Physical Performance, Cardiovascular Health and Psychosocial Wellbeing in Older Adults Compared to Oldest-Old Residential Seniors
Abstract
1. Introduction
2. Methods
2.1. Study Design and Study Population
2.2. Procedures
2.3. Short Physical Performance Battery
2.4. Handgrip Strength
2.5. Cardiovascular Health
2.6. Questionnaires
2.7. Statistical Analysis
3. Results
3.1. Study Population
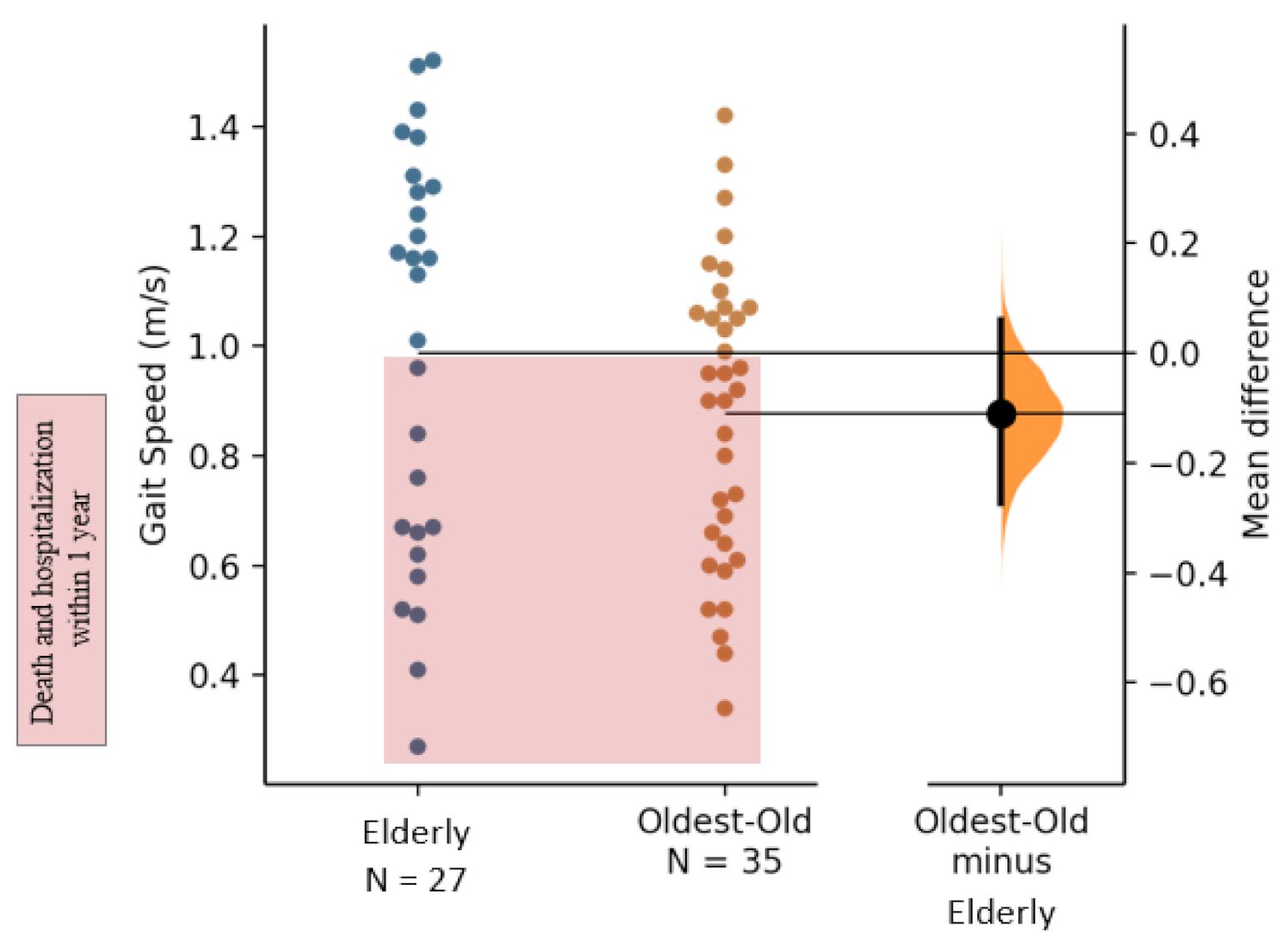
3.2. Physical Functioning
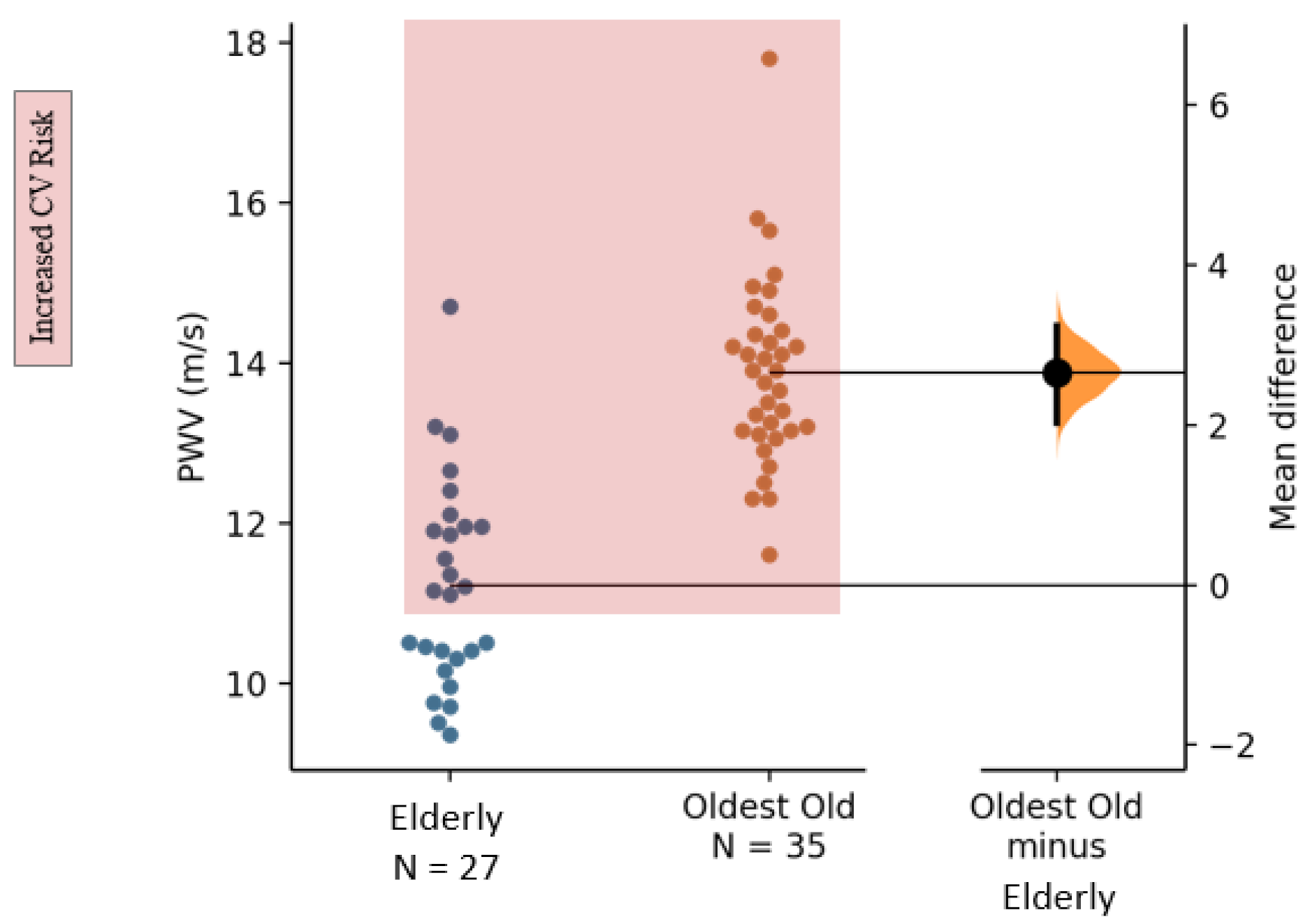
3.3. Cardiovascular Health
3.4. Psychosocial Wellbeing
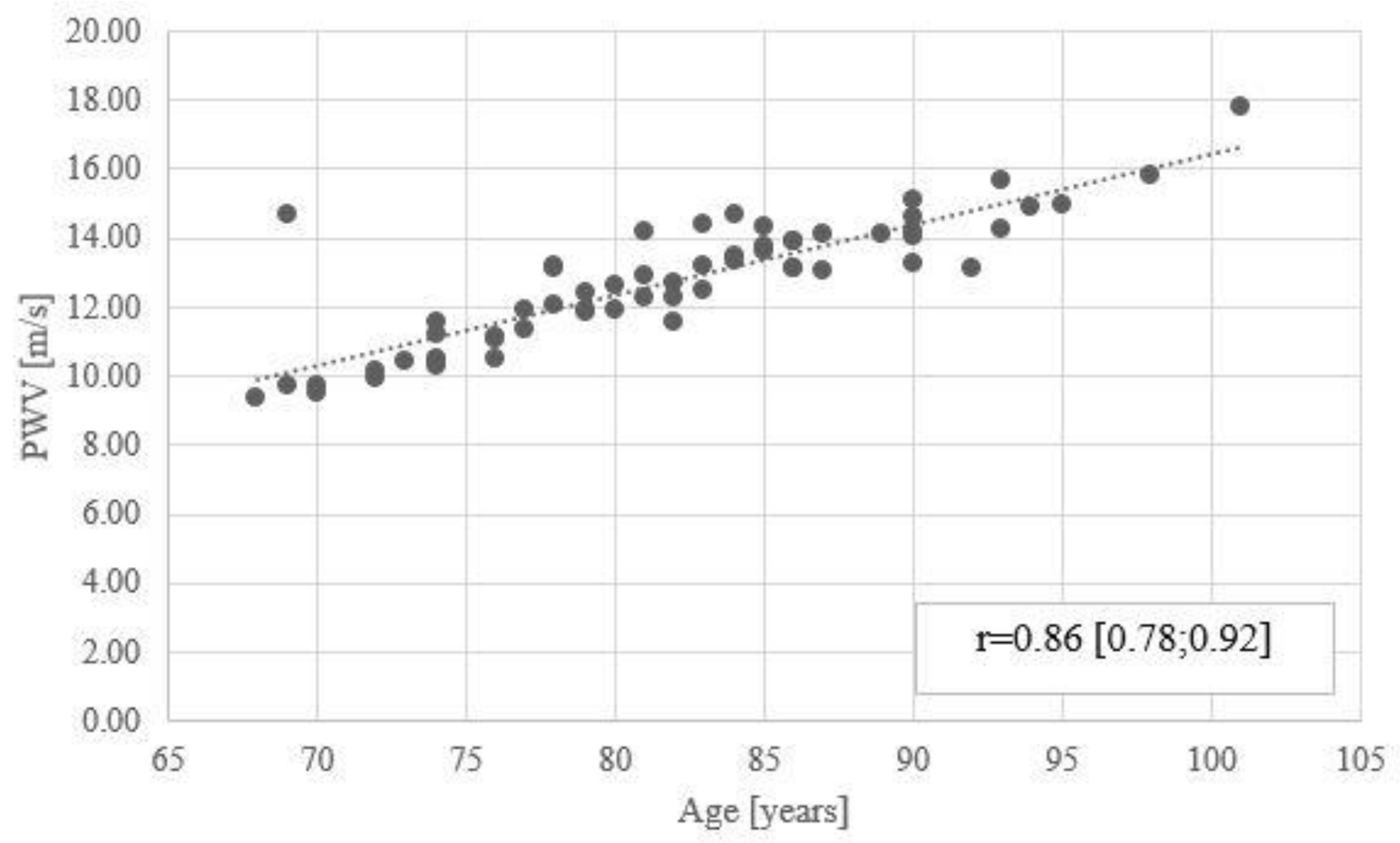
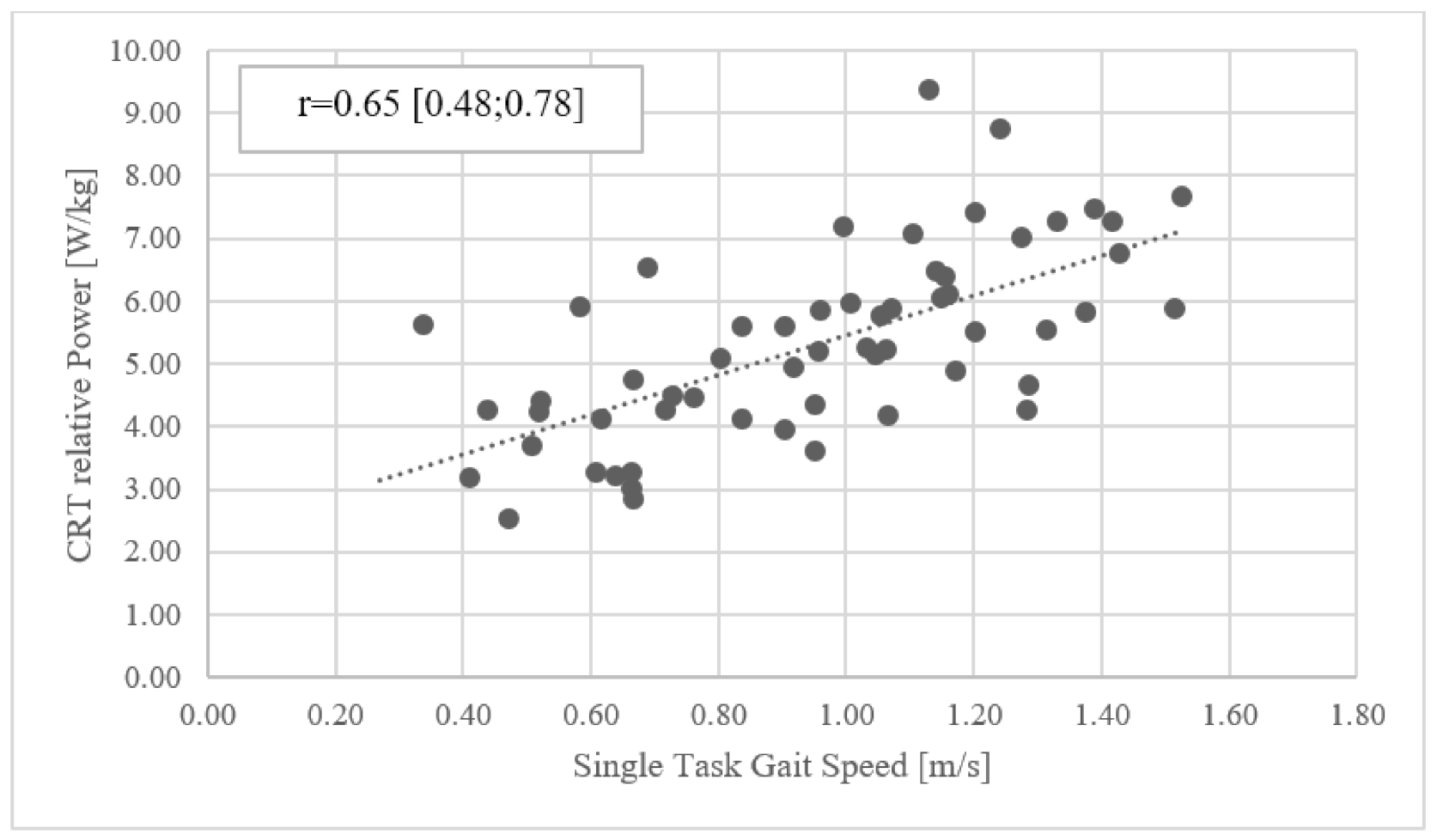
3.5. Correlations
4. Discussion
4.1. Methodological Considerations
4.2. Conclusions and Practical Applications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
Appendix A
| Older Adults | Oldest-Old | t-Test | ||||
|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean Difference [95% CI] | Hedge’s g [95% CI] | |||
| SF-36: General Health | 67 (19) | 64 (17) | −2.8 | [−11.8; 6.4] | −0.15 | [−0.68; 0.36] |
| SF-36: Physical Functioning | 68 (28) | 61 (27) | −6.3 | [−19.5; 7.5] | −0.23 | [−0.73; 0.28] |
| SF-36: Physical Limitations | 77 (33) | 75 (38) | −1.9 | [−19.1; 16.5] | −0.05 | [−0.51; 0.49] |
| SF-36: Emotional Limitations | 83 (33) | 79 (38) | −3.7 | [−20.6; 14.8] | −0.10 | [−0.56; 0.44] |
| SF-36: Vitality | 76 (16) | 68 (22) | −8.6 | [−17.6; 0.8] | −0.43 | [−0.91; 0.06] |
| SF-36: Mental Health | 78 (12) | 75 (17) | −2.5 | [−9.6; 4.6] | −0.16 | [−0.66; 0.33] |
| SF-36: Social Functioning | 89 (18) | 88 (15) | −0.7 | [−8.4; 8.2] | −0.04 | [−0.54; 0.49] |
| SF-36: Bodily Pain | 75 (27) | 74 (22) | −0.7 | [−12.2; 13.1] | −0.02 | [−0.53; 0.51] |
| AQoL: Independent Living | 7.6 (3.3) | 8.6 (3.3) | 0.9 | [−0.76; 2.50] | 0.28 | [−0.26; 0.74] |
| AQoL: Pain | 5.6 (2.7) | 5.7 (2.3) | 0.10 | [−1.21; 1.26] | 0.04 | [−0.49; 0.55] |
| AQoL: Mental Health | 16.0 (3.6) | 15.3 (3.9) | −0.62 | [−2.43; 1.23] | −0.16 | [−0.65; 0.34] |
| AQoL: Life satisfaction | 8.4 (2.0) | 8.0 (2.2) | −0.42 | [−1.48; 0.55] | −0.20 | [−0.70; 0.29] |
| AQoL: Self-worth | 6.0 (1.9) | 4.9 (1.7) | −1.12 | [−2.05; −0.30] | −0.63 | [−1.12; −0.15] |
| AQoL: Coping | 6.2 (1.8) | 5.8 (2.0) | −0.35 | [−1.22; 0.67] | −0.18 | [−0.67; 0.35] |
| AQoL: Relationships | 11.8 (2.4) | 11.7 (2.9) | −0.16 | [−1.48; 1.13] | −0.06 | [−0.60; 0.43] |

References
- United Nations, Department of Economic and Social Affairs. Social Affairs, Population Division. In World Population Ageing 2019; Department of Economic and Social Affairs, Population Division, Eds.; United Nations: New York, NY, USA, 2020; Volume ST/ESA/SER.A/444. [Google Scholar]
- BFS, Bundesamt für Statistik. Sozialmedizinische Betreuung in Institutionen und zu Hause 2018; Gesundheitsversorgung, G.d.B., Ed.; Bundesamt für Statistik: Neuchâtel, Switzerland, 2019. [Google Scholar]
- Hunter, S.K.; Pereira, H.M.; Keenan, K.G. The aging neuromuscular system and motor performance. J. Appl. Physiol. 2016, 121, 982–995. [Google Scholar] [CrossRef]
- Peiffer, A.M.; Hugenschmidt, C.E.; Maldjian, J.A.; Casanova, R.; Srikanth, R.; Hayasaka, S.; Burdette, J.H.; Kraft, R.A.; Laurienti, P.J. Aging and the interaction of sensory cortical function and structure. Hum. Brain Mapp. 2009, 30, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Naro, F.; Venturelli, M.; Monaco, L.; Toniolo, L.; Muti, E.; Milanese, C.; Zhao, J.; Richardson, R.S.; Schena, F.; Reggiani, C. Skeletal muscle fiber size and gene expression in the oldest-old with differing degrees of mobility. Front. Physiol. 2019, 10, 313. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.J.; Piasecki, M.; Atherton, P.J. The age-related loss of skeletal muscle mass and function: Measurement and physiology of muscle fibre atrophy and muscle fibre loss in humans. Ageing Res. Rev. 2018, 47, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Rubenstein, L.Z. Falls in older people: Epidemiology, risk factors and strategies for prevention. Age Ageing 2006, 35 (Suppl. 2), ii37–ii41. [Google Scholar] [CrossRef] [PubMed]
- BFS, Bundesamt für Statistik. Aktives Altern. In Statistik der Schweiz; Statistik, B.F., Ed.; Bundesamt für Statistik: Neuchâtel, Switzerland, 2018. [Google Scholar]
- Bohl, A.A.; Fishman, P.A.; Ciol, M.A.; Williams, B.; Logerfo, J.; Phelan, E.A. A longitudinal analysis of total 3-year healthcare costs for older adults who experience a fall requiring medical care. J. Am. Geriatr. Soc. 2010, 58, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Laina, A.; Stellos, K.; Stamatelopoulos, K. Vascular ageing: Underlying mechanisms and clinical implications. Exp. Gerontol. 2018, 109, 16–30. [Google Scholar] [CrossRef] [PubMed]
- Baulmann, J.; Nürnberger, J.; Slany, J.; Schmieder, R.; Schmidt-Trucksass, A.; Baumgart, D.; Weber, T. Arterielle gefäßsteifigkeit und pulswellenanalyse. DMW-Deutsche Medizinische Wochenschrift 2010, 135, 4–9. [Google Scholar] [CrossRef]
- Murphy, S.L.; Xu, J.; Kochanek, K.D.; Arias, E. Mortality in the United States, 2017. In NCHS Data Brief; no 328; National Center for Health Statistics: Hyattsville, MD, USA, 2018. [Google Scholar]
- Berlau, D.J.; Corrada, M.M.; Kawas, C. The prevalence of disability in the oldest—Old is high and continues to increase with age: Findings from the 90+ study. Int. J. Geriatr. Psychiatry 2009, 24, 1217–1225. [Google Scholar] [CrossRef]
- Yumin, E.T.; Simsek, T.T.; Sertel, M.; Ozturk, A.; Yumin, M. The effect of functional mobility and balance on health-related quality of life (hrqol) among elderly people living at home and those living in nursing home. Arch. Gerontol. Geriatr. 2011, 52, e180–e184. [Google Scholar] [CrossRef]
- Berlau, D.J.; Corrada, M.M.; Peltz, C.B.; Kawas, C.H. Disability in the oldest-old: Incidence and risk factors in the 90+ study. Am. J. Geriatr. Psychiatry 2012, 20, 159–168. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef]
- Ramirez-Velez, R.; Lopez Saez de Asteasu, M.; Morley, J.E.; Cano-Gutierrez, C.A.; Izquierdo, M. Performance of the short physical performance battery in identifying the frailty phenotype and predicting geriatric syndromes in community-dwelling elderly. J. Nutr. Health Aging 2021, 25, 209–217. [Google Scholar] [CrossRef]
- Steiber, N. Strong or weak handgrip? Normative reference values for the german population across the life course stratified by sex, age, and body height. PLoS ONE 2016, 11, e0163917. [Google Scholar] [CrossRef]
- Hametner, B.; Wassertheurer, S.; Kropf, J.; Mayer, C.; Holzinger, A.; Eber, B.; Weber, T. Wave reflection quantification based on pressure waveforms alone—Methods, comparison, and clinical covariates. Comput. Methods Programs Biomed. 2013, 109, 250–259. [Google Scholar] [CrossRef]
- Wassertheurer, S.; Kropf, J.; Weber, T.; van der Giet, M.; Baulmann, J.; Ammer, M.; Hametner, B.; Mayer, C.C.; Eber, B.; Magometschnigg, D. A new oscillometric method for pulse wave analysis: Comparison with a common tonometric method. J. Hum. Hypertens 2010, 24, 498–504. [Google Scholar] [CrossRef]
- Ware, J.E., Jr. Sf-36 health survey update. Spine 2000, 25, 3130–3139. [Google Scholar] [CrossRef]
- Richardson, J.; Iezzi, A.; Khan, M.A.; Maxwell, A. Validity and reliability of the assessment of quality of life (aqol)-8d multi-attribute utility instrument. Patient 2014, 7, 85–96. [Google Scholar] [CrossRef]
- Yardley, L.; Beyer, N.; Hauer, K.; Kempen, G.; Piot-Ziegler, C.; Todd, C. Development and initial validation of the falls efficacy scale-international (fes-i). Age Ageing 2005, 34, 614–619. [Google Scholar] [CrossRef]
- Greenland, S.; Senn, S.J.; Rothman, K.J.; Carlin, J.B.; Poole, C.; Goodman, S.N.; Altman, D.G. Statistical tests, p values, confidence intervals, and power: A guide to misinterpretations. Eur. J. Epidemiol. 2016, 31, 337–350. [Google Scholar] [CrossRef]
- Cohen, J. A power primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef]
- Ho, J.; Tumkaya, T.; Aryal, S.; Choi, H.; Claridge-Chang, A. Moving beyond p values: Data analysis with estimation graphics. Nat. Methods 2019, 16, 565–566. [Google Scholar] [CrossRef]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive statistics for studies in sports medicine and exercise science. Med. Sci. Sports Exerc. 2009, 41, 3. [Google Scholar] [CrossRef]
- Peeters, G.; Dobson, A.J.; Deeg, D.J.; Brown, W.J. A life-course perspective on physical functioning in women. Bull. World Health Organ. 2013, 91, 661–670. [Google Scholar] [CrossRef]
- Cabrero-Garcia, J.; Munoz-Mendoza, C.L.; Cabanero-Martinez, M.J.; Gonzalez-Llopis, L.; Ramos-Pichardo, J.D.; Reig-Ferrer, A. Short physical performance battery reference values for patients 70 years-old and over in primary health care. Aten Primaria 2012, 44, 540–548. [Google Scholar] [CrossRef]
- Syddall, H.; Cooper, C.; Martin, F.; Briggs, R.; Aihie Sayer, A. Is grip strength a useful single marker of frailty? Age Ageing 2003, 32, 650–656. [Google Scholar] [CrossRef]
- Ling, C.H.Y.; Gussekloo, J.; Trompet, S.; Meskers, C.G.M.; Maier, A.B. Clinical determinants of low handgrip strength and its decline in the oldest old: The leiden 85-plus study. Aging Clin. Exp. Res. 2020, 33, 1307–1313. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, K.S.; Dias, J.M.; Bastone Ade, C.; Vieira, R.A.; Andrade, A.C.; Perracini, M.R.; Guerra, R.O.; Dias, R.C. Handgrip strength cutoff points to identify mobility limitation in community—Dwelling older people and associated factors. J. Nutr. Health Aging 2016, 20, 306–315. [Google Scholar] [CrossRef]
- Abellan van Kan, G.; Rolland, Y.; Andrieu, S.; Bauer, J.; Beauchet, O.; Bonnefoy, M.; Cesari, M.; Donini, L.M.; Gillette Guyonnet, S.; Inzitari, M.; et al. Gait speed at usual pace as a predictor of adverse outcomes in community—Dwelling older people an international academy on nutrition and aging (iana) task force. J. Nutr. Health Aging 2009, 13, 881–889. [Google Scholar] [CrossRef]
- Sun, Z. Aging, arterial stiffness, and hypertension. Hypertension 2015, 65, 252–256. [Google Scholar] [CrossRef]
- Sawabe, M.; Takahashi, R.; Matsushita, S.; Ozawa, T.; Arai, T.; Hamamatsu, A.; Nakahara, K.; Chida, K.; Yamanouchi, H.; Murayama, S.; et al. Aortic pulse wave velocity and the degree of atherosclerosis in the elderly: A pathological study based on 304 autopsy cases. Atherosclerosis 2005, 179, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Hanssen, H.; Cordes, M.; Rossmeissl, A.; Endes, S.; Schmidt-Trucksass, A. Aerobic, resistance and combined exercise training on arterial stiffness in normotensive and hypertensive adults: A review. Eur. J. Sport Sci. 2015, 15, 443–457. [Google Scholar] [CrossRef] [PubMed]
- van de Laar, R.J.; Ferreira, I.; van Mechelen, W.; Prins, M.H.; Twisk, J.W.; Stehouwer, C.D. Habitual physical activity and peripheral arterial compliance in young adults: The Amsterdam growth and health longitudinal study. Am. J. Hypertens. 2011, 24, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Muratani, H. Improvement of arterial stiffness by aerobic exercise in elderly subjects. Hypertens. Res. 2007, 30, 875–876. [Google Scholar] [CrossRef][Green Version]
- Deiseroth, A.; Streese, L.; Kochli, S.; Wust, R.S.; Infanger, D.; Schmidt-Trucksass, A.; Hanssen, H. Exercise and arterial stiffness in the elderly: A combined cross-sectional and randomized controlled trial (examin age). Front. Physiol. 2019, 10, 1119. [Google Scholar] [CrossRef]
- Reference Values for Arterial Stiffness, C. Determinants of pulse wave velocity in healthy people and in the presence of cardiovascular risk factors: Establishing normal and reference values. Eur. Heart J. 2010, 31, 2338–2350. [Google Scholar] [CrossRef]
- Burholt, V.; Nash, P. Short form 36 (sf-36) health survey questionnaire: Normative data for wales. J. Public Health 2011, 33, 587–603. [Google Scholar] [CrossRef]
- Sackley, C.; Levin, S.; Cardoso, K.; Hoppitt, T. Observations of activity levels and social interaction in a residential care setting. Int. J. Ther. Rehabil. 2014, 13, 370–373. [Google Scholar] [CrossRef]
- Wiesmann, U.; Niehorster, G.; Hannich, H.J. Subjective health in old age from a salutogenic perspective. Br. J. Health Psychol. 2009, 14, 767–787. [Google Scholar] [CrossRef]
- Crowe, E.M.; Ball, S.D. Effectiveness of advanced stay strong, stay healthy in community settings. Gerontol. Geriatr. Med. 2015, 1, 1–7. [Google Scholar] [CrossRef]
- Syed-Abdul, M.M.; McClellan, C.L.; Parks, E.J.; Ball, S.D. Effects of a resistance training community programme in older adults. Ageing Soc. 2021, 1–16. [Google Scholar] [CrossRef]
- Izquierdo, M.; Morley, J.E.; Lucia, A. Exercise in people over 85. BMJ 2020, 368, 1–2. [Google Scholar] [CrossRef]
- Neviani, F.; Belvederi Murri, M.; Mussi, C.; Triolo, F.; Toni, G.; Simoncini, E.; Tripi, F.; Menchetti, M.; Ferrari, S.; Ceresini, G.; et al. Physical exercise for late life depression: Effects on cognition and disability. Int. Psychogeriatr. 2017, 29, 1105–1112. [Google Scholar] [CrossRef]
- Jansen, C.P.; Classen, K.; Wahl, H.W.; Hauer, K. Effects of interventions on physical activity in nursing home residents. Eur. J. Ageing 2015, 12, 261–271. [Google Scholar] [CrossRef]
- Distefano, G.; Goodpaster, B.H. Effects of exercise and aging on skeletal muscle. Cold Spring Harb. Perspect. Med. 2018, 8, a029785. [Google Scholar] [CrossRef]
- Melov, S.; Tarnopolsky, M.A.; Beckman, K.; Felkey, K.; Hubbard, A. Resistance exercise reverses aging in human skeletal muscle. PLoS ONE 2007, 2, e465. [Google Scholar] [CrossRef]
- Syed-Abdul, M.M. Benefits of resistance training in older adults. Curr. Aging Sci. 2021, 14, 5–9. [Google Scholar] [CrossRef]
- Smith, J.; Borchelt, M.; Maier, H.; Jopp, D. Health and well-being in the young old and oldest old. J. Soc. Issues 2002, 58, 715–732. [Google Scholar] [CrossRef]
| Older Adults | Oldest-Old | t-Test | ||||
|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean Difference [95% CI] | Hedge’s g [95% CI] | |||
| SPPB Gait Score | 3.3 (1.0) | 3.4 (0.9) | 0.10 | [−0.35; 0.59] | 0.11 | [−0.40; 0.63] |
| SPPB CRT Score | 1.7 (1.1) | 1.5 (0.9) | −0.25 | [−0.76; 0.23] | −0.25 | [−0.75; 0.26] |
| SPPB Balance Score | 2.9 (1.3) | 2.7 (1.1) | −0.24 | [−0.82; 0.40] | −0.20 | [−0.73; 0.32] |
| SPPB Total Score | 7.9 (2.6) | 7.5 (2.1) | −0.38 | [−1.52; 0.86] | −0.16 | [−0.68; 0.39] |
| Gait Speed Single-task (m/s) | 0.99 (0.37) | 0.88 (0.27) | −0.11 | [−0.28; 0.05] | −0.34 | [−0.89; 0.20] |
| Gait Speed Dual-task (m/s) | 0.89 (0.39) | 0.77 (0.31) | −0.12 | [−0.28; 0.05] | −0.37 | [−0.92; 0.17] |
| CRT (s) | 18.4 (10.1) | 19.0 (7.9) | 0.66 | [−5.46; 3.72] | 0.08 | [−0.56; 0.65] |
| Rel. Pmax CRT (W/kg) | 5.4 (1.9) | 5.2 (2.0) | −0.20 | [−1.00; 0.54] | −0.13 | [−0.66; 0.39] |
| Grip Strength (N) | 181 (73) | 150 (54) | −31.3 | [−66.3; −1.7] | −0.49 | [−0.97; 0.03] |
| cSBP (mmHg) | 120 (17) | 126 (20) | 6.9 | [−2.9; 15.4] | 0.36 | [−0.17; 0.88] |
| cDBP (mmHg) | 81 (8) | 82 (11) | 0.6 | [−4.1; 5.5] | 0.06 | [−0.42; 0.56] |
| AIx@75 (%) | 29.0 (11.1) | 32.3 (9.6) | 3.35 | [−1.94; 8.37] | 0.32 | [−0.21; 0.85] |
| PWV (m/s) | 11.2 (1.3) | 13.9 (1.2) | 2.65 | [2.04; 3.26] | 2.14 | [1.36; 2.81] |
| SF-36: Total Score | 76.5 (16.6) | 73.2 (18.5) | −3.38 | [−11.80; 5.35] | −0.19 | [−0.67; 0.35] |
| AQoL-8D: Total Score | 61.6 (12.8) | 60.1 (13.4) | −1.54 | [−8.0; 4.9] | −0.12 | [−0.39; 0.61] |
| FES | 22.9 (8.0) | 24.3 (8.5) | 1.38 | [−2.87; 5.28] | −0.16 | [−0.37; 0.63] |
| AIx@75 [%] | PWV [m/s] | Grip [N] | Gait ST [m/s] | Gait DT [m/s] | CRT [s] | CRT [W/kg] | SPPB Balance | SPPB Gait | SPPB CRT | SPPB Total | SF-36 | AQoL | |
| PWV [m/s] | 0.34 [0.13; 0.55] | ||||||||||||
| Grip [N] | −0.12 [−0.36; 0.13] | −0.22 [−0.45; 0.03] | |||||||||||
| Gait ST [m/s] | −0.09 [−0.34; 0.16] | −0.21 [−0.43; 0.05] | 0.51 [0.30; 0.68] | ||||||||||
| Gait DT [m/s] | −0.14 [−0.38; 0.12] | −0.21 [−0.44; 0.05] | 0.55 [0.35; 0.71] | 0.95 [0.91; 0.97] | |||||||||
| CRT [s] | 0.10 [−0.17; 0.35] | 0.23 [−0.03; 0.46] | −0.24 [−0.47; 0.02] | −0.41 [−0.60; −0.16] | −0.43 [−0.62; −0.19] | ||||||||
| CRT [W/kg] | −0.19 [−0.43; 0.07] | −0.23 [−0.46; 0.03] | 0.54 [0.33; 0.70] | 0.65 [0.48; 0.78] | 0.69 [0.52; 0.80] | −0.46 [−0.64; −0.23] | |||||||
| SPPB Balance | 0.06 [−0.20; 0.30] | −0.09 [−0.34; 0.16] | 0.38 [0.14; 0.58] | 0.41 [0.18; 0.60] | 0.40 [0.16; 0.59] | −0.40 [−0.60; −0.16] | 0.16 [−0.11; 0.40] | ||||||
| SPPB Gait | −0.02 [−0.27; 0.23] | −0.01 [−0.26; 0.24] | 0.35 [0.11; 0.55] | 0.78 [0.66; 0.86] | 0.69 [0.52; 0.80] | −0.24 [−0.47; 0.02] | 0.57 [0.37; 0.72] | 0.30 [0.06; 0.51] | |||||
| SPPB CRT | −0.16 [−0.40; 0.09] | −0.17 [−0.41; 0.08] | 0.32 [0.07; 0.52] | 0.49 [0.27; 0.66] | 0.43 [0.20; 0.62] | −0.65 [−0.78; −0.47] | 0.56 [0.36; 0.72] | 0.25 [0.00; 0.47] | 0.41 [0.17; 0.60] | ||||
| SPPB Total | −0.05 [−0.29; 0.21] | −0.13 [−0.37; 0.13] | 0.47 [0.25; 0.65] | 0.74 [0.60; 0.83] | 0.67 [0.51; 0.79] | −0.60 [−0.74; −0.40] | 0.55 [0.35; 0.71] | 0.75 [0.62; 0.85] | 0.74 [0.59; 0.83] | 0.72 [0.58; 0.83] | |||
| SF-36 | 0.01 [−0.24; 0.26] | −0.09 [−0.33; 0.16] | 0.10 [−0.15; 0.34] | 0.26 [0.01; 0.48] | 0.30 [0.05; 0.51] | −0.29 [−0.51; −0.03] | 0.28 [0.02; 0.50] | 0.16 [−0.09; 0.40] | 0.06 [−0.19; 0.31] | 0.27 [0.02; 0.48] | 0.22 [−0.03; 0.45] | ||
| AQoL | −0.21 [−0.44; 0.04] | −0.07 [−0.32; 0.18] | −0.25 [−0.47; 0.00] | −0.38 [−0.58; −0.15] | −0.44 [−0.62; −0.20] | 0.29 [0.03; 0.51] | −0.47 [−0.65; −0.24] | −0.01 [−0.26; 0.24] | −0.23 [−0.45; 0.04] | −0.30 [−0.51; −0.06] | −0.23 [−0.45; 0.03] | −0.64 [−0.76; −0.46] | |
| FES | −0.15 [−0.32; 0.11] | 0.04 [−0.21; 0.29] | −0.29 [−0.51; −0.05] | −0.41 [−0.60; −0.18] | −0.41 [−0.60; −0.17] | 0.29 [0.04; 0.51] | −0.39 [−0.59; −0.15] | −0.11 [−0.35; 0.15] | −0.36 [−0.56; −0.12] | −0.33 [−0.53; −0.08] | −0.34 [−0.54; −0.10] | −0.57 [−0.72; −0.37] | 0.56 [0.36; 0.71] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Minghetti, A.; Donath, L.; Hanssen, H.; Roth, R.; Lichtenstein, E.; Zahner, L.; Faude, O. Physical Performance, Cardiovascular Health and Psychosocial Wellbeing in Older Adults Compared to Oldest-Old Residential Seniors. Int. J. Environ. Res. Public Health 2022, 19, 1451. https://doi.org/10.3390/ijerph19031451
Minghetti A, Donath L, Hanssen H, Roth R, Lichtenstein E, Zahner L, Faude O. Physical Performance, Cardiovascular Health and Psychosocial Wellbeing in Older Adults Compared to Oldest-Old Residential Seniors. International Journal of Environmental Research and Public Health. 2022; 19(3):1451. https://doi.org/10.3390/ijerph19031451
Chicago/Turabian StyleMinghetti, Alice, Lars Donath, Henner Hanssen, Ralf Roth, Eric Lichtenstein, Lukas Zahner, and Oliver Faude. 2022. "Physical Performance, Cardiovascular Health and Psychosocial Wellbeing in Older Adults Compared to Oldest-Old Residential Seniors" International Journal of Environmental Research and Public Health 19, no. 3: 1451. https://doi.org/10.3390/ijerph19031451
APA StyleMinghetti, A., Donath, L., Hanssen, H., Roth, R., Lichtenstein, E., Zahner, L., & Faude, O. (2022). Physical Performance, Cardiovascular Health and Psychosocial Wellbeing in Older Adults Compared to Oldest-Old Residential Seniors. International Journal of Environmental Research and Public Health, 19(3), 1451. https://doi.org/10.3390/ijerph19031451







