Impact of Contaminants on Microbiota: Linking the Gut–Brain Axis with Neurotoxicity
Abstract
:1. Introduction
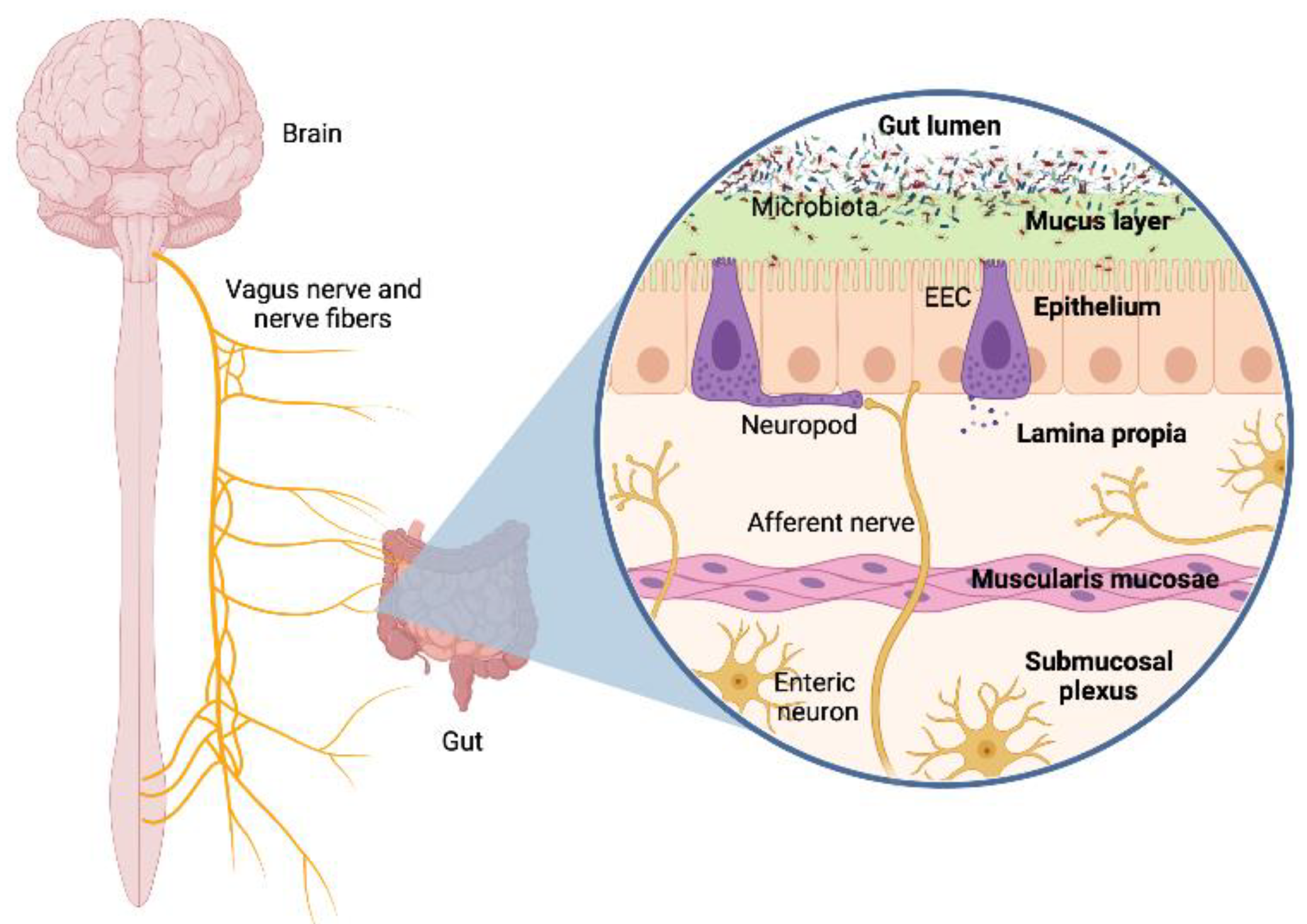
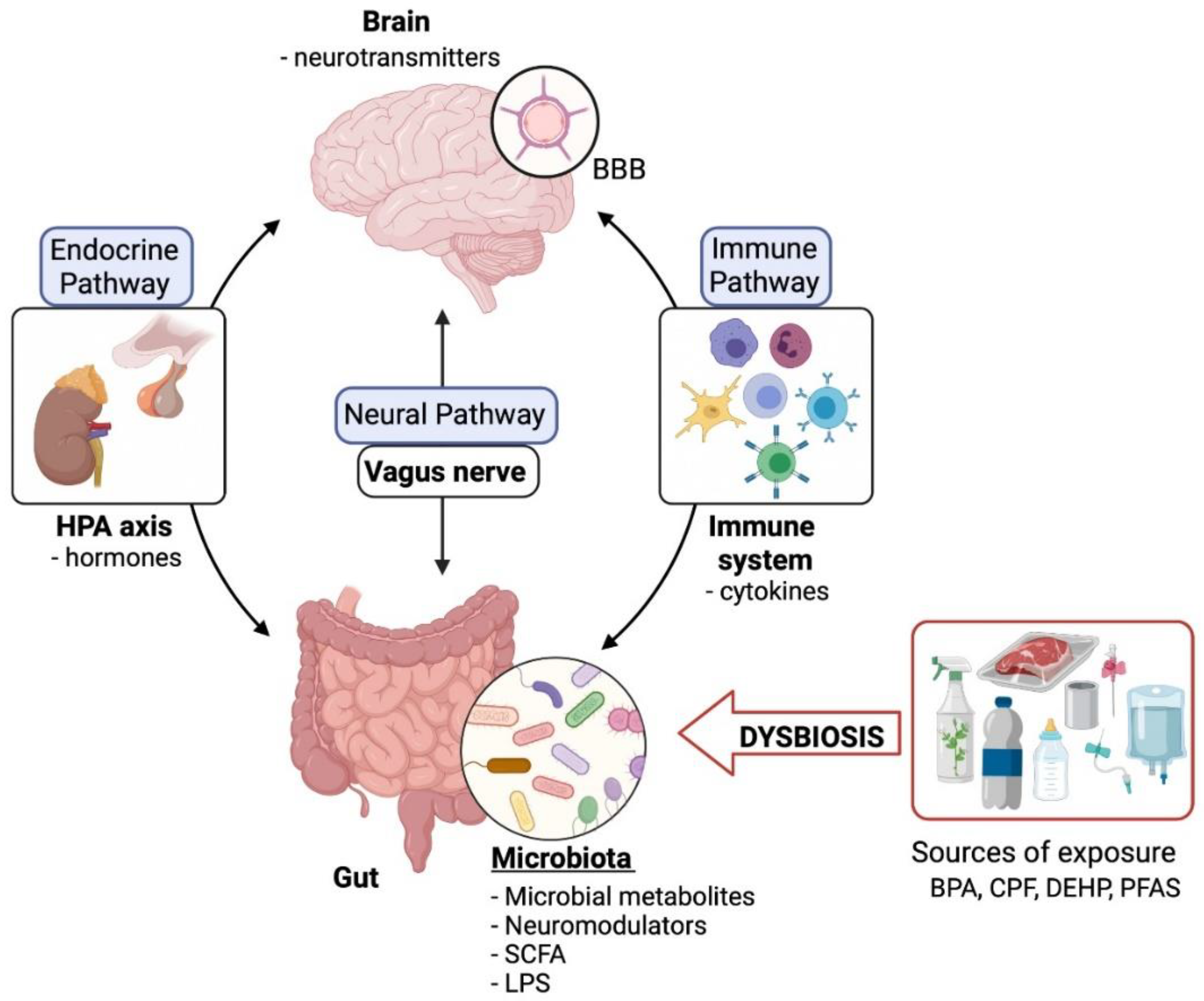
2. Crosstalk between Microbiota and Nervous System: Gut-Brain Axis
2.1. Neural Pathway: Vagus Nerve and Neurotransmitters
2.2. Endocrine Pathway: Hypothalamic-Pituitary-Adrenal Axis and Hormones
2.3. Immune Pathway: Microglia, Lymphocytes, and Cytokines
3. Contaminants, Microbiota and Neurotoxicity
3.1. Endocrine Disruptors
3.1.1. Bisphenol A
| BPA Exposure | ||||||
|---|---|---|---|---|---|---|
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| HepG2 (Human) | 25 μg/L, 250 μg/L and 2500 μg/L | 10 days | Lactobacilllus | Firmicutes | ↑ | [73] |
| Alcaligenes | Proteobacteria | ↑ | ||||
| Mycobacterium | Actinobacteria | ↑ | ||||
| Male CD-1 mice | 0.5 mg/kg of food | 24 weeks | Proteobacteria | ↑ | [78] | |
| Akkermansia | Verrucomicrobia | ↓ | ||||
| Rikenella | Bacteroidetes | ↑ | ||||
| Male CD-1 mice | 20 mg/10 g body weight | 10 weeks | Helicobacter | Proteobacteria | ↑ | [79] |
| TM7/Saccharibacteria | ↑ | |||||
| Coprococcus | Firmicutes | ↓ | ||||
| Firmicutes (Clostridia) | ↓ | |||||
| Eubacterium | Firmicutes | ↓ | ||||
| Lactobacillus | Firmicutes | ↓ | ||||
| Tenericutes | ↓ | |||||
| Male C57BL/6J | 0.05, 0.5, 5 and 50 mg/kg/day (females only 50 mg/kg/day) | 22 weeks | Oscillibacter | Firmicutes | ↑ | [80] |
| Tyzzerella | Firmicutes | ↑ | ||||
| Firmicutes (Ruminococcaceae NK4A214 group) | ↑ | |||||
| Alloprevotella | Bacteroidetes | ↓ | ||||
| Ruminococcus | Firmicutes | ↓ | ||||
| Parabacteroides | Bacteroidetes | ↓ | ||||
| Akkermansia | Verrucomicrobia | ↓ | ||||
| Male and female C57BL/6J | Allobaculum | Firmicutes | ↓ | |||
| Muribaculum | Bacteroidetes | ↓ | ||||
| Ruminiclostridium | Firmicutes | ↑ | ||||
| Desulfovibrio | Proteobacteria | ↑ | ||||
| Female C57BL/6J | Bilophila | Proteobacteria | ↑ | |||
| Peptococcus | Firmicutes | ↑ | ||||
| Bacteroides | Bacteroidetes | ↓ | ||||
| Parasutterella | Proteobacteria | ↓ | ||||
| Akkermansia | Verrucomicrobia | ↓ | ||||
| Rikenella | Bacteroidetes | ↑ | ||||
3.1.2. Chlorpyrifos
| CPF Exposure | ||||||
|---|---|---|---|---|---|---|
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Pregnant females Hannover Wistar rat and their offspring and SHIME | 1 mg/kg body weight/day | Gestation and 60 post-natal days | Enterococcus | Firmicutes | ↑ | [68] |
| Bacteroides | Bacteroidetes | ↑ | ||||
| Lactobacilllus | Firmicutes | ↓ | ||||
| Bifidobacterium | Actinobacteria | ↓ | ||||
| Caco-2/TC7 cells And SHIME | 3.5 mg/day | 21–23 days post-seeding | Lactobacilllus | Firmicutes | ↓ | [85] |
| Bifidobacterium | Actinobacteria | ↓ | ||||
| Male Wistar rats | 0.3 mg/kg bw/day | 20–25 weeks | Streptococcus | Firmicutes | ↑ | [86] |
| Ruminiclostridium | Firmicutes | ↑ | ||||
| Actinobacteria | ↑ | |||||
| Romboutsia | Firmicutes | ↓ | ||||
| Turicibacter | Firmicutes | ↓ | ||||
| Clostridium | Firmicutes | ↓ | ||||
| Akkermansia | Verrucomicrobia | ↓ | ||||
| Luteolibacter | Verrucomicrobia | ↓ | ||||
| Prosthecobacter | Verrucomicrobia | ↓ | ||||
| Coraliomargarita | Verrucomicrobia | ↓ | ||||
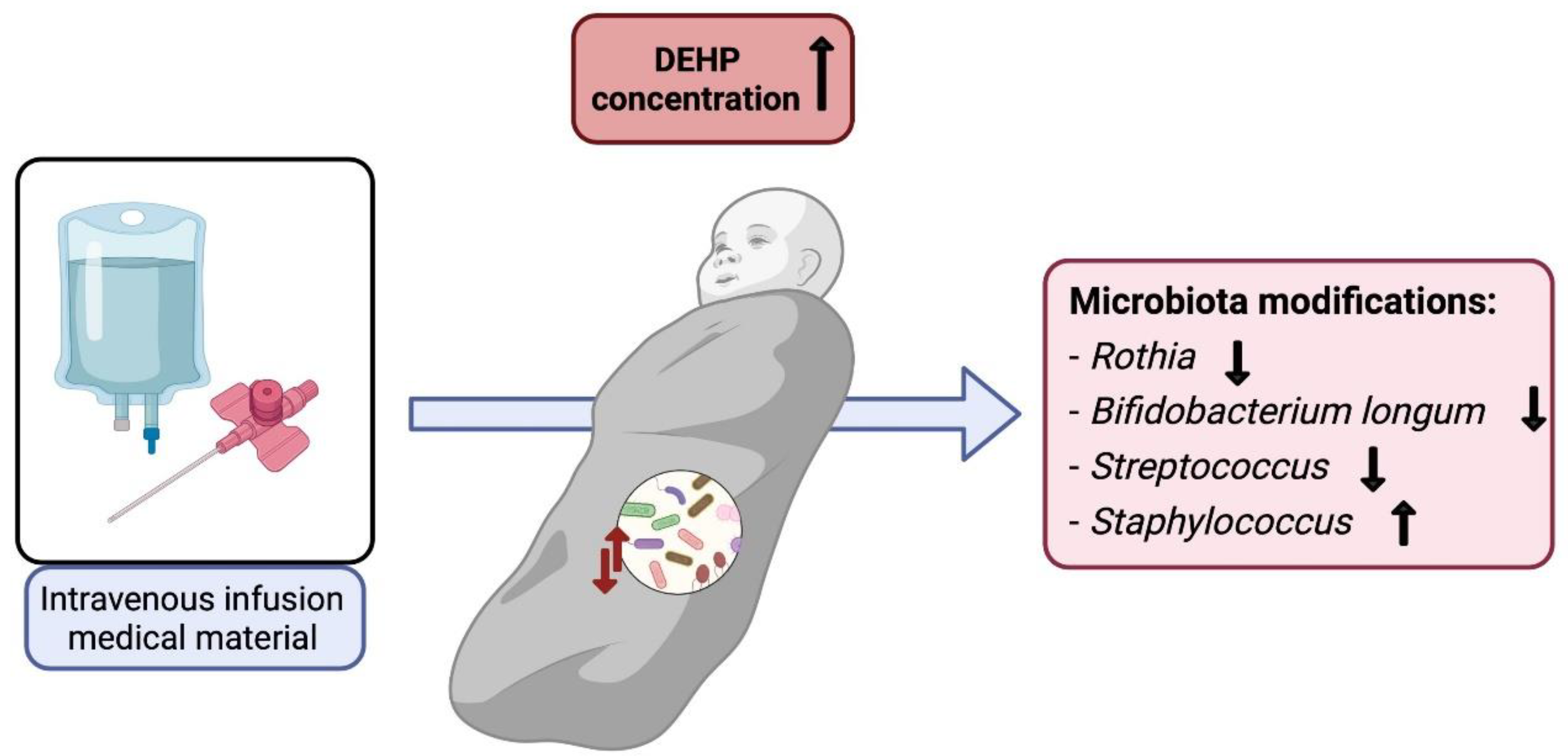
3.1.3. Diethylhexyl Phthalate
3.1.4. Perfluoroalkylated and Polyfluoroalkylated Substances (PFAS)
3.2. Early-Exposure and Developmental Neurotoxicity
| BPA Exposure | ||||||
|---|---|---|---|---|---|---|
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Male and female offspring of California mice dams | 5 mg/kg feed weight (LD) and 50 mg/kg feed weight (UD) (administered to dams) | Two weeks prior to breeding, throughout gestation, and lactation. | Blautia | Firmicutes | [105] | |
| Female offspring | Desulfovibrio | Proteobacteria | ↑ | |||
| Male offspring | Firmicutes (Clostridiales) | ↑ | ||||
| Bacteroidetes (Bacteroidales) | ↓ | |||||
| Male offspring of pregnant females Sprague Dawley rats | 50 μg/kg/day (administered to dams) | 30 days of gestation and 21 days of lactation | Blautia | Firmicutes | ↑ | [106] |
| Prevotella | Bacteroidetes | ↑ | ||||
| Allobaculum | Firmicutes | ↑ | ||||
| Firmicutes (lactobacillaceae) | ↑ | |||||
| Adlercreutzia | Actinobacteria | ↓ | ||||
| Oscillospira | Firmicutes | ↓ | ||||
| CPF Exposure | ||||||
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Male and female offspring of pregnant female Wistar rats | 1 mg/kg/mL/day | Six days (PND10-PND15) | Anaerobranca | Firmicutes | ↑ | [109] |
| Borrelia | Spirochaetes | ↑ | ||||
| Brevundimonas | Proteobacteria | ↑ | ||||
| Butyrivibrio | Firmicutes | ↑ | ||||
| Candidatus Endobugula | Firmicutes | ↑ | ||||
| Mogibacterium | Firmicutes | ↑ | ||||
| Pelagicoccus | Verrucomicrobia | ↑ | ||||
| Candidatus Contubernalis | Firmicutes | ↓ | ||||
| Hyphomicrobium | Proteobacteria | ↓ | ||||
| Nitrincola | Proteobacteria | ↓ | ||||
| Paracoccus | Proteobacteria | ↓ | ||||
| Rhizobium | Proteobacteria | ↓ | ||||
| Vogesella | Proteobacteria | ↓ | ||||
| Male C57BL/6, apoE3 and apoE4 mice (homozygous for the ε3 and ε4 allele) | 1 mg/kg/mL/day | Six days (PND10-PND15) | Streptococcus | Firmicutes | ↓ | [110] |
| Akkermansia | Verrucomicrobia | ↓ | ||||
| Luteolibacter | Verrucomicrobia | ↓ | ||||
| Prosthecobacter | Verrucomicrobia | ↓ | ||||
| Coraliomargarita | Verrucomicrobia | ↓ | ||||
| DEHP Exposure | ||||||
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Female C57BL/6 mice | 1 and 10 mg/kg body weight/day | 14 days | Lachnoclostridium | Firmicutes | ↑ | [112] |
| Akkermansia | Verrucomicrobia | ↓ | ||||
| Odoribacter | Bacteroidetes | ↓ | ||||
| Clostridium | Firmicutes | ↓ | ||||
| Anaerobic culture of caecal microbiota | 10 and 100 μM | Seven days | Parabacteroidetes | ↑ | ||
| Lactobacilllus | Firmicutes | ↓ | ||||
| Firmicutes | ↓ | |||||
| PFAS Exposure | ||||||
| PFOS | ||||||
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Male C57BL/6J mice | 0.3, 3 and 30 μg/g BW/day | 16 days | Bacteroidetes (Rikenellaceae) | ↑ | [113] | |
| Proteobacteria (Gammaproteobacteria) | ↓ | |||||
| Blautia | Firmicutes | ↓ | ||||
| PFOA | ||||||
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Juvenile C57BL/6J mice | 3 and 30 mg/kg BW (Subacute exposure) | 14 days | Bacteroidetes (Porphyromonadaceae) | ↑ | [114] | |
| Parabacteroides | Bacteroidetes | ↑ | ||||
| 2.5, 5 and 10 mg/kg BW (Subchronic exposure) | 30 days | Bacteroidetes | ↑ | |||
| Firmicutes | ↓ | |||||
| Both doses | Respective days | Lactobacilllus | Firmicutes | ↓ | ||
| Bifidobacterium | Actinobacteria | ↓ | ||||
| Dehalobacterium | Firmicutes | ↓ | ||||
4. Existing Gaps in the Microbiota Research
5. In-Silico Models for Microbiota-Chemical Interaction and Future Prospects
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Nishino, R.; Mikami, K.; Takahashi, H.; Tomonaga, S.; Furuse, M.; Hiramoto, T.; Aiba, Y.; Koga, Y.; Sudo, N. Commensal Microbiota Modulate Murine Behaviors in a Strictly Contamination-Free Environment Confirmed by Culture-Based Methods. Neurogastroenterol. Motil. 2013, 25, 521-e371. [Google Scholar] [CrossRef] [PubMed]
- Bezek, K.; Petelin, A.; Pražnikar, J.; Nova, E.; Redondo, N.; Marcos, A.; Pražnikar, Z.J. Obesity Measures and Dietary Parameters as Predictors of Gut Microbiota Phyla in Healthy Individuals. Nutrients 2020, 12, 2695. [Google Scholar] [CrossRef] [PubMed]
- Arumugam, M.; Raes, J.; Pelletier, E.; le Paslier, D.; Yamada, T.; Mende, D.R.; Fernandes, G.R.; Tap, J.; Bruls, T.; Batto, J.M.; et al. Enterotypes of the Human Gut Microbiome. Nature 2011, 473, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Davenport, E.R.; Sanders, J.G.; Song, S.J.; Amato, K.R.; Clark, A.G.; Knight, R. The Human Microbiome in Evolution. BMC Biol. 2017, 15, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Vaga, S.; Lee, S.; Ji, B.; Andreasson, A.; Talley, N.J.; Agréus, L.; Bidkhori, G.; Kovatcheva-Datchary, P.; Park, J.; Lee, D.; et al. Compositional and Functional Differences of the Mucosal Microbiota along the Intestine of Healthy Individuals. Sci. Rep. 2020, 10, 14977. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Hayes, R.B. Environmental Influences on the Human Microbiome and Implications for Noncommunicable Disease. Annu. Rev. Public Health 2021, 42, 277–292. [Google Scholar] [CrossRef]
- Magueresse-Battistoni, B. le Adipose Tissue and Endocrine-Disrupting Chemicals: Does Sex Matter? Int. J. Environ. Res. Public Health 2020, 17, 9403. [Google Scholar] [CrossRef]
- Long, D.; Liu, M.; Li, H.; Song, J.; Jiang, X.; Wang, G.; Yang, X. Dysbacteriosis Induces Abnormal Neurogenesis via LPS in a Pathway Requiring NF-ΚB/IL-6. Pharmacol. Res. 2021, 167, 105543. [Google Scholar] [CrossRef]
- Gubert, C.; Kong, G.; Renoir, T.; Hannan, A.J. Exercise, Diet and Stress as Modulators of Gut Microbiota: Implications for Neurodegenerative Diseases. Neurobiol. Dis. 2020, 134, 104621. [Google Scholar] [CrossRef]
- Man, A.W.C.; Zhou, Y.; Xia, N.; Li, H. Involvement of Gut Microbiota, Microbial Metabolites and Interaction with Polyphenol in Host Immunometabolism. Nutrients 2020, 12, 3054. [Google Scholar] [CrossRef]
- Clark, A.; Mach, N. Exercise-Induced Stress Behavior, Gut-Microbiota-Brain Axis and Diet: A Systematic Review for Athletes. J. Int. Soc. Sports Nutr. 2016, 13, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gentile, F.; Doneddu, P.E.; Riva, N.; Nobile-Orazio, E.; Quattrini, A. Diet, Microbiota and Brain Health: Unraveling the Network Intersecting Metabolism and Neurodegeneration. Int. J. Mol. Sci. 2020, 21, 7471. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, M.; Gálvez-Ontiveros, Y.; Rivas, A. Endobolome, a New Concept for Determining the Influence of Microbiota Disrupting Chemicals (MDC) in Relation to Specific Endocrine Pathogenesis. Front. Microbiol. 2020, 11, 578007. [Google Scholar] [CrossRef] [PubMed]
- Raja, G.L.; Subhashree, K.D.; Kantayya, K.E. In Utero Exposure to Endocrine Disruptors and Developmental Neurotoxicity: Implications for Behavioural and Neurological Disorders in Adult Life. Environ. Res. 2022, 203, 111829. [Google Scholar] [CrossRef]
- Vilcins, D.; Cortes-Ramirez, J.; Currie, D.; Preston, P. Early Environmental Exposures and Life-Long Risk of Chronic Non-Respiratory Disease. Paediatr. Respir. Rev. 2021, 40, 33–38. [Google Scholar] [CrossRef]
- Heinken, A.; Basile, A.; Thiele, I. Advances in Constraint-Based Modelling of Microbial Communities. Curr. Opin. Syst. Biol. 2021, 27, 100346. [Google Scholar] [CrossRef]
- Merten, C.; Schoonjans, R.; di Gioia, D.; Peláez, C.; Sanz, Y.; Maurici, D.; Robinson, T. Editorial: Exploring the Need to Include Microbiomes into EFSA’s Scientific Assessments. EFSA J. 2020, 18, e18061. [Google Scholar] [CrossRef]
- Spencer, N.J.; Hu, H. Enteric Nervous System: Sensory Transduction, Neural Circuits and Gastrointestinal Motility. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 338–351. [Google Scholar] [CrossRef]
- Kulkarni, S.; Ganz, J.; Bayrer, J.; Becker, L.; Bogunovic, M.; Rao, M. Advances in Enteric Neurobiology: The “Brain” in the Gut in Health and Disease. J. Neurosci. 2018, 38, 9346–9354. [Google Scholar] [CrossRef] [Green Version]
- D’alessandro, G.; Lauro, C.; Quaglio, D.; Ghirga, F.; Botta, B.; Trettel, F.; Limatola, C. Neuro-signals from Gut Microbiota: Perspectives for Brain Glioma. Cancers 2021, 13, 2810. [Google Scholar] [CrossRef]
- Erny, D.; Dokalis, N.; Mezö, C.; Castoldi, A.; Mossad, O.; Staszewski, O.; Frosch, M.; Villa, M.; Fuchs, V.; Mayer, A.; et al. Microbiota-Derived Acetate Enables the Metabolic Fitness of the Brain Innate Immune System during Health and Disease. Cell Metab. 2021, 33, 2260–2276.e7. [Google Scholar] [CrossRef] [PubMed]
- Chevalier, G.; Siopi, E.; Guenin-Macé, L.; Pascal, M.; Laval, T.; Rifflet, A.; Boneca, I.G.; Demangel, C.; Colsch, B.; Pruvost, A.; et al. Effect of Gut Microbiota on Depressive-like Behaviors in Mice Is Mediated by the Endocannabinoid System. Nat. Commun. 2020, 11, 6363. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.; Wang, W.; Guo, R.; Liu, H. Faecalibacterium Prausnitzii (ATCC 27766) Has Preventive and Therapeutic Effects on Chronic Unpredictable Mild Stress-Induced Depression-like and Anxiety-like Behavior in Rats. Psychoneuroendocrinology 2019, 104, 132–142. [Google Scholar] [CrossRef]
- Sudo, N.; Chida, Y.; Aiba, Y.; Sonoda, J.; Oyama, N.; Yu, X.N.; Kubo, C.; Koga, Y. Postnatal Microbial Colonization Programs the Hypothalamic-Pituitary-Adrenal System for Stress Response in Mice. J. Physiol. 2004, 558, 263–275. [Google Scholar] [CrossRef] [PubMed]
- McVey Neufeld, K.A.; Bienenstock, J.; Bharwani, A.; Champagne-Jorgensen, K.; Mao, Y.K.; West, C.; Liu, Y.; Surette, M.G.; Kunze, W.; Forsythe, P. Oral Selective Serotonin Reuptake Inhibitors Activate Vagus Nerve Dependent Gut-Brain Signalling. Sci. Rep. 2019, 9, 14290. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yarandi, S.S.; Kulkarni, S.; Saha, M.; Sylvia, K.E.; Sears, C.L.; Pasricha, P.J. Intestinal Bacteria Maintain Adult Enteric Nervous System and Nitrergic Neurons via Toll-like Receptor 2-Induced Neurogenesis in Mice. Gastroenterology 2020, 159, 200–213.e8. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, J.; Chen, Y. Regulation of Neurotransmitters by the Gut Microbiota and Effects on Cognition in Neurological Disorders. Nutrients 2021, 13, 2099. [Google Scholar] [CrossRef]
- Kaelberer, M.M.; Rupprecht, L.E.; Liu, W.W.; Weng, P.; Bohorquez, D.V. Neuropod Cells: The Emerging Biology of Gut-Brain Sensory Transduction. Annu. Rev. Neurosci. 2020, 43, 337–353. [Google Scholar] [CrossRef] [Green Version]
- Cohen Kadosh, K.; Basso, M.; Knytl, P.; Johnstone, N.; Lau, J.Y.F.; Gibson, G.R. Psychobiotic Interventions for Anxiety in Young People: A Systematic Review and Meta-Analysis, with Youth Consultation. Transl. Psychiatry 2021, 11, 352. [Google Scholar] [CrossRef]
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W.J. Psychobiotics and the Manipulation of Bacteria–Gut–Brain Signals. Trends Neurosci. 2016, 39, 763–781. [Google Scholar] [CrossRef] [Green Version]
- Venegas, D.P.; de La Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.M.; Faber, K.N.; Hermoso, M.A. Short Chain Fatty Acids (SCFAs)Mediated Gut Epithelial and Immune Regulation and Its Relevance for Inflammatory Bowel Diseases. Front. Immunol. 2019, 10, 277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karunaratne, T.B.; Okereke, C.; Seamon, M.; Purohit, S.; Wakade, C.; Sharma, A. Niacin and Butyrate: Nutraceuticals Targeting Dysbiosis and Intestinal Permeability in Parkinson’s Disease. Nutrients 2021, 13, 28. [Google Scholar] [CrossRef] [PubMed]
- Bourassa, M.W.; Alim, I.; Bultman, S.J.; Ratan, R.R. Butyrate, Neuroepigenetics and the Gut Microbiome: Can a High Fiber Diet Improve Brain Health? Neurosci. Lett. 2016, 625, 56–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Zheng, D.; Weng, F.; Jin, Y.; He, L. Sodium Butyrate Ameliorates the Cognitive Impairment of Alzheimer’s Disease by Regulating the Metabolism of Astrocytes. Psychopharmacology 2021, 239, 215–227. [Google Scholar] [CrossRef] [PubMed]
- Paley, E.L.; Perry, G. Towards an Integrative Understanding of TRNA Aminoacylation-Diet-Host-Gut Microbiome Interactions in Neurodegeneration. Nutrients 2018, 10, 410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wan, L.; Ge, W.R.; Zhang, S.; Sun, Y.L.; Wang, B.; Yang, G. Case-Control Study of the Effects of Gut Microbiota Composition on Neurotransmitter Metabolic Pathways in Children with Attention Deficit Hyperactivity Disorder. Front. Neurosci. 2020, 14, 127. [Google Scholar] [CrossRef] [Green Version]
- Hata, T.; Asano, Y.; Yoshihara, K.; Kimura-Todani, T.; Miyata, N.; Zhang, X.T.; Takakura, S.; Aiba, Y.; Koga, Y.; Sudo, N. Regulation of Gut Luminal Serotonin by Commensal Microbiota in Mice. PLoS ONE 2017, 12, e0180745. [Google Scholar] [CrossRef]
- Keskitalo, A.; Aatsinki, A.K.; Kortesluoma, S.; Pelto, J.; Korhonen, L.; Lahti, L.; Lukkarinen, M.; Munukka, E.; Karlsson, H.; Karlsson, L. Gut Microbiota Diversity but Not Composition Is Related to Saliva Cortisol Stress Response at the Age of 2.5 Months. Stress 2021, 24, 551–560. [Google Scholar] [CrossRef]
- Modasia, A.; Parker, A.; Jones, E.; Stentz, R.; Brion, A.; Goldson, A.; Defernez, M.; Wileman, T.; Ashley Blackshaw, L.; Carding, S.R. Regulation of Enteroendocrine Cell Networks by the Major Human Gut Symbiont Bacteroides Thetaiotaomicron. Front. Microbiol. 2020, 11, 575595. [Google Scholar] [CrossRef]
- Yoon, K.; Kim, N. Roles of Sex Hormones and Gender in the Gut Microbiota. J. Neurogastroenterol. Motil. 2021, 27, 314–325. [Google Scholar] [CrossRef]
- Shin, J.H.; Park, Y.H.; Sim, M.; Kim, S.A.; Joung, H.; Shin, D.M. Serum Level of Sex Steroid Hormone Is Associated with Diversity and Profiles of Human Gut Microbiome. Res. Microbiol. 2019, 170, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Uren Webster, T.M.; Rodriguez-Barreto, D.; Consuegra, S.; Garcia de Leaniz, C. Cortisol-Related Signatures of Stress in the Fish Microbiome. Front. Microbiol. 2020, 11, 1621. [Google Scholar] [CrossRef] [PubMed]
- Rosin, S.; Xia, K.; Azcarate-Peril, M.A.; Carlson, A.L.; Propper, C.B.; Thompson, A.L.; Grewen, K.; Knickmeyer, R.C. A Preliminary Study of Gut Microbiome Variation and HPA Axis Reactivity in Healthy Infants. Psychoneuroendocrinology 2021, 124, 105046. [Google Scholar] [CrossRef] [PubMed]
- Jahnke, J.R.; Roach, J.; Azcarate-Peril, M.A.; Thompson, A.L. Maternal Precarity and HPA Axis Functioning Shape Infant Gut Microbiota and HPA Axis Development in Humans. PLoS ONE 2021, 16, e0251782. [Google Scholar] [CrossRef]
- Hantsoo, L.; Jašarević, E.; Criniti, S.; McGeehan, B.; Tanes, C.; Sammel, M.D.; Elovitz, M.A.; Compher, C.; Wu, G.; Epperson, C.N. Childhood Adversity Impact on Gut Microbiota and Inflammatory Response to Stress during Pregnancy. Brain Behav. Immun. 2019, 75, 240–250. [Google Scholar] [CrossRef]
- Chassaing, B.; Kumar, M.; Baker, M.T.; Singh, V.; Vijay-Kumar, M. Mammalian Gut Immunity. Biomed. J. 2014, 37, 246–258. [Google Scholar]
- Teratani, T.; Mikami, Y.; Nakamoto, N.; Suzuki, T.; Harada, Y.; Okabayashi, K.; Hagihara, Y.; Taniki, N.; Kohno, K.; Shibata, S.; et al. The Liver–Brain–Gut Neural Arc Maintains the Treg Cell Niche in the Gut. Nature 2020, 585, 591–596. [Google Scholar] [CrossRef]
- Chung, T.W.; Li, Y.R.; Huang, W.Y.; Su, J.H.; Chan, H.L.; Lin, S.H.; Liu, C.S.; Lin, S.C.; Lin, C.C.; Lin, C.H. Sinulariolide Suppresses LPS-Induced Phenotypic and Functional Maturation of Dendritic Cells. Mol. Med. Rep. 2017, 16, 6992–7000. [Google Scholar] [CrossRef] [Green Version]
- Mol, S.; Hafkamp, F.M.J.; Varela, L.; Simkhada, N.; Taanman-Kueter, E.W.; Tas, S.W.; Wauben, M.H.M.; Kormelink, T.G.; de Jong, E.C. Efficient Neutrophil Activation Requires Two Simultaneous Activating Stimuli. Int. J. Mol. Sci. 2021, 22, 10106. [Google Scholar] [CrossRef]
- Liu, Y.; Sanderson, D.; Mian, M.F.; McVey Neufeld, K.-A.; Forsythe, P. Loss of Vagal Integrity Disrupts Immune Components of the Microbiota-Gut-Brain Axis and Inhibits the Effect of Lactobacillus Rhamnosus on Behavior and the Corticosterone Stress Response. Neuropharmacology 2021, 195, 108682. [Google Scholar] [CrossRef]
- Erny, D.; de Angelis, A.L.H.; Jaitin, D.; Wieghofer, P.; Staszewski, O.; David, E.; Keren-Shaul, H.; Mahlakoiv, T.; Jakobshagen, K.; Buch, T.; et al. Host Microbiota Constantly Control Maturation and Function of Microglia in the CNS. Nat. Neurosci. 2015, 18, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Li, K.; Li, X.; Xu, L.; Yang, Z. Sodium Butyrate Ameliorates the Impairment of Synaptic Plasticity by Inhibiting the Neuroinflammation in 5XFAD Mice. Chem. Biol. Interact. 2021, 341, 109452. [Google Scholar] [CrossRef] [PubMed]
- Nakkarach, A.; Foo, H.L.; Song, A.A.L.; Mutalib, N.E.A.; Nitisinprasert, S.; Withayagiat, U. Anti-Cancer and Anti-Inflammatory Effects Elicited by Short Chain Fatty Acids Produced by Escherichia coli Isolated from Healthy Human Gut Microbiota. Microb. Cell Factories 2021, 20, 36. [Google Scholar] [CrossRef] [PubMed]
- Stengel, A.; Taché, Y. Corticotropin-Releasing Factor Signaling and Visceral Response to Stress. In Proceedings of the Experimental Biology and Medicine; SAGE Publications Inc.: Thousand Oaks, CA, USA, 2010; Volume 235, pp. 1168–1178. [Google Scholar]
- Deleu, S.; Machiels, K.; Raes, J.; Verbeke, K.; Vermeire, S. Short Chain Fatty Acids and Its Producing Organisms: An Overlooked Therapy for IBD? EBioMedicine 2021, 66, 103293. [Google Scholar] [CrossRef]
- Fragiadakis, G.K.; Smits, S.A.; Sonnenburg, E.D.; van Treuren, W.; Reid, G.; Knight, R.; Manjurano, A.; Changalucha, J.; Dominguez-Bello, M.G.; Leach, J.; et al. Links between Environment, Diet, and the Hunter-Gatherer Microbiome. Gut Microbes 2019, 10, 216–227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Wu, W.; Wu, S.; Zheng, H.M.; Li, P.; Sheng, H.F.; Chen, M.X.; Chen, Z.H.; Ji, G.Y.; Zheng, Z.D.X.; et al. Linking Gut Microbiota, Metabolic Syndrome and Economic Status Based on a Population-Level Analysis. Microbiome 2018, 6, 172. [Google Scholar] [CrossRef] [PubMed]
- Moszak, M.; Szulińska, M.; Bogdański, P. You Are What You Eat—the Relationship between Diet, Microbiota, and Metabolic Disorders—A Review. Nutrients 2020, 12, 1096. [Google Scholar] [CrossRef] [Green Version]
- Cani, P.D. Human Gut Microbiome: Hopes, Threats and Promises. Gut 2018, 67, 1716–1725. [Google Scholar] [CrossRef]
- Collins, S.L.; Patterson, A.D. The Gut Microbiome: An Orchestrator of Xenobiotic Metabolism. Acta Pharm. Sin. B 2020, 10, 19–32. [Google Scholar] [CrossRef]
- Średnicka, P.; Juszczuk-Kubiak, E.; Wójcicki, M.; Akimowicz, M.; Roszko, M. Probiotics as a Biological Detoxification Tool of Food Chemical Contamination: A Review. Food Chem. Toxicol. 2021, 153, 112306. [Google Scholar] [CrossRef]
- Velmurugan, G. Gut Microbiota in Toxicological Risk Assessment of Drugs and Chemicals: The Need of Hour. Gut Microbes 2018, 9, 465–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kabir, E.R.; Rahman, M.S.; Rahman, I. A Review on Endocrine Disruptors and Their Possible Impacts on Human Health. Environ. Toxicol. Pharmacol. 2015, 40, 241–258. [Google Scholar] [CrossRef] [PubMed]
- Lazarevic, N.; Barnett, A.G.; Sly, P.D.; Knibbs, L.D. Statistical Methodology in Studies of Prenatal Exposure to Mixtures of Endocrine-Disrupting Chemicals: A Review of Existing Approaches and New Alternatives. Environ. Health Perspect. 2019, 127, 26001. [Google Scholar] [CrossRef] [PubMed]
- Walker, D.M.; Gore, A.C. Epigenetic Impacts of Endocrine Disruptors in the Brain. Front. Neuroendocrinol. 2017, 44, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Perera, B.P.U.; Faulk, C.; Svoboda, L.K.; Goodrich, J.M.; Dolinoy, D.C. The Role of Environmental Exposures and the Epigenome in Health and Disease. Environ. Mol. Mutagenesis 2020, 61, 176–192. [Google Scholar] [CrossRef] [Green Version]
- Lin, Z.; Wang, L.; Jia, Y.; Zhang, Y.; Dong, Q.; Huang, C. A Study on Environmental Bisphenol A Pollution in Plastics Industry Areas. Water Air Soil Pollut. 2017, 228, 98. [Google Scholar] [CrossRef]
- Joly, C.; Gay-Quéheillard, J.; Léké, A.; Chardon, K.; Delanaud, S.; Bach, V.; Khorsi-Cauet, H. Impact of Chronic Exposure to Low Doses of Chlorpyrifos on the Intestinal Microbiota in the Simulator of the Human Intestinal Microbial Ecosystem (SHIME®) and in the Rat. Environ. Sci. Pollut. Res. 2013, 20, 2726–2734. [Google Scholar] [CrossRef]
- García-Fabila, M.M.; Chávez, A.A.; Meza, J.C.S.; Montes, L.P.B.; García, A.R. Phthalates in the Diet of Mexican Children of School Age. Risk Analysis. Toxicol. Rep. 2020, 7, 1487–1494. [Google Scholar] [CrossRef]
- Meneguzzi, A.; Fava, C.; Castelli, M.; Minuz, P. Exposure to Perfluoroalkyl Chemicals and Cardiovascular Disease: Experimental and Epidemiological Evidence. Front. Endocrinol. 2021, 12, 850. [Google Scholar] [CrossRef]
- Papadopoulou, E.; Haug, L.S.; Sakhi, A.K.; Andrusaityte, S.; Basagaña, X.; Brantsaeter, A.L.; Casas, M.; Fernández-Barrés, S.; Grazuleviciene, R.; Knutsen, H.K.; et al. Diet as a Source of Exposure to Environmental Contaminants for Pregnant Women and Children from Six European Countries. Environ. Health Perspect. 2019, 127, 107005. [Google Scholar] [CrossRef]
- Alla, L.N.R.; Monshi, M.; Siddiqua, Z.; Shields, J.; Alame, K.; Wahls, A.; Akemann, C.; Meyer, D.; Crofts, E.J.; Saad, F.; et al. Detection of Endocrine Disrupting Chemicals in Danio Rerio and Daphnia Pulex: Step-One, Behavioral Screen. Chemosphere 2021, 271, 129442. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Rui, M.; Nie, Y.; Lu, G. Influence of Gastrointestinal Tract on Metabolism of Bisphenol A as Determined by in Vitro Simulated System. J. Hazard. Mater. 2018, 355, 111–118. [Google Scholar] [CrossRef] [PubMed]
- López-Moreno, A.; Torres-Sánchez, A.; Acuña, I.; Suárez, A.; Aguilera, M. Representative Bacillus sp. AM1 from Gut Microbiota Harbor Versatile Molecular Pathways for Bisphenol A Biodegradation. Int. J. Mol. Sci. 2021, 22, 4952. [Google Scholar] [CrossRef] [PubMed]
- Sirim, D.; Widmann, M.; Wagner, F.; Pleiss, J. Prediction and Analysis of the Modular Structure of Cytochrome P450 Monooxygenases. BMC Struct. Biol. 2010, 10, 34. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Yu, H.; Qin, H.; Long, Y.; Ye, J.; Qu, Y. Bisphenol A Degradation Pathway and Associated Metabolic Networks in Escherichia coli Harboring the Gene Encoding CYP450. J. Hazard. Mater. 2020, 388, 121737. [Google Scholar] [CrossRef]
- Esteves, F.; Rueff, J.; Kranendonk, M. The Central Role of Cytochrome P450 in Xenobiotic Metabolism—A Brief Review on a Fascinating Enzyme Family. J. Xenobiotics 2021, 11, 94–114. [Google Scholar] [CrossRef]
- Feng, D.; Zhang, H.; Jiang, X.; Zou, J.; Li, Q.; Mai, H.; Su, D.; Ling, W.; Feng, X. Bisphenol A Exposure Induces Gut Microbiota Dysbiosis and Consequent Activation of Gut-Liver Axis Leading to Hepatic Steatosis in CD-1 Mice. Environ. Pollut. 2020, 265, 114880. [Google Scholar] [CrossRef]
- Lai, K.P.; Chung, Y.T.; Li, R.; Wan, H.T.; Wong, C.K.C. Bisphenol A Alters Gut Microbiome: Comparative Metagenomics Analysis. Environ. Pollut. 2016, 218, 923–930. [Google Scholar] [CrossRef]
- Ni, Y.; Hu, L.; Yang, S.; Ni, L.; Ma, L.; Zhao, Y.; Zheng, A.; Jin, Y.; Fu, Z. Bisphenol A Impairs Cognitive Function and 5-HT Metabolism in Adult Male Mice by Modulating the Microbiota-Gut-Brain Axis. Chemosphere 2021, 282, 130952. [Google Scholar] [CrossRef]
- Abreu-Villaça, Y.; Levin, E.D. Developmental Neurotoxicity of Succeeding Generations of Insecticides. Environ. Int. 2017, 99, 55–77. [Google Scholar] [CrossRef] [Green Version]
- EFSA Statement on the Available Outcomes of the Human Health Assessment in the Context of the Pesticides Peer Review of the Active Substance Chlorpyrifos. EFSA J. 2019, 17, 5809. [CrossRef]
- Mendler, A.; Geier, F.; Haange, S.B.; Pierzchalski, A.; Krause, J.L.; Nijenhuis, I.; Froment, J.; Jehmlich, N.; Berger, U.; Ackermann, G.; et al. Mucosal-Associated Invariant T-Cell (MAIT) Activation Is Altered by Chlorpyrifos- and Glyphosate-Treated Commensal Gut Bacteria. J. Immunotoxicol. 2020, 17, 10–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reygner, J.; Condette, C.J.; Bruneau, A.; Delanaud, S.; Rhazi, L.; Depeint, F.; Abdennebi-Najar, L.; Bach, V.; Mayeur, C.; Khorsi-Cauet, H. Changes in Composition and Function of Human Intestinal Microbiota Exposed to Chlorpyrifos in Oil as Assessed by the SHIME® Model. Int. J. Environ. Res. Public Health 2016, 13, 1088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Réquilé, M.; Gonzàlez Alvarez, D.O.; Delanaud, S.; Rhazi, L.; Bach, V.; Depeint, F.; Khorsi-Cauet, H. Use of a Combination of in Vitro Models to Investigate the Impact of Chlorpyrifos and Inulin on the Intestinal Microbiota and the Permeability of the Intestinal Mucosa. Environ. Sci. Pollut. Res. 2018, 25, 22529–22540. [Google Scholar] [CrossRef] [PubMed]
- Li, J.W.; Fang, B.; Pang, G.F.; Zhang, M.; Ren, F.Z. Age- and Diet-Specific Effects of Chronic Exposure to Chlorpyrifos on Hormones, Inflammation and Gut Microbiota in Rats. Pestic. Biochem. Physiol. 2019, 159, 68–79. [Google Scholar] [CrossRef]
- U.S. EPA. Available online: https://www.epa.gov/sites/default/files/2019-08/documents/di-ethylhexyl_phthalate_117-81-7_proposeddesignation_082219.pdf (accessed on 1 December 2021).
- U.S. FDA. Available online: https://www.fda.gov/media/114001/download (accessed on 1 December 2021).
- U.S. FDA. Available online: https://www.gmp-compliance.org/files/guidemgr/UCM294086.pdf (accessed on 1 December 2021).
- Ramadan, M.; Cooper, B.; Posnack, N.G. Bisphenols and Phthalates: Plastic Chemical Exposures Can Contribute to Adverse Cardiovascular Health Outcomes. Birth Defects Res. 2020, 112, 1362–1385. [Google Scholar] [CrossRef]
- Fu, X.; Han, H.; Li, Y.; Xu, B.; Dai, W.; Zhang, Y.; Zhou, F.; Ma, H.; Pei, X. Di-(2-Ethylhexyl) Phthalate Exposure Induces Female Reproductive Toxicity and Alters the Intestinal Microbiota Community Structure and Fecal Metabolite Profile in Mice. Environ. Toxicol. 2021, 36, 1226–1242. [Google Scholar] [CrossRef]
- Wang, G.; Chen, Q.; Tian, P.; Wang, L.; Li, X.; Lee, Y.K.; Zhao, J.; Zhang, H.; Chen, W. Gut Microbiota Dysbiosis Might Be Responsible to Different Toxicity Caused by Di-(2-Ethylhexyl) Phthalate Exposure in Murine Rodents. Environ. Pollut. 2020, 261. [Google Scholar] [CrossRef]
- Zhao, T.X.; Wei, Y.X.; Wang, J.K.; Han, L.D.; Sun, M.; Wu, Y.H.; Shen, L.J.; Long, C.L.; Wu, S.; Wei, G.H. The Gut-Microbiota-Testis Axis Mediated by the Activation of the Nrf2 Antioxidant Pathway Is Related to Prepuberal Steroidogenesis Disorders Induced by Di-(2-Ethylhexyl) Phthalate. Environ. Sci. Pollut. Res. 2020, 27, 35261–35271. [Google Scholar] [CrossRef]
- Yu, Z.; Shi, Z.; Zheng, Z.; Han, J.; Yang, W.; Lu, R.; Lin, W.; Zheng, Y.; Nie, D.; Chen, G. DEHP Induce Cholesterol Imbalance via Disturbing Bile Acid Metabolism by Altering the Composition of Gut Microbiota in Rats. Chemosphere 2021, 263, 127959. [Google Scholar] [CrossRef]
- Bhargava, P.; Smith, M.D.; Mische, L.; Harrington, E.; Fitzgerald, K.C.; Martin, K.; Kim, S.; Reyes, A.A.; Gonzalez-Cardona, J.; Volsko, C.; et al. Bile Acid Metabolism Is Altered in Multiple Sclerosis and Supplementation Ameliorates Neuroinflammation. J. Clin. Investig. 2020, 130, 3467–3482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Podder, A.; Sadmani, A.H.M.A.; Reinhart, D.; Chang, N.-B.; Goel, R. Per and Poly-Fluoroalkyl Substances (PFAS) as a Contaminant of Emerging Concern in Surface Water: A Transboundary Review of Their Occurrences and Toxicity Effects. J. Hazard. Mater. 2021, 419, 126361. [Google Scholar] [CrossRef] [PubMed]
- Gardner, C.M.; Hoffman, K.; Stapleton, H.M.; Gunsch, C.K. Exposures to Semivolatile Organic Compounds in Indoor Environments and Associations with the Gut Microbiomes of Children. Environ. Sci. Technol. Lett. 2021, 8, 73–79. [Google Scholar] [CrossRef]
- Zhang, L.; Rimal, B.; Nichols, R.G.; Tian, Y.; Smith, P.B.; Hatzakis, E.; Chang, S.C.; Butenhoff, J.L.; Peters, J.M.; Patterson, A.D. Perfluorooctane Sulfonate Alters Gut Microbiota-Host Metabolic Homeostasis in Mice. Toxicology 2020, 431, 152365. [Google Scholar] [CrossRef]
- Shi, L.; Zheng, J.; Yan, S.; Li, Y.; Wang, Y.; Liu, X.; Xiao, C. Exposure to Perfluorooctanoic Acid Induces Cognitive Deficits via Altering Gut Microbiota Composition, Impairing Intestinal Barrier Integrity, and Causing Inflammation in Gut and Brain. J. Agric. Food Chem. 2020, 68, 13916–13928. [Google Scholar] [CrossRef] [PubMed]
- Coscia, A.; Bardanzellu, F.; Caboni, E.; Fanos, V.; Peroni, D.G. When a Neonate Is Born, so Is a Microbiota. Life 2021, 11, 148. [Google Scholar] [CrossRef]
- Parkin, K.; Christophersen, C.T.; Verhasselt, V.; Cooper, M.N.; Martino, D. Risk Factors for Gut Dysbiosis in Early Life. Microorganisms 2021, 9, 2066. [Google Scholar] [CrossRef]
- Berghuis, S.A.; Roze, E. Prenatal Exposure to PCBs and Neurological and Sexual/Pubertal Development from Birth to Adolescence. Curr. Probl. Pediatric Adolesc. Health Care 2019, 49, 133–159. [Google Scholar] [CrossRef] [Green Version]
- Sarkar, A.; Yoo, J.Y.; Valeria Ozorio Dutra, S.; Morgan, K.H.; Groer, M. The Association between Early-Life Gut Microbiota and Long-Term Health and Diseases. J. Clin. Med. 2021, 10, 459. [Google Scholar] [CrossRef]
- Calatayud Arroyo, M.; García Barrera, T.; Callejón Leblic, B.; Arias Borrego, A.; Collado, M.C. A Review of the Impact of Xenobiotics from Dietary Sources on Infant Health: Early Life Exposures and the Role of the Microbiota. Environ. Pollut. 2021, 269. [Google Scholar] [CrossRef]
- Kaur, S.; Sarma, S.J.; Marshall, B.L.; Liu, Y.; Kinkade, J.A.; Bellamy, M.M.; Mao, J.; Helferich, W.G.; Schenk, A.K.; Bivens, N.J.; et al. Developmental Exposure of California Mice to Endocrine Disrupting Chemicals and Potential Effects on the Microbiome-Gut-Brain Axis at Adulthood. Sci. Rep. 2020, 10, 10902. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.X.; Chen, Y.W.; Shih, M.K.; Tain, Y.L.; Yeh, Y.T.; Chiu, M.H.; Chang, S.K.C.; Hou, C.Y. Resveratrol Butyrate Esters Inhibit Bpa-induced Liver Damage in Male Offspring Rats by Modulating Antioxidant Capacity and Gut Microbiota. Int. J. Mol. Sci. 2021, 22, 5273. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.R.; Zheng, H.M.; Zhang, G.X.; Chen, F.L.; Chen, L.D.; Yang, Z.C. High Oscillospira Abundance Indicates Constipation and Low BMI in the Guangdong Gut Microbiome Project. Sci. Rep. 2020, 10, 9364. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Jiang, Y.; Xu, K.; Cui, M.; Ye, W.; Zhao, G.; Jin, L.; Chen, X. The Progress of Gut Microbiome Research Related to Brain Disorders. J. Neuroinflammation 2020, 17, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perez-Fernandez, C.; Morales-Navas, M.; Guardia-Escote, L.; Garrido-Cárdenas, J.A.; Colomina, M.T.; Giménez, E.; Sánchez-Santed, F. Long-Term Effects of Low Doses of Chlorpyrifos Exposure at the Preweaning Developmental Stage: A Locomotor, Pharmacological, Brain Gene Expression and Gut Microbiome Analysis. Food Chem. Toxicol. 2020, 135, 110865. [Google Scholar] [CrossRef] [PubMed]
- Guardia-Escote, L.; Basaure, P.; Biosca-Brull, J.; Cabré, M.; Blanco, J.; Pérez-Fernández, C.; Sánchez-Santed, F.; Domingo, J.L.; Colomina, M.T. APOE Genotype and Postnatal Chlorpyrifos Exposure Modulate Gut Microbiota and Cerebral Short-Chain Fatty Acids in Preweaning Mice. Food Chem. Toxicol. 2020, 135, 110872. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.N.; Yang, Y.C.S.H.; Lin, I.H.; Chen, Y.Y.; Lin, H.Y.; Wu, C.Y.; Su, Y.T.; Yang, Y.J.; Yang, S.N.; Suen, J.L. Phthalate Exposure Alters Gut Microbiota Composition and IgM Vaccine Response in Human Newborns. Food Chem. Toxicol. 2019, 132, 110700. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.; Menon, R.; Manteiga, S.; Alden, N.; Hunt, C.; Alaniz, R.C.; Lee, K.; Jayaraman, A. Environmental Chemical Diethylhexyl Phthalate Alters Intestinal Microbiota Community Structure and Metabolite Profile in Mice. Msystems 2019, 4, e00724-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, G.; Sun, S.; Wu, X.; Yang, S.; Wu, Y.; Zhao, J.; Zhang, H.; Chen, W. Intestinal Environmental Disorders Associate with the Tissue Damages Induced by Perfluorooctane Sulfonate Exposure. Ecotoxicol. Environ. Saf. 2020, 197, 110590. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Pan, R.; Liang, X.; Wu, X.; Wu, Y.; Zhang, H.; Zhao, J.; Chen, W. Perfluorooctanoic Acid-Induced Liver Injury Is Potentially Associated with Gut Microbiota Dysbiosis. Chemosphere 2021, 266, 129004. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, R.; Wang, S.; Solberg Woods, L.C.; Seshie, O.; Chung, S.T.; Shively, C.A.; Register, T.C.; Craft, S.; McClain, D.A.; Yadav, H. Comparative Microbiome Signatures and Short-Chain Fatty Acids in Mouse, Rat, Non-Human Primate, and Human Feces. Front. Microbiol. 2018, 9, 2897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, H.; Guo, R.; Zhu, J.; Wang, Q.; Ju, Y.; Xie, Y.; Zheng, Y.; Wang, Z.; Li, T.; Liu, Z.; et al. A Gene Catalogue of the Sprague-Dawley Rat Gut Metagenome. GigaScience 2018, 7, giy055. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manor, O.; Dai, C.L.; Kornilov, S.A.; Smith, B.; Price, N.D.; Lovejoy, J.C.; Gibbons, S.M.; Magis, A.T. Health and Disease Markers Correlate with Gut Microbiome Composition across Thousands of People. Nat. Commun. 2020, 11, 5206. [Google Scholar] [CrossRef] [PubMed]
- Olovo, C.V.; Huang, X.; Zheng, X.; Xu, M. Faecal Microbial Biomarkers in Early Diagnosis of Colorectal Cancer. J. Cell. Mol. Med. 2021, 25, 10783–10797. [Google Scholar] [CrossRef] [PubMed]
- Aho, V.T.E.; Houser, M.C.; Pereira, P.A.B.; Chang, J.; Rudi, K.; Paulin, L.; Hertzberg, V.; Auvinen, P.; Tansey, M.G.; Scheperjans, F. Relationships of Gut Microbiota, Short-Chain Fatty Acids, Inflammation, and the Gut Barrier in Parkinson’s Disease. Mol. Neurodegener. 2021, 16, 6. [Google Scholar] [CrossRef]
- King, C.H.; Desai, H.; Sylvetsky, A.C.; LoTempio, J.; Ayanyan, S.; Carrie, J.; Crandall, K.A.; Fochtman, B.C.; Gasparyan, L.; Gulzar, N.; et al. Baseline Human Gut Microbiota Profile in Healthy People and Standard Reporting Template. PLoS ONE 2019, 14, e0206484. [Google Scholar] [CrossRef] [Green Version]
- Agudelo-Ochoa, G.M.; Valdés-Duque, B.E.; Giraldo-Giraldo, N.A.; Jaillier-Ramírez, A.M.; Giraldo-Villa, A.; Acevedo-Castaño, I.; Yepes-Molina, M.A.; Barbosa-Barbosa, J.; Benítez-Paéz, A. Gut Microbiota Profiles in Critically Ill Patients, Potential Biomarkers and Risk Variables for Sepsis. Gut Microbes 2020, 12, 1707610. [Google Scholar] [CrossRef] [Green Version]
- Hawkins, K.G.; Casolaro, C.; Brown, J.A.; Edwards, D.A.; Wikswo, J.P. The Microbiome and the Gut-Liver-Brain Axis for Central Nervous System Clinical Pharmacology: Challenges in Specifying and Integrating In Vitro and In Silico Models. Clin. Pharmacol. Ther. 2020, 108, 929–948. [Google Scholar] [CrossRef]
- Bauer, E.; Thiele, I. From Network Analysis to Functional Metabolic Modeling of the Human Gut Microbiota. mSystems 2018, 3, e00209-17. [Google Scholar] [CrossRef] [Green Version]
- Altamirano, Á.; Saa, P.A.; Garrido, D. Inferring Composition and Function of the Human Gut Microbiome in Time and Space: A Review of Genome-Scale Metabolic Modelling Tools. Comput. Struct. Biotechnol. J. 2020, 18, 3897–3904. [Google Scholar] [CrossRef]
- Zimmermann-Kogadeeva, M.; Zimmermann, M.; Goodman, A.L. Insights from Pharmacokinetic Models of Host-Microbiome Drug Metabolism. Gut Microbes 2020, 11, 587–596. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thiele, I.; Clancy, C.M.; Heinken, A.; Fleming, R.M.T. Quantitative Systems Pharmacology and the Personalized Drug-Microbiota-Diet Axis. Curr. Opin. Syst. Biol. 2017, 4, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Jansma, J.; el Aidy, S. Understanding the Host-Microbe Interactions Using Metabolic Modeling. Microbiome 2021, 9, 16. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Figeys, D. Proteomics and Metaproteomics Add Functional, Taxonomic and Biomass Dimensions to Modeling the Ecosystem at the Mucosal-Luminal Interface. Mol. Cell. Proteom. 2020, 19, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Gutierrez, E.; Cotter, P.D. Relevance of Organ(s)-on-a-Chip Systems to the Investigation of Food-Gut Microbiota-Host Interactions. Crit. Rev. Microbiol. 2021, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Signore, M.A.; de Pascali, C.; Giampetruzzi, L.; Siciliano, P.A.; Francioso, L. Gut-on-Chip Microphysiological Systems: Latest Advances in the Integration of Sensing Strategies and Adoption of Mature Detection Mechanisms. Sens. Bio-Sens. Res. 2021, 33, 100443. [Google Scholar] [CrossRef]


| DEHP Exposure | ||||||
|---|---|---|---|---|---|---|
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Female ICR mice | 500 and 1500 mg/kg bw/day | 30 days | Turicibacter | Firmicutes | ↑ | [91] |
| Akkermansia | Verrucomicrobia | ↑ | ||||
| Romboutsia | Firmicutes | ↑ | ||||
| Bacteroidetes | ↓ | |||||
| Epsilonbacteraeota | ↓ | |||||
| Actinobacteria | ↓ | |||||
| Male SD rats | 0, 300, 1000 and 3000 mg/kg bw/day | 30 days | Proteobacteria | ↑ | [92] | |
| Mycoplasma | Tenericutes | ↑ | ||||
| Actinomyces | Actinobacteria | ↑ | ||||
| Porphyromonas | Bacteroidetes | ↑ | ||||
| Peptostreptococcaceae | Firmicutes | ↑ | ||||
| Sutterella | Proteobacteria | ↑ | ||||
| Male SD rats | 500 mg/kg bw/day | 14 days | Tenericutes (Mollicutes) | ↑ | [93] | |
| Allobaculum | Firmicutes | ↓ | ||||
| Female SD rats | 0.5 mg/kg bw/day | 23 weeks | Akkermansia | Verrucomicrobia | ↑ | [94] |
| Oscillibacter | Firmicutes | ↑ | ||||
| Pseudoflavonifractor | Firmicutes | ↑ | ||||
| Proteobacteria (Desulfovibrionaceae) | ↑ | |||||
| Firmicutes (Ruminococcaceae) | ↑ | |||||
| Acetivibrio | Firmicutes | ↓ | ||||
| Alloprevotella | Bacteroidetes | ↓ | ||||
| Barnesiella | Bacteroidetes | ↓ | ||||
| Clostridium | Firmicutes | ↓ | ||||
| Firmicutes (Lachnospiraceae) | ↓ | |||||
| Lactobacilllus | Firmicutes | ↓ | ||||
| Prevotella | Bacteroidetes | ↓ | ||||
| Roseburia | Firmicutes | ↓ | ||||
| Ruminococcus | Firmicutes | ↓ | ||||
| Bacteroidetes (Porphyromonadaceae) | ↓ | |||||
| PFAS Exposure | ||||||
|---|---|---|---|---|---|---|
| PFOS | ||||||
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Male C57BL/6J mice | 0, 0.003%, 0.006%, and 0.012% | 3 weeks | Clostridium | Firmicutes | ↑ | [98] |
| Streptococcus | Firmicutes | ↑ | ||||
| Bacteroidetes | ↑ | |||||
| Flavonifractor | Firmicutes | ↓ | ||||
| Alistipes | Bacteroidetes | ↓ | ||||
| PFOA | ||||||
| Cell Line/Species of Animal | Dose | Duration | Bacterial Genus | Bacterial Phylum | Impact * | Reference |
| Male C57BL/6J mice | 0, 0.5, 1, and 3 mg/kg (bw)/day | 35 days | Bacteroidetes | ↑ | [99] | |
| Akkermansia | Verrucomicrobia | ↓ | ||||
| Bifidobacterium | Actinobacteria | ↓ | ||||
| Ruminococcus | Firmicutes | ↓ | ||||
| Anoxybacillus | Firmicutes | ↓ | ||||
| Gemmiger | Firmicutes | ↓ | ||||
| Parabacteroides | Bacteroidetes | ↓ | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balaguer-Trias, J.; Deepika, D.; Schuhmacher, M.; Kumar, V. Impact of Contaminants on Microbiota: Linking the Gut–Brain Axis with Neurotoxicity. Int. J. Environ. Res. Public Health 2022, 19, 1368. https://doi.org/10.3390/ijerph19031368
Balaguer-Trias J, Deepika D, Schuhmacher M, Kumar V. Impact of Contaminants on Microbiota: Linking the Gut–Brain Axis with Neurotoxicity. International Journal of Environmental Research and Public Health. 2022; 19(3):1368. https://doi.org/10.3390/ijerph19031368
Chicago/Turabian StyleBalaguer-Trias, Jordina, Deepika Deepika, Marta Schuhmacher, and Vikas Kumar. 2022. "Impact of Contaminants on Microbiota: Linking the Gut–Brain Axis with Neurotoxicity" International Journal of Environmental Research and Public Health 19, no. 3: 1368. https://doi.org/10.3390/ijerph19031368
APA StyleBalaguer-Trias, J., Deepika, D., Schuhmacher, M., & Kumar, V. (2022). Impact of Contaminants on Microbiota: Linking the Gut–Brain Axis with Neurotoxicity. International Journal of Environmental Research and Public Health, 19(3), 1368. https://doi.org/10.3390/ijerph19031368









