The Biological Role of Vitamins in Athletes’ Muscle, Heart and Microbiota
Abstract
:1. Introduction
2. Role and Function of Vitamins in Biological Processes
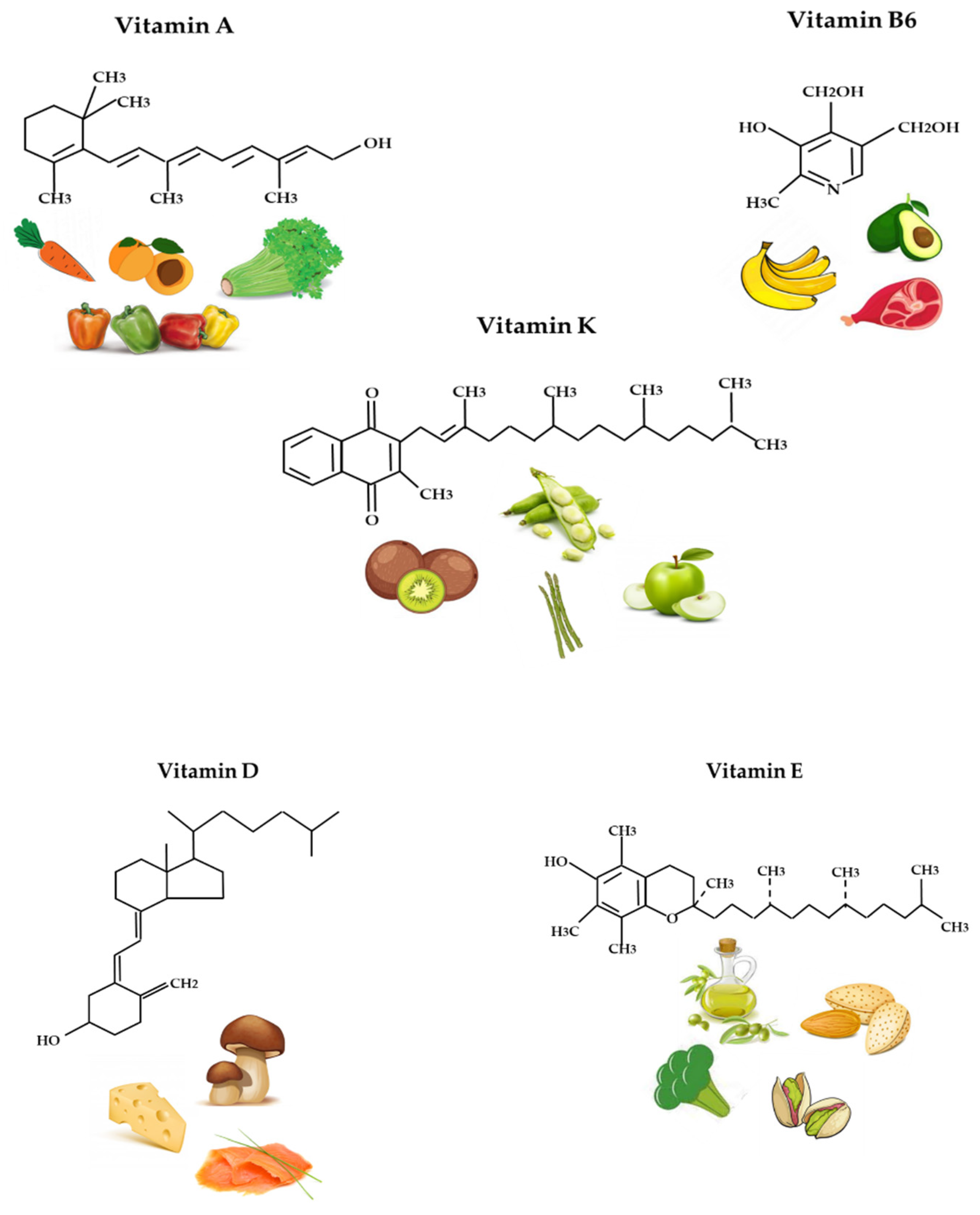
2.1. Vitamin A
2.2. Vitamin D
- Hypovitaminosis D: A diet lacking in vitamin D in combination with inadequate sun exposure causes osteomalacia in adults and rickets in children, which consists of the rarefaction of bone tissue [37,38]. Today, in the Western world, these conditions are extremely rare. However, vitamin D deficiency has become a global problem for the elderly population, and remains common in children and adults in less developed countries. The low content of calcifediol (25-hydroxy-vitamin D) derives mainly from poor sun exposure, and deficiency leads to reduced bone mineralization and damage to the skeleton, leading to the aforementioned diseases [88].
- Hypervitaminosis D is caused by increased intestinal absorption and bone resorption of calcium, with consequent hypercalcemia, which is easily identifiable by the increase in urination and thirst [89].
2.3. Vitamin E
2.4. Vitamin K
2.5. Vitamin B6
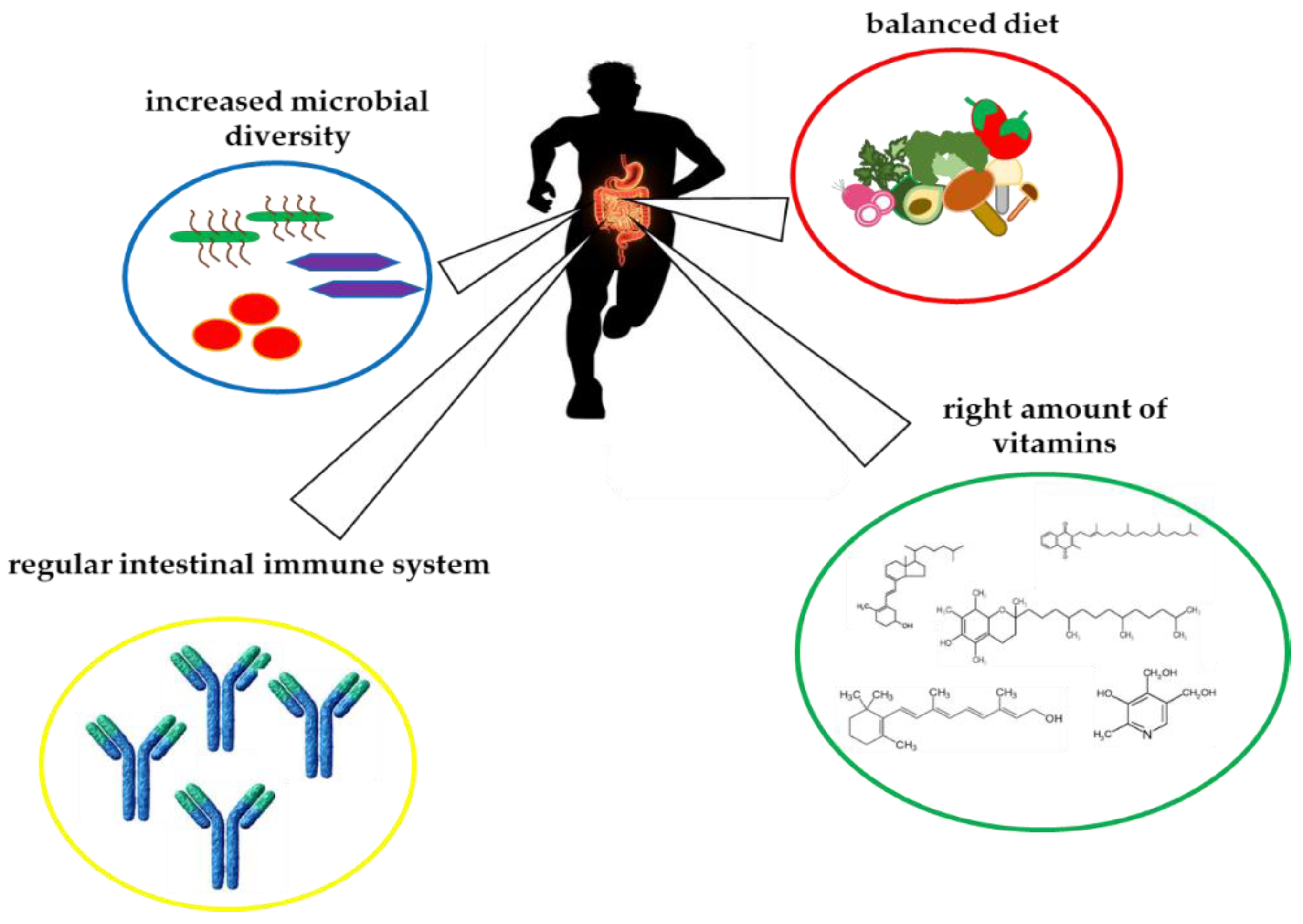
3. Crosstalk between Vitamins and Gut Microbiota in Athletes
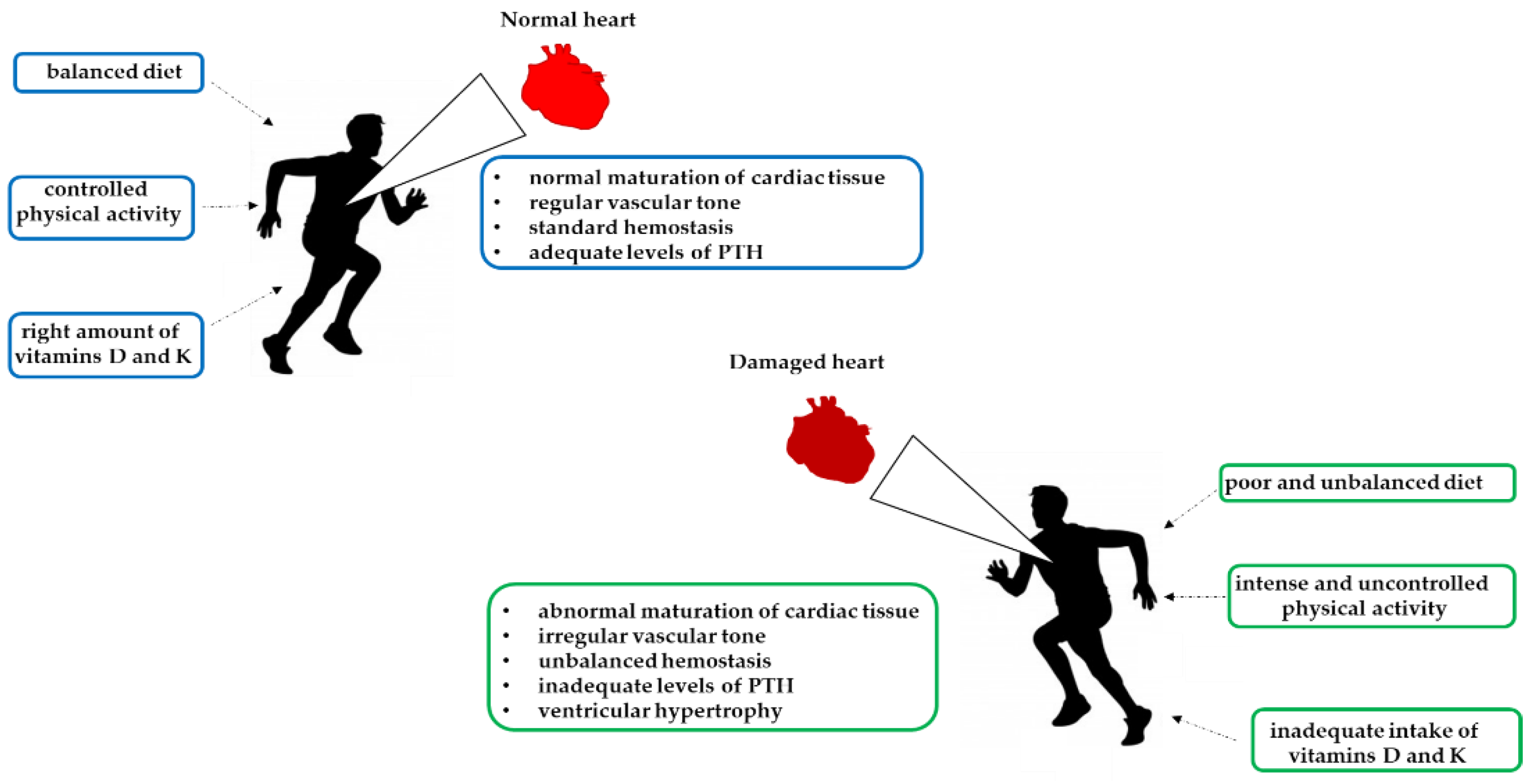
4. Vitamin Deficiency of Athletes’ Cardiac Disorders
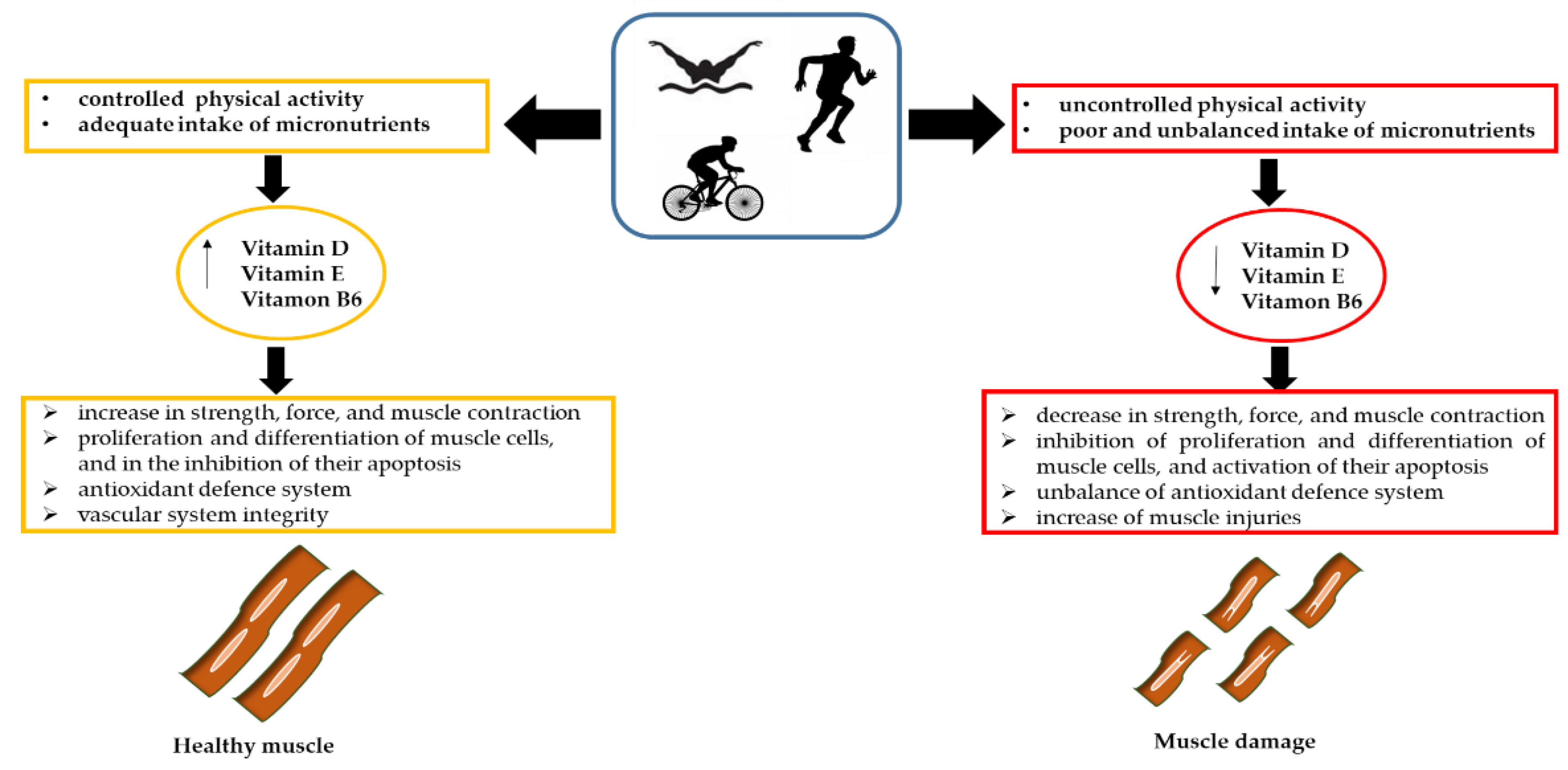
5. The Influence of Vitamins on Athletes’ Muscle Damage
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, F.F.; Barr, S.I.; McNulty, H.; Li, D.; Blumberg, J.B. Health effects of vitamin and mineral supplements. BMJ 2020, 369, m2511. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; National Academies Press: Washington, DC, USA, 1997. [Google Scholar]
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes and its Panel on Folate, Other B Vitamins, and Choline. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press: Washington, DC, USA, 1998. [Google Scholar]
- Institute of Medicine (US) Panel on Dietary Antioxidants and Related Compounds. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids; National Academies Press: Washington, DC, USA, 2000. [Google Scholar]
- Institute of Medicine (US) Panel on Micronutrients. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc; National Academies Press: Washington, DC, USA, 2001. [Google Scholar]
- Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium. Dietary Reference Intakes for Calcium and Vitamin D; Ross, A.C., Taylor, C.L., Yaktine, A.L., Del Valle, H.B., Eds.; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Chawla, J.; Kvarnberg, D. Hydrosoluble vitamins. Handb. Clin. Neurol. 2014, 120, 891–914. [Google Scholar] [PubMed]
- Stevens, S.L. Fat-soluble vitamins. Nurs. Clin. North Am. 2021, 56, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Dakshinamurti, K. Vitamins and their derivatives in the prevention and treatment of metabolic syndrome diseases (diabetes). Can. J. Physiol. Pharmacol. 2015, 93, 355–362. [Google Scholar] [CrossRef] [Green Version]
- Kallis, P.J.; Price, A.; Dosal, J.R.; Nichols, A.J.; Keri, J. A biologically based approach to acne and rosacea. J. Drugs Dermatol. 2018, 17, 611–617. [Google Scholar]
- Zinder, R.; Cooley, R.; Vlad, L.G.; Molnar, J.A. Vitamin A and wound healing. Nutr. Clin. Pract. 2019, 34, 839–849. [Google Scholar] [CrossRef]
- Huang, Z.R.; Lin, Y.K.; Fang, J.Y. Biological and pharmacological activities of squalene and related compounds: Potential uses in cosmetic dermatology. Molecules 2009, 14, 540–554. [Google Scholar] [CrossRef]
- Rúa-Figueroa Fernández de Larrinoa, I. What is new in systemic lupus erythematosus. Reumatol. ClíNica 2015, 11, 27–32. [Google Scholar] [CrossRef]
- Dommisch, H.; Kuzmanova, D.; Jönsson, D.; Grant, M.; Chapple, I. Effect of micronutrient malnutrition on periodontal disease and periodontal therapy. Periodontology 2000 2018, 78, 129–153. [Google Scholar] [CrossRef]
- Zasada, M.; Budzisz, E. Retinoids: Active molecules influencing skin structure formation in cosmetic and dermatological treatments. Postepy Dermatol. Alergol. 2019, 36, 392–397. [Google Scholar] [CrossRef] [Green Version]
- Palace, V.P.; Khaper, N.; Qin, Q.; Singal, P.K. Antioxidant potentials of vitamin A and carotenoids and their relevance to heart disease. Free. Radic. Biol. Med. 1999, 26, 746–761. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Prakash, M.; Apostolopoulos, V. The effects of vitamin B on the immune/cytokine network and their involvement in depression. Maturitas 2017, 96, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Huskisson, E.; Maggini, S.; Ruf, M. The role of vitamins and minerals in energy metabolism and well-being. J. Int. Med. Res. 2007, 35, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Katz, A.; Wang, Y.; Eck, P.; Kwon, O.; Lee, J.H.; Chen, S.; Corpe, C.; Dutta, A.; Dutta, S.K.; et al. Vitamin C as an antioxidant: Evaluation of its role in disease prevention. J. Am. Coll. Nutr. 2003, 22, 18–35. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G.; Stevens, J.F. Vitamins C and E: Beneficial effects from a mechanistic perspective. Free Radic. Biol. Med. 2011, 51, 1000–1013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carr, A.C.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aranow, C. Vitamin D and the immune system. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef] [Green Version]
- Umar, M.; Sastry, K.S.; Chouchane, A.I. Role of vitamin D beyond the skeletal function: A review of the molecular and clinical studies. Int. J. Mol. Sci. 2018, 19, 1618. [Google Scholar] [CrossRef] [Green Version]
- Botelho, J.; Machado, V.; Proença, L.; Delgado, A.S.; Mendes, J.J. Vitamin D deficiency and oral health: A comprehensive review. Nutrients 2020, 12, 1471. [Google Scholar] [CrossRef]
- Vardi, M.; Levy, N.S.; Levy, A.P. Vitamin E in the prevention of cardiovascular disease: The importance of proper patient selection. J. Lipid Res. 2013, 54, 2307–2314. [Google Scholar] [CrossRef] [Green Version]
- Van Ballegooijen, A.J.; Beulens, J.W. The role of vitamin K status in cardiovascular health: Evidence from observational and clinical studies. Curr. Nutr. Rep. 2017, 6, 197–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vermeer, C. Vitamin K: The effect on health beyond coagulation—An overview. Food Nutr. Res. 2012, 56, 5329. [Google Scholar] [CrossRef] [PubMed]
- Yasin, M.; Butt, M.S.; Yasmin, A.; Bashir, S. Chemical, antioxidant and sensory profiling of vitamin K-rich dietary sources. J. Korean Soc. Appl. Biol. Chem. 2014, 57, 153–160. [Google Scholar] [CrossRef]
- Pavlov, C.S.; Damulin, I.V.; Shulpekova, Y.O.; Andreev, E.A. Neurological disorders in vitamin B12 deficiency. Ter. Arkh. 2019, 91, 122–129. [Google Scholar] [CrossRef]
- Thomas-Valdés, S.; Tostes, M.D.G.V.; Anunciação, P.C.; da Silva, B.P.; Sant’Ana, H.M.P. Association between vitamin deficiency and metabolic disorders related to obesity. Crit. Rev. Food Sci. Nutr. 2017, 57, 3332–3343. [Google Scholar] [CrossRef]
- Allgrove, J.; Shaw, N.J. A practical approach to vitamin D deficiency and rickets. Endocr. Dev. 2015, 28, 119–133. [Google Scholar]
- Clifford, L.J.; Turnbull, A.M.J.; Denning, A.M. Reversible night blindness—A reminder of the increasing importance of vitamin A deficiency in the developed world. J. Optom. 2013, 6, 173–174. [Google Scholar] [CrossRef] [Green Version]
- Evain-Brion, D.; Porquet, D.; Thérond, P.; Fjellestad-Paulsen, A.; Grenèche, M.O.; François, L.; Czernichow, P. Vitamin A deficiency and nocturnal growth hormone secretion in short children. Lancet 1994, 343, 87–88. [Google Scholar] [CrossRef]
- Clagett-Dame, M.; Knutson, D. Vitamin A in reproduction and development. Nutrients 2011, 3, 385–428. [Google Scholar] [CrossRef] [Green Version]
- Blair, M.G.; Pigman, W.; Holly, H.L. Vitamin C and diseases of the connective tissues. I. Rheumatism 1957, 13, 52–88. [Google Scholar]
- Wang, K.; Jiang, H.; Li, W.; Qiang, M.; Dong, T.; Li, H. Role of vitamin C in skin diseases. Front. Physiol. 2018, 9, 819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahay, M.; Sahay, R. Rickets-vitamin D deficiency and dependency. Indian J. Endocrinol. Metab. 2012, 16, 164–176. [Google Scholar] [CrossRef] [PubMed]
- Sitta Mdo, C.; Cassis, S.V.; Horie, N.C.; Moyses, R.M.; Jorgetti, V.; Garcez-Leme, L.E. Osteomalacia and vitamin D deficiency in the elderly. Clinics 2009, 64, 156–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Staff, N.P.; Windebank, A.J. Peripheral neuropathy due to vitamin deficiency, toxins, and medications. Continuum 2014, 20, 1293–1306. [Google Scholar] [CrossRef] [Green Version]
- Armutcu, F.; Coskun, O.; Gürel, A.; Sahin, S.; Kanter, M.; Cihan, A.; Numanoglu, K.V.; Altinyazar, C. Vitamin E protects against acetone-induced oxidative stress in rat red blood cells. Cell Biol. Toxicol. 2005, 21, 53–60. [Google Scholar] [CrossRef]
- Sutor, A.H. Vitamin K deficiency bleeding in infants and children. Semin. Thromb. Hemost. 1995, 21, 317–329. [Google Scholar] [CrossRef]
- Kato, N. Role of vitamin B6 in skin health and diseases. In Handbook of Diet, Nutrition and the Skin; Preedy, V.R., Ed.; Human Health Handbooks; Wageningen Academic Publishers: Wageningen, The Netherlands, 2012. [Google Scholar]
- Young, G. Leg cramps. BMJ Clin. Evid. 2015, 2015, 1113. [Google Scholar]
- Razzaque, M.S. Can adverse effects of excessive vitamin D supplementation occur without developing hypervitaminosis D? J Steroid Biochem. Mol. Biol. 2018, 180, 81–86. [Google Scholar] [CrossRef]
- Silverman, A.K.; Ellis, C.N.; Voorhees, J.J. Hypervitaminosis a syndrome: A paradigm of retinoid side effects. J. Am. Acad. Dermatol. 1987, 16, 1027–1039. [Google Scholar] [CrossRef]
- Sünder, A.; Halle, I.; Flachowsky, G. Vitamin E hypervitaminosis in laying hens. Arch. Tierernahr. 1999, 52, 185–194. [Google Scholar] [CrossRef]
- Bastos Maia, S.; Rolland Souza, A.S.; Costa Caminha, M.F.; da Silva, S.L.; de Sá Barreto Luna Callou Cruz, R.; dos Santos, C.C.; Filho, M.B. Vitamin A and pregnancy: A narrative review. Nutrients 2019, 11, 681. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, J.; Zhang, Z.; Li, D.; Wong, K.E.; Zhang, Y.; Szeto, F.L.; Musch, M.W.; Li, Y.C. Loss of vitamin D receptor produces polyuria by increasing thirst. JASN 2008, 19, 2396–2405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sung, K.C.; Chang, Y.; Ryu, S.; Chung, H.K. High levels of serum vitamin D are associated with a decreased risk of metabolic diseases in both men and women, but an increased risk for coronary artery calcification in Korean men. Cardiovasc Diabetol. 2016, 15, 112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calderón-Ospina, C.A.; Nava-Mesa, M.O. B Vitamins in the nervous system: Current knowledge of the biochemical modes of action and synergies of thiamine, pyridoxine, and cobalamin. CNS Neurosci. Ther. 2020, 26, 5–13. [Google Scholar] [CrossRef] [Green Version]
- Allison, S. Ascorbic acid and risk of kidney stones. Nat. Rev. Nephrol. 2013, 9, 187. [Google Scholar] [CrossRef]
- Traber, M.G.; Buettner, G.R.; Bruno, R.S. The relationship between vitamin C status, the gut-liver axis, and metabolic syndrome. Redox Biol. 2019, 21, 101091. [Google Scholar] [CrossRef] [PubMed]
- Traber, M.G. Mechanisms for the prevention of vitamin E excess. J. Lipid Res. 2013, 54, 2295–2306. [Google Scholar] [CrossRef] [Green Version]
- Merli, G.J.; Fink, J. Vitamin K and thrombosis. Vitam. Horm. 2008, 78, 265–279. [Google Scholar] [PubMed]
- Scudiero, O.; Lombardo, B.; Brancaccio, M.; Mennitti, C.; Cesaro, A.; Fimiani, F.; Gentile, L.; Moscarella, E.; Amodio, F.; Ranieri, A.; et al. Exercise, immune system, nutrition, respiratory and cardiovascular diseases during COVID-19: A complex combination. Int. J. Environ. Res. Public Health 2021, 18, 904. [Google Scholar] [CrossRef]
- Jovic, T.H.; Ali, S.R.; Ibrahim, N.; Jessop, Z.M.; Tarassoli, S.P.; Dobbs, T.D.; Holford, P.; Thornton, C.A.; Whitaker, I.S. Could Vitamins Help in the Fight Against COVID-19? Nutrients 2020, 12, 2550. [Google Scholar] [CrossRef]
- Scudiero, O.; Pero, R.; Ranieri, A.; Terracciano, D.; Fimiani, F.; Cesaro, A.; Gentile, L.; Leggiero, E.; Laneri, S.; Moscarella, E.; et al. Childhood obesity: An overview of laboratory medicine, exercise and microbiome. Clin. Chem. Lab. Med. 2019, 58, 1385–1406. [Google Scholar] [CrossRef] [PubMed]
- Koduah, P.; Paul, F.; Dörr, J.M. Vitamin D in the prevention, prediction and treatment of neurodegenerative and neuroinflammatory diseases. EPMA J. 2017, 8, 313–325. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brancaccio, M.; Mennitti, C.; Cesaro, A.; Fimiani, F.; Moscarella, E.; Caiazza, M.; Gragnano, F.; Ranieri, A.; D’Alicandro, G.; Tinto, N.; et al. Dietary thiols: A potential supporting strategy against oxidative stress in heart failure and muscular damage during sports activity. Int. J. Environ. Res. Public Health 2020, 17, 9424. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. American college of sports medicine joint position statement. Nutrition and athletic performance. Med. Sci. Sport. Exerc. 2016, 48, 543–568. [Google Scholar]
- Brancaccio, M.; Mennitti, C.; Gentile, A.; Correale, L.; Buzzachera, C.F.; Ferraris, C.; Montomoli, C.; Frisso, G.; Borrelli, P.; Scudiero, O. Effects of the COVID-19 pandemic on job activity, dietary behaviours and physical activity habits of University Population of Naples, Federico II-Italy. Int. J. Environ. Res. Public Health 2021, 18, 1502. [Google Scholar] [CrossRef]
- Kerksick, C.M.; Wilborn, C.D.; Roberts, M.D.; Smith-Ryan, A.; Kleiner, S.M.; Jäger, R.; Collins, R.; Cooke, M.; Davis, J.N.; Galvan, E.; et al. ISSN exercise & sports nutrition review update: Research & recommendations. J. Int. Soc. Sports Nutr. 2018, 15, 38. [Google Scholar]
- Mennitti, C.; Brancaccio, M.; Gentile, L.; Ranieri, A.; Terracciano, D.; Cennamo, M.; La Civita, E.; Liotti, A.; D’Alicandro, G.; Mazzaccara, C.; et al. Athlete’s passport: Prevention of infections, inflammations, injuries and cardiovascular diseases. J. Clin. Med. 2020, 9, 2540. [Google Scholar] [CrossRef]
- Brancaccio, M.; Mennitti, C.; Laneri, S.; Franco, A.; De Biasi, M.G.; Cesaro, A.; Fimiani, F.; Moscarella, E.; Gragnano, F.; Mazzaccara, C.; et al. Methicillin-resistant staphylococcus aureus: Risk for general infection and endocarditis among athletes. Antibiotics 2020, 9, 332. [Google Scholar] [CrossRef]
- Scudiero, O.; Brancaccio, M.; Mennitti, C.; Laneri, S.; Lombardo, B.; De Biasi, M.G.; De Gregorio, E.; Pagliuca, C.; Colicchio, R.; Salvatore, P.; et al. Human defensins: A novel approach in the fight against skin colonizing staphylococcus aureus. Antibiotics 2020, 9, 198. [Google Scholar] [CrossRef] [Green Version]
- Pero, R.; Brancaccio, M.; Mennitti, C.; Gentile, L.; Arpino, S.; De Falco, R.; Leggiero, E.; Ranieri, A.; Pagliuca, C.; Colicchio, R.; et al. Urinary biomarkers: Diagnostic tools for monitoring athletes’ health status. Int. J. Environ. Res. Public Health 2020, 17, 6065. [Google Scholar] [CrossRef]
- Pero, R.; Brancaccio, M.; Mennitti, C.; Gentile, L.; Franco, A.; Laneri, S.; De Biasi, M.G.; Pagliuca, C.; Colicchio, R.; Salvatore, P.; et al. HNP-1 and HBD-1 as biomarkers for the immune systems of elite basketball athletes. Antibiotics 2020, 9, 306. [Google Scholar] [CrossRef] [PubMed]
- Mohr, A.E.; Jäger, R.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Townsend, J.R.; West, N.P.; Black, K.; Gleeson, M.; Pyne, D.B.; et al. The athletic gut microbiota. J. Int. Soc. Sports Nutr. 2020, 17, 24. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Liu, Y.; Qi, G.; Brand, D.; Zheng, S.G. Role of vitamin A in the immune system. J. Clin. Med. 2018, 7, 258. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russeau, A.-S.; Hininger, I.; Palazzetti, S.; Faure, H.; Roussel, A.-M.; Margaritis, I. Antioxidant vitamin status in high exposure to oxidative stress in competitive athletes. Br. J. Nutr. 2004, 92, 461–468. [Google Scholar]
- Parra, M.; Stahl, S.; Hellmann, H. Vitamin B₆ and its role in cell metabolism and physiology. Cells 2018, 7, 84. [Google Scholar] [CrossRef] [Green Version]
- Manore, M.M. Effect of physical activity on thiamine, riboflavin, and vitamin B-6 requirements. Am. J. Clin. Nutr. 2000, 72 (Suppl. 2), 598S–606S. [Google Scholar] [CrossRef] [Green Version]
- Larson-Meyer, D.E.; Willis, K.S. Vitamin D and athletes. Curr. Sports Med. Rep. 2010, 9, 220–226. [Google Scholar] [CrossRef] [Green Version]
- De la Puente Yagüe, M.; Collado Yurrita, L.; Ciudad Cabañas, M.J.; Cuadrado Cenzual, M.A. Role of vitamin D in athletes and their performance: Current concepts and new trends. Nutrients 2020, 12, 579. [Google Scholar] [CrossRef] [Green Version]
- Rizvi, S.; Raza, S.T.; Ahmed, F.; Ahmad, A.; Abbas, S.; Mahdi, F. The role of vitamin E in human health and some diseases. Sultan Qaboos Univ. Med. J. 2014, 14, e157–e165. [Google Scholar]
- Abdul Rahim, N.; Mohamad Shalan, N.A.A. The potential effects of vitamin E in sport performance. Int. J. Curr. Res. Biosci. Plantbiol. 2018, 5, 17–27. [Google Scholar] [CrossRef]
- Volpe, S.L. Vitamin K, osteoarthritis, and athletic performance. ACSM Health Fit. J. 2016, 20, 32–33. [Google Scholar] [CrossRef]
- Sumida, S.; Iwamoto, J.; Kamide, N.; Otani, T. Evaluation of bone, nutrition, and physical function in Shorinji Kempo athletes. Open Access J. Sport. Med. 2012, 3, 107–114. [Google Scholar] [PubMed] [Green Version]
- Mora, J.R.; Iwata, M.; von Andrian, U.H. Vitamin effects on the immune system: Vitamins A and D take centre stage. Nat. Rev. Immunol. 2008, 8, 685–698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ross, A.C.; Moran, N.E. Our current dietary reference intakes for vitamin A—Now 20 years old. Curr. Develop. Nutr. 2020, 4, nzaa096. [Google Scholar] [CrossRef]
- Penniston, K.L.; Tanumihardjo, S.A. The acute and chronic toxic effects of vitamin A. Am. J. Clin. Nutr. 2006, 83, 191–201. [Google Scholar] [CrossRef]
- Henning, P.; Conaway, H.H.; Lerner, U.H. Retinoid receptors in bone and their role in bone remodeling. Front. Endocrinol. 2015, 6, 31. [Google Scholar] [CrossRef] [Green Version]
- Olson, J.M.; Ameer, M.A.; Goyal, A. Vitamin A toxicity. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Oruch, R.; Pryme, I.F. The biological significance of vitamin A in humans: A review of nutritional aspects and clinical considerations. ScienceJet 2012, 1, 19. [Google Scholar]
- Stevens, G.A.; Bennett, J.E.; Hennocq, Q.; Lu, Y.; De-Regil, L.M.; Rogers, L.; Danaei, G.; Li, G.; White, R.A.; Flaxman, S.R.; et al. Trends and mortality effects of vitamin A deficiency in children in 138 low-income and middle-income countries between 1991 and 2013: A pooled analysis of population-based surveys. Lancet Glob. Health 2015, 3, e528–e536. [Google Scholar] [CrossRef] [Green Version]
- Veldurthy, V.; Wei, R.; Oz, L.; Dhawan, P.; Jeon, Y.H.; Christakos, S. Vitamin D, calcium homeostasis and aging. Bone Res. 2016, 4, 16041. [Google Scholar] [CrossRef] [Green Version]
- Institute of Medicine, Food and Nutrition Board. Dietary Reference Intakes for Calcium and Vitamin D; National Academy Press: Washington, DC, USA, 2010. [Google Scholar]
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: A worldwide problem with health consequences. Am. J. Clin. Nutr. 2008, 87, 1080S–1086S. [Google Scholar] [CrossRef] [Green Version]
- Marcinowska-Suchowierska, E.; Kupisz-Urbańska, M.; Łukaszkiewicz, J.; Płudowski, P.; Jones, G. Vitamin D toxicity—A clinical perspective. Front. Endocrinol. 2018, 9, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamilton, B. Vitamin D and human skeletal muscle. Scand. J. Med. Sci. Sports 2010, 20, 182–190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lappe, J.M.; Travers-Gustafson, D.; Michael Davies, K.; Recker, R.R.; Heaney, R.P. Vitamin D and calcium supplementation reduces cancer risk: Results of a randomized trial. Am. J. Clin. Nutr. 2007, 85, 1586–1591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cannell, J.J.; Hollis, B.W.; Sorenson, M.B.; Taft, T.N.; Anderson, J.J. Athletic performance and vitamin D. Med. Sci. Sports Exerc. 2009, 41, 1102–1110. [Google Scholar] [CrossRef]
- Rejnmark, L. Effects of vitamin d on muscle function and performance: A review of evidence from randomized controlled trials. Ther. Adv. Chronic. Dis. 2011, 2, 25–37. [Google Scholar] [CrossRef] [Green Version]
- Engin, K.N. Alpha-tocopherol: Looking beyond an antioxidant. Mol. Vis. 2009, 15, 855–860. [Google Scholar]
- Pazdro, R.; Burgess, J.R. The role of vitamin E and oxidative stress in diabetes complications. Mech. Age. Develop. 2010, 131, 276–286. [Google Scholar] [CrossRef]
- Saremi, A.; Arora, R. Vitamin E and cardiovascular disease. Am. J. Ther. 2010, 17, e56–e65. [Google Scholar] [CrossRef]
- Ricciarelli, R.; Argellati, F.; Pronzato, M.A.; Domenicotti, C. Vitamin E and neurodegenerative diseases. Mol. Asp. Med. 2007, 28, 591–606. [Google Scholar] [CrossRef]
- Alkhenizan, A.; Hafez, K. The role of vitamin E in the prevention of cancer: A meta-analysis of randomized controlled trials. Ann. Saudi. Med. 2007, 27, 409–414. [Google Scholar]
- Takasaki, M.; Yanagawa, K.; Shinozaki, K.; Fujii, H.; Shibuya, T.; Takeda, H.; Matsumiya, T.; Egashira, T. Relationship between aging and vitamin E. Niho. Rone. Igakkai Zasshi. 2002, 39, 494–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takanami, Y.; Iwane, H.; Kawai, Y.; Shimomitsu, T. Vitamin E supplementation and endurance exercise: Are there benefits? Sport. Med. 2000, 29, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Vitamin E, Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/ (accessed on 26 March 2021).
- Khadangi, F.; Azzi, A. Vitamin E—The next 100 years. IUBMB Life 2019, 71, 411–415. [Google Scholar] [CrossRef] [PubMed]
- Weber, P. Vitamin K and bone health. Nutrition 2001, 17, 880–887. [Google Scholar] [CrossRef]
- Buitenhuis, H.C.; Soute, B.A.; Vermeer, C. Comparison of the vitamins K1, K2 and K3 as cofactors for the hepatic vitamin K-dependent carboxylase. Biochim. Biophys. Acta 1990, 1034, 170–175. [Google Scholar] [CrossRef]
- Suttie, J.W.; Booth, S.L. Vitamin K. Adv. Nutr. 2011, 2, 440–441. [Google Scholar] [CrossRef]
- Vitamin K, Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/VitaminK-HealthProfessional/ (accessed on 29 March 2021).
- Rodríguez-Olleros Rodríguez, C.; Díaz Curiel, M. Vitamin K and bone health: A review on the effects of vitamin K deficiency and supplementation and the effect of non-vitamin K antagonist oral anticoagulants on different bone parameters. J. Osteoporos. 2019, 2019, 2069176. [Google Scholar] [CrossRef]
- Schwalfenberg, G.K. Vitamins K1 and K2: The emerging group of vitamins required for human health. J. Nutr. Metab. 2017, 2017, 6254836. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; Bhutani, J.; O’Keefe, J.H. The health benefits of vitamin K. Open Heart 2015, 2, e000300. [Google Scholar] [CrossRef]
- McFarlin, B.K.; Henning, A.L.; Venable, A.S. Oral consumption of vitamin K2 for 8 weeks associated with increased maximal cardiac output during exercise. Altern. Ther. Health Med. 2017, 23, 26–32. [Google Scholar]
- Craciun, A.M.; Wolf, J.; Knapen, M.H.; Brouns, F.; Vermeer, C. Improved bone metabolism in female elite athletes after vitamin K supplementation. Int. J. Sports Med. 1998, 19, 479–484. [Google Scholar] [CrossRef] [PubMed]
- Romo, A.J.; Liu, H.W. Mechanisms and structures of vitamin B(6)-dependent enzymes involved in deoxy sugar biosynthesis. Biochim. Biophys. Acta. 2011, 1814, 1534–1547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pregnolato, P.; Maranesi, M.; Marchetti, M.; Barzanti, V.; Bergami, R.; Tolomelli, B. Interaction among dietary vitamin B6, proteins and lipids: Effects on liver lipids in rats. Int. J. Vitam. Nutr. Res. 1994, 64, 263–269. [Google Scholar]
- Brown, M.J.; Ameer, M.A.; Beier, K. Vitamin B6 Deficiency; StatPearls: Treasure Island, FL, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK470579/ (accessed on 29 December 2021).
- Vitamin B6. Fact Sheet for Health Professional. Available online: https://ods.od.nih.gov/factsheets/VitaminB6-HealthProfessional/#en. (accessed on 26 March 2021).
- Bendich, A.; Cohen, M. Vitamin B6 safety issues. Ann. N. Y. Acad. Sci. 1990, 585, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Woolf, K.; Hahn, N.L.; Christensen, M.M.; Carlson-Phillips, A.; Hansen, C.M. Nutrition assessment of B-vitamins in highly active and sedentary women. Nutrients 2017, 9, 329. [Google Scholar] [CrossRef] [PubMed]
- Miodownik, C.; Lerner, V.; Vishne, T.; Sela, B.A.; Levine, J. High-dose vitamin B6 decreases homocysteine serum levels in patients with schizophrenia and schizoaffective disorders: A preliminary study. Clin. Neuropharmacol. 2007, 30, 13–17. [Google Scholar] [CrossRef]
- D’Argenio, V. Human microbiome acquisition and bioinformatic challenges in metagenomic studies. Int. J. Mol. Sci. 2018, 19, 383. [Google Scholar] [CrossRef] [Green Version]
- Clark, A.; Mach, N. Exercise-induced stress behavior, gut-microbiota-brain axis and diet: A systematic review for athletes. J. Int. Soc. Sports Nutr. 2016, 13, 43. [Google Scholar] [CrossRef] [Green Version]
- Allen, J.M.; Berg Miller, M.E.; Pence, B.D.; Whitlock, K.; Nehra, V.; Gaskins, H.R.; White, B.A.; Fryer, J.D.; Woods, J.A. Voluntary and forced exercise differentially alters the gut microbiome in C57BL/6J mice. J. Appl. Physiol. 1985 2015, 118, 1059–1066. [Google Scholar] [CrossRef] [Green Version]
- Sohail, M.U.; Yassine, H.M.; Sohail, A.; Thani, A.A.A. Impact of physical exercise on gut microbiome, inflammation, and the pathobiology of metabolic disorders. Rev. Diabet. Stud. 2019, 15, 35–48. [Google Scholar] [CrossRef] [Green Version]
- Campbell, S.C.; Wisniewski, P.J. Exercise is a novel promoter of intestinal health and microbial diversity. Exerc. Sport. Sci. Rev. 2017, 45, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Gommerman, J.L.; Rojas, O.L.; Fritz, J.H. Re-thinking the functions of IgA(þ) plasma cells. Gut Microbes 2014, 5, 652–662. [Google Scholar] [CrossRef] [Green Version]
- Bivona, G.; Agnello, L.; Ciaccio, M. The immunological implication of the new vitamin D metabolism. Cent. Eur. J. Immunol. 2018, 43, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Bashir, M.; Prietl, B.; Tauschmann, M.; Mautner, S.I.; Kump, P.K.; Treiber, G.; Wurm, P.; Gorkiewicz, G.; Hogenauer, C.; Pieber, T.R. Effects of high doses of vitamin D3 on mucosa-associated gut microbiome vary between regions of the human gastrointestinal tract. Eur. J. Nutr. 2016, 55, 1479–1489. [Google Scholar] [CrossRef] [Green Version]
- Selhub, J.; Byun, A.; Liu, Z.; Mason, J.B.; Bronson, R.T.; Crott, J.W. Dietary vitamin B6 intake modulates colonic inflammation in the IL10−/− model of inflammatory bowel disease. J. Nutr. Biochem. 2013, 24, 2138. [Google Scholar] [CrossRef] [Green Version]
- Ellis, J.L.; Karl, J.P.; Oliverio, A.M.; Fu, X.; Soares, J.W.; Wolfe, B.E.; Hernandez, C.J.; Mason, J.B.; Booth, S.L. Dietary vitamin K is remodeled by gut microbiota and influences community composition. Gut Microbes 2021, 13, 1–16. [Google Scholar] [CrossRef]
- Petersen, L.M.; Bautista, E.J.; Nguyen, H.; Hanson, B.M.; Chen, L.; Lek, S.H.; Sodergren, E.; Weinstock, G.M. Community characteristics of the gut microbiomes of competitive cyclists. Microbiome 2017, 5, 98. [Google Scholar] [CrossRef]
- O’Donovan, C.M.; Madigan, S.M.; Garcia-Perez, I.; Rankin, A.; O’ Sullivan, O.; Cotter, P.D. Distinct microbiome composition and metabolome exists across subgroups of elite Irish athletes. J. Sci. Med. Sport. 2020, 23, 63–68. [Google Scholar] [CrossRef] [Green Version]
- Bressa, C.; Bailén-Andrino, M.; Pérez-Santiago, J.; González-Soltero, R.; Pérez, M.; Montalvo-Lominchar, M.G.; Maté-Muñoz, J.L.; Domínguez, R.; Moreno, D.; Larrosa, M. Differences in gut microbiota profile between women with active lifestyle and sedentary women. PLoS ONE 2017, 12, e0171352. [Google Scholar] [CrossRef] [Green Version]
- Bermon, S.; Petriz, B.; Kajėnienė, A.; Prestes, J.; Castell, L.; Franco, O.L. The microbiota: An exercise immunology perspective. Exerc. Immunol. Rev. 2015, 21, 70–79. [Google Scholar]
- Derman, W.E.; Lambert, M.I. The differential diagnosis and clinical approach to the athlete with chronic fatigue. Int. Fed. Sport. Med. 2000, 1, 3. Available online: https://hdl.handle.net/10520/EJC48433 (accessed on 1 January 2000).
- Heap, L.C.; Peters, T.J.; Wessely, S. Vitamin B status in patients with chronic fatigue syndrome. J. R. Soc. Med. 1999, 92, 183–185. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miwa, K.; Fujita, M. Fluctuation of serum vitamin E (alpha-tocopherol) concentrations during exacerbation and remission phases in patients with chronic fatigue syndrome. Heart Vessels 2010, 25, 319–323. [Google Scholar] [CrossRef]
- Grant, J.E.; Veldee, M.S.; Buchwald, D. Analysis of dietary intake and selected nutrient concentrations in patients with chronic fatigue syndrome. J. Am. Diet. Assoc. 1996, 96, 383–386. [Google Scholar] [CrossRef]
- Rossi, M.; Amaretti, A.; Raimondi, S. Folate production by probiotic bacteria. Nutrients 2011, 3, 118–134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barkhidarian, B.; Roldos, L.; Iskandar, M.M.; Saedisomeolia, A.; Kubow, S. Probiotic supplementation and micronutrient status in healthy subjects: A systematic review of clinical trials. Nutrients 2021, 13, 3001. [Google Scholar] [CrossRef] [PubMed]
- Allison, R.J.; Close, G.L.; Farooq, A.; Riding, N.R.; Salah, O.; Hamilton, B.; Wilson, M.G. Severely vitamin D-deficient athletes present smaller hearts than sufficient athletes. Eur. J. Prev. Cardiol. 2015, 22, 535–542. [Google Scholar] [CrossRef]
- Hilger, J.; Friedel, A.; Herr, R.; Rausch, T.; Roos, F.; Wahl, D.A.; Pierroz, D.D.; Weber, P.; Hoffmann, K. A systematic review of vitamin D status in populations worldwide. Br. J. Nutr. 2014, 111, 23–45. [Google Scholar] [CrossRef] [Green Version]
- Lombardi, G.; Vitale, J.A.; Logoluso, S.; Logoluso, G.; Cocco, N.; Cocco, G.; Cocco, A.; Banfi, G. Circannual rhythm of plasmatic vitamin D levels and the association with markers of psychophysical stress in a cohort of Italian professional soccer players. Chronobiol. Int. 2017, 34, 471–479. [Google Scholar] [CrossRef]
- Chatard, J.C.; Mujika, I.; Goiriena, J.J.; Carré, F. Screening young athletes for prevention of sudden cardiac death: Practical recommendations for sports physicians. Scand. J. Med. Sci. Sports 2016, 26, 362–374. [Google Scholar] [CrossRef]
- Chen, S.; Glenn, D.J.; Ni, W.; Grigsby, C.L.; Olsen, K.; Nishimoto, M.; Law, C.S.; Gardner, D.G. Expression of the vitamin d receptor is increased in the hypertrophic heart. Hypertension 2008, 52, 1106–1112. [Google Scholar] [CrossRef] [Green Version]
- Cozzolino, M.; Ketteler, M.; Zehnder, D. The vitamin D system: A crosstalk between the heart and kidney. Eur. J. Heart Fail. 2010, 12, 1031–1041. [Google Scholar] [CrossRef] [Green Version]
- Girgis, C.M.; Mokbel, N.; Cha, K.M.; Houweling, P.J.; Abboud, M.; Fraser, D.R.; Mason, R.S.; Clifton-Bligh, R.J.; Gunton, J.E. The vitamin D receptor (VDR) is expressed in skeletal muscle of male mice and modulates 25-hydroxyvitamin D (25OHD) uptake in myofibers. Endocrinology 2014, 155, 3227–3237. [Google Scholar] [CrossRef] [Green Version]
- Pilz, S.; Tomaschitz, A.; Drechsler, C.; Dekker, J.M.; März, W. Vitamin D deficiency and myocardial diseases. Mol. Nutr. Food Res. 2010, 54, 1103–1113. [Google Scholar] [CrossRef]
- Willis, K.S.; Smith, D.T.; Broughton, K.S.; Larson-Meyer, D.E. Vitamin D status and biomarkers of inflammation in runners. Open Access J. Sports Med. 2012, 3, 35–42. [Google Scholar]
- Lavie, C.J.; Dinicolantonio, J.J.; Milani, R.V.; O’Keefe, J.H. Vitamin D and cardiovascular health. Circulation 2013, 128, 2404–2406. [Google Scholar] [CrossRef] [Green Version]
- Chandra, N.; Bastiaenen, R.; Papadakis, M.; Sharma, S. Sudden cardiac death in young athletes: Practical challenges and diagnostic dilemmas. J. Am. Coll. Cardiol. 2013, 61, 1027–1040. [Google Scholar] [CrossRef] [Green Version]
- Ragozzino, E.; Brancaccio, M.; Di Costanzo, A.; Scalabri, F.; Andolfi, G.; Wanderlingh, L.G.; Patriarca, E.J.; Minchiotti, G.; Altamura, S.; Varrone, F.; et al. 6-Bromoindirubin- 3’-oxime intercepts GSK3 signaling to promote and enhance skeletal muscle differentiation affecting miR-206 expression in mice. Sci. Rep. 2019, 9, 18091. [Google Scholar] [CrossRef]
- Abrams, G.D.; Feldman, D.; Safran, M.R. Effects of vitamin D on skeletal muscle and athletic performance. J. Am. Acad. Orthop. Surg. 2018, 26, 278–285. [Google Scholar] [CrossRef]
- Zsiazek, A.; Zagrodna, A.; Slowinska-Lisowska, M. Vitamin D, skeletal muscle function and athletic performance in athletes—A narrative review. Nutrients 2019, 11, 1800. [Google Scholar] [CrossRef] [Green Version]
- Gunton, J.E.; Girgis, C.M. Vitamin D and muscle. Bone Rep. 2018, 8, 163–167. [Google Scholar] [CrossRef]
- Zittermann, A. Vitamin D in preventive medicine: Are we ignoring the evidence? Br. J. Nutr. 2003, 89, 552–572. [Google Scholar] [CrossRef]
- Dzik, K.P.; Kaczor, J.J. Mechanisms of vitamin D on skeletal muscle function: Oxidative stress, energy metabolism and anabolic state. Eur. J. Appl. Phys. 2019, 119, 825–839. [Google Scholar] [CrossRef] [Green Version]
- Bhat, M.; Kalam, R.; Qadri, S.S.Y.H.; Madabushi, S.; Ismail, A. Vitamin D deficiency-induced muscle wasting occurs through the ubiquitin proteasome pathway and is partially corrected by calcium in male rats. Endocrinology 2013, 154, 4018–4029. [Google Scholar] [CrossRef] [Green Version]
- Farrokhyar, F.; Sivakumar, G.; Savage, K.; Koziarz, A.; Jamshidi, S.; Ayeni, O.R.; Peterson, D.; Bhandari, M. Effects of vitamin D supplementation on serum 25- hydroxyvitamin D concentrations and physical performance in athletes: A systematic review and meta-analysis of randomized controlled trials. Sport. Med. 2017, 47, 2323–2339. [Google Scholar] [CrossRef]
- Ogan, D.; Pritchett, K. Vitamin D and the athlete: Risks, recommendations, and benefits. Nutrients 2013, 5, 1856–1868. [Google Scholar] [CrossRef] [Green Version]
- Powers, S.K.; Jackson, M.J. Exercise-induced oxidative stress: Cellular mechanisms and impact on muscle force production. Physiol. Rev. 2008, 88, 1243–1276. [Google Scholar] [CrossRef] [Green Version]
- Phung, C.D.; Ezieme, J.A.; Turrens, J.F. Hydrogen peroxide metabolism in skeletal muscle mitochondria. Arch. Biochem. Biophys. 1994, 315, 479–482. [Google Scholar] [CrossRef]
- Sacheck, J.M.; Blumberg, J.B. Role of vitamin E and oxidative stress in exercise. Nutrition 2001, 17, 809–814. [Google Scholar] [CrossRef]
- Davies, K.J.A.; Quintanilha, A.T.; Brooks, G.A.; Parker, L. Free radicals and tissue damage produced by exercise. Biochem. Biophys. Res. Commun. 1982, 107, 1198–1205. [Google Scholar] [CrossRef]
- Gornicka, M.; Ciecierska, A.; Hamulka, J.; Drywien, M.E.; Frackiewicz, J.; Gornicki, K.; Wawrzyniak, A. α-tocopherol protects the heart, muscles, and testes from lipid peroxidation in growing male rats subjected to physical efforts. Oxid. Med. Cell. Longev. 2019, 2019, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robertson, J.D.; Maughan, R.J.; Duthie, G.G.; Morrice, P.C. Increased blood antioxidant systems of runners in response to training load. Clin. Sci. 1991, 80, 611. [Google Scholar] [CrossRef] [PubMed]
- Meydani, M.; Fielding, R.A.; Cannon, J.G.; Blumberg, J.B.; Evans, W.J. Muscle uptake of vitamin E and its association with muscle fiber type. J. Nutr. Biochem. 1997, 8, 74–78. [Google Scholar] [CrossRef]
- Maxwell, S.R.J.; Jakeman, P.; Thomason, H.; Leguen, C.; Thorpe, G.H.G. Changes in plasma antioxidant status during eccentric exercise and the effect on vitamin supplementation. Free Radic. Res. Commun. 1993, 19, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Kaikkonen, J.; Kosonen, L.; Nyyssonen, K.; Porkkala-Sarataho, E.; Salonen, R.; Korpela, H.; Salonen, J. Effect of combined coenzyme Q10 and d-Alpha-tocopheryl acetate supplementation on exercise-induced lipid peroxidation and muscular damage: A placebo-controlled double-blind study in marathon runners. Free Rad. Res. 1998, 29, 85–92. [Google Scholar] [CrossRef]
- Manore, M.M. Vitamin B-6 and exercise. Int. J. Sport Nutr. 1994, 4, 89–103. [Google Scholar] [CrossRef]
- Steed, M.M.; Tyagi, S.C. Mechanisms of cardiovascular remodeling in hyperhomocysteinemia. Antioxid. Redox Signal. 2011, 15, 1927–1943. [Google Scholar] [CrossRef] [Green Version]
- Kanwar, Y.; Manaligod, J.; Wong, P. Morphologic studies in a patient with homocystinuria due to 5,10-methylenetetrahydrofolate reductase deficiency. Pediatr. Res. 1976, 10, 598–609. [Google Scholar] [CrossRef] [Green Version]
- Veeranki, S.; Tyagi, S.C. Defective homocysteine metabolism: Potential implications for skeletal muscle malfunction. Int. J. Mol. Sci. 2013, 14, 15074–15091. [Google Scholar] [CrossRef] [Green Version]
- Woolf, K.; Manore, M.M. B-Vitamins and Exercise: Does Exercise Alter Requirements? Int. J. Sport Nutr. Exerc. Metab. 2006, 16, 453–484. [Google Scholar] [CrossRef]
- Frisso, G. Vitamine. In Nutrizione Umana; Edizioni Idelson-Gnocchi: Naples, Italy, 2017; pp. 33–58. [Google Scholar]
- Limongelli, G.; Nunziato, M.; D’Argenio, V.; Esposito, M.V.; Monda, E.; Mazzaccara, C.; Caiazza, M.; D’Aponte, A.; D’Andrea, A.; Bossone, E.; et al. Yield and clinical significance of genetic screening in elite and amateur athletes. Eur. J. Prev. Cardiol. 2020, 2, 2047487320934265. [Google Scholar] [CrossRef] [PubMed]
| Micronutrient | Physiological Functions | References |
|---|---|---|
| Vitamin A | Improvement of vision, antioxidant, maintenance of the immune system, maintenance of healthy skin. | [15,16] |
| Vitamin B | Energy production, nucleic acid, protein, sugar and fat metabolism, maintenance of the immune system, psychological functions. | [17,18] |
| Vitamin C | Antioxidant, wound healing, maintenance of the immune system, maintenance of healthy skin, teeth, and gums. | [19,20,21] |
| Vitamin D | Healthy bones and tissues, modulation of cell growth, maintenance of the immune system, teeth growth. | [22,23,24] |
| Vitamin E | Antioxidant, maintenance of the immune system, prevention of cardiovascular diseases, vision protection. | [20,25] |
| Vitamin K | Blood clotting, bone strengthening, cardiovascular disease prevention, antioxidant. | [26,27,28] |
| Micronutrient | Disorders | References |
|---|---|---|
| Vitamin A | Night blindness, growth disturbance, dysfunctions to the reproductive system; dysfunctions of the immune response. | [32,33,34] |
| Vitamin C | Scurvy, connective tissue disorders. | [35,36] |
| Vitamin D | Rickets, osteomalacia. | [37,38] |
| Vitamin E | Muscle metabolism disorders, neuropathy, oxidative hemolysis. | [39,40] |
| Vitamin K | Delayed coagulation, Hemorrhagic disease of the newborn. | [41] |
| Vitamin B6 | Dermatitis, polyneuritis, muscle spasms. | [39,42,43] |
| Micronutrient | Diseases | References |
|---|---|---|
| Vitamin A | Headache, vomiting and numbness, anemia, teratogen in the fetus. | [39,47] |
| Vitamin D | Vomiting, headache, diarrhea, polyuria, calcinosis, fatigue. | [48,49] |
| Vitamin B6 | Damage to the nervous system. | [50] |
| Vitamin C | Kidney stones, intestinal disorders. | [51,52] |
| Vitamin E | Absorption reduction in other liposoluble vitamins. | [53] |
| Vitamin K | Anemia, vomiting, thrombosis, excessive sweating. | [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brancaccio, M.; Mennitti, C.; Cesaro, A.; Fimiani, F.; Vano, M.; Gargiulo, B.; Caiazza, M.; Amodio, F.; Coto, I.; D’Alicandro, G.; et al. The Biological Role of Vitamins in Athletes’ Muscle, Heart and Microbiota. Int. J. Environ. Res. Public Health 2022, 19, 1249. https://doi.org/10.3390/ijerph19031249
Brancaccio M, Mennitti C, Cesaro A, Fimiani F, Vano M, Gargiulo B, Caiazza M, Amodio F, Coto I, D’Alicandro G, et al. The Biological Role of Vitamins in Athletes’ Muscle, Heart and Microbiota. International Journal of Environmental Research and Public Health. 2022; 19(3):1249. https://doi.org/10.3390/ijerph19031249
Chicago/Turabian StyleBrancaccio, Mariarita, Cristina Mennitti, Arturo Cesaro, Fabio Fimiani, Martina Vano, Biagio Gargiulo, Martina Caiazza, Federica Amodio, Iolanda Coto, Giovanni D’Alicandro, and et al. 2022. "The Biological Role of Vitamins in Athletes’ Muscle, Heart and Microbiota" International Journal of Environmental Research and Public Health 19, no. 3: 1249. https://doi.org/10.3390/ijerph19031249
APA StyleBrancaccio, M., Mennitti, C., Cesaro, A., Fimiani, F., Vano, M., Gargiulo, B., Caiazza, M., Amodio, F., Coto, I., D’Alicandro, G., Mazzaccara, C., Lombardo, B., Pero, R., Terracciano, D., Limongelli, G., Calabrò, P., D’Argenio, V., Frisso, G., & Scudiero, O. (2022). The Biological Role of Vitamins in Athletes’ Muscle, Heart and Microbiota. International Journal of Environmental Research and Public Health, 19(3), 1249. https://doi.org/10.3390/ijerph19031249

















