Bioactive Compounds for Fibromyalgia-like Symptoms: A Narrative Review and Future Perspectives
Abstract
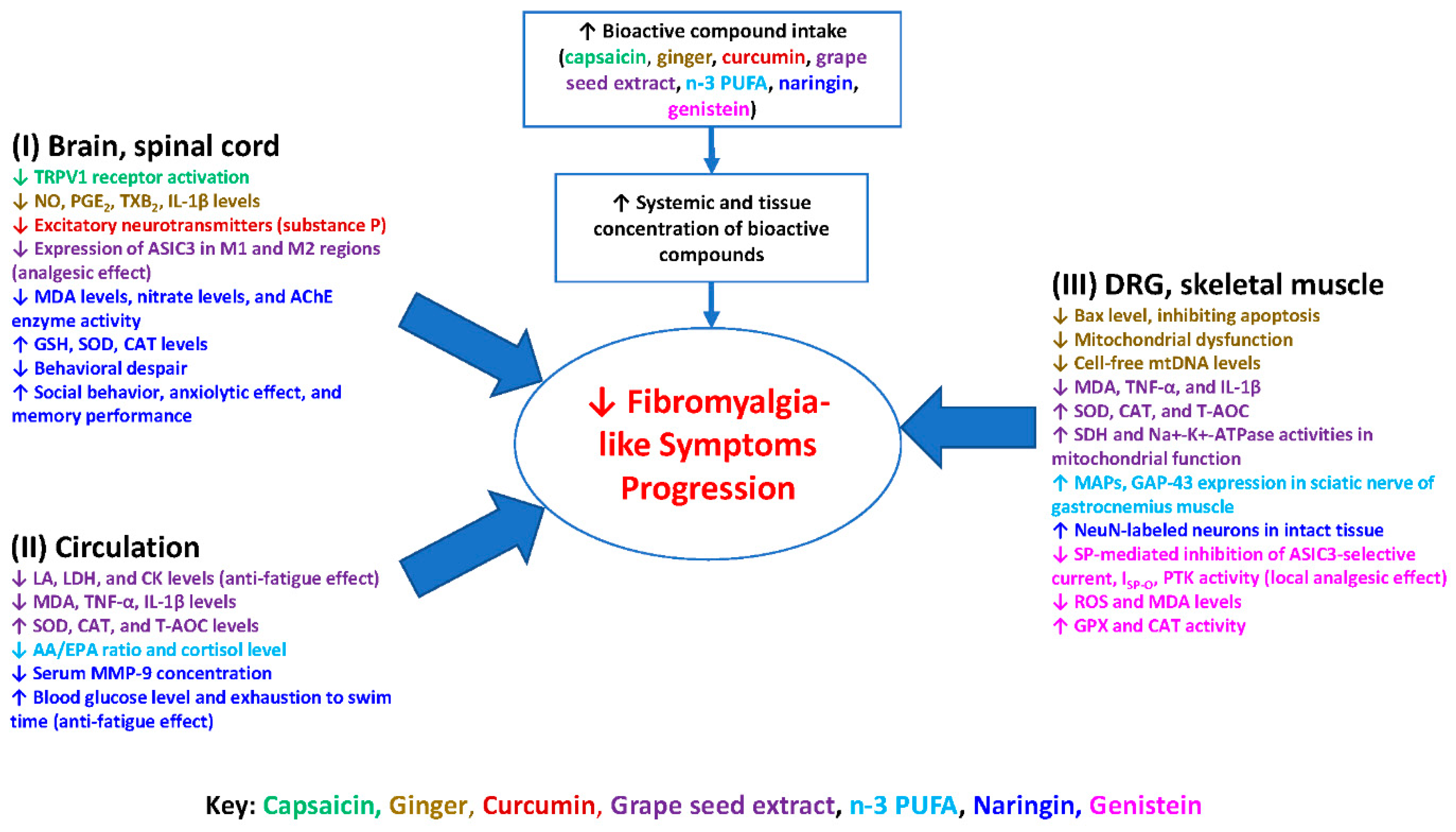
1. Introduction
2. Capsaicin
| First Author, Year [ref] | Experimental Design and Treatments | Results |
|---|---|---|
| In vitro studies | ||
| Shang, 2017 [24] | Myoblast cells pre-exposed to capsaicin (50, 100 μM) overnight and then treated with LPS (100 μg/mL) | ↓ LPS-induced inflammation |
| Vos, 2006 [26] | HEK293 cells transiently transfected with full--length TRPV1 and activated by capsaicin (1 µm) | ↓ TRPV1 channel function in a dose-dependent manner |
| Animal studies | ||
| Scheich, 2017 [28] | Chronic restraint stress-induced FM model | Compared to control group, RTX group: |
| CD1 male mice (12-week-old, n = 9–11/group) | ↓ mechanical hyperalgesia | |
| ↑ basal noxious heat threshold | ||
| Treatments: Capsaicin analogue RTX (10, 20, 70, 100 μg/kg) s.c. once daily for four consecutive days | ↔ anxiety, depression, or peripheral inflammatory changes | |
| Human studies | ||
| McCarty, 1994 [31] | Randomized double-blind, vehicle-controlled trial with primary | Compared to control group, capsaicin group: |
| FM patients (n = 45, 44 women, 1 man) | ↑ grip strength at week 2 ↓ tenderness in tender points at week 4 | |
| Treatments: capsaicin (0.025%) cream applied as thin layer to tender points on left or right side of upper body as directed by the tube label for 4 weeks | ↔ Visual Analog Scale of pain scores | |
| ↑ minor side effects including transient burning at application site | ||
| Casanueva, 2013 [32] | Randomized controlled trial with FM patients | Compared to control group, capsaicin group: |
| ↑ desensitization to pain with repeated capsaicin application | ||
| (n = 130, 126 women, 4 men) | ↓ Fibromyalgia Impact Questionnaire | |
| ↓ Fatigue Severity Scale scores | ||
| Treatments: capsaicin (0.075%) cream applied topically over 18 tender points 3x/day for 6 weeks | ↓ myalgia score | |
| ↓ pressure pain threshold | ||
| ↓ role limitations due to emotional problems | ||
| ↓ Visual Analogue Scale of depression | ||
| Chrubasik, 2010 [33] | Randomized double-blind, placebo-controlled trial with chronic soft tissue/back pain patients (n = 281, 174 women, 107 men) Treatments: capsaicin (0.05%) cream applied as a thin layer to painful area 3x/day for 3 weeks | Compared to control group, capsaicin group: |
| ↓ pain sum scores | ||
| ↑ minor side effects including local warmth and pruritis |
3. Ginger
4. Curcumin
5. Grape Seed Extract
6. N-3 Polyunsaturated Fatty Acids
7. Naringin
8. Genistein
9. Possible Molecular Mechanisms
10. Summary and Future Direction
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Clauw, D.J.; Arnold, L.M.; McCarberg, B.H.; FibroCollaborative. The science of fibromyalgia. Mayo Clin. Proc. 2011, 86, 907–911. [Google Scholar] [CrossRef]
- Goldenberg, D.L. Diagnosis and differential diagnosis of fibromyalgia. Am. J. Med. 2009, 122, S14–S21. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, R.C.; Felson, D.T.; Helmick, C.G.; Arnold, L.M.; Choi, H.; Deyo, R.A.; Gabriel, S.; Hirsch, R.; Hochberg, M.C.; Hunder, G.G.; et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008, 58, 26–35. [Google Scholar] [CrossRef]
- Wolfe, F.; Walitt, B.; Perrot, S.; Rasker, J.J.; Hauser, W. Fibromyalgia diagnosis and biased assessment: Sex, prevalence and bias. PLoS ONE 2018, 13, e0203755. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, F.; Ross, K.; Anderson, J.; Russell, I.J.; Hebert, L. The prevalence and characteristics of fibromyalgia in the general population. Arthritis Rheum. 1995, 38, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Prados, G.; Miro, E.; Martinez, M.P.; Sanchez, A.I.; Lopez, S.; Saez, G. Fibromyalgia: Gender differences and sleep-disordered breathing. Clin. Exp. Rheumatol. 2013, 31, S102–S110. [Google Scholar] [PubMed]
- Mease, P. Fibromyalgia syndrome: Review of clinical presentation, pathogenesis, outcome measures, and treatment. J. Rheumatol. Suppl. 2005, 75, 6–21. [Google Scholar]
- Talotta, R.; Bazzichi, L.; Di Franco, M.; Casale, R.; Batticciotto, A.; Gerardi, M.C.; Sarzi-Puttini, P. One year in review 2017: Fibromyalgia. Clin. Exp. Rheumatol. 2017, 35 (Suppl. 105), 6–12. [Google Scholar] [PubMed]
- Schmidt-Wilcke, T.; Clauw, D.J. Fibromyalgia: From pathophysiology to therapy. Nat. Rev. Rheumatol. 2011, 7, 518–527. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; Diaz-Parrado, E.; Carrion, A.M.; Alfonsi, S.; Sanchez-Alcazar, J.A.; Bullon, P.; Battino, M.; de Miguel, M. Is inflammation a mitochondrial dysfunction-dependent event in fibromyalgia? Antioxid. Redox Signal. 2013, 18, 800–807. [Google Scholar] [CrossRef]
- Meeus, M.; Nijs, J.; Hermans, L.; Goubert, D.; Calders, P. The role of mitochondrial dysfunctions due to oxidative and nitrosative stress in the chronic pain or chronic fatigue syndromes and fibromyalgia patients: Peripheral and central mechanisms as therapeutic targets? Expert Opin. Ther. Targets 2013, 17, 1081–1089. [Google Scholar] [CrossRef]
- Wang, Z.Q.; Porreca, F.; Cuzzocrea, S.; Galen, K.; Lightfoot, R.; Masini, E.; Muscoli, C.; Mollace, V.; Ndengele, M.; Ischiropoulos, H.; et al. A newly identified role for superoxide in inflammatory pain. J. Pharmacol. Exp. Ther. 2004, 309, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Li, Q.; Zhang, M.-T.; Mao-Ying, Q.-L.; Hu, L.-Y.; Wu, G.-C.; Mi, W.-L.; Wang, Y.-Q. Curcumin ameliorates neuropathic pain by down-regulating spinal IL-1β via suppressing astroglial NALP1 inflammasome and JAK2-STAT3 signalling. Sci. Rep. 2016, 6, 28956. [Google Scholar] [CrossRef]
- Ji, G.; Li, Z.; Neugebauer, V. Reactive oxygen species mediate visceral pain-related amygdala plasticity and behaviors. Pain 2015, 156, 825–836. [Google Scholar] [CrossRef]
- Li, Z.; Ji, G.; Neugebauer, V. Mitochondrial reactive oxygen species are activated by mGluR5 through IP3 and activate ERK and PKA to increase excitability of amygdala neurons and pain behavior. J. Neurosci. 2011, 31, 1114–1127. [Google Scholar] [CrossRef] [PubMed]
- Ji, G.; Neugebauer, V. Reactive oxygen species are involved in group I mGluR-mediated facilitation of nociceptive processing in amygdala neurons. J. Neurophysiol. 2010, 104, 218–229. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Littlejohn, G. Neurogenic neuroinflammation in fibromyalgia and complex regional pain syndrome. Nat. Rev. Rheumatol. 2015, 11, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Romano, G.F.; Tomassi, S.; Russell, A.; Mondelli, V.; Pariante, C.M. Fibromyalgia and chronic fatigue: The underlying biology and related theoretical issues. Adv. Psychosom. Med. 2015, 34, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Cordero, M.D.; de Miguel, M.; Carmona-Lopez, I.; Bonal, P.; Campa, F.; Moreno-Fernandez, A.M. Oxidative stress and mitochondrial dysfunction in fibromyalgia. Neuro Endocrinol. Lett. 2010, 31, 169–173. [Google Scholar] [PubMed]
- Siracusa, R.; Paola, R.D.; Cuzzocrea, S.; Impellizzeri, D. Fibromyalgia: Pathogenesis, Mechanisms, Diagnosis and Treatment Options Update. Int. J. Mol. Sci. 2021, 22, 3891. [Google Scholar] [CrossRef]
- Lindman, R.; Hagberg, M.; Angqvist, K.A.; Soderlund, K.; Hultman, E.; Thornell, L.E. Changes in muscle morphology in chronic trapezius myalgia. Scand. J. Work. Environ. Health 1991, 17, 347–355. [Google Scholar] [CrossRef]
- Jegede, A.B.; Gilbert, C.; Tulkin, S.R. Muscle characteristics of persons with fibromyalgia syndrome. Neuro Rehabil. 2008, 23, 217–230. [Google Scholar] [CrossRef]
- Valera-Calero, J.A.; Ubeda-D’Ocasar, E.; Caballero-Corella, M.; Fernandez-de-Las-Penas, C.; Sendarrubias, G.M.G.; Arias-Buria, J.L. Cervical Multifidus Morphology and Quality is not Associated with Clinical Variables in Women with Fibromyalgia: An Observational Study. Pain Med. 2021, pnab297. [Google Scholar] [CrossRef]
- De Santana, J.M.; da Cruz, K.M.; Sluka, K.A. Animal models of fibromyalgia. Arthritis Res. Ther. 2013, 15, 222. [Google Scholar] [CrossRef] [PubMed]
- Clauw, D.J. Fibromyalgia: A clinical review. JAMA 2014, 311, 1547–1555. [Google Scholar] [CrossRef] [PubMed]
- Nagakura, Y. Challenges in drug discovery for overcoming ‘dysfunctional pain’: An emerging category of chronic pain. Expert Opin. Drug Discov. 2015, 10, 1043–1045. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bhargava, J.; Hurley, J.A. Fibromyalgia; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Krupkova, O.; Sekiguchi, M.; Klasen, J.; Hausmann, O.; Konno, S.; Ferguson, S.J.; Wuertz-Kozak, K. Epigallocatechin 3-gallate suppresses interleukin-1beta-induced inflammatory responses in intervertebral disc cells in vitro and reduces radiculopathic pain in rats. Eur. Cell Mater. 2014, 28, 372–386. [Google Scholar] [CrossRef] [PubMed]
- Fattori, V.; Hohmann, M.S.; Rossaneis, A.C.; Pinho-Ribeiro, F.A.; Verri, W.A. Capsaicin: Current Understanding of Its Mechanisms and Therapy of Pain and Other Pre-Clinical and Clinical Uses. Molecules 2016, 21, 844. [Google Scholar] [CrossRef] [PubMed]
- Shang, K.; Amna, T.; Amina, M.; Al-Musayeib, N.M.; Al-Deyab, S.S.; Hwang, I. Influence of Capsaicin on Inflammatory Cytokines Induced by Lipopolysaccharide in Myoblast Cells Under In vitro Environment. Pharmacogn. Mag. 2017, 13, S26–S32. [Google Scholar] [CrossRef]
- Chaudhary, A.; Gour, J.K.; Rizvi, S.I. Capsaicin has potent anti-oxidative effects in vivo through a mechanism which is non-receptor mediated. Arch. Physiol. Biochem. 2019, 128, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Vos, M.H.; Neelands, T.R.; McDonald, H.A.; Choi, W.; Kroeger, P.E.; Puttfarcken, P.S.; Faltynek, C.R.; Moreland, R.B.; Han, P. TRPV1b overexpression negatively regulates TRPV1 responsiveness to capsaicin, heat and low pH in HEK293 cells. J. Neurochem. 2006, 99, 1088–1102. [Google Scholar] [CrossRef] [PubMed]
- Ilie, M.A.; Caruntu, C.; Tampa, M.; Georgescu, S.R.; Matei, C.; Negrei, C.; Ion, R.M.; Constantin, C.; Neagu, M.; Boda, D. Capsaicin: Physicochemical properties, cutaneous reactions and potential applications in painful and inflammatory conditions. Exp. Ther. Med. 2019, 18, 916–925. [Google Scholar] [CrossRef]
- Scheich, B.; Vincze, P.; Szoke, E.; Borbely, E.; Hunyady, A.; Szolcsanyi, J.; Denes, A.; Kornyei, Z.; Gaszner, B.; Helyes, Z. Chronic stress-induced mechanical hyperalgesia is controlled by capsaicin-sensitive neurones in the mouse. Eur. J. Pain 2017, 21, 1417–1431. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Mendez, M.E.; Castro-Sanchez, L.A.; Dagnino-Acosta, A.; Aguilar-Martinez, I.; Perez-Burgos, A.; Vazquez-Jimenez, C.; Moreno-Galindo, E.G.; Alvarez-Cervera, F.J.; Gongora-Alfaro, J.L.; Navarro-Polanco, R.A.; et al. Capsaicin produces antidepressant-like effects in the forced swimming test and enhances the response of a sub-effective dose of amitriptyline in rats. Physiol. Behav. 2018, 195, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Ramamurthy, S.; Prihoda, T.J.; Eckmann, M.S. Effect of delayed intrathecal administration of capsaicin on neuropathic pain induced by chronic constriction injury of the sciatic nerve in rats. J. Pain Res. 2014, 7, 547–554. [Google Scholar] [CrossRef][Green Version]
- McCarty, D.J. Treatment of pain due to fibromyalgia with topical capsaicin: A pilot study. Semin. Arthritis Reum. 1994, 23, 41–47. [Google Scholar] [CrossRef]
- Casanueva, B.; Rodero, B.; Quintial, C.; Llorca, J.; Gonzalez-Gay, M.A. Short-term efficacy of topical capsaicin therapy in severely affected fibromyalgia patients. Rheumatol. Int. 2013, 33, 2665–2670. [Google Scholar] [CrossRef]
- Chrubasik, S.; Weiser, T.; Beime, B. Effectiveness and safety of topical capsaicin cream in the treatment of chronic soft tissue pain. Phytother. Res. 2010, 24, 1877–1885. [Google Scholar] [CrossRef]
- McCleane, G. Topical application of doxepin hydrochloride, capsaicin and a combination of both produces analgesia in chronic human neuropathic pain: A randomized, double-blind, placebo-controlled study. Br. J. Clin. Pharmacol. 2000, 49, 574–579. [Google Scholar] [CrossRef]
- Mao, Q.Q.; Xu, X.Y.; Cao, S.Y.; Gan, R.Y.; Corke, H.; Beta, T.; Li, H.B. Bioactive Compounds and Bioactivities of Ginger (Zingiber officinale Roscoe). Foods 2019, 8, 185. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.K.; Moon, E.; Ju, M.S.; Kim, D.H.; Ryu, J.H.; Oh, M.S.; Kim, S.Y. 6-Shogaol, a ginger product, modulates neuroinflammation: A new approach to neuroprotection. Neuropharmacology 2012, 63, 211–223. [Google Scholar] [CrossRef]
- Hosseinzadeh, A.; Bahrampour Juybari, K.; Fatemi, M.J.; Kamarul, T.; Bagheri, A.; Tekiyehmaroof, N.; Sharifi, A.M. Protective Effect of Ginger (Zingiber officinale Roscoe) Extract against Oxidative Stress and Mitochondrial Apoptosis Induced by Interleukin-1beta in Cultured Chondrocytes. Cells Tissues Organs 2017, 204, 241–250. [Google Scholar] [CrossRef]
- Montserrat-de la Paz, S.; Garcia-Gimenez, M.D.; Quilez, A.M.; De la Puerta, R.; Fernandez-Arche, A. Ginger rhizome enhances the anti-inflammatory and anti-nociceptive effects of paracetamol in an experimental mouse model of fibromyalgia. Inflammopharmacology 2018, 26, 1093–1101. [Google Scholar] [CrossRef]
- Fajrin, F.A.; Purwandhono, A.; Christianty, F.M.; Sulistyaningrum, G.D.; Afifah; Imandasari, N.; Barki, T. Antihyperalgesia potency of Zingiber officinale var. Rubrum in inflammatory and neuropathy-induced chronic pain condition in mice. Pak. J. Pharm. Sci. 2019, 32, 1663–1669. [Google Scholar]
- Abolaji, A.O.; Ojo, M.; Afolabi, T.T.; Arowoogun, M.D.; Nwawolor, D.; Farombi, E.O. Protective properties of 6-gingerol-rich fraction from Zingiber officinale (Ginger) on chlorpyrifos-induced oxidative damage and inflammation in the brain, ovary and uterus of rats. Chem. Biol. Interact. 2017, 270, 15–23. [Google Scholar] [CrossRef]
- Zhang, M.; Viennois, E.; Prasad, M.; Zhang, Y.; Wang, L.; Zhang, Z.; Han, M.K.; Xiao, B.; Xu, C.; Srinivasan, S.; et al. Edible ginger-derived nanoparticles: A novel therapeutic approach for the prevention and treatment of inflammatory bowel disease and colitis-associated cancer. Biomaterials 2016, 101, 321–340. [Google Scholar] [CrossRef] [PubMed]
- Mun, H.-I. Analgesic Effect of Grape Seed Proanthocyanidin Extract in Fibromyalgia Animal Model. J. Life Sci. 2010, 20, 496–502. [Google Scholar] [CrossRef][Green Version]
- Yang, M.; Wang, J.; Yang, C.; Han, H.; Rong, W.; Zhang, G. Oral administration of curcumin attenuates visceral hyperalgesia through inhibiting phosphorylation of TRPV1 in rat model of ulcerative colitis. Mol. Pain 2017, 13, 1744806917726416. [Google Scholar] [CrossRef]
- Zhi, L.; Dong, L.; Kong, D.; Sun, B.; Sun, Q.; Grundy, D.; Zhang, G.; Rong, W. Curcumin acts via transient receptor potential vanilloid-1 receptors to inhibit gut nociception and reverses visceral hyperalgesia. Neurogastroenterol. Motil. 2013, 25, e429–e440. [Google Scholar] [CrossRef]
- Fattori, V.; Pinho-Ribeiro, F.A.; Borghi, S.M.; Alves-Filho, J.C.; Cunha, T.M.; Cunha, F.Q.; Casagrande, R.; Verri, W.A., Jr. Curcumin inhibits superoxide anion-induced pain-like behavior and leukocyte recruitment by increasing Nrf2 expression and reducing NF-kappaB activation. Inflamm. Res. 2015, 64, 993–1003. [Google Scholar] [CrossRef] [PubMed]
- Arora, V.; Kuhad, A.; Tiwari, V.; Chopra, K. Curcumin ameliorates reserpine-induced pain-depression dyad: Behavioural, biochemical, neurochemical and molecular evidences. Psychoneuroendocrinology 2011, 36, 1570–1581. [Google Scholar] [CrossRef]
- Fujishita, K.; Ozawa, T.; Shibata, K.; Tanabe, S.; Sato, Y.; Hisamoto, M.; Okuda, T.; Koizumi, S. Grape seed extract acting on astrocytes reveals neuronal protection against oxidative stress via interleukin-6-mediated mechanisms. Cell Mol. Neurobiol. 2009, 29, 1121–1129. [Google Scholar] [CrossRef]
- Narita, K.; Hisamoto, M.; Okuda, T.; Takeda, S. Differential neuroprotective activity of two different grape seed extracts. PLoS ONE 2011, 6, e14575. [Google Scholar] [CrossRef] [PubMed]
- Xianchu, L.; Ming, L.; Xiangbin, L.; Lan, Z. Grape seed proanthocyanidin extract supplementation affects exhaustive exercise-induced fatigue in mice. Food Nutr. Res. 2018, 62. [Google Scholar] [CrossRef]
- Edwards, A.M. Food Supplements in the Treatment of Primary Fibromyalgia: A Double-blind, Crossover Trial of Anthocyanidins and Placebo. J. Nutr. Environ. Med. 2009, 10, 189–199. [Google Scholar] [CrossRef]
- Manzhulo, I.V.; Ogurtsova, O.S.; Lamash, N.E.; Latyshev, N.A.; Kasyanov, S.P.; Dyuizen, I.V. Analgetic effect of docosahexaenoic acid is mediated by modulating the microglia activity in the dorsal root ganglia in a rat model of neuropathic pain. Acta Histochem. 2015, 117, 659–666. [Google Scholar] [CrossRef]
- King, V.R. Omega-3 Fatty Acids Improve Recovery, whereas Omega-6 Fatty Acids Worsen Outcome, after Spinal Cord Injury in the Adult Rat. J. Neurosci. 2006, 26, 4672–4680. [Google Scholar] [CrossRef]
- Veigas, J.M.; Williams, P.J.; Halade, G.; Rahman, M.M.; Yoneda, T.; Fernandes, G. Fish oil concentrate delays sensitivity to thermal nociception in mice. Pharmacol. Res. 2011, 63, 377–382. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fontani, G. Administration of Omega-3 Fatty Acids Reduces Positive Tender Point Count in Chronic Musculoskeletal Pain Patients. J. Complementary Integr. Med. 2010, 7. [Google Scholar] [CrossRef]
- Silva, R.V.; Oliveira, J.T.; Santos, B.L.R.; Dias, F.C.; Martinez, A.M.B.; Lima, C.K.F.; Miranda, A.L.P. Long-Chain Omega-3 Fatty Acids Supplementation Accelerates Nerve Regeneration and Prevents Neuropathic Pain Behavior in Mice. Front. Pharmacol. 2017, 8, 723. [Google Scholar] [CrossRef] [PubMed]
- Ben-Azu, B.; Nwoke, E.E.; Aderibigbe, A.O.; Omogbiya, I.A.; Ajayi, A.M.; Olonode, E.T.; Umukoro, S.; Iwalewa, E.O. Possible neuroprotective mechanisms of action involved in the neurobehavioral property of naringin in mice. Biomed. Pharm. 2019, 109, 536–546. [Google Scholar] [CrossRef]
- Pinho-Ribeiro, F.A.; Zarpelon, A.C.; Fattori, V.; Manchope, M.F.; Mizokami, S.S.; Casagrande, R.; Verri, W.A., Jr. Naringenin reduces inflammatory pain in mice. Neuropharmacology 2016, 105, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Huang, Z.; Chen, X.; Jia, G.; Zhao, H.; Liu, G. Naringin induces skeletal muscle fiber type transformation via AMPK/PGC-1alpha signaling pathway in mice and C2C12 myotubes. Nutr. Res. 2021, 92, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Zamanian, M.; Hajizadeh, M.; Shamsizadeh, A.; Moemenzadeh, M.; Amirteimouri, M.; Elshiekh, M.; Allahtavakoli, M. Effects of naringin on physical fatigue and serum MMP-9 concentration in female rats. Pharm. Biol. 2017, 55, 423–427. [Google Scholar] [CrossRef]
- Xue, N.; Wu, X.; Wu, L.; Li, L.; Wang, F. Antinociceptive and anti-inflammatory effect of Naringenin in different nociceptive and inflammatory mice models. Life Sci. 2019, 217, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.C.; Chen, W.N.; Chen, C.J.; Lin, Y.W.; Zimmer, A.; Chen, C.C. An antinociceptive role for substance P in acid-induced chronic muscle pain. Proc. Natl. Acad. Sci. USA 2012, 109, E76–E83. [Google Scholar] [CrossRef] [PubMed]
- Jie, H.F.; Yang, G.J.; Bi, R.Y.; Mo, S.Y.; Gan, Y.H.; Xie, Q.F. Genistein Antagonizes 17beta-Estradiol Effects on Glutamate-Evoked Masseter Muscle Hypernociception in Rats. Front. Neurol. 2018, 9, 649. [Google Scholar] [CrossRef] [PubMed]
- Fischer, S.; Doerr, J.M.; Strahler, J.; Mewes, R.; Thieme, K.; Nater, U.M. Stress exacerbates pain in the everyday lives of women with fibromyalgia syndrome—The role of cortisol and alpha-amylase. Psychoneuroendocrinology 2016, 63, 68–77. [Google Scholar] [CrossRef]
| First Author, Year [ref] | Experimental Design and Treatments | Results |
|---|---|---|
| In vitro study | ||
| Ha, 2012 [36] | BV-2 cells were cultured and activated with LPS (1 mg/mL) for 12 h. LPS was removed from the cells and indicated 6-shogaol (1, 5, 10 μM) was added. NMMA, an inhibitor of iNOS enzyme activity, was used as a positive control | Compared to control group, 6-shogaol groups: |
| ↓ iNOS expression and release of NO | ||
| ↓ microglial activation | ||
| Hosseinzadeh, 2017 [37] | C28I2 human chondrocytes pretreated with GE (5, 25 μg/mL) for 24 h, followed by incubation with IL-1β (10 ng/mL) for 24 h | Compared to vehicle group, GE groups: |
| ↑ anti-oxidant enzyme gene expression | ||
| ↓ reactive oxygen species | ||
| ↓ lipid peroxidation | ||
| ↓ Bax/Bcl ratio, inhibiting apoptosis | ||
| ↓ caspase-3 activity | ||
| Animal studies | ||
| Montserrat-de la Paz, 2018 [38] | Intermittent cold stress (ICS)-induced FM model | Compared to control group, GR groups: |
| Female C57BL/6 J mice (n = 48, 5-week-old) | ↓ mechanical and thermal allodynia | |
| ↓ mechanical hyperalgesia | ||
| Treatments: healthy control, ICS group, ICS + APAP (40 mg/kg/day), ICS + GR (0.5%, 1%), and ICS + GR (0.5%) + APAP (40 mg/kg/day) supplemented with standard diet for 8 weeks | ↑ improved behavioral changes related to cognitive disturbances associated with pain, anxiety, and depression ↓ proinflammatory mediators such as NO, PGE2, TXB2, IL-1β in LPS-stimulated macrophages | |
| ↑ synergism with APAP leading to further reduction in FM-like symptoms | ||
| Fajrin, 2019 [39] | Complete Freud’s Adjuvant (CFA)-induced FM-like model | Compared to control group, RGO groups: |
| Male mice (n = 48, 8-week-old) | ↓ hyperalgesia in a dose-dependent manner | |
| Treatments: sham, negative control, RGO doses at 100, 200, 400 and 600 mg/kg orally 1x/day for 7 days | ↑ thermal response threshold | |
| Abolaji, 2017 [40] | Chlorpyrifos (CPF)-induced FM-like model | Compared to the control group, 6-GRF groups: |
| Female Wistar rats (n = 70, 100–125 g) | ↓ H2O2 and MDA levels | |
| ↑ catalase, SOD, GPX activity | ||
| Treatments: control with corn oil only (2 mL/kg BW), 6-GRF (100 mg/kg BW), CPF dissolved in corn oil (5 mg/kg BW), CPF (5 mg/kg BW) and 6-GRF (50 mg/kg BW) concomitantly, CPF (5 mg/kg BW) and 6-GRF (100 mg/kg BW) concomitantly 1x/day for 35 days after CPF toxicity | ↓ NO, MPO, TNF-α | |
| ↓ caspase-3 | ||
| Zhang, 2016 [41] | DSS-induced chronic colitis model | Compared to control group, GDNP group: |
| Female FVB/NJ mice (n = 7, 6–8-week-old) | ↑ healing in wounded intestinal epithelial monolayers | |
| ↑ proliferation of IECs | ||
| Groups including control with normal feeding and GDNPs 2 (300 μL of 1 mg/mL solution) by oral gavage 1x/day for 18 weeks | ↓ TNF-α, IL-6, IL-1β, MPO | |
| ↑ IL-10, IL-22 |
| First Author, Year [ref] | Experimental Design and Treatments | Results |
|---|---|---|
| Animal studies | ||
| Yang, 2017 [43] | Dextran sodium sulfate (DSS)-induced FM-like model | Compared to naive control group, curcumin group: |
| Male SD rats (n = 57, 190–210 g) | ↓ visceral hyperalgesia in dose-dependent manner (no significant effect with lower curcumin dose) | |
| Treatments: naive control, DSS + saline, and DSS + curcumin (20, 60 mg/kg) by oral gavage 1x/day for 10 days, beginning 3 days after initiation of DSS | ↓ TRPV1 expression in DRG neurons | |
| ↓ TRPV1 protein level in inflamed colon | ||
| Zhi, 2013 [44] | Colorectal distension (CRD)-induced FM-like (VMR) model | Compared to vehicle group, curcumin group: |
| ↓ CRD-induced VMRs indicating a decrease in GI nociception | ||
| Adult male SD rats (n = 4, 250–300 g) | ↓ visceral hyperalgesia | |
| Treatments: control, vehicle, and curcumin (4 mg/kg⋅min) infusion for 3 min | ↓ TRPV1 activation in primary afferent neurons in concentration-dependent manner | |
| Jejunal afferent firing in ex vivo jejunum preparations model | Compared to control group, curcumin groups: | |
| Adult male Kunming mice (n = 25, 20–30 g) | ↓ TNBS-induced hypersensitivity of jejunal afferents | |
| Treatments: control, curcumin at 1, 3, 10, and 30 μmol/L given extra- and intra-luminally in naïve and TNBS-treated mouse jejunum | ↓ capsaicin-induced rise in intracellular calcium and inward currents in mouse or rat DRG neurons | |
| Fattori, 2015 [45] | KO2-induced superoxide anion-induced FM-like model | Compared to control group, curcumin groups: |
| ↓ mechanical and thermal hyperalgesia | ||
| Male Swiss mice (25–30 g) | ↓ superoxide anion-induced leukocyte recruitment in peritoneal cavity | |
| ↓ MPO activity, oxidative stress, IL-1β and TNF-α production in paw skin | ||
| Treatments: vehicle (2% DMSO in saline) i.pl., KO2 i.pl + saline s.c., KO2 i.pl. + curcumin (3, 10, or 30 mg/kg) s.c. 1 h before KO2 stimulus | ↓ NF-κB activation in paw skin | |
| ↑ IL-10 production, and HO-1 and Nrf2 mRNA expression in paw skin |
| First Author, Year [ref] | Experimental Design and Treatments | Results |
|---|---|---|
| In vitro studies | ||
| Fujishita, 2009 [47] | Oxidative stress-induced hippocampal neuronal cell death model | Compared to vehicle group, the GSE groups: |
| Neurons treated with GSE (0–100 µg/mL) for 2, 6 and 12 h. Neurons pre-incubated for 24 h with GSE-treated ACM in presence or absence of anti-IL-6 antibodies (50 ng/mL). Then, neurons were stimulated with H2O2 in presence or absence of IL-6 with and without anti-IL-6 antibody for 2 h | ↑ mRNA expression of IL-6, COX-2, IL-1α, | |
| in astrocytes with oxidative stress status | ||
| Protected against neuronal death induced by | ||
| oxidative stress | ||
| Narita, 2011 [48] | Hippocampal neurons at 8 DIV treated with 50 µM glutamate for 30 min, in presence or absence of 0.01, 0.1, 1.0, or 10 ng/mL of GSE | Compared to vehicle group, the GSE groups: |
| Protected Erk1/2 phosphorylation | ||
| Protected dendritic arborization and augmented cell survival | ||
| ↓ caspase-3 activity | ||
| Animal studies | ||
| Mun, 2010 [42] | i.p. injection in gastrocnemius muscle of acidic saline-induced FM-like model | Compared to control group, proanthrocyanidin group: |
| (n = 15) | ||
| Female male SD rats (n = 15, 250–320 g) | ↑ anti-hyperalgesic effect in injected paw and contralateral paw | |
| Treatments: control, acidic saline i.p. injection, oligomeric proanthrocyanidin complexes at 300 mg/kg injection i.p. on day 7 | ↓ expression of acid sensing ion channel 3 in brain M1 and motor cortex area | |
| Xianchu, 2018 [49] | Exhaustive exercise-induced FM model | Compared to control group, GSPE-M and GSPE-H groups, |
| ↓ fatigue by prolonging the time to exhaustion in forced swimming test | ||
| ↓ lactic acid, LDH, and CK in serum | ||
| Male ICR mice (n = 32, 8-week-old, 25 g) | ↓ MDA, TNF-α, and IL-1β in serum and skeletal | |
| muscle of mice | ||
| ↑ SOD, CAT, and T-AOC in serum and skeletal | ||
| Treatments: control, low-dose GSPE-L group (1 mg/kg/day), medium-dose GSPE-M group (50 mg/kg/day), and high-dose GSPE-H group (100 mg/kg/day) for 28 days | muscle of mice | |
| ↑ SDH and Na+-K+-ATPase activities in | ||
| mitochondrial function of skeletal muscle | ||
| Human study | ||
| Edwards, 2000 [50] | Double-blind, randomized, crossover trial with moderate to severe primary FM (n = 12) | Compared to the placebo group, anthocyanidins at 80 mg daily group: |
| Treatments: placebo, 40 mg, 80 mg, and 120 mg anthocyanidins daily. Each dose for 3 months | ↓ fatigue and sleep disturbance in 80 mg dose group |
| First Author, Year [ref] | Experiment Design and Treatment | Results |
|---|---|---|
| Animal studies | ||
| Veigas, 2011 [53] | Heat-induced FM-like model | Compared to SO group, CFO groups |
| C57BL/6J male mice (n = 60) | ↓ sensitivity to heat induced pain in plantar paw region | |
| Treatments: 4% regular fish oil diet (FO), 4% concentrated fish oil diet (CFO), 5% safflower oil diet (SO) ad libitum for 6 months | ↓ c-fos protein immunoreactivity and mRNA expression of ASIC1a, ASIC13, and TRPV1 in DRGs | |
| Human study | ||
| Fontani, 2010 [54] | Double-blind placebo-controlled design | Compared to placebo group, n-3 PUFA group: |
| ↓ pain intensity | ||
| ↓ number of positive tender points | ||
| Treatments: placebo group (n = 23): 4 g of oleic sunflower oil daily for 35 days and n-3 PUFA group (n = 23): 4 g oil (2.8 g of omega-3 polyunsaturated fatty acids, EPA +DHA in a 2:1 ratio; 1,60 g EPA, 0.8 g DHA, 0.4 g of other types of omega-3 polyunsaturated fatty acids: alpha linolenic, stearidonic, eicosatetraenoic and ocosapentaenoic acid) daily for 35 days | ↓ pain intensity | |
| ↔ superficial thermal (heat and cold) | ||
| ↓ anger-hostility and depression↓ in AA/EPA, AA, cortisol↑ EPA, pain area (%) |
| First Author, Year [ref] | Experimental Design and Treatments | Results |
|---|---|---|
| Animal studies | ||
| Ben-Azu, 2019 [56] | Forced swim-induced FM-like model Male Swiss mice (n = 50) Treatments: vehicle (10 mL/kg BW, i.p.), naringin (2.5, 5 and 10 mg/kg BW, i.p.), diazepam (2 mg/kg BW, i.p.), donepezil (1 mg/kg BW, i.p.) and imipramine (15 mg/kg BW, i.p.) for 7 days | Compared to vehicle group, naringin groups: |
| ↑ locomotor activity | ||
| ↓ depressant and anxiety | ||
| ↑ % social preference | ||
| ↑ cognitive performance | ||
| ↓ AChE enzyme activity in brain | ||
| ↑ GSH, SOD, CAT levels in brain | ||
| ↓ MDA and nitrate levels in brain | ||
| Pinho-Ribeiro, 2016 [57] | Acetic acid- and PBQ-induced FM-like visceral pain model Male Swiss mice Treatments: vehicle, Nar (16.7–150 mg/kg p.o.) given 30 min before stimulus Formalin-induced FM-like pain model Male Swiss mice Treatments: vehicle, Nar (50 mg/kg, p.o.) given 30 min before stimulus Capsaicin- and CFA-induced overt FM-like pain models Male Swiss mice Treatments: vehicle, Nar (50 mg/kg, p.o.) given 30 min before nociceptive stimulus. Carrageenan-, CFA-, capsaicin-, and PGE2-induced mechanical hyperalgesia Male Swiss mice Treatments: vehicle, Nar p.o. given 30 min before stimulus | Compared to vehicle group, Nar groups: |
| ↑ antinociceptive effect induced by acid, formalin, and capsaicin- and CFA | ||
| ↓ mechanical hyperalgesia induced by carrageenan, capsaicin, CFA, and PGE2 | ||
| ↑ NO/cGMP/PKG/ATP sensitive K+ channel signaling pathway | ||
| ↑ GSH production of plantar skin tissue | ||
| ↓ IL-33, TNF-α, IL-1β production, and NF-κB activation of paw skin tissues | ||
| Zamanian, 2016 [59] | Exercise-induced fatigue-induced FM model Female Wistar rats (n = 50) Treatments: control, vehicle, 40, 80, and 160 mg naringin/kg/day for 30 days | Compared to vehicle group, naringin groups: |
| ↑ exhaustion swimming time | ||
| ↓ LDH activity and serum MMP-9 levels | ||
| ↑ blood glucose levels | ||
| Xue, 2019 [60] | Hot plate-induced FM-like pain model | Compared to vehicle group, Nar groups: |
| Acetic acid-, glutamate-, capsaicin-, capsaicin-, and formalin-induced FM-like pain models | ↑ antinociceptive effect against thermal and chemical-induced pain | |
| Treatments: vehicles, Nar (25–75 mg/kg) p.o., before stimulus | ↓ capsaicin-induced paw licking number | |
| Carrageenan-induced FM-like inflammatory model | ↓ carrageenan-induced peritoneal leukocyte infiltration of paw edema | |
| Treatments: vehicles, Nar (25–75 mg/kg) p.o., given 30 min before stimulus | ↓ TNF-α, IL-1β, and IL-6 levels of skin |
| First Author, Year [ref] | Experimental Design and Treatment | Results |
|---|---|---|
| Animal studies | ||
| Lin, 2011 [61] | Acetic acid-induced FM-like pain model | Compared to vehicle group, genistein group: |
| C57/BL6 mice (8–12-week-old) | ||
| Treatments: vehicle treated with substance P (SP to induce current), genistein (30 µM), SP + genistein, SP + daidzein (30 µM) for 10 min on DRG neurons | ↓ SP-mediated inhibition of ASIC3-selective current and PTK activity of muscle dorsal root ganglion neuron | |
| Jie, 2018 [68] | Glutamate-induced masseter muscle FM-like pain model | Compared to control group, genistein groups: |
| ↓ mechanical hypernociception at high genistein dose | ||
| Female Sprague-Dawley OVX rats | Partially reversed E2-potentiated glutamate-evoked hypernociception of masseter muscle | |
| Treatments: no injections (control); E2 vehicle + genistein, E2 + genistein vehicle, and E2 + genistein (2–60 mg/kg) for 12 days. | ↓ pNR2B and pERK1/2 expression in hippocampus |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shen, C.-L.; Schuck, A.; Tompkins, C.; Dunn, D.M.; Neugebauer, V. Bioactive Compounds for Fibromyalgia-like Symptoms: A Narrative Review and Future Perspectives. Int. J. Environ. Res. Public Health 2022, 19, 4148. https://doi.org/10.3390/ijerph19074148
Shen C-L, Schuck A, Tompkins C, Dunn DM, Neugebauer V. Bioactive Compounds for Fibromyalgia-like Symptoms: A Narrative Review and Future Perspectives. International Journal of Environmental Research and Public Health. 2022; 19(7):4148. https://doi.org/10.3390/ijerph19074148
Chicago/Turabian StyleShen, Chwan-Li, Alexis Schuck, Christina Tompkins, Dale M. Dunn, and Volker Neugebauer. 2022. "Bioactive Compounds for Fibromyalgia-like Symptoms: A Narrative Review and Future Perspectives" International Journal of Environmental Research and Public Health 19, no. 7: 4148. https://doi.org/10.3390/ijerph19074148
APA StyleShen, C.-L., Schuck, A., Tompkins, C., Dunn, D. M., & Neugebauer, V. (2022). Bioactive Compounds for Fibromyalgia-like Symptoms: A Narrative Review and Future Perspectives. International Journal of Environmental Research and Public Health, 19(7), 4148. https://doi.org/10.3390/ijerph19074148







