolerance [83]. | n = 1339 | Observational study, men and non-pregnant women. Level of education, current smoking, previously diagnosed hypertension, diabetes, treatment. Level of education. Blood pressure and random plasma glucose. Height, weight, BMI, and waist circumference. | The lack of anthropometric data at birth and in infancy. |
| Tangshan earthquake, China (1976) | Natural | Higher serum uric-acid concentrations; 70% more likely to develop hyperuricemia approximately 33 years after the event, independent of traditional risk factors. [130] | n = 536 | Collection of blood samples for uric acid measurements. Questionnaire for covariates: demographic, socioeconomic, and medical data (age, sex, smoking status, alcohol drinking status, education, and physical activity). Blood pressure. Fasting blood glucose. | Results may not be generalizable to other populations with different diet patterns, lifestyles, and genetic backgrounds; participants were young, so there was a low prevalence of major chronic disease. | |
| Quebec Ice Storm (1998) | ±6 h to 5 weeks | Natural | Negative impact on cognitive and language development of the unborn child, independent of maternal personality factors [59]. The level of prenatal maternal stress is associated with higher BMI levels and adiposity in children of ages 5.5, 8.5, 13.5, and 15.5 years [60]. DNA methylation mediates the impact of exposure to prenatal maternal stress on BMI and central adiposity in children at age 13.5 years [61]. | I. n = 150 [59] II. n = 386 [60] III. n = 66 [61] | I. Lacking data on the immediate biological processes. II. No control group exists; sample was skewed to the middle-upper and upper classes and relatively small. III. Small sample size. | |
| 9/11 Terrorist attacks (2001) | ±1 day | Manmade | Offspring at one year of age of mothers exposed to attacks on the World Trade Center during pregnancy show lower salivary cortisol levels [84]. | n = 38 | Probable PTSD and PTSD severity by PTSD checklist. Severity of depression assessment with Beck Depression Index. Demographic and medical information, exposure and pregnancy outcome. Salivary sample collection + determination free cortisol levels. Relationship maternal PTSD—cortisol and infant cortisol levels, impact of pregnancy trimester of exposure. | Small sample size. Maternal age, ethnicity, BMI, hours of sleep, wakefulness, and breastfeeding. |
| Hurricane Katrina, Louisiana (2005) | ±1 day | Natural | No association seen between maternal stress and child temperament [63]. | n = 288 | [63] Interview 8–10 weeks postpartum. Temperament interview 1 year postpartum. Delivery questionnaire (basic demographic information, hurricane stress exposure, and social support). Interview information on living conditions before the storm, at evacuation and currently. PTSD Checklist—civilian-version screening tool. Edinburgh Postnatal Depression Scale. Symptoms Cheklist-090-Revised. | The evaluation of the infants’ temperament was performed by the mother, the means of the EITQ were consistently lower than the means provided by the authors, and study limited to English-speaking women. |
| Iowa Flood (2008) | ±3 weeks | Natural | Greater experienced prenatal maternal stress predicted higher BMI at 30 months in offspring [69]. | n = 103 | Small sample size; sample consisted of primarily Caucasian women of relatively high SES; and no control group. | |
| The QF2011 Queensland Flood, Brisbane (2011) | ±36 h | Natural | Increased 4-year child anxiety symptoms. [36] Association with prenatal maternal stress and alterations in placental glucocorticoid system [37]. | I. n = 181 [36] II. n = 230 [37] | I. Small sample size. II. Small sample size, no placentas of women not exposed to the flood, and no placentas of women exposed in the third trimester. | |
| Superstorm Sandy (2012) | ±1 day | Natural | Prenatal maternal depression amplified by Sandy; association with lower emotion regulation and greater distress at 6 months in neonates [66,67]. | I. n = 408 [66] II. n = 318 [67] | I. Infant temperament was based on maternal report; maternal mood may introduce a potential bias and potential unknown confounders; the statistical design of our study does not allow for an analysis of the impact of the exact timing of exposure to Sandy in relation to the trimester of exposure. II. Temperament was measured solely via maternal report. Trimester-specific effects of Sandy on temperament were not examined. | |
| Tailings dam breakage, Brazil (2015) | ± hours [breakage itself] | Manmade | Increased premature birth [85]. | n = 914,795 | ||
| COVID-19 pandemic (2019) | Years | Natural and manmade | ? |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Van den Bergh, B.R.H.; van den Heuvel, M.I.; Lahti, M.; Braeken, M.; de Rooij, S.R.; Entringer, S.; Hoyer, D.; Roseboom, T.; Raikkonen, K.; King, S.; et al. Prenatal developmental origins of behavior and mental health: The influence of maternal stress in pregnancy. Neurosci. Biobehav. Rev. 2020, 117, 26–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Browne, P.D.; Bossenbroek, R.; Kluft, A.; van Tetering, E.M.A.; de Weerth, C. Prenatal Anxiety and Depression: Treatment Uptake, Barriers, and Facilitators in Midwifery Care. J. Womens Health 2021, 30, 1116–1126. [Google Scholar] [CrossRef] [PubMed]
- Missler, M.; van Straten, A.; Denissen, J.; Donker, T.; Beijers, R. Effectiveness of a psycho-educational intervention for expecting parents to prevent postpartum parenting stress, depression and anxiety: A randomized controlled trial. BMC Pregnancy Childbirth 2020, 20, 658. [Google Scholar] [CrossRef]
- Barzilay, R.; Moore, T.M.; Greenberg, D.M.; DiDomenico, G.E.; Brown, L.A.; White, L.K.; Gur, R.C.; Gur, R.E. Resilience, COVID-19-related stress, anxiety and depression during the pandemic in a large population enriched for healthcare providers. Transl. Psychiatry 2020, 10, 291. [Google Scholar] [CrossRef] [PubMed]
- Ghazanfarpour, M.; Bahrami, F.; Rashidi Fakari, F.; Ashrafinia, F.; Babakhanian, M.; Dordeh, M.; Abdi, F. Prevalence of anxiety and depression among pregnant women during the COVID-19 pandemic: A meta-analysis. J. Psychosom. Obstet. Gynaecol. 2021, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Vacaru, S.; Beijers, R.; Browne, P.D.; Cloin, M.; van Bakel, H.; van den Heuvel, M.I.; de Weerth, C. The risk and protective factors of heightened prenatal anxiety and depression during the COVID-19 lockdown. Sci. Rep. 2021, 11, 20261. [Google Scholar] [CrossRef]
- Been, J.V.; Burgos Ochoa, L.; Bertens, L.C.M.; Schoenmakers, S.; Steegers, E.A.P.; Reiss, I.K.M. Impact of COVID-19 mitigation measures on the incidence of preterm birth: A national quasi-experimental study. Lancet Public Health 2020, 5, e604–e611. [Google Scholar] [CrossRef]
- Assiri, A.M.; Midgley, C.M.; Abedi, G.R.; Bin Saeed, A.; Almasri, M.M.; Lu, X.; Al-Abdely, H.M.; Abdalla, O.; Mohammed, M.; Algarni, H.S.; et al. Epidemiology of a Novel Recombinant Middle East Respiratory Syndrome Coronavirus in Humans in Saudi Arabia. J. Infect. Dis. 2016, 214, 712–721. [Google Scholar] [CrossRef] [Green Version]
- Wong, S.F.; Chow, K.M.; de Swiet, M. Severe Acute Respiratory Syndrome and pregnancy. BJOG 2003, 110, 641–642. [Google Scholar] [CrossRef]
- COVID-19 and pregnancy. BMJ 2020, 369, m1672.
- Narang, K.; Ibirogba, E.R.; Elrefaei, A.; Trad, A.T.A.; Theiler, R.; Nomura, R.; Picone, O.; Kilby, M.; Escuriet, R.; Suy, A.; et al. SARS-CoV-2 in Pregnancy: A Comprehensive Summary of Current Guidelines. J. Clin. Med. 2020, 9, 1521. [Google Scholar] [CrossRef] [PubMed]
- Verweij, E.J.; M’Hamdi, H.I.; Steegers, E.A.P.; Reiss, I.K.M.; Schoenmakers, S. Collateral damage of the COVID-19 pandemic: A Dutch perinatal perspective. BMJ 2020, 369, m2326. [Google Scholar] [CrossRef] [PubMed]
- Zambrano, L.D.; Ellington, S.; Strid, P.; Galang, R.R.; Oduyebo, T.; Tong, V.T.; Woodworth, K.R.; Nahabedian, J.F., 3rd; Azziz-Baumgartner, E.; Gilboa, S.M.; et al. Update: Characteristics of Symptomatic Women of Reproductive Age with Laboratory-Confirmed SARS-CoV-2 Infection by Pregnancy Status—United States, January 22-October 3, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1641–1647. [Google Scholar] [CrossRef]
- Husen, M.F.; van der Meeren, L.E.; Verdijk, R.M.; Fraaij, P.L.A.; van der Eijk, A.A.; Koopmans, M.P.G.; Freeman, L.; Bogers, H.; Trietsch, M.D.; Reiss, I.K.M.; et al. Unique Severe COVID-19 Placental Signature Independent of Severity of Clinical Maternal Symptoms. Viruses 2021, 13, 1670. [Google Scholar] [CrossRef] [PubMed]
- DeSisto, C.L.; Wallace, B.; Simeone, R.M.; Polen, K.; Ko, J.Y.; Meaney-Delman, D.; Ellington, S.R. Risk for Stillbirth Among Women With and Without COVID-19 at Delivery Hospitalization—United States, March 2020–September 2021. MMWR Morb. Mortal. Wkly. Rep. 2021, 70, 1640–1645. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.A.; Avvad-Portari, E.; Babal, P.; Baldewijns, M.; Blomberg, M.; Bouachba, A.; Camacho, J.; Collardeau-Frachon, S.; Colson, A.; Dehaene, I.; et al. Placental Tissue Destruction and Insufficiency from COVID-19 Causes Stillbirth and Neonatal Death from Hypoxic-Ischemic Injury: A Study of 68 Cases with SARS-CoV-2 Placentitis from 12 Countries. Arch. Pathol. Lab. Med. 2022. [Google Scholar] [CrossRef]
- Goncu Ayhan, S.; Oluklu, D.; Atalay, A.; Menekse Beser, D.; Tanacan, A.; Moraloglu Tekin, O.; Sahin, D. COVID-19 vaccine acceptance in pregnant women. Int. J. Gynaecol. Obstet. 2021, 154, 291–296. [Google Scholar] [CrossRef]
- Riad, A.; Jouzova, A.; Ustun, B.; Lagova, E.; Hruban, L.; Janku, P.; Pokorna, A.; Klugarova, J.; Koscik, M.; Klugar, M. COVID-19 Vaccine Acceptance of Pregnant and Lactating Women (PLW) in Czechia: An Analytical Cross-Sectional Study. Int. J. Environ. Res. Public Health 2021, 18, 13373. [Google Scholar] [CrossRef]
- Skjefte, M.; Ngirbabul, M.; Akeju, O.; Escudero, D.; Hernandez-Diaz, S.; Wyszynski, D.F.; Wu, J.W. COVID-19 vaccine acceptance among pregnant women and mothers of young children: Results of a survey in 16 countries. Eur. J. Epidemiol. 2021, 36, 197–211. [Google Scholar] [CrossRef]
- Wilson, R.J.; Paterson, P.; Jarrett, C.; Larson, H.J. Understanding factors influencing vaccination acceptance during pregnancy globally: A literature review. Vaccine 2015, 33, 6420–6429. [Google Scholar] [CrossRef] [Green Version]
- Van Spall, H.G.C. Exclusion of pregnant and lactating women from COVID-19 vaccine trials: A missed opportunity. Eur. Heart J. 2021, 42, 2724–2726. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.D.; Pippen, J.L.; Adesomo, A.A.; Rood, K.M.; Landon, M.B.; Costantine, M.M. Exclusion of Pregnant Women from Clinical Trials during the Coronavirus Disease 2019 Pandemic: A Review of International Registries. Am. J. Perinatol. 2020, 37, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Moro, P.L.; Panagiotakopoulos, L.; Oduyebo, T.; Olson, C.K.; Myers, T. Monitoring the safety of COVID-19 vaccines in pregnancy in the US. Hum. Vaccin. Immunother. 2021, 17, 4705–4713. [Google Scholar] [CrossRef] [PubMed]
- Oliver, S.E.; Gargano, J.W.; Marin, M.; Wallace, M.; Curran, K.G.; Chamberland, M.; McClung, N.; Campos-Outcalt, D.; Morgan, R.L.; Mbaeyi, S.; et al. The Advisory Committee on Immunization Practices’ Interim Recommendation for Use of Pfizer-BioNTech COVID-19 Vaccine—United States, December 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 1922–1924. [Google Scholar] [CrossRef]
- Madigan, S.; Oatley, H.; Racine, N.; Fearon, R.M.P.; Schumacher, L.; Akbari, E.; Cooke, J.E.; Tarabulsy, G.M. A Meta-Analysis of Maternal Prenatal Depression and Anxiety on Child Socioemotional Development. J. Am. Acad. Child. Adolesc. Psychiatry 2018, 57, 645–657.e648. [Google Scholar] [CrossRef]
- Manzari, N.; Matvienko-Sikar, K.; Baldoni, F.; O’Keeffe, G.W.; Khashan, A.S. Prenatal maternal stress and risk of neurodevelopmental disorders in the offspring: A systematic review and meta-analysis. Soc. Psychiatry Psychiatr. Epidemiol. 2019, 54, 1299–1309. [Google Scholar] [CrossRef] [PubMed]
- Barker, D.J. The developmental origins of adult disease. Eur. J. Epidemiol. 2003, 18, 733–736. [Google Scholar] [CrossRef]
- Barker, D.J. The origins of the developmental origins theory. J. Intern. Med. 2007, 261, 412–417. [Google Scholar] [CrossRef]
- Barouki, R.; Gluckman, P.D.; Grandjean, P.; Hanson, M.; Heindel, J.J. Developmental origins of non-communicable disease: Implications for research and public health. Environ. Health 2012, 11, 42. [Google Scholar] [CrossRef] [Green Version]
- Gluckman, P.D.; Hanson, M.A.; Mitchell, M.D. Developmental origins of health and disease: Reducing the burden of chronic disease in the next generation. Genome Med. 2010, 2, 14. [Google Scholar] [CrossRef] [Green Version]
- Beijers, R.; Buitelaar, J.K.; de Weerth, C. Mechanisms underlying the effects of prenatal psychosocial stress on child outcomes: Beyond the HPA axis. Eur. Child Adolesc. Psychiatry 2014, 23, 943–956. [Google Scholar] [CrossRef] [PubMed]
- Cook, C.J.; Fletcher, J.M.; Forgues, A. Multigenerational Effects of Early-Life Health Shocks. Demography 2019, 56, 1855–1874. [Google Scholar] [CrossRef] [PubMed]
- Helgertz, J.; Bengtsson, T. The Long-Lasting Influenza: The Impact of Fetal Stress During the 1918 Influenza Pandemic on Socioeconomic Attainment and Health in Sweden, 1968–2012. Demography 2019, 56, 1389–1425. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, M.J.; Liu, E.M. Does in utero exposure to Illness matter? The 1918 influenza epidemic in Taiwan as a natural experiment. J. Health Econ. 2014, 37, 152–163. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mazumder, B.; Almond, D.; Park, K.; Crimmins, E.M.; Finch, C.E. Lingering prenatal effects of the 1918 influenza pandemic on cardiovascular disease. J. Dev. Orig. Health Dis. 2010, 1, 26–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McLean, M.A.; Cobham, V.E.; Simcock, G.; Kildea, S.; King, S. Toddler Temperament Mediates the Effect of Prenatal Maternal Stress on Childhood Anxiety Symptomatology: The QF2011 Queensland Flood Study. Int. J. Environ. Res. Public Health 2019, 16, 1998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- St-Pierre, J.; Laplante, D.P.; Elgbeili, G.; Dawson, P.A.; Kildea, S.; King, S.; Vaillancourt, C. Natural disaster-related prenatal maternal stress is associated with alterations in placental glucocorticoid system: The QF2011 Queensland Flood Study. Psychoneuroendocrinology 2018, 94, 38–48. [Google Scholar] [CrossRef] [Green Version]
- Reduction UNOfDR. Available online: https://www.unisdr.org/files/7817_UNISDRTerminologyEnglish.pdf (accessed on 26 February 2022).
- Makwana, N. Disaster and its impact on mental health: A narrative review. J. Family Med. Prim. Care 2019, 8, 3090–3095. [Google Scholar] [CrossRef]
- Beijers, R.; Hartman, S.; Shalev, I.; Hastings, W.; Mattern, B.C.; de Weerth, C.; Belsky, J. Testing three hypotheses about effects of sensitive-insensitive parenting on telomeres. Dev. Psychol. 2020, 56, 237–250. [Google Scholar] [CrossRef] [Green Version]
- Grande, L.A.; Swales, D.A.; Sandman, C.A.; Glynn, L.M.; Davis, E.P. Maternal caregiving ameliorates the consequences of prenatal maternal psychological distress on child development. Dev. Psychopathol. 2021, 1–10. [Google Scholar] [CrossRef]
- Kildea, S.; Simcock, G.; Liu, A.; Elgbeili, G.; Laplante, D.P.; Kahler, A.; Austin, M.P.; Tracy, S.; Kruske, S.; Tracy, M.; et al. Continuity of midwifery carer moderates the effects of prenatal maternal stress on postnatal maternal wellbeing: The Queensland flood study. Arch. Womens Ment. Health 2018, 21, 203–214. [Google Scholar] [CrossRef] [PubMed]
- Simcock, G.; Kildea, S.; Kruske, S.; Laplante, D.P.; Elgbeili, G.; King, S. Disaster in pregnancy: Midwifery continuity positively impacts infant neurodevelopment, QF2011 study. BMC Pregnancy Childbirth 2018, 18, 309. [Google Scholar] [CrossRef] [PubMed]
- Almond, D. Is the 1918 influenza pandemic over? Long-term effects of in utero influenza exposure in the post-1940 US population. J. Polit. Econ. 2006, 114, 672–712. [Google Scholar] [CrossRef] [Green Version]
- Almond, D.; Mazumder, B. The 1918 influenza pandemic and subsequent health outcomes: An analysis of SIPP data. Am. Econ. Rev. 2005, 95, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Bos, M.J.; Koudstaal, P.J.; Hofman, A.; Witteman, J.C.; Breteler, M.M. Uric acid is a risk factor for myocardial infarction and stroke: The Rotterdam study. Stroke 2006, 37, 1503–1507. [Google Scholar] [CrossRef] [Green Version]
- Eisen, A.; Benderly, M.; Goldbourt, U.; Haim, M. Is serum uric acid level an independent predictor of heart failure among patients with coronary artery disease? Clin. Cardiol. 2013, 36, 110–116. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Iribarren, C.; McCulloch, C.E.; Darbinian, J.; Go, A.S. Risk factors for end-stage renal disease: 25-year follow-up. Arch. Intern. Med. 2009, 169, 342–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, M.; Yang, F.; Yang, I.; Yin, Y.; Luo, J.J.; Wang, H.; Yang, X.F. Uric acid, hyperuricemia and vascular diseases. Front. Biosci. 2012, 17, 656–669. [Google Scholar] [CrossRef] [Green Version]
- Lv, Q.; Meng, X.F.; He, F.F.; Chen, S.; Su, H.; Xiong, J.; Gao, P.; Tian, X.J.; Liu, J.S.; Zhu, Z.H.; et al. High serum uric acid and increased risk of type 2 diabetes: A systemic review and meta-analysis of prospective cohort studies. PLoS ONE 2013, 8, e56864. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Zhang, C.; Song, X.; Lin, H.; Zhang, D.; Meng, W.; Zhang, Y.; Zhu, Z.; Tang, F.; Liu, L.; et al. A longitudinal cohort based association study between uric acid level and metabolic syndrome in Chinese Han urban male population. BMC Public Health 2012, 12, 419. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Bian, L.; Choi, Y. Serum uric acid: A marker of metabolic syndrome and subclinical atherosclerosis in Korean men. Angiology 2012, 63, 420–428. [Google Scholar] [CrossRef]
- An, C.; Zhang, Y.; Yu, L.; Li, N.; Song, M.; Wang, L.; Zhao, X.; Gao, Y.; Wang, X. Long-term impact of earthquake stress on fasting glucose control and diabetes prevalence among Chinese adults of Tangshan. Int. J. Clin. Exp. Med. 2014, 7, 4441–4447. [Google Scholar] [PubMed]
- Guo, C.; He, P.; Song, X.; Zheng, X. Long-term effects of prenatal exposure to earthquake on adult schizophrenia. Br. J. Psychiatry 2019, 215, 730–735. [Google Scholar] [CrossRef]
- Li, N.; Song, M.; Wang, L.; Zhao, X.C.; Wang, R.; Gao, Y.Y.; Yu, L.L.; An, C.X.; Wang, X.Y. Experiencing earthquake in the first trimester of the fetal life increases subsequent diabetes risk in the adulthood: A cross-sectional study. Reprod. Biol. Endocrinol. 2020, 18, 109. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Wang, Y.; Zhao, X.; Gao, Y.; Song, M.; Yu, L.; Wang, L.; Li, N.; Chen, Q.; Li, Y.; et al. Long-term effect of early-life stress from earthquake exposure on working memory in adulthood. Neuropsychiatr. Dis. Treat. 2015, 11, 2959–2965. [Google Scholar] [PubMed] [Green Version]
- Song, M.; Wang, L.; Zhao, X.C.; Li, N.; Wang, R.; Gao, Y.Y.; Yu, L.L.; An, C.X.; Wang, X.Y. The prevalence and risk of DSM-IV axis I disorder in adults with early-life earthquake stress: A cross-sectional study. Int. J. Dev. Neurosci. 2019, 76, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; An, C.; Wang, J.; Wang, Y.; Song, M.; Li, N.; Chen, Y.; Sun, F.; Chen, X.; Wang, X. Earthquake Experience at Different Trimesters during Pregnancy Is Associated with Leukocyte Telomere Length and Long-term Health in Adulthood. Front. Psychiatry 2017, 8, 208. [Google Scholar] [CrossRef] [Green Version]
- King, S.; Laplante, D.P. The effects of prenatal maternal stress on children’s cognitive development: Project Ice Storm. Stress 2005, 8, 35–45. [Google Scholar] [CrossRef]
- Liu, G.T.; Dancause, K.N.; Elgbeili, G.; Laplante, D.P.; King, S. Disaster-related prenatal maternal stress explains increasing amounts of variance in body composition through childhood and adolescence: Project Ice Storm. Environ. Res. 2016, 150, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Cao-Lei, L.; Dancause, K.N.; Elgbeili, G.; Massart, R.; Szyf, M.; Liu, A.; Laplante, D.P.; King, S. DNA methylation mediates the impact of exposure to prenatal maternal stress on BMI and central adiposity in children at age 13(1/2) years: Project Ice Storm. Epigenetics 2015, 10, 749–761. [Google Scholar] [CrossRef]
- Yong Ping, E.; Laplante, D.P.; Elgbeili, G.; Jones, S.L.; Brunet, A.; King, S. Disaster-related prenatal maternal stress predicts HPA reactivity and psychopathology in adolescent offspring: Project Ice Storm. Psychoneuroendocrinology 2020, 117, 104697. [Google Scholar] [CrossRef]
- Tees, M.T.; Harville, E.W.; Xiong, X.; Buekens, P.; Pridjian, G.; Elkind-Hirsch, K. Hurricane Katrina-related maternal stress, maternal mental health, and early infant temperament. Matern. Child Health J. 2010, 14, 511–518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oni, O.; Harville, E.; Xiong, X.; Buekens, P. Relationships among stress coping styles and pregnancy complications among women exposed to Hurricane Katrina. J. Obstet. Gynecol. Neonatal. Nurs. 2015, 44, 256–267. [Google Scholar] [CrossRef] [Green Version]
- Londono Tobon, A.; Diaz Stransky, A.; Ross, D.A.; Stevens, H.E. Effects of Maternal Prenatal Stress: Mechanisms, Implications, and Novel Therapeutic Interventions. Biol. Psychiatry 2016, 80, e85–e87. [Google Scholar] [CrossRef] [Green Version]
- Nomura, Y.; Davey, K.; Pehme, P.M.; Finik, J.; Glover, V.; Zhang, W.; Huang, Y.; Buthmann, J.; Dana, K.; Yoshida, S.; et al. Influence of in utero exposure to maternal depression and natural disaster-related stress on infant temperament at 6 months: The children of Superstorm Sandy. Infant. Ment. Health J. 2019, 40, 204–216. [Google Scholar] [CrossRef]
- Zhang, W.; Rajendran, K.; Ham, J.; Finik, J.; Buthmann, J.; Davey, K.; Pehme, P.M.; Dana, K.; Pritchett, A.; Laws, H.; et al. Prenatal exposure to disaster-related traumatic stress and developmental trajectories of temperament in early childhood: Superstorm Sandy pregnancy study. J. Affect. Disord. 2018, 234, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Dancause, K.N.; Laplante, D.P.; Hart, K.J.; O’Hara, M.W.; Elgbeili, G.; Brunet, A.; King, S. Prenatal stress due to a natural disaster predicts adiposity in childhood: The Iowa Flood Study. J. Obes. 2015, 2015, 570541. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kroska, E.B.; O’Hara, M.W.; Elgbeili, G.; Hart, K.J.; Laplante, D.P.; Dancause, K.N.; King, S. The impact of maternal flood-related stress and social support on offspring weight in early childhood. Arch. Womens Ment. Health 2018, 21, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Laplante, D.P.; Hart, K.J.; O’Hara, M.W.; Brunet, A.; King, S. Prenatal maternal stress is associated with toddler cognitive functioning: The Iowa Flood Study. Early Hum. Dev. 2018, 116, 84–92. [Google Scholar] [CrossRef] [PubMed]
- McLean, M.A.; Simcock, G.; Elgbeili, G.; Laplante, D.P.; Kildea, S.; Hurrion, E.; Lequertier, B.; Cobham, V.E.; King, S. Disaster-related prenatal maternal stress, and childhood HPA-axis regulation and anxiety: The QF2011 Queensland Flood Study. Psychoneuroendocrinology 2020, 118, 104716. [Google Scholar] [CrossRef]
- Yong Ping, E.; Laplante, D.P.; Elgbeili, G.; Hillerer, K.M.; Brunet, A.; O’Hara, M.W.; King, S. Prenatal maternal stress predicts stress reactivity at 2(1/2) years of age: The Iowa Flood Study. Psychoneuroendocrinology 2015, 56, 62–78. [Google Scholar] [CrossRef] [PubMed]
- Roseboom, T.; de Rooij, S.; Painter, R. The Dutch famine and its long-term consequences for adult health. Early Hum. Dev. 2006, 82, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Kyle, U.G.; Pichard, C. The Dutch Famine of 1944-1945: A pathophysiological model of long-term consequences of wasting disease. Curr. Opin. Clin. Nutr. Metab. Care 2006, 9, 388–394. [Google Scholar] [CrossRef]
- Neugebauer, R.; Hoek, H.W.; Susser, E. Prenatal exposure to wartime famine and development of antisocial personality disorder in early adulthood. JAMA 1999, 282, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Susser, E.; Neugebauer, R.; Hoek, H.W.; Brown, A.S.; Lin, S.; Labovitz, D.; Gorman, J.M. Schizophrenia after prenatal famine. Further evidence. Arch. Gen. Psychiatry 1996, 53, 25–31. [Google Scholar] [CrossRef]
- Susser, E.S.; Lin, S.P. Schizophrenia after prenatal exposure to the Dutch Hunger Winter of 1944-1945. Arch. Gen. Psychiatry 1992, 49, 983–988. [Google Scholar] [CrossRef]
- Painter, R.C.; Roseboom, T.J.; Bleker, O.P. Prenatal exposure to the Dutch famine and disease in later life: An overview. Reprod. Toxicol. 2005, 20, 345–352. [Google Scholar] [CrossRef]
- Li, Y.; Jaddoe, V.W.; Qi, L.; He, Y.; Lai, J.; Wang, J.; Zhang, J.; Hu, Y.; Ding, E.L.; Yang, X.; et al. Exposure to the Chinese famine in early life and the risk of hypertension in adulthood. J. Hypertens. 2011, 29, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Liu, S.; Li, S.; Feng, R.; Na, L.; Chu, X.; Wu, X.; Niu, Y.; Sun, Z.; Han, T.; et al. Prenatal exposure to famine and the development of hyperglycemia and type 2 diabetes in adulthood across consecutive generations: A population-based cohort study of families in Suihua, China. Am. J. Clin. Nutr. 2017, 105, 221–227. [Google Scholar] [CrossRef] [Green Version]
- Song, S.; Wang, W.; Hu, P. Famine, death, and madness: Schizophrenia in early adulthood after prenatal exposure to the Chinese Great Leap Forward Famine. Soc. Sci. Med. 2009, 68, 1315–1321. [Google Scholar] [CrossRef]
- Li, J.; Yang, Q.; An, R.; Sesso, H.D.; Zhong, V.W.; Chan, K.H.K.; Madsen, T.E.; Papandonatos, G.D.; Zheng, T.; Wu, W.C.; et al. Famine and Trajectories of Body Mass Index, Waist Circumference, and Blood Pressure in Two Generations: Results From the CHNS From 1993–2015. Hypertension 2022, 79, 518–531. [Google Scholar] [CrossRef] [PubMed]
- Hult, M.; Tornhammar, P.; Ueda, P.; Chima, C.; Bonamy, A.K.; Ozumba, B.; Norman, M. Hypertension, diabetes and overweight: Looming legacies of the Biafran famine. PLoS ONE 2010, 5, e13582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yehuda, R.; Engel, S.M.; Brand, S.R.; Seckl, J.; Marcus, S.M.; Berkowitz, G.S. Transgenerational effects of posttraumatic stress disorder in babies of mothers exposed to the World Trade Center attacks during pregnancy. J. Clin. Endocrinol. Metab. 2005, 90, 4115–4118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mrejen, M.; Perelman, J.; Machado, D.C. Environmental disasters and birth outcomes: Impact of a tailings dam breakage in Brazil. Soc. Sci. Med. 2020, 250, 112868. [Google Scholar] [CrossRef]
- Wu, Y.; Lu, Y.C.; Jacobs, M.; Pradhan, S.; Kapse, K.; Zhao, L.; Niforatos-Andescavage, N.; Vezina, G.; du Plessis, A.J.; Limperopoulos, C. Association of Prenatal Maternal Psychological Distress With Fetal Brain Growth, Metabolism, and Cortical Maturation. JAMA Netw. Open 2020, 3, e1919940. [Google Scholar] [CrossRef]
- Dawson, G.; Ashman, S.B.; Carver, L.J. The role of early experience in shaping behavioral and brain development and its implications for social policy. Dev. Psychopathol. 2000, 12, 695–712. [Google Scholar] [CrossRef] [Green Version]
- Gemmill, A.; Catalano, R.; Casey, J.A.; Karasek, D.; Alcala, H.E.; Elser, H.; Torres, J.M. Association of Preterm Births Among US Latina Women With the 2016 Presidential Election. JAMA Netw. Open 2019, 2, e197084. [Google Scholar] [CrossRef]
- Yehuda, R. Current status of cortisol findings in post-traumatic stress disorder. Psychiatr. Clin. N. Am. 2002, 25, 341–368. [Google Scholar] [CrossRef]
- Catalani, A.; Alema, G.S.; Cinque, C.; Zuena, A.R.; Casolini, P. Maternal corticosterone effects on hypothalamus-pituitary-adrenal axis regulation and behavior of the offspring in rodents. Neurosci. Biobehav. Rev. 2011, 35, 1502–1517. [Google Scholar] [CrossRef] [PubMed]
- Gitau, R.; Cameron, A.; Fisk, N.M.; Glover, V. Fetal exposure to maternal cortisol. Lancet 1998, 352, 707–708. [Google Scholar] [CrossRef]
- Kapoor, A.; Petropoulos, S.; Matthews, S.G. Fetal programming of hypothalamic-pituitary-adrenal (HPA) axis function and behavior by synthetic glucocorticoids. Brain Res. Rev. 2008, 57, 586–595. [Google Scholar] [CrossRef]
- Zijlmans, M.A.; Riksen-Walraven, J.M.; de Weerth, C. Associations between maternal prenatal cortisol concentrations and child outcomes: A systematic review. Neurosci. Biobehav. Rev. 2015, 53, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Jasarevic, E.; Rodgers, A.B.; Bale, T.L. A novel role for maternal stress and microbial transmission in early life programming and neurodevelopment. Neurobiol. Stress 2015, 1, 81–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hechler, C.; Borewicz, K.; Beijers, R.; Saccenti, E.; Riksen-Walraven, M.; Smidt, H.; de Weerth, C. Association between Psychosocial Stress and Fecal Microbiota in Pregnant Women. Sci. Rep. 2019, 9, 4463. [Google Scholar] [CrossRef] [Green Version]
- Carlson, A.L.; Xia, K.; Azcarate-Peril, M.A.; Goldman, B.D.; Ahn, M.; Styner, M.A.; Thompson, A.L.; Geng, X.; Gilmore, J.H.; Knickmeyer, R.C. Infant Gut Microbiome Associated with Cognitive Development. Biol. Psychiatry 2018, 83, 148–159. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef]
- Brunton, P.J.; Russell, J.A. Neuroendocrine control of maternal stress responses and fetal programming by stress in pregnancy. Prog. Neuropsychopharmacol. Biol. Psychiatry 2011, 35, 1178–1191. [Google Scholar] [CrossRef]
- Jahnke, J.R.; Teran, E.; Murgueitio, F.; Cabrera, H.; Thompson, A.L. Maternal stress, placental 11beta-hydroxysteroid dehydrogenase type 2, and infant HPA axis development in humans: Psychosocial and physiological pathways. Placenta 2021, 104, 179–187. [Google Scholar] [CrossRef]
- Welberg, L.A.; Thrivikraman, K.V.; Plotsky, P.M. Chronic maternal stress inhibits the capacity to up-regulate placental 11beta-hydroxysteroid dehydrogenase type 2 activity. J. Endocrinol. 2005, 186, R7–R12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wroble-Biglan, M.C.; Dietz, L.J.; Pienkosky, T.V. Prediction of infant temperament from catecholamine and self-report measures of maternal stress during pregnancy. J. Reprod. Infant. Psychol. 2009, 27, 374–389. [Google Scholar] [CrossRef]
- Glover, V.; O’Donnell, K.J.; O’Connor, T.G.; Fisher, J. Prenatal maternal stress, fetal programming, and mechanisms underlying later psychopathology–A global perspective. Dev. Psychopathol. 2018, 30, 843–854. [Google Scholar] [CrossRef]
- Almeida, M.; Shrestha, A.D.; Stojanac, D.; Miller, L.J. The impact of the COVID-19 pandemic on women’s mental health. Arch. Womens Ment. Health 2020, 23, 741–748. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.S.; Schaefer, C.A.; Wyatt, R.J.; Goetz, R.; Begg, M.D.; Gorman, J.M.; Susser, E.S. Maternal exposure to respiratory infections and adult schizophrenia spectrum disorders: A prospective birth cohort study. Schizophr. Bull. 2000, 26, 287–295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chmielewska, B.; Barratt, I.; Townsend, R.; Kalafat, E.; van der Meulen, J.; Gurol-Urganci, I.; O’Brien, P.; Morris, E.; Draycott, T.; Thangaratinam, S.; et al. Effects of the COVID-19 pandemic on maternal and perinatal outcomes: A systematic review and meta-analysis. Lancet Glob. Health 2021, 9, e759–e772. [Google Scholar] [CrossRef]
- Hornig, M.; Bresnahan, M.A.; Che, X.; Schultz, A.F.; Ukaigwe, J.E.; Eddy, M.L.; Hirtz, D.; Gunnes, N.; Lie, K.K.; Magnus, P.; et al. Prenatal fever and autism risk. Mol. Psychiatry 2018, 23, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Lambregtse-van den Berg, M.; Quinlivan, J. Identifying pregnant women at risk of developing COVID-19 related mental health problems—A call for enhanced psychoeducation and social support. J. Psychosom. Obstet. Gynaecol. 2020, 41, 249–250. [Google Scholar] [CrossRef] [PubMed]
- Lebel, C.; Walton, M.; Letourneau, N.; Giesbrecht, G.F.; Kaplan, B.J.; Dewey, D. Prepartum and Postpartum Maternal Depressive Symptoms Are Related to Children’s Brain Structure in Preschool. Biol. Psychiatry 2016, 80, 859–868. [Google Scholar] [CrossRef]
- Mednick, S.A.; Machon, R.A.; Huttunen, M.O.; Bonett, D. Adult schizophrenia following prenatal exposure to an influenza epidemic. Arch. Gen. Psychiatry 1988, 45, 189–192. [Google Scholar] [CrossRef]
- Norris, F.H.; Friedman, M.J.; Watson, P.J.; Byrne, C.M.; Diaz, E.; Kaniasty, K. 60,000 disaster victims speak: Part I. An empirical review of the empirical literature, 1981-2001. Psychiatry 2002, 65, 207–239. [Google Scholar] [CrossRef] [Green Version]
- O’Callaghan, E.; Sham, P.; Takei, N.; Glover, G.; Murray, R.M. Schizophrenia after prenatal exposure to 1957 A2 influenza epidemic. Lancet 1991, 337, 1248–1250. [Google Scholar] [CrossRef]
- Rasmussen, S.A.; Jamieson, D.J. Pregnancy, Postpartum Care, and COVID-19 Vaccination in 2021. JAMA 2021, 325, 1099–1100. [Google Scholar] [CrossRef] [PubMed]
- Trocado, V.; Silvestre-Machado, J.; Azevedo, L.; Miranda, A.; Nogueira-Silva, C. Pregnancy and COVID-19: A systematic review of maternal, obstetric and neonatal outcomes. J. Matern. Fetal-Neonatal. Med. 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, L.; Wu, T.; Shi, H.; Li, Q.; Jiang, H.; Zheng, D.; Wang, X.; Wei, Y.; Zhao, Y.; et al. Impact of COVID-19 in pregnancy on mother’s psychological status and infant’s neurobehavioral development: A longitudinal cohort study in China. BMC Med. 2020, 18, 347. [Google Scholar] [CrossRef] [PubMed]
- Abdoli, A.; Falahi, S.; Kenarkoohi, A.; Shams, M.; Mir, H.; Jahromi, M.A.M. The COVID-19 pandemic, psychological stress during pregnancy, and risk of neurodevelopmental disorders in offspring: A neglected consequence. J. Psychosom. Obstet. Gynaecol. 2020, 41, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Hsiao, E.Y.; Patterson, P.H. Activation of the maternal immune system induces endocrine changes in the placenta via IL-6. Brain Behav. Immun. 2011, 25, 604–615. [Google Scholar] [CrossRef] [Green Version]
- Lebel, C.; MacKinnon, A.; Bagshawe, M.; Tomfohr-Madsen, L.; Giesbrecht, G. Elevated depression and anxiety symptoms among pregnant individuals during the COVID-19 pandemic. J. Affect. Disord. 2020, 277, 5–13. [Google Scholar] [CrossRef]
- Smith, S.E.; Li, J.; Garbett, K.; Mirnics, K.; Patterson, P.H. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 2007, 27, 10695–10702. [Google Scholar] [CrossRef] [Green Version]
- Tomfohr-Madsen, L.M.; Racine, N.; Giesbrecht, G.F.; Lebel, C.; Madigan, S. Depression and anxiety in pregnancy during COVID-19: A rapid review and meta-analysis. Psychiatry Res. 2021, 300, 113912. [Google Scholar] [CrossRef]
- Werenberg Dreier, J.; Nybo Andersen, A.M.; Hvolby, A.; Garne, E.; Kragh Andersen, P.; Berg-Beckhoff, G. Fever and infections in pregnancy and risk of attention deficit/hyperactivity disorder in the offspring. J. Child Psychol. Psychiatry 2016, 57, 540–548. [Google Scholar] [CrossRef]
- Zawawi, A.; Naser, A.Y.; Alwafi, H.; Minshawi, F. Profile of Circulatory Cytokines and Chemokines in Human Coronaviruses: A Systematic Review and Meta-Analysis. Front. Immunol. 2021, 12, 666223. [Google Scholar] [CrossRef]
- Brooks, S.K.; Webster, R.K.; Smith, L.E.; Woodland, L.; Wessely, S.; Greenberg, N.; Rubin, G.J. The psychological impact of quarantine and how to reduce it: Rapid review of the evidence. Lancet 2020, 395, 912–920. [Google Scholar] [CrossRef] [Green Version]
- Brooks, S.K.; Weston, D.; Greenberg, N. Psychological impact of infectious disease outbreaks on pregnant women: Rapid evidence review. Public Health 2020, 189, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Coussons-Read, M.E. Effects of prenatal stress on pregnancy and human development: Mechanisms and pathways. Obstet. Med. 2013, 6, 52–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wei, S.Q.; Bilodeau-Bertrand, M.; Liu, S.; Auger, N. The impact of COVID-19 on pregnancy outcomes: A systematic review and meta-analysis. CMAJ 2021, 193, E540–E548. [Google Scholar] [CrossRef]
- Villar, J.; Ariff, S.; Gunier, R.B.; Thiruvengadam, R.; Rauch, S.; Kholin, A.; Roggero, P.; Prefumo, F.; do Vale, M.S.; Cardona-Perez, J.A.; et al. Maternal and Neonatal Morbidity and Mortality Among Pregnant Women With and Without COVID-19 Infection: The INTERCOVID Multinational Cohort Study. JAMA Pediatr. 2021, 175, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Mullins, E.; Hudak, M.L.; Banerjee, J.; Getzlaff, T.; Townson, J.; Barnette, K.; Playle, R.; Perry, A.; Bourne, T.; Lees, C.C.; et al. Pregnancy and neonatal outcomes of COVID-19: Coreporting of common outcomes from PAN-COVID and AAP-SONPM registries. Ultrasound Obstet. Gynecol. 2021, 57, 573–581. [Google Scholar] [CrossRef]
- Goldstein, J.M.; Cohen, J.E.; Mareckova, K.; Holsen, L.; Whitfield-Gabrieli, S.; Gilman, S.E.; Buka, S.L.; Hornig, M. Impact of prenatal maternal cytokine exposure on sex differences in brain circuitry regulating stress in offspring 45 years later. Proc. Natl. Acad. Sci. USA 2021, 118, 15. [Google Scholar] [CrossRef]
- Giesbrecht, G.F.; Rojas, L.; Patel, S.; Kuret, V.; MacKinnon, A.L.; Tomfohr-Madsen, L.; Lebel, C. Fear of COVID-19, mental health, and pregnancy outcomes in the pregnancy during the COVID-19 pandemic study: Fear of COVID-19 and pregnancy outcomes. J. Affect. Disord. 2022, 299, 483–491. [Google Scholar] [CrossRef]
- Ji, C.; Li, Y.; Cui, L.; Cai, J.; Shi, J.; Cheng, F.W.; Li, Y.; Curhan, G.C.; Wu, S.; Gao, X. Prenatal Earthquake Exposure and Midlife Uric Acid Levels Among Chinese Adults. Arthritis Care Res. 2017, 69, 703–708. [Google Scholar] [CrossRef] [Green Version]
- Provenzi, L.; Mambretti, F.; Villa, M.; Grumi, S.; Citterio, A.; Bertazzoli, E.; Biasucci, G.; Decembrino, L.; Falcone, R.; Gardella, B.; et al. Hidden pandemic: COVID-19-related stress, SLC6A4 methylation, and infants’ temperament at 3 months. Sci. Rep. 2021, 11, 15658. [Google Scholar] [CrossRef]
- Cacioppo, J.T.; Cacioppo, S.; Capitanio, J.P.; Cole, S.W. The neuroendocrinology of social isolation. Annu. Rev. Psychol. 2015, 66, 733–767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, J.; Huang, Y.; Liu, J.; Zheng, Z.; Xu, X.; Zhou, Y.; Wang, J. COVID-19 Related Stress and Mental Health Outcomes 1 Year After the Peak of the Pandemic Outbreak in China: The Mediating Effect of Resilience and Social Support. Front. Psychiatry 2022, 13, 828379. [Google Scholar] [CrossRef] [PubMed]
- Ye, Z.; Yang, X.; Zeng, C.; Wang, Y.; Shen, Z.; Li, X.; Lin, D. Resilience, Social Support, and Coping as Mediators between COVID-19-related Stressful Experiences and Acute Stress Disorder among College Students in China. Appl. Psychol. Health Well-Being 2020, 12, 1074–1094. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Dong, G.; Meng, W.; Chen, Z.; Cao, Y.; Zhang, M. Perceived Stress and Psychological Impact Among Healthcare Workers at a Tertiaty Hospital in China During the COVID-19 Outbreak: The Moderating Role of Resilience and Social Support. Front. Psychiatry 2021, 12, 570971. [Google Scholar] [CrossRef] [PubMed]
- Strelkovskii, N.; Rovenskaya, E.; Ilmola-Sheppard, L.; Bartmann, R.; Rein-Sapir, Y.; Feitelson, E. Implications of COVID-19 Mitigation Policies for National Well-Being: A Systems Perspective. Sustainability 2022, 14, 433. [Google Scholar] [CrossRef]
- Mazza, M.; Marano, G.; Lai, C.; Janiri, L.; Sani, G. Danger in danger: Interpersonal violence during COVID-19 quarantine. Psychiatry Res. 2020, 289, 113046. [Google Scholar] [CrossRef]
- Donovan, B.M.; Spracklen, C.N.; Schweizer, M.L.; Ryckman, K.K.; Saftlas, A.F. Intimate partner violence during pregnancy and the risk for adverse infant outcomes: A systematic review and meta-analysis. BJOG 2016, 123, 1289–1299. [Google Scholar] [CrossRef]
- Richter, J.; Bittner, A.; Petrowski, K.; Junge-Hoffmeister, J.; Bergmann, S.; Joraschky, P.; Weidner, K. Effects of an early intervention on perceived stress and diurnal cortisol in pregnant women with elevated stress, anxiety, and depressive symptomatology. J. Psychosom. Obstet. Gynaecol. 2012, 33, 162–170. [Google Scholar] [CrossRef]
- Zarenejad, M.; Yazdkhasti, M.; Rahimzadeh, M.; Mehdizadeh Tourzani, Z.; Esmaelzadeh-Saeieh, S. The effect of mindfulness-based stress reduction on maternal anxiety and self-efficacy: A randomized controlled trial. Brain Behav. 2020, 10, e01561. [Google Scholar] [CrossRef] [Green Version]
- Thomas, M.; Vieten, C.; Adler, N.; Ammondson, I.; Coleman-Phox, K.; Epel, E.; Laraia, B. Potential for a stress reduction intervention to promote healthy gestational weight gain: Focus groups with low-income pregnant women. Womens Health Issues 2014, 24, e305–e311. [Google Scholar] [CrossRef] [Green Version]
- Bleker, L.S.; Milgrom, J.; Parker, D.; Gemmill, A.W.; Holt, C.J.; Connelly, A.; Burger, H.; Roseboom, T.J.; de Rooij, S.R. Brain Magnetic Resonance Imaging Findings in Children after Antenatal Maternal Depression Treatment, a Longitudinal Study Built on a Pilot Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2019, 16, 1816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van den Heuvel, M.I.; Donkers, F.C.; Winkler, I.; Otte, R.A.; Van den Bergh, B.R. Maternal mindfulness and anxiety during pregnancy affect infants’ neural responses to sounds. Soc. Cogn. Affect. Neurosci. 2015, 10, 453–460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boserup, B.; McKenney, M.; Elkbuli, A. Alarming trends in US domestic violence during the COVID-19 pandemic. Am. J. Emerg. Med. 2020, 38, 2753–2755. [Google Scholar] [CrossRef] [PubMed]
- Kwong, A.S.F.; Pearson, R.M.; Adams, M.J.; Northstone, K.; Tilling, K.; Smith, D.; Fawns-Ritchie, C.; Bould, H.; Warne, N.; Zammit, S.; et al. Mental health before and during the COVID-19 pandemic in two longitudinal UK population cohorts. Br. J. Psychiatry 2021, 218, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Roberton, T.; Carter, E.D.; Chou, V.B.; Stegmuller, A.R.; Jackson, B.D.; Tam, Y.; Sawadogo-Lewis, T.; Walker, N. Early estimates of the indirect effects of the COVID-19 pandemic on maternal and child mortality in low-income and middle-income countries: A modelling study. Lancet Glob. Health 2020, 8, e901–e908. [Google Scholar] [CrossRef]
- Abrams, E.M.; Szefler, S.J. COVID-19 and the impact of social determinants of health. Lancet Respir. Med. 2020, 8, 659–661. [Google Scholar] [CrossRef]
- Bann, D.; Villadsen, A.; Maddock, J.; Hughes, A.; Ploubidis, G.B.; Silverwood, R.; Patalay, P. Changes in the behavioural determinants of health during the COVID-19 pandemic: Gender, socioeconomic and ethnic inequalities in five British cohort studies. J. Epidemiol. Community Health 2021, 75, 1136–1142. [Google Scholar] [CrossRef]
- Politi, J.; Martin-Sanchez, M.; Mercuriali, L.; Borras-Bermejo, B.; Lopez-Contreras, J.; Vilella, A.; Villar, J.; COVID-19 Surveillance Working Group of Barcelona; Orcau, A.; de Olalla, P.G.; et al. Epidemiological characteristics and outcomes of COVID-19 cases: Mortality inequalities by socio-economic status, Barcelona, Spain, 24 February to 4 May 2020. Eurosurveill 2021, 26, 2001138. [Google Scholar] [CrossRef]
- Sweeney, S.; Capeding, T.P.J.; Eggo, R.; Huda, M.; Jit, M.; Mudzengi, D.; Naylor, N.R.; Procter, S.; Quaife, M.; Serebryakova, L.; et al. Exploring equity in health and poverty impacts of control measures for SARS-CoV-2 in six countries. BMJ Glob. Health 2021, 6, e005521. [Google Scholar] [CrossRef]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef]
- Jacob, C.M.; Briana, D.D.; Di Renzo, G.C.; Modi, N.; Bustreo, F.; Conti, G.; Malamitsi-Puchner, A.; Hanson, M. Building resilient societies after COVID-19: The case for investing in maternal, neonatal, and child health. Lancet Public Health 2020, 5, e624–e627. [Google Scholar] [CrossRef]
| Type of Disaster | Subtypes | Examples |
|---|---|---|
| Natural | Biological | Viral pan-/epidemic |
| Geological | Earthquake, vulcanic eruptions, tsunamis | |
| Hydrometerological | Floods versus droughts, storms | |
| Manmade | Societal, hazardous, transportation, environmental | Terrorism, war, wildfires, Chernobyl meltdown, dam failure, ship wreckage, oil spillage |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schoenmakers, S.; Verweij, E.J.; Beijers, R.; Bijma, H.H.; Been, J.V.; Steegers-Theunissen, R.P.M.; Koopmans, M.P.G.; Reiss, I.K.M.; Steegers, E.A.P. The Impact of Maternal Prenatal Stress Related to the COVID-19 Pandemic during the First 1000 Days: A Historical Perspective. Int. J. Environ. Res. Public Health 2022, 19, 4710. https://doi.org/10.3390/ijerph19084710
Schoenmakers S, Verweij EJ, Beijers R, Bijma HH, Been JV, Steegers-Theunissen RPM, Koopmans MPG, Reiss IKM, Steegers EAP. The Impact of Maternal Prenatal Stress Related to the COVID-19 Pandemic during the First 1000 Days: A Historical Perspective. International Journal of Environmental Research and Public Health. 2022; 19(8):4710. https://doi.org/10.3390/ijerph19084710
Chicago/Turabian StyleSchoenmakers, Sam, E. J. (Joanne) Verweij, Roseriet Beijers, Hilmar H. Bijma, Jasper V. Been, Régine P. M. Steegers-Theunissen, Marion P. G. Koopmans, Irwin K. M. Reiss, and Eric A. P. Steegers. 2022. "The Impact of Maternal Prenatal Stress Related to the COVID-19 Pandemic during the First 1000 Days: A Historical Perspective" International Journal of Environmental Research and Public Health 19, no. 8: 4710. https://doi.org/10.3390/ijerph19084710
APA StyleSchoenmakers, S., Verweij, E. J., Beijers, R., Bijma, H. H., Been, J. V., Steegers-Theunissen, R. P. M., Koopmans, M. P. G., Reiss, I. K. M., & Steegers, E. A. P. (2022). The Impact of Maternal Prenatal Stress Related to the COVID-19 Pandemic during the First 1000 Days: A Historical Perspective. International Journal of Environmental Research and Public Health, 19(8), 4710. https://doi.org/10.3390/ijerph19084710






