Sarcopenia Is Associated with Metabolic Syndrome in Korean Adults Aged over 50 Years: A Cross-Sectional Study
Abstract
:1. Introduction
2. Materials and Methods
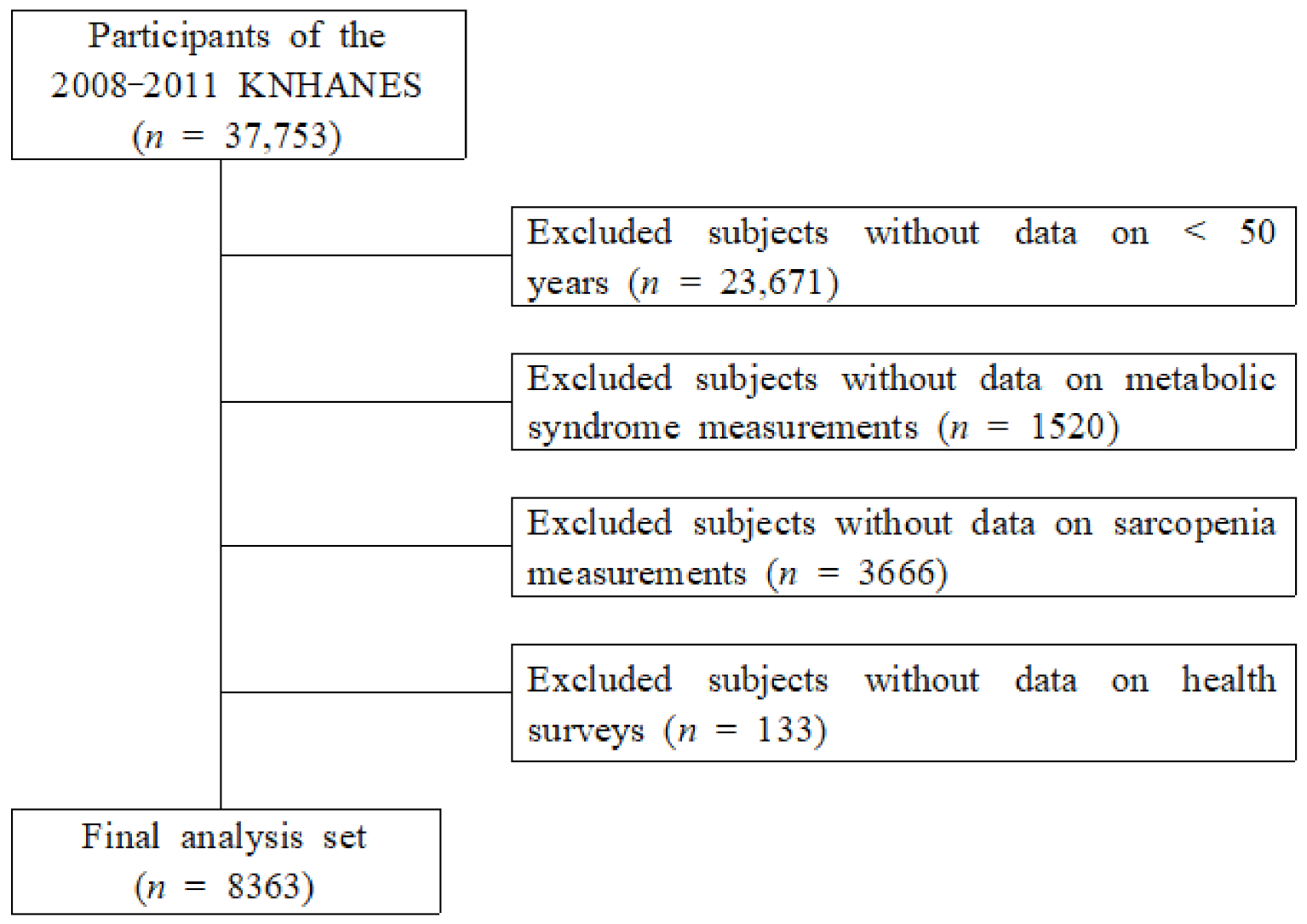
2.1. Data Source and Sampling
2.2. Measurement of Variables
2.2.1. Covariates
2.2.2. Sarcopenia
2.2.3. Metabolic Syndrome
2.3. Data Analysis
3. Results
3.1. Characteristics of Participants According to Sarcopenia and Sex
3.2. Characteristics of Participants According to MetS and Sex
3.3. MetS Components According to Sarcopenia and Sex
3.4. Odds Ratios for Sarcopenia According to MetS Stratified by Sex
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rosenberg, I.H. Sarcopenia: Origins and clinical relevance. J. Nutr. 1997, 127, 990s–991s. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, C.; Bai, L. Sarcopenia in the elderly: Basic and clinical issues. Geriatr. Gerontol. Int. 2012, 12, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Lim, S.; Choi, S.H.; Kim, J.H.; Shin, C.S.; Park, K.S.; Jang, H.C. Cardiometabolic implication of sarcopenia: The Korea National Health and Nutrition Examination Study (KNHANES) 2008–2010. IJC Metab. Endocr. 2014, 4, 63–69. [Google Scholar] [CrossRef] [Green Version]
- Janssen, I. Influence of sarcopenia on the development of physical disability: The Cardiovascular Health Study. J. Am. Geriatr. Soc. 2006, 54, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Bae, E.J.; Kim, Y.H. Factors Affecting Sarcopenia in Korean Adults by Age Groups. Osong. Public. Health Res. Perspect. 2017, 8, 169–178. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [Green Version]
- Janssen, I.; Shepard, D.S.; Katzmarzyk, P.T.; Roubenoff, R. The healthcare costs of sarcopenia in the United States. J. Am. Geriatr. Soc. 2004, 52, 80–85. [Google Scholar] [CrossRef]
- Isomaa, B.; Almgren, P.; Tuomi, T.; Forsén, B.; Lahti, K.; Nissén, M.; Taskinen, M.R.; Groop, L. Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 2001, 24, 683–689. [Google Scholar] [CrossRef] [Green Version]
- Gami, A.S.; Witt, B.J.; Howard, D.E.; Erwin, P.J.; Gami, L.A.; Somers, V.K.; Montori, V.M. Metabolic syndrome and risk of incident cardiovascular events and death: A systematic review and meta-analysis of longitudinal studies. J. Am. Coll. Cardiol. 2007, 49, 403–414. [Google Scholar] [CrossRef] [Green Version]
- Reaven, G.M.; Laws, A. Insulin resistance, compensatory hyperinsulinaemia, and coronary heart disease. Diabetologia 1994, 37, 948–952. [Google Scholar] [CrossRef]
- Samuel, V.T.; Shulman, G.I. The pathogenesis of insulin resistance: Integrating signaling pathways and substrate flux. J. Clin. Investig. 2016, 126, 12–22. [Google Scholar] [CrossRef] [Green Version]
- Doğan, M.H.; Karadag, B.; Ozyigit, T.; Kayaoglu, S.; Ozturk, A.O.; Altuntas, Y. Correlations between sarcopenia and hypertensive target organ damage in a Turkish cohort. Acta Clin. Belg. 2012, 67, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Khamseh, M.E.; Malek, M.; Aghili, R.; Emami, Z. Sarcopenia and diabetes: Pathogenesis and consequences. Br. J. Diabetes Vasc. Dis. 2011, 11, 230–234. [Google Scholar] [CrossRef]
- Waters, D.L.; Baumgartner, R.N. Sarcopenia and obesity. Clin. Geriatr. Med. 2011, 27, 401–421. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.H.; Hwang, H.J.; Han, C.H.; Son, B.S.; Kim, D.H.; Park, M.S. Association between sarcopenia and metabolic syndrome in chronic obstructive pulmonary disease: The Korea National Health and Nutrition Examination Survey (KNHANES) from 2008 to 2011. COPD 2015, 12, 82–89. [Google Scholar] [CrossRef]
- Moon, S.-S. Low skeletal muscle mass is associated with insulin resistance, diabetes, and metabolic syndrome in the Korean population: The Korea National Health and Nutrition Examination Survey (KNHANES) 2009–2010. Endocr. J. 2013, 61, EJ13-0244. [Google Scholar] [CrossRef] [Green Version]
- Ishii, S.; Tanaka, T.; Akishita, M.; Ouchi, Y.; Tuji, T.; Iijima, K.; Investigators, K.S. Metabolic syndrome, sarcopenia and role of sex and age: Cross-sectional analysis of Kashiwa cohort study. PLoS ONE 2014, 9, e112718. [Google Scholar] [CrossRef]
- Proctor, D.N.; Balagopal, P.; Nair, K.S. Age-related sarcopenia in humans is associated with reduced synthetic rates of specific muscle proteins. J. Nutr. 1998, 128, 351s–355s. [Google Scholar] [CrossRef] [Green Version]
- Keller, K.; Engelhardt, M. Strength and muscle mass loss with aging process. Age and strength loss. Muscles Ligaments Tendons J. 2013, 3, 346–350. [Google Scholar] [CrossRef]
- Petersen, K.F.; Dufour, S.; Savage, D.B.; Bilz, S.; Solomon, G.; Yonemitsu, S.; Cline, G.W.; Befroy, D.; Zemany, L.; Kahn, B.B.; et al. The role of skeletal muscle insulin resistance in the pathogenesis of the metabolic syndrome. Proc. Natl. Acad. Sci. USA 2007, 104, 12587–12594. [Google Scholar] [CrossRef] [Green Version]
- Balagopal, P.; Rooyackers, O.E.; Adey, D.B.; Ades, P.A.; Nair, K.S. Effects of aging on in vivo synthesis of skeletal muscle myosin heavy-chain and sarcoplasmic protein in humans. Am. J. Physiol. 1997, 273, E790-800. [Google Scholar] [CrossRef] [PubMed]
- Romeo, G.R.; Lee, J.; Shoelson, S.E. Metabolic syndrome, insulin resistance, and roles of inflammation--mechanisms and therapeutic targets. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1771–1776. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petersen, K.F.; Shulman, G.I. Pathogenesis of skeletal muscle insulin resistance in type 2 diabetes mellitus. Am. J. Cardiol. 2002, 90, 11g–18g. [Google Scholar] [CrossRef]
- Wu, H.; Ballantyne, C.M. Skeletal muscle inflammation and insulin resistance in obesity. J. Clin. Investig. 2017, 127, 43–54. [Google Scholar] [CrossRef]
- Eckardt, K.; Görgens, S.W.; Raschke, S.; Eckel, J. Myokines in insulin resistance and type 2 diabetes. Diabetologia 2014, 57, 1087–1099. [Google Scholar] [CrossRef] [PubMed]
- Roubenoff, R. Sarcopenic obesity: Does muscle loss cause fat gain? Lessons from rheumatoid arthritis and osteoarthritis. Ann. N. Y. Acad. Sci. 2000, 904, 553–557. [Google Scholar] [CrossRef]
- Reaven, G.M. Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef]
- Schrager, M.A.; Metter, E.J.; Simonsick, E.; Ble, A.; Bandinelli, S.; Lauretani, F.; Ferrucci, L. Sarcopenic obesity and inflammation in the InCHIANTI study. J. Appl. Physiol. 2007, 102, 919–925. [Google Scholar] [CrossRef]
- Walsh, K. Adipokines, myokines and cardiovascular disease. Circ. J. 2009, 73, 13–18. [Google Scholar] [CrossRef] [Green Version]
| Variables | Males | Females | ||||
|---|---|---|---|---|---|---|
| Sarcopenia (n = 681) | Non-Sarcopenia (n = 2946) | p | Sarcopenia (n = 951) | Non-Sarcopenia (n = 3785) | p | |
| Age (y) | 64.75 ± 0.41 | 59.93 ± 0.19 | <0.0001 | 66.10 ± 0.42 | 61.04 ± 0.19 | <0.0001 |
| MetS, n (%) | 331 (49.6) | 978 (35.0) | <0.0001 | 527 (55.1) | 1372 (35.9) | <0.0001 |
| BMI (kg/m2), mean (SD) | 25.01 ± 0.13 | 23.61 ± 0.07 | <0.0001 | 26.07 ± 0.16 | 23.81 ± 0.06 | <0.0001 |
| <18.5 (underweight), n (%) | 8 (1.2) | 120 (3.4) | 3 (0.7) | 118 (2.9) | ||
| <25 (normal-weight), n (%) | 346 (48.3) | 1982 (65.4) | 375 (40.8) | 2454 (64.7) | ||
| ≥25 (overweight), n (%) | 327 (50.5) | 844 (1.0) | 573 (58.5) | 1213 (32.4) | ||
| Smoking status, (%) (current-/ex-/non-smoker) | 56.4/31.1/12.4 | 60.3/23.4/16.2 | 0.002 | 6.5/2.7/90.9 | 7.2/1.6/91.2 | 0.200 |
| Drinking status (%) (current-/non-drinking) | 64.5/35.5 | 71.6/28.4 | 0.002 | 22.9/77.1 | 28.4/71.6 | 0.005 |
| Marital status, (%) (living with spouse) | 88.3 | 92.7 | 0.003 | 55.0 | 70.3 | <0.0001 |
| Income (individual) | ||||||
| Q1 (lowest) Q2 Q3 Q4 (highest) | 25.7 27.2 26.3 20.8 | 24.0 25.5 24.0 26.5 | 0.121 | 26.5 23.6 27.9 21.9 | 24.4 25.2 25.5 24.9 | 0.221 |
| Height (cm) | 160.85 ± 0.25 | 168.35 ± 0.12 | <0.0001 | 148.14 ± 0.22 | 155.07 ± 0.12 | <0.0001 |
| Weight (kg) | 64.91 ± 0.43 | 67.05 ± 0.24 | <0.0001 | 57.36 ± 0.41 | 57.36 ± 0.17 | 0.996 |
| Fasting glucose (mg/dL) | 109.79 ± 1.48 | 104.53 ± 0.55 | 0.001 | 103.81 ± 1.03 | 100.49 ± 0.45 | 0.003 |
| Waist circumference (cm) | 87.98 ± 0.37 | 84.95 ± 0.20 | <0.0001 | 86.42 ± 0.42 | 81.43 ± 0.21 | <0.0001 |
| Triglyceride | 183.29 ± 6.95 | 160.78 ± 3.33 | 0.005 | 148.50 ± 3.14 | 133.89 ± 1.78 | <0.0001 |
| Systolic BP (mmHg) | 130.44 ± 0.87 | 126.27 ± 0.44 | <0.0001 | 131.30 ± 0.74 | 126.06 ± 0.42 | <0.0001 |
| Diastolic BP (mmHg) | 79.90 ± 0.57 | 80.78 ± 0.29 | 0.143 | 77.94 ± 0.39 | 77.83 ± 0.23 | 0.802 |
| HDL-C | 43.82 ± 0.53 | 45.42 ± 0.28 | 0.005 | 47.94 ± 0.46 | 48.70 ± 0.26 | <0.0001 |
| Total cholesterol | 185.81 ± 1.97 | 187.59 ± 0.79 | 0.400 | 205.42 ± 1.36 | 199.76 ± 0.78 | <0.0001 |
| Skeletal muscle index | 0.74 ± 0.00 | 0.91 ± 0.00 | <0.0001 | 0.48 ± 0.00 | 0.61 ± 0.00 | <0.0001 |
| Variables | Males | Females | ||||
|---|---|---|---|---|---|---|
| MetS (n = 1309) | Non-MetS (n = 2318) | p | MetS (n = 1899) | Non-MetS (n = 2837) | p | |
| Age (y) | 60.38 ± 0.26 | 60.94 ± 0.22 | 0.097 | 64.47 ± 0.28 | 60.43 ± 0.21 | <0.0001 |
| Sarcopenia, n (%) | 331 (22.0) | 350 (13.4) | <0.0001 | 527 (27.3) | 424 (14.7) | <0.0001 |
| BMI (kg/m2), mean (SD) | 25.37 ± 0.10 | 22.93 ± 0.08 | <0.0001 | 25.80 ± 0.10 | 23.23 ± 0.06 | <0.0001 |
| <18.5 (underweight), n (%) | 9 (0.6) | 119 (4.5) | 12 (0.7) | 109 (3.6) | ||
| <25 (normal-weight), n (%) | 581 (43.1) | 1747 (74.1) | 759 (40.9) | 2070 (72.6) | ||
| ≥25 (overweight), n (%) | 719 (56.3) | 452 (21.4) | 1128 (58.4) | 658 (23.8) | ||
| Smoking status, (%) (current-/ex-/non-smoker) | 61.2/23.7/15.1 | 58.8/25.4/15.9 | 0.463 | 7.7/1.8/90.5 | 6.6/1.8/91.6 | 0.528 |
| Drinking status (%) (current-/non-drinking) | 71.6/28.4 | 69.7/30.3 | 0.344 | 23.0/77.0 | 30.2/69.8 | <0.0001 |
| Marital status, (%) (living with spouse) | 90.9 | 92.7 | 0.085 | 60.4 | 71.9 | <0.0001 |
| Income (individual) | ||||||
| Q1 (lowest) Q2 Q3 Q4 (highest) | 24.6 25.0 23.8 26.6 | 24.1 26.3 24.7 24.9 | 0.733 | 26.6 26.6 24.3 22.6 | 23.6 23.7 27.2 25.5 | 0.014 |
| Height (cm) | 167.63 ± 0.212 | 166.79 ± 0.16 | 0.002 | 153.30 ± 0.18 | 153,98 ± 0.15 | 0.003 |
| Weight (kg) | 71.39 ± 0.35 | 63.89 ± 0.25 | <0.0001 | 60.73 ± 0.26 | 55.14 ± 0.18 | <0.0001 |
| Fasting glucose (mg/dL) | 116.13 ± 0.21 | 98.99 ± 0.55 | <0.0001 | 111.38 ± 0.82 | 94.41 ± 0.37 | <0.0001 |
| Waist circumference (cm) | 90.30 ± 0.28 | 82.55 ± 0.21 | <0.0001 | 87.76 ± 0.26 | 78.89 ± 0.21 | <0.0001 |
| Triglyceride | 230.53 ± 5.13 | 125.05 ± 3.16 | <0.0001 | 184.88 ± 2.84 | 105.06 ± 1.29 | <0.0001 |
| Systolic BP (mmHg) | 133.58 ± 0.55 | 123.01 ± 0.47 | <0.0001 | 135.61 ± 0.46 | 121.48 ± 0.45 | <0.0001 |
| Diastolic BP (mmHg) | 84.31 ± 0.38 | 78.43 ± 0.30 | <0.0001 | 80.91 ± 0.29 | 75.83 ± 0.26 | <0.0001 |
| HDL-cholesterol | 39.79 ± 0.33 | 48.36 ± 0.32 | <0.0001 | 42.74 ± 0.25 | 52.37 ± 0.29 | <0.0001 |
| Total cholesterol | 188.97 ± 1.34 | 186.30 ± 0.85 | 0.088 | 202.74 ± 1.17 | 199.65 ± 0.84 | 0.030 |
| Skeletal muscle index | 0.86 ± 0.00 | 0.90 ± 0.00 | <0.0001 | 0.56 ± 0.00 | 0.60 ± 0.00 | <0.0001 |
| Variables | Males | p | Females | p | ||
|---|---|---|---|---|---|---|
| Sarcopenia | Non-Sarcopenia | Sarcopenia | Non-Sarcopenia | |||
| a High fasting glucose | 55.3 ± 2.2 | 44.9 ± 1.1 | <0.0001 | 46.2 ± 2.1 | 34.9 ± 0.9 | <0.0001 |
| b Abdominal obesity | 41.5 ± 2.4 | 27.5 ± 1.0 | <0.0001 | 54.3 ± 2.2 | 34.4 ± 1.1 | <0.0001 |
| c High triglyceride | 47.3 ± 2.1 | 38.4 ± 1.1 | <0.0001 | 40.3 ± 2.0 | 30.0 ± 1.0 | <0.0001 |
| d High blood pressure | 56.7 ± 2.5 | 48.4 ± 1.2 | 0.002 | 55.9 ± 2.1 | 44.9 ± 1.1 | <0.0001 |
| e Low HDL-C | 45.0 ± 2.5 | 36.9 ± 1.2 | 0.002 | 62.0 ± 1.9 | 59.3 ± 1.1 | 0.213 |
| MetS | 49.6 ± 2.3 | 35.0 ± 1.1 | <0.0001 | 55.1 ± 2.0 | 35.9 ± 1.0 | <0.0001 |
| MetS | OR (95% CI) | p | |
|---|---|---|---|
| Crude | Males | 1.827 (1.496–2.231) | <0.0001 |
| Females | 2.189 (1.818–2.635) | <0.0001 | |
| Total | 2.029 (1.766–2.331) | <0.0001 | |
| Model 1 | Males | 1.970 (1.597–2.430) | <0.0001 |
| Females | 1.792 (1.489–2.157) | <0.0001 | |
| Total | 1.849 (1.608–2.126) | <0.0001 | |
| Model 2 | Males | 1.972 (1.599–2.432) | <0.0001 |
| Females | 1.794 (1.489–2.160) | <0.0001 | |
| Total | 1.822 (1.586–2.095) | <0.0001 | |
| Model 3 | Males | 1.957 (1.587–2.413) | <0.0001 |
| Females | 1.779 (1.478–2.141) | <0.0001 | |
| Total | 1.822 (1.586–2.095) | <0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, D.-Y.; Shin, S. Sarcopenia Is Associated with Metabolic Syndrome in Korean Adults Aged over 50 Years: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2022, 19, 1330. https://doi.org/10.3390/ijerph19031330
Lee D-Y, Shin S. Sarcopenia Is Associated with Metabolic Syndrome in Korean Adults Aged over 50 Years: A Cross-Sectional Study. International Journal of Environmental Research and Public Health. 2022; 19(3):1330. https://doi.org/10.3390/ijerph19031330
Chicago/Turabian StyleLee, Do-Youn, and Sunghoon Shin. 2022. "Sarcopenia Is Associated with Metabolic Syndrome in Korean Adults Aged over 50 Years: A Cross-Sectional Study" International Journal of Environmental Research and Public Health 19, no. 3: 1330. https://doi.org/10.3390/ijerph19031330
APA StyleLee, D.-Y., & Shin, S. (2022). Sarcopenia Is Associated with Metabolic Syndrome in Korean Adults Aged over 50 Years: A Cross-Sectional Study. International Journal of Environmental Research and Public Health, 19(3), 1330. https://doi.org/10.3390/ijerph19031330






