The COVID-19 Pandemic Lowers Active Behavior of Patients with Cardiovascular Diseases, Healthy Peoples and Athletes
Abstract
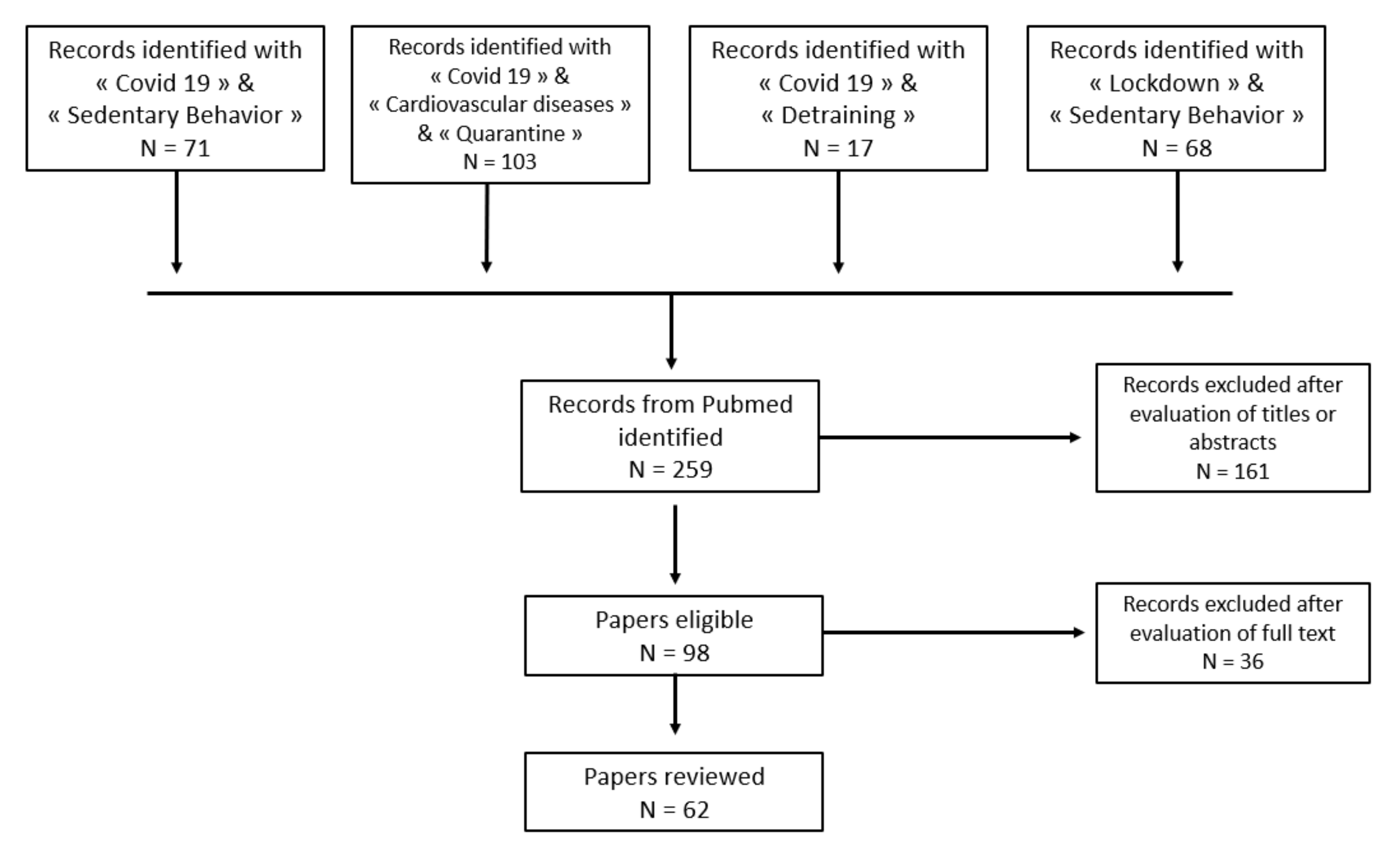
:1. Introduction
2. Materials and Methods
3. Results
3.1. Impact of the COVID-19 Lockdown in CVD Patients
3.2. Impact of the COVID-19 Lockdown in Healthy Subjects
3.3. Physical and Physiological Impacts of Training Cessation in Athletes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, J.; Siegrist, J. Physical Activity and Risk of Cardiovascular Disease—A Meta-Analysis of Prospective Cohort Studies. Int. J. Environ. Res. Public Health 2012, 9, 391–407. [Google Scholar] [CrossRef] [PubMed]
- Pescatello, L.S.; MacDonald, H.V.; Lamberti, L.; Johnson, B.T. Exercise for Hypertension: A Prescription Update Integrating Existing Recommendations with Emerging Research. Curr. Hypertens. Rep. 2015, 17, 87. [Google Scholar] [CrossRef] [Green Version]
- Nystoriak, M.A.; Bhatnagar, A. Cardiovascular Effects and Benefits of Exercise. Front. Cardiovasc. Med. 2018, 5, 135. [Google Scholar] [CrossRef] [Green Version]
- Blair, S.N.; Kampert, J.B.; Kohl, H.W.; Barlow, C.E.; Macera, C.A.; Paffenbarger, R.S.; Gibbons, L.W. Influences of Cardiorespiratory Fitness and Other Precursors on Cardiovascular Disease and All-Cause Mortality in Men and Women. JAMA 1996, 276, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Lavie, C.J.; Ozemek, C.; Carbone, S.; Katzmarzyk, P.T.; Blair, S.N. Sedentary Behavior, Exercise, and Cardiovascular Health. Circ. Res. 2019, 124, 799–815. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.S.; Colley, R.C.; Saunders, T.J.; Healy, G.N.; Owen, N. Physiological and Health Implications of a Sedentary Lifestyle. Appl. Physiol. Nutr. Metab. 2010, 35, 725–740. [Google Scholar] [CrossRef]
- Long, L.; Mordi, I.R.; Bridges, C.; Sagar, V.A.; Davies, E.J.; Coats, A.J.; Dalal, H.; Rees, K.; Singh, S.J.; Taylor, R.S. Exercise-Based Cardiac Rehabilitation for Adults with Heart Failure. Cochrane Database Syst. Rev. 2019, 1, CD003331. [Google Scholar] [CrossRef] [Green Version]
- Pelliccia, A.; Sharma, S.; Gati, S.; Bäck, M.; Börjesson, M.; Caselli, S.; Collet, J.-P.; Corrado, D.; Drezner, J.A.; Halle, M.; et al. 2020 ESC Guidelines on Sports Cardiology and Exercise in Patients with Cardiovascular Disease. Eur. Heart J. 2021, 42, 17–96. [Google Scholar] [CrossRef] [PubMed]
- Vetrovsky, T.; Frybova, T.; Gant, I.; Semerad, M.; Cimler, R.; Bunc, V.; Siranec, M.; Miklikova, M.; Vesely, J.; Griva, M.; et al. The Detrimental Effect of COVID-19 Nationwide Quarantine on Accelerometer-Assessed Physical Activity of Heart Failure Patients. ESC Heart Fail. 2020, 7, 2093–2097. [Google Scholar] [CrossRef]
- Al Fagih, A.; Al Onazi, M.; Al Basiri, S.; Al-Kaf, F.; Dagriri, K.; Al Hebaishi, Y.; Samargandy, S.; Al Shengeiti, L. Remotely Monitored Inactivity Due to COVID-19 Lockdowns. Potential Hazard for Heart Failure Patients. Saudi Med. J. 2020, 41, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Sassone, B.; Mandini, S.; Grazzi, G.; Mazzoni, G.; Myers, J.; Pasanisi, G. Impact of COVID-19 Pandemic on Physical Activity in Patients With Implantable Cardioverter-Defibrillators. J. Cardiopulm. Rehabil. Prev. 2020, 40, 285–286. [Google Scholar] [CrossRef]
- Chagué, F.; Boulin, M.; Eicher, J.-C.; Bichat, F.; Saint Jalmes, M.; Cransac-Miet, A.; Soudry-Faure, A.; Danchin, N.; Cottin, Y.; Zeller, M. Impact of Lockdown on Patients with Congestive Heart Failure during the Coronavirus Disease 2019 Pandemic. ESC Heart Fail. 2020, 7, 4420–4423. [Google Scholar] [CrossRef] [PubMed]
- Van Bakel, B.M.A.; Bakker, E.A.; de Vries, F.; Thijssen, D.H.J.; Eijsvogels, T.M.H. Impact of COVID-19 Lockdown on Physical Activity and Sedentary Behaviour in Dutch Cardiovascular Disease Patients. Neth. Heart J. Mon. J. Neth. Soc. Cardiol. Neth. Heart Found. 2021, 29, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Romero-Blanco, C.; Rodríguez-Almagro, J.; Onieva-Zafra, M.D.; Parra-Fernández, M.L.; Prado-Laguna, M.D.C.; Hernández-Martínez, A. Physical Activity and Sedentary Lifestyle in University Students: Changes during Confinement Due to the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2020, 17, 6567. [Google Scholar] [CrossRef]
- Zheng, C.; Huang, W.Y.; Sheridan, S.; Sit, C.H.-P.; Chen, X.-K.; Wong, S.H.-S. COVID-19 Pandemic Brings a Sedentary Lifestyle in Young Adults: A Cross-Sectional and Longitudinal Study. Int. J. Environ. Res. Public Health 2020, 17, 6035. [Google Scholar] [CrossRef] [PubMed]
- Castañeda-Babarro, A.; Arbillaga-Etxarri, A.; Gutiérrez-Santamaría, B.; Coca, A. Physical Activity Change during COVID-19 Confinement. Int. J. Environ. Res. Public Health 2020, 17, 6878. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.; Brach, M.; Trabelsi, K.; Chtourou, H.; Boukhris, O.; Masmoudi, L.; Bouaziz, B.; Bentlage, E.; How, D.; Ahmed, M.; et al. Effects of COVID-19 Home Confinement on Eating Behaviour and Physical Activity: Results of the ECLB-COVID19 International Online Survey. Nutrients 2020, 12, 1583. [Google Scholar] [CrossRef]
- Huber, B.C.; Steffen, J.; Schlichtiger, J.; Graupe, T.; Deuster, E.; Strouvelle, V.P.; Fischer, M.R.; Massberg, S.; Brunner, S. Alteration of Physical Activity during COVID-19 Pandemic Lockdown in Young Adults. J. Transl. Med. 2020, 18, 410. [Google Scholar] [CrossRef]
- Mujika, I.; Padilla, S. Detraining: Loss of Training-Induced Physiological and Performance Adaptations. Part I: Short Term Insufficient Training Stimulus. Sports Med. Auckl. NZ 2000, 30, 79–87. [Google Scholar] [CrossRef]
- Coyle, E.F.; Hemmert, M.K.; Coggan, A.R. Effects of Detraining on Cardiovascular Responses to Exercise: Role of Blood Volume. J. Appl. Physiol. Bethesda Md 1985 1986, 60, 95–99. [Google Scholar] [CrossRef]
- Martin, W.H.; Coyle, E.F.; Bloomfield, S.A.; Ehsani, A.A. Effects of Physical Deconditioning after Intense Endurance Training on Left Ventricular Dimensions and Stroke Volume. J. Am. Coll. Cardiol. 1986, 7, 982–989. [Google Scholar] [CrossRef] [Green Version]
- Raven, P.B.; Drinkwater, B.L.; Horvath, S.M. Cardiovascular Responses of Young Female Track Athletes during Exercise. Med. Sci. Sports 1972, 4, 205–209. [Google Scholar] [PubMed]
- Houmard, J.A.; Hortobágyi, T.; Johns, R.A.; Bruno, N.J.; Nute, C.C.; Shinebarger, M.H.; Welborn, J.W. Effect of Short-Term Training Cessation on Performance Measures in Distance Runners. Int. J. Sports Med. 1992, 13, 572–576. [Google Scholar] [CrossRef] [PubMed]
- Thompson, P.D.; Cullinane, E.M.; Eshleman, R.; Sady, S.P.; Herbert, P.N. The Effects of Caloric Restriction or Exercise Cessation on the Serum Lipid and Lipoprotein Concentrations of Endurance Athletes. Metabolism 1984, 33, 943–950. [Google Scholar] [CrossRef]
- Cullinane, E.M.; Sady, S.P.; Vadeboncoeur, L.; Burke, M.; Thompson, P.D. Cardiac Size and VO2max Do Not Decrease after Short-Term Exercise Cessation. Med. Sci. Sports Exerc. 1986, 18, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Raven, P.B.; Welch-O’Connor, R.M.; Shi, X. Cardiovascular Function Following Reduced Aerobic Activity. Med. Sci. Sports Exerc. 1998, 30, 1041–1052. [Google Scholar] [CrossRef] [PubMed]
- Coyle, E.F.; Martin, W.H.; Sinacore, D.R.; Joyner, M.J.; Hagberg, J.M.; Holloszy, J.O. Time Course of Loss of Adaptations after Stopping Prolonged Intense Endurance Training. J. Appl. Physiol. 1984, 57, 1857–1864. [Google Scholar] [CrossRef] [PubMed]
- Coyle, E.F.; Martin, W.H.; Bloomfield, S.A.; Lowry, O.H.; Holloszy, J.O. Effects of Detraining on Responses to Submaximal Exercise. J. Appl. Physiol. Bethesda Md 1985 1985, 59, 853–859. [Google Scholar] [CrossRef]
- Wibom, R.; Hultman, E.; Johansson, M.; Matherei, K.; Constantin-Teodosiu, D.; Schantz, P.G. Adaptation of Mitochondrial ATP Production in Human Skeletal Muscle to Endurance Training and Detraining. J. Appl. Physiol. Bethesda Md 1985 1992, 73, 2004–2010. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, J.; Reitman, J.S. Time Course of Changes in Human Skeletal Muscle Succinate Dehydrogenase and Cytochrome Oxidase Activities and Maximal Oxygen Uptake with Physical Activity and Inactivity. Acta Physiol. Scand. 1977, 99, 91–97. [Google Scholar] [CrossRef]
- Moore, R.L.; Thacker, E.M.; Kelley, G.A.; Musch, T.I.; Sinoway, L.I.; Foster, V.L.; Dickinson, A.L. Effect of Training/Detraining on Submaximal Exercise Responses in Humans. J. Appl. Physiol. Bethesda Md 1985 1987, 63, 1719–1724. [Google Scholar] [CrossRef]
- Bosquet, L.; Berryman, N.; Dupuy, O.; Mekary, S.; Arvisais, D.; Bherer, L.; Mujika, I. Effect of Training Cessation on Muscular Performance: A Meta-Analysis. Scand. J. Med. Sci. Sports 2013, 23, e140–e149. [Google Scholar] [CrossRef]
- Klausen, K.; Andersen, L.B.; Pelle, I. Adaptive Changes in Work Capacity, Skeletal Muscle Capillarization and Enzyme Levels during Training and Detraining. Acta Physiol. Scand. 1981, 113, 9–16. [Google Scholar] [CrossRef]
- Psilander, N.; Eftestøl, E.; Cumming, K.T.; Juvkam, I.; Ekblom, M.M.; Sunding, K.; Wernbom, M.; Holmberg, H.-C.; Ekblom, B.; Bruusgaard, J.C.; et al. Effects of Training, Detraining, and Retraining on Strength, Hypertrophy, and Myonuclear Number in Human Skeletal Muscle. J. Appl. Physiol. Bethesda Md 1985 2019, 126, 1636–1645. [Google Scholar] [CrossRef]
- Häkkinen, K.; Alén, M.; Komi, P.V. Changes in Isometric Force- and Relaxation-Time, Electromyographic and Muscle Fibre Characteristics of Human Skeletal Muscle during Strength Training and Detraining. Acta Physiol. Scand. 1985, 125, 573–585. [Google Scholar] [CrossRef] [PubMed]
- Houston, M.E.; Bentzen, H.; Larsen, H. Interrelationships between Skeletal Muscle Adaptations and Performance as Studied by Detraining and Retraining. Acta Physiol. Scand. 1979, 105, 163–170. [Google Scholar] [CrossRef]
- Hortobágyi, T.; Houmard, J.A.; Stevenson, J.R.; Fraser, D.D.; Johns, R.A.; Israel, R.G. The Effects of Detraining on Power Athletes. Med. Sci. Sports Exerc. 1993, 25, 929–935. [Google Scholar]
- Fringer, M.N.; Stull, G.A. Changes in Cardiorespiratory Parameters during Periods of Training and Detraining in Young Adult Females. Med. Sci. Sports 1974, 6, 20–25. [Google Scholar]
- Giada, F.; Bertaglia, E.; De Piccoli, B.; Franceschi, M.; Sartori, F.; Raviele, A.; Pascotto, P. Cardiovascular Adaptations to Endurance Training and Detraining in Young and Older Athletes. Int. J. Cardiol. 1998, 65, 149–155. [Google Scholar] [CrossRef]
- Leitão, L.; Pereira, A.; Mazini, M.; Venturini, G.; Campos, Y.; Vieira, J.; Novaes, J.; Vianna, J.; da Silva, S.; Louro, H. Effects of Three Months of Detraining on the Health Profile of Older Women after a Multicomponent Exercise Program. Int. J. Environ. Res. Public Health 2019, 16, 3881. [Google Scholar] [CrossRef] [Green Version]
- Nolan, P.B.; Keeling, S.M.; Robitaille, C.A.; Buchanan, C.A.; Dalleck, L.C. The Effect of Detraining after a Period of Training on Cardiometabolic Health in Previously Sedentary Individuals. Int. J. Environ. Res. Public Health 2018, 15, 2303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petibois, C.; Déléris, G. Effects of Short- and Long-Term Detraining on the Metabolic Response to Endurance Exercise. Int. J. Sports Med. 2003, 24, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Heath, G.W.; Gavin, J.R.; Hinderliter, J.M.; Hagberg, J.M.; Bloomfield, S.A.; Holloszy, J.O. Effects of Exercise and Lack of Exercise on Glucose Tolerance and Insulin Sensitivity. J. Appl. Physiol. 1983, 55, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Giada, F.; Vigna, G.B.; Vitale, E.; Baldo-Enzi, G.; Bertaglia, M.; Crecca, R.; Fellin, R. Effect of Age on the Response of Blood Lipids, Body Composition, and Aerobic Power to Physical Conditioning and Deconditioning. Metabolism 1995, 44, 161–165. [Google Scholar] [CrossRef]
- Madsen, K.; Pedersen, P.K.; Djurhuus, M.S.; Klitgaard, N.A. Effects of Detraining on Endurance Capacity and Metabolic Changes during Prolonged Exhaustive Exercise. J. Appl. Physiol. Bethesda Md 1985 1993, 75, 1444–1451. [Google Scholar] [CrossRef]
- Li, B.; Yang, J.; Zhao, F.; Zhi, L.; Wang, X.; Liu, L.; Bi, Z.; Zhao, Y. Prevalence and Impact of Cardiovascular Metabolic Diseases on COVID-19 in China. Clin. Res. Cardiol. Off. J. Ger. Card. Soc. 2020, 109, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.; Oldridge, N.; Thompson, D.R.; Zwisler, A.-D.; Rees, K.; Martin, N.; Taylor, R.S. Exercise-Based Cardiac Rehabilitation for Coronary Heart Disease: Cochrane Systematic Review and Meta-Analysis. J. Am. Coll. Cardiol. 2016, 67, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, P.; Mao, L.; Nassis, G.P.; Harmer, P.; Ainsworth, B.E.; Li, F. Coronavirus Disease (COVID-19): The Need to Maintain Regular Physical Activity While Taking Precautions. J. Sport Health Sci. 2020, 9, 103–104. [Google Scholar] [CrossRef]
- Cook, R.; Davidson, P.; Martin, R. NIHR Dissemination Centre Cardiac Rehabilitation for Heart Failure Can Improve Quality of Life and Fitness. BMJ 2019, 367, l5456. [Google Scholar] [CrossRef]
- Frederix, I.; Vanhees, L.; Dendale, P.; Goetschalckx, K. A Review of Telerehabilitation for Cardiac Patients. J. Telemed. Telecare 2015, 21, 45–53. [Google Scholar] [CrossRef]
- Thomas, R.J.; Beatty, A.L.; Beckie, T.M.; Brewer, L.C.; Brown, T.M.; Forman, D.E.; Franklin, B.A.; Keteyian, S.J.; Kitzman, D.W.; Regensteiner, J.G.; et al. Home-Based Cardiac Rehabilitation: A Scientific Statement From the American Association of Cardiovascular and Pulmonary Rehabilitation, the American Heart Association, and the American College of Cardiology. Circulation 2019, 140, e69–e89. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.; Sharp, G.A.; Norton, R.J.; Dalal, H.; Dean, S.G.; Jolly, K.; Cowie, A.; Zawada, A.; Taylor, R.S. Home-Based versus Centre-Based Cardiac Rehabilitation. Cochrane Database Syst. Rev. 2017, 6, CD007130. [Google Scholar] [CrossRef] [PubMed]
- Frederix, I.; Caiani, E.G.; Dendale, P.; Anker, S.; Bax, J.; Böhm, A.; Cowie, M.; Crawford, J.; de Groot, N.; Dilaveris, P.; et al. ESC E-Cardiology Working Group Position Paper: Overcoming Challenges in Digital Health Implementation in Cardiovascular Medicine. Eur. J. Prev. Cardiol. 2019, 26, 1166–1177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scherrenberg, M.; Wilhelm, M.; Hansen, D.; Völler, H.; Cornelissen, V.; Frederix, I.; Kemps, H.; Dendale, P. The Future Is Now: A Call for Action for Cardiac Telerehabilitation in the COVID-19 Pandemic from the Secondary Prevention and Rehabilitation Section of the European Association of Preventive Cardiology. Eur. J. Prev. Cardiol. 2021, 28, 524–540. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, A.; Takayama, N.; Kobayashi, M.; Hyodo, K.; Maeshima, N.; Takayuki, F.; Morita, H.; Komuro, I. Remote Cardiac Rehabilitation Is a Good Alternative of Outpatient Cardiac Rehabilitation in the COVID-19 Era. Environ. Health Prev. Med. 2020, 25, 48. [Google Scholar] [CrossRef]
- Lamberti, N.; Straudi, S.; Manfredini, R.; De Giorgi, A.; Gasbarro, V.; Zamboni, P.; Manfredini, F. Don’t Stop Walking: The in-Home Rehabilitation Program for Peripheral Artery Disease Patients during the COVID-19 Pandemic. Intern. Emerg. Med. 2021, 16, 1307–1315. [Google Scholar] [CrossRef]
- Ricci, F.; Izzicupo, P.; Moscucci, F.; Sciomer, S.; Maffei, S.; Di Baldassarre, A.; Mattioli, A.V.; Gallina, S. Recommendations for Physical Inactivity and Sedentary Behavior During the Coronavirus Disease (COVID-19) Pandemic. Front. Public Health 2020, 8, 199. [Google Scholar] [CrossRef] [PubMed]
- Chaabene, H.; Prieske, O.; Herz, M.; Moran, J.; Höhne, J.; Kliegl, R.; Ramirez-Campillo, R.; Behm, D.G.; Hortobágyi, T.; Granacher, U. Home-Based Exercise Programmes Improve Physical Fitness of Healthy Older Adults: A PRISMA-Compliant Systematic Review and Meta-Analysis with Relevance for COVID-19. Ageing Res. Rev. 2021, 67, 101265. [Google Scholar] [CrossRef]
- Loh, R.; Stamatakis, E.; Folkerts, D.; Allgrove, J.E.; Moir, H.J. Effects of Interrupting Prolonged Sitting with Physical Activity Breaks on Blood Glucose, Insulin and Triacylglycerol Measures: A Systematic Review and Meta-Analysis. Sports Med. Auckl. NZ 2020, 50, 295–330. [Google Scholar] [CrossRef] [Green Version]
- Kraus, W.E.; Janz, K.F.; Powell, K.E.; Campbell, W.W.; Jakicic, J.M.; Troiano, R.P.; Sprow, K.; Torres, A.; Piercy, K.L.; 2018 Physical Activity Guidelines Advisory Committee. Daily Step Counts for Measuring Physical Activity Exposure and Its Relation to Health. Med. Sci. Sports Exerc. 2019, 51, 1206–1212. [Google Scholar] [CrossRef]
- Menz, V.; Marterer, N.; Amin, S.B.; Faulhaber, M.; Hansen, A.B.; Lawley, J.S. Functional Vs. Running Low-Volume High-Intensity Interval Training: Effects on VO2max and Muscular Endurance. J. Sports Sci. Med. 2019, 18, 497–504. [Google Scholar] [PubMed]
- Ross, L.M.; Porter, R.R.; Durstine, J.L. High-Intensity Interval Training (HIIT) for Patients with Chronic Diseases. J. Sport Health Sci. 2016, 5, 139–144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Girardi, M.; Casolo, A.; Nuccio, S.; Gattoni, C.; Capelli, C. Detraining Effects Prevention: A New Rising Challenge for Athletes. Front. Physiol. 2020, 11, 588784. [Google Scholar] [CrossRef] [PubMed]
| References | First Author (Year) | Outcome Measures | Results | Conclusion |
|---|---|---|---|---|
| [9] | Vetrovsky (2020) | The daily numbers of steps in 26 heart failure (HF) patients | A 16.2% decrease of daily steps was found. | Quarantine had a detrimental effect on the level of the habitual physical activity in HF patients. |
| [10] | Al Faghi (2020) | HF patients with cardiac implantable electronic devices (CIEDs) activity as hours per day from 2 February to 19 April 2020 | A 27.1% decline in physical activity was found. The median physical activity significantly declined from 2.4 to 1.8 h/day. | There was a significant decline in the physical activity due to the pandemic. |
| [11] | Sassone (2020) | The daily physical activities in patients with implantable cardioverter defibrillators (ICDs) | A 25% reduction of the physical activity (1.2 ± 0.3 h/day during the confinement vs. 1.6 ± 0.5 h/day before the confinement) | The COVID-19 pandemic led to an abrupt and statistically significant reduction of the physical activity in patients with primary prevention ICDs. |
| [12] | Chagué (2020) | The physical activities, the lifestyle behaviors, and the psychological states of 150 randomly selected chronic-heart-failure (CHF) patients | A 41.9% decrease in the physical activity and a 21.8% increase in HF symptoms were found. | The current pandemic had negative effects on lifestyle behaviors such as reduced physical activity. |
| [13] | Van Bakel (2020) | The physical activity and the sedentary behavior before and during the COVID-19 lockdown period | The time spent exercising declined from 1.0 to 0.0 h/week. The sedentary time increased from 7.8 to 8.9 h/day. The sedentary behavior increased by 55 min/day. | The increase in the sedentary time induced a net reduction in habitual physical activity levels in Dutch cardiovascular diseases (CVDs) patients (48% myocardial infarction) during the first-wave COVID-19 lockdown. |
| References | First Author (Year) | Outcome Measures | Results | Conclusion |
|---|---|---|---|---|
| [14] | Romero Blanco (2020) | The weekly sitting times in 213 university students | The weekly sitting time increased by 106.76 min. | The confinement changed the physical activities, and the sedentary lifestyles in university students. |
| [15] | Zheng (2020) | The physical activity levels, the sedentary behaviors, and the sleep in 631 young adults during the COVID-19 epidemic | Walking significantly declined from 39.7 to 19.8 min/day. The times spent in the sedentary behavior and the sleep significantly increased from 7.8 to 10 and 7.7 to 8.4 h/day, respectively. | A significant reduction in physical behaviors and significant increases in the sedentary behavior and the sleep duration of young adults during the COVID-19 epidemic were identified. |
| [16] | Castaneda-Babarro (2020) | The physical activities, the walking times, and the sedentary times in 3800 healthy adults during confinement | The physical activity and the walking time decreased by 16.8% and 58.2%, respectively. The sedentary time increased by 23.8%. | Healthy adults decreased the daily physical activity and increased the sedentary time during the COVID-19 confinement. |
| [17] | Ammar (2020) | The physical activities, the lifestyle behaviors, the daily sitting times, and the walking times of 1047 randomly selected adults | The physical activity decreased from five to three days/week. The daily sitting time increased from 5 to 8 h/day. The number of minutes/day walking decreased by 34%. | Home confinement had negative effects on the physical activity with a significant increase in the sitting time, indicative of a more sedentary lifestyle. |
| [18] | Huber (2020) | The physical activities during the COVID-19 lockdown measures in 1980 students | The physical activity decreased in 44.5% of the participants. The daily step count decreased by 25%. | The COVID-19 crisis led to changes in the physical activity among young adults. |
| References | First Author (Year) | Outcome Measures | Results | Conclusion |
|---|---|---|---|---|
| [19] | Mujika (2000) | The VO2max, the blood volumes, and the maximal cardiac outputs in highly trained athletes after a short-term detraining | Declines in the maximal oxygen uptake (VO2max) and the blood volume were found. A reduction of the maximal cardiac output | Short-term detraining induced losses of training-induced physiological and performance adaptations |
| [20] | Coyle (1986) | The VO2max, the cardiac outputs, and the blood volumes in endurance-trained men who stopped training for a few weeks | by 9% in blood volume (5.177 to 4.692 mL), a 12% reduction of the stroke volume, and a 6% reduction of VO2max were found. | The decline in the cardiovascular function following a few weeks of detraining is largely due to a reduction in blood volume. |
| [21] | Martin (1986) | The oxygen uptakes, the cardiac outputs, the heart rates in 6 exercise-trained endurance athletes after deconditioning | A reduction in stroke volume and a 20% decrease in the left ventricular mass were found. | Inactivity resulted in losses of adaptations such as a greater stroke volume and a regression of left ventricular hypertrophy. |
| [22] | Raven (1972) | The cardiac outputs and the cardiorespiratory parameters in young females athletes | A reduction of the cardiac output was found. | Three months without formal training sessions reduced the cardiorespiratory fitness of young females athletes. |
| [23] | Houmard (1992) | The VO2max, the resting plasma volumes, and the maximal heart rates in 12 distance runners after 14 days of training cessation | The VO2max decreased by 3 mL/kg/min. The maximal heart rate increased by 9 beats per minute. The resting plasma volume decreased by 5%. | Training cessation affected measures associated with the distance. The running performance was affected by short-term (14 days) training cessation. |
| [24] | Thompson (1984) | The low-density lipoprotein cholesterol levels of men running 16 km daily after exercise cessation | Low-density lipoprotein cholesterol decreased by 10% to 15%. A 5% decrease in the plasma volume was found. | Exercise cessation led to a reduction in the plasma volume |
| [25] | Cullinane (1986) | The maximum oxygen uptakes, the estimated changes in the plasma volume, and the cardiac dimensions of 15 male competitive distance runners before and after 10 days of exercise cessation | The plasma volume decreased by 5%. The resting heart rate, blood pressure, and cardiac dimensions remained unchanged with the physical inactivity. | Short periods of the exercise cessation decrease estimated the plasma volume and increased the maximum exercise heart rate of endurance athletes but did not alter their cardiac dimensions. |
| [26] | Raven (1998) | The VO2max and the lower body negative pressures in 19 volunteers after an 8-week physical deconditioning | The VO2max and the lower body negative pressure tolerance decreased by 7% and 13%, respectively. | The functional modification of the cardiac pressure–volume relationship resulted in the reduced lower body negative pressure tolerance. |
| [27] | Coyle (1984) | The maximal heart rates, the stroke volumes, and the VO2max in 7 endurance exercise-trained subjects after the cessation of training | VO2max declined by 7% during the first 21 day of inactivity. An increase of 4% in the maximal heart rate was found. A decrease of 10% of the stroke volume was identified. | Loss of adaptations after stopping prolonged intense endurance training occurred from 21 days. |
| [28] | Coyle (1985) | The heart rates, the ventilations, the respiratory exchange ratios, and the blood lactate concentrations in 7 endurance-exercise-trained subjects after the cessation of training | After 84 days of detraining, experimental subjects’ muscle mitochondrial enzyme levels were still 50% above, and the lactate dehydrogenase (LDH) activity was 22% below sedentary control levels. | Adaptations to prolonged endurance training (responsible for the higher lactate threshold) persisting for a long time after training were stopped. |
| [29] | Wibom (1992) | The mitochondrial ATP production rates in 9 men after 3 weeks of detraining | The mitochondrial ATP production rate decreased by 12–28%. | Mitochondrial ATP production rate decreased with detraining. |
| [30] | Henriksson (1977) | Succinate dehydrogenase (SDH) and cytochrome oxidase activities during a 6-week period without training | SDH and cytochrome activities returned to the pre-training level. | The fast return to the pre-training levels of both SDH and cytochrome oxidase activities indicated a high turnover rate of enzymes in the TCA cycle as well as the respiratory chain. |
| [31] | Moore (1987) | The VO2max and the citrate synthase (CS) activities in trained subjects after 3 weeks of inactivity | A decrease in CS activity to 80 ± 14.6 nmol/mg protein/min was found. | The mitochondrial content of working skeletal muscle is an important determinant of the substrate utilization during submaximal exercise. |
| [32] | Bosquet (2013) | Meta analysis to assess the effect of resistance training cessation on the strength performance | The submaximal strength, the maximal force, and the maximal power declined. | Resistance training cessation had detrimental effects on all components of muscular performance. |
| [33] | Klausen (1981) | The numbers of capillaries per mm2 and the numbers of capillaries per fiber in 6 male subjects after 8 weeks of detraining | The number of capillaries per fiber decreased. | Eight-week detraining had negative effects on muscle capillarization. |
| [34] | Psilander (1985) | The myonuclear numbers, the fiber volumes, and the cross-sectional areas (CSAs) assessed in 19 subjects after 20 weeks of detraining | The CSA decreased to 17%. | Long detraining periods led to a decrease of the mean muscle fiber areas. |
| [35] | Häkkinen (1981) | The maximal isometric strengths, the strengths correlated, and neural activations in 11 males after 12 weeks of detraining | A decrease of the maximal isometric strength and a decrease of the mean muscle-fiber areas of both fiber types were identified. | Detraining affected muscle hypertrophy. |
| [36] | Houston (1979) | Activities of SDH and LDH, the VO2max, and the muscle fiber areas in 6 well-trained runners after 15 days of detraining | SDH and LDH activities decreased by 24% and 13%, respectively. The VO2max decreased by 4%. The muscle fiber areas became larger. | Short periods of detraining resulted in significant changes in indices of physiological capacity and function. |
| [37] | Hortobagyi (1993) | The performances, the surface EMG activities, and the types of fibers in 12 power athletes after 14 days of detraining | The performances declined. Type II fiber area decreased by 6.4%. | Short-term detraining affected the size of the type II muscle fibers. |
| [38] | Fringer (1974) | The pulmonary ventilations, the oxygen uptakes, the oxygen pulses, the heart rates, and the total work outputs in 44 trained women after 5 or 10 weeks of detraining | Increases in the resting heart rate and the maximal ventilation equivalent were found. Decreases in the total work, the pulmonary ventilation, the oxygen uptake, and the oxygen pulse were identified. | Losses in the maximal values for the oxygen uptake, the oxygen pulse, and the ventilation equivalent were greater for 10 weeks of detraining than for 5 weeks of detraining. |
| [39] | Giada (1998) | The left ventricle morphologies, systolic functions, and diastolic filling patterns of 24 male cyclists, 12 young, and 12 older, after a 2-month detraining | The wall thicknesses decreased only in young athletes, while the left ventricular mass and the end-diastolic diameter and volume reduced only in older athletes. | Detraining induced greater left ventricular morphological modifications in older athletes. |
| [40] | Leitão (2019) | The oxygen uptake (VO2) and health profile assessments in 47 older trained women after 3 months of detraining | Increases of the resting heart rate and the systolic and diastolic blood pressures were found. Decreases of the pulmonary ventilation and the VO2/heart rate were identified. | Detraining induced greater declines in the total health profile and in VO2 after a training particularly developed for older women. |
| [41] | Nolan (2018) | The VO2max, the body fat percentage, the mean arterial pressure, and the HDL cholesterol and triglycerides levels after a 13-week training program followed by detraining | The VO2max and the body fat percentage, along with the mean arterial pressure and HDL cholesterol and triglycerides levels, significantly worsened. | These novel findings underscored the importance of sustained and uninterrupted exercise training. |
| [42] | Petitbois (2003) | The VO2max and the metabolic responses in 10 trained rowers after detraining | A lower adipose tissue triglyceride delivery during exercise was found. The total fatty acid concentration decreased. | Alterations of the metabolic adaptations to training may become rapidly chronic after such a detraining. |
| [43] | Heath (1983) | The VO2max values, the glucose tolerances, and the insulin sensitivities in 8 well-trained subjects who stopped training for 10 days | The maximum rise in the plasma insulin concentration was 100%. Blood glucose concentrations higher | Detraining induced decreased the insulin sensitivity and the glucose tolerance |
| [44] | Giada (1995) | The VO2max, the total, LDL, and HDL cholesterol level, and the triglycerides levels in 24 males cyclists after a 2-month detraining | The VO2max decreased. The triglycerides and LDL cholesterol levels increased. | Detraining induced changes in metabolic response to exercise. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirsch, M.; Vitiello, D. The COVID-19 Pandemic Lowers Active Behavior of Patients with Cardiovascular Diseases, Healthy Peoples and Athletes. Int. J. Environ. Res. Public Health 2022, 19, 1108. https://doi.org/10.3390/ijerph19031108
Kirsch M, Vitiello D. The COVID-19 Pandemic Lowers Active Behavior of Patients with Cardiovascular Diseases, Healthy Peoples and Athletes. International Journal of Environmental Research and Public Health. 2022; 19(3):1108. https://doi.org/10.3390/ijerph19031108
Chicago/Turabian StyleKirsch, Marine, and Damien Vitiello. 2022. "The COVID-19 Pandemic Lowers Active Behavior of Patients with Cardiovascular Diseases, Healthy Peoples and Athletes" International Journal of Environmental Research and Public Health 19, no. 3: 1108. https://doi.org/10.3390/ijerph19031108
APA StyleKirsch, M., & Vitiello, D. (2022). The COVID-19 Pandemic Lowers Active Behavior of Patients with Cardiovascular Diseases, Healthy Peoples and Athletes. International Journal of Environmental Research and Public Health, 19(3), 1108. https://doi.org/10.3390/ijerph19031108






