Exposure to Inorganic Arsenic in Rice in Brazil: A Human Health Risk Assessment
Abstract
1. Introduction
2. Materials and Methods
2.1. Average Daily Dose, Cancer Risk, and Hazard Quotient
2.2. Concentration of iAs in Brazilian Rice
2.3. Rice Consumption
- From 4 months to 1 year and 5 to 10 years—the estimated rice consumption was based on the quantity recommended by the São Paulo Municipal Department of Education for consumption in schools and daycare centers [31,32]. Since we noticed that the recommended consumption was overestimated in comparison with the actual consumption for children 1 to 5 years of age (see below), we estimated that the actual rice consumption for children 4 months to 1 year and 5 to 10 years of age was 39.8% less than that. In the school diet, rice is served cooked (as a side dish, in soups, or as a dessert) and the quantities were registered in grams of raw rice. Students from 6 to 10 years of age have a part-time school period, having only one meal at school (lunch or dinner), and daily rice consumption for that age group was, therefore, estimated on the basis of the recommended quantity.
- From 1 to 5 years—data were obtained from a study involving 64 children at 2 daycare centers in the city of São Paulo [33], in which the portions of 24-h duplicate diet samples were recorded, including the food consumed at the daycare center and that consumed at home. The consumption of rice and soup containing rice was evaluated. Household measures (e.g., tablespoons) were converted to grams in accordance with nutrition guidelines [34,35].
- From 10 to 70 years—data were obtained from a study conducted by the Brazilian Institute of Geography and Statistics [13], in which the consumption of cooked rice in the last 48 h was determined on the basis of self-reports by interviewees in all Brazilian states, from 10 to 70 years.
2.4. Body Weight
2.5. Exposure Frequency
2.6. Statistical Analysis
2.7. Hypothetical Scenarios of Risk Reduction
3. Results
3.1. Cancer Risk Associated with Exposure to iAs in Rice
3.2. Non-Cancer Risk of Exposure to iAs in Rice
3.3. Exposure to iAs in Rice
3.4. Reducing the Risk
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IARC. Arsenic and Arsenic Compounds Monograph; IARC: Lyon, France, 2012; Volume 100C. [Google Scholar]
- WHO. List of Classifications; WHO: Geneva, Switzerland, 2012; Volume 1–119. [Google Scholar]
- EFSA Panel on Contaminants in the Food Chain. Statement on Tolerable Weekly Intake for Cadmium. Off. J. Eur. Union 2014, 9, 1975. [Google Scholar] [CrossRef]
- U.S. FDA. Arsenic in Rice and Rice Products Risk Assessment Report; Center for Food Safety and Applied Nutrition, Food and Drug Administration, U.S. Department of Health and Human Services: College Park, MD, USA, 2016. [Google Scholar]
- Joint FAO/WHO. Code of Practice for the Prevention and Reduction of Arsenic Contamination in Rice; Joint FAO/WHO: Geneva, Switzerland, 2017; Volume 1984. [Google Scholar]
- Zhao, F.-J.; McGrath, S.P.; Meharg, A.A. Arsenic as a Food Chain Contaminant: Mechanisms of Plant Uptake and Metabolism and Mitigation Strategies. Annu. Rev. Plant Biol. 2010, 61, 535–559. [Google Scholar] [CrossRef]
- Fu, Q.L.; Li, L.; Achal, V.; Jiao, A.Y.; Liu, Y. Concentrations of Heavy Metals and Arsenic in Market Rice Grain and Their Potential Health Risks to the Population of Fuzhou, China. Hum. Ecol. Risk Assess. 2015, 21, 117–128. [Google Scholar] [CrossRef]
- Gross, B.L.; Zhao, Z. Archaeological and Genetic Insights into the Origins of Domesticated Rice. Proc. Natl. Acad. Sci. USA 2014, 111, 6190–6197. [Google Scholar] [CrossRef]
- Ma, L.; Wang, L.; Tang, J.; Yang, Z. Arsenic Speciation and Heavy Metal Distribution in Polished Rice Grown in Guangdong Province, Southern China. Food Chem. 2017, 233, 110–116. [Google Scholar] [CrossRef]
- EFSA. Dietary Exposure to Inorganic Arsenic in the European Population; EFSA: Parma, Italy, 2014. [Google Scholar]
- Joint FAO/WHO. Evaluation of Certain Food Additives and Contaminants; Joint FAO/WHO: Geneva, Switzerland, 2011; Volume 14. [Google Scholar]
- Al-Saleh, I.; Abduljabbar, M. Heavy Metals (Lead, Cadmium, Methylmercury, Arsenic) in Commonly Imported Rice Grains (Oryza Sativa) Sold in Saudi Arabia and Their Potential Health Risk. Int. J. Hyg. Environ. Health 2017, 220, 1168–1178. [Google Scholar] [CrossRef]
- IBGE. Pesquisa de Orçamentos Familiares: 2008–2009. In Análise Do Consumo Alimentar Pessoal No Brasil; IBGE: Rio de Janeiro, Brazil, 2011. [Google Scholar]
- Li, G.; Sun, G.X.; Williams, P.N.; Nunes, L.; Zhu, Y.G. Inorganic Arsenic in Chinese Food and Its Cancer Risk. Environ. Int. 2011, 37, 1219–1225. [Google Scholar] [CrossRef]
- Ciminelli, V.S.T.; Gasparon, M.; Ng, J.C.; Silva, G.C.; Caldeira, C.L. Dietary Arsenic Exposure in Brazil: The Contribution of Rice and Beans. Chemosphere 2017, 168, 996–1003. [Google Scholar] [CrossRef]
- ATSDR. Toxicological Profile For Arsenic; ATSDR: Atlanta, GA, USA, 2007. [Google Scholar]
- JECFA. Who Food Additives Series 64: Safety Evaluation of Certain Food Additives and Contaminants, Prepared by the Seventy-Third Meeting of JECFA Lead; JECFA: Geneva, Switzerland, 2011. [Google Scholar]
- FAO. Guidance for Risk Management Options in Light of Different Risk Assessment Outcomes; FAO: Kanagawa, Japan, 2015. [Google Scholar]
- Batista, B.L.; Souza, J.M.O.; De Souza, S.S.; Barbosa, F. Speciation of Arsenic in Rice and Estimation of Daily Intake of Different Arsenic Species by Brazilians through Rice Consumption. J. Hazard. Mater. 2011, 191, 342–348. [Google Scholar] [CrossRef]
- Santos, L.M.G.; Barata-Silva, C.; Neto, S.A.V.; Magalhães, C.D.; Moreira, J.C.; Jacob, S.C. Analysis and Risk Assessment of Arsenic in Rice from Different Regions of Brazil. J. Food Compos. Anal. 2021, 99, 103853. [Google Scholar] [CrossRef]
- de Oliveira, V.F.; Busanello, C.; Viana, V.E.; Stafen, C.F.; Pedrolo, A.M.; Paniz, F.P.; Pedron, T.; Pereira, R.M.; Rosa, S.A.; de Magalhães Junior, A.M.; et al. Assessing Mineral and Toxic Elements Content in Rice Grains Grown in Southern Brazil. J. Food Compos. Anal. 2021, 100, 103914. [Google Scholar] [CrossRef]
- Paulelli, A.C.C.; Martins, A.C.; Batista, B.L.; Barbosa, F. Evaluation of Uptake, Translocation, and Accumulation of Arsenic Species by Six Different Brazilian Rice (Oryza sativa L.) Cultivars. Ecotoxicol. Environ. Saf. 2019, 169, 376–382. [Google Scholar] [CrossRef]
- Kato, L.S.; De Nadai Fernandes, E.A.; Raab, A.; Bacchi, M.A.; Feldmann, J. Arsenic and Cadmium Contents in Brazilian Rice from Different Origins Can Vary More than Two Orders of Magnitude. Food Chem. 2019, 286, 644–650. [Google Scholar] [CrossRef]
- Segura, F.R.; de Oliveira Souza, J.M.; De Paula, E.S.; da Cunha Martins, A.; Paulelli, A.C.C.; Barbosa, F.; Batista, B.L. Arsenic Speciation in Brazilian Rice Grains Organically and Traditionally Cultivated: Is There Any Difference in Arsenic Content? Food Res. Int. 2016, 89, 169–176. [Google Scholar] [CrossRef] [PubMed]
- U.S.EPA. Chemical Assessment Summary: Arsenic (Inorganic); U.S.EPA: Washington, DC, USA, 1995. [Google Scholar]
- U.S. EPA. Risk Assessment Guidance for Superfund Volume I Human Health Evaluation Manual (Part A); U.S.EPA: Washington, DC, USA, 1989; Volume I. [Google Scholar]
- U.S. EPA. Guidance on Selecting Age Groups for Monitoring and Assessing Childhood Exposures to Environmental Contaminants; U.S.EPA: Washington, DC, USA, 2005; p. 50. [Google Scholar]
- Pelletier, B.G.; Box, P.O. YASAIw Xla—A Modified Version of an Open—Source Add—in for Excel to Provide Additional Functions for Monte Carlo Simulation; Installing YASAIw. Wash. Dep. Ecol. Rep. 2009, 7710, 1–17. [Google Scholar]
- Batista, L.B. Arsenic and Rice: Monitoring and (Bio)Remediation Studies for Food Safety; FAPESP: Santo André, Brazil, 2015. [Google Scholar]
- Paniz, F.P.; Pedron, T.; Freire, B.M.; Torres, D.P.; Silva, F.F.; Batista, B.L. Effective Procedures for the Determination of As, Cd, Cu, Fe, Hg, Mg, Mn, Ni, Pb, Se, Th, Zn, U and Rare Earth Elements in Plants and Foodstuffs. Anal. Methods 2018, 10, 4094–4103. [Google Scholar] [CrossRef]
- Manual de Orientação Para Centros de Educação Infantil—CEI. Esquema Alimentar e Porcionamentos; SME: Sao Paulo, Brazil, 2011.
- Manual de Orientação Para EMEI e EMEF. Esquema Alimentar e Porcionamentos; SME: Sao Paulo, Brazil, 2011.
- Leroux, I.N.; da Silva Ferreira, A.P.S.; Paniz, F.P.; Pedron, T.; Salles, F.J.; da Silva, F.F.; Maltez, H.F.; Batista, B.L.; Olympio, K.P.K. Lead, Cadmium, and Arsenic Bioaccessibility of 24 h Duplicate Diet Ingested by Preschool Children Attending Day Care Centers in Brazil. Int. J. Environ. Res. Public Health 2018, 15, 1778. [Google Scholar] [CrossRef]
- Bompem, K.C.M.; Canella, D.C.; Bandoni, D.H.; Jaime, P.C. Manual de Medidas Caseiras e Receitas Para Cálculo Dietéticos; M. Books do Brasil Ltd.: São Paulo, Brazil, 2012; ISBN 978-85-7680-137-5. [Google Scholar]
- Tomita, L.Y.; Cardoso, M.A. Relação de Medidas Caseiras, Composição Química e Receitas de Alimentos Nipo-Brasileiros; Faculdade de Medicina de São José do Rio Preto: São José do Rio Preto, Brazil, 2000. [Google Scholar]
- IBGE. Pesquisa de Orçamentos Familiares 2008–2009. In Antropometria e Estado Nutricinal de Crianças, Adolescentes e Adultos No Brasil; IBGE: Rio de Janeiro, Brazil, 2010; Volume 46. [Google Scholar]
- U.S. EPA. Risk Assessment Guidance for Superfund (RAGS) Volume III—Part A: Process for Conducting Probabilistic Risk Assessment, Appendix B. U.S. Environ. Prot. Agency 2001, III, B1–B51. [Google Scholar]
- Ministry of Health. RDC N° 42 de 29 de Agosto de 2013; Ministry of Health: Chiyoda, Tokyo, 2013. [Google Scholar]
- Joint FAO/WHO. Report of the 12th Session of the Codex Committee on Contaminants in Foods; Joint FAO/WHO: Geneva, Switzerland, 2018. [Google Scholar]
- EC. Amending Regulation (EC) No 1881/2006 as Regards Maximum Levels of Inorganic Arsenic in Foodstuffs; EC: Brussels, Belgium, 2015; Volume 58. [Google Scholar]
- Ministry of Health of China. China’s Maximum Levels for Contaminants in Foods China; Ministry of Health of China: Beijing, China, 2014. [Google Scholar]
- Joint FAO/WHO. Joint FAO/WHO Codex Alimentarius Commission; Joint FAO/WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Althobiti, R.A.; Sadiq, N.W.; Beauchemin, D. Realistic Risk Assessment of Arsenic in Rice. Food Chem. 2018, 257, 230–236. [Google Scholar] [CrossRef]
- Conab. A Cultura Do Arroz; Conab: Brasília, Brazil, 2015. [Google Scholar]
- Ministry of Agriculture. Relatório de Gestão—Exercício 2018; Ministry of Agriculture: New Delhi, India, 2019; Volume 53. [Google Scholar]
- Batista, B.L.; Nigar, M.; Mestrot, A.; Rocha, B.A.; Júnior, F.B.; Price, A.H.; Raab, A.; Feldmann, J. Identification and Quantification of Phytochelatins in Roots of Rice to Long-Term Exposure: Evidence of Individual Role on Arsenic Accumulation and Translocation. J. Exp. Bot. 2014, 65, 1467–1479. [Google Scholar] [CrossRef]
- Sharma, S.; Kumar, R.; Sahoo, P.K.; Mittal, S. Geochemical Relationship and Translocation Mechanism of Arsenic in Rice Plants: A Case Study from Health Prone South West Punjab, India. Groundw. Sustain. Dev. 2020, 10, 100333. [Google Scholar] [CrossRef]
- Shibata, T.; Meng, C.; Umoren, J.; West, H. Risk Assessment of Arsenic in Rice Cereal and Other Dietary Sources for Infants and Toddlers in the U.S. Int. J. Environ. Res. Public Health 2016, 13, 361. [Google Scholar] [CrossRef] [PubMed]
- Segura, F.R.; Franco, D.F.; da Silva, J.J.C.; Batista, B.L. Variations in Total As and As Species in Rice Indicate the Need for Crop-Tracking. J. Food Compos. Anal. 2020, 86, 103392. [Google Scholar] [CrossRef]
- Senado Federal. Constituição Da República Federativa Do Brasil; Senado Federal, Centro Gráfico: Brasília, Brazil, 2016; Volume 6. [Google Scholar]
- WHO. Guideline: Protecting, Promoting and Supporting Breastfeeding in Facilities Providing Maternity and Newborn Services; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Senado Federal. Lei n 11.770 de 9 de Setembro de 2008; Senado Federal: Brasília, Brazil, 2008; Volume 2, p. 2008. [Google Scholar]
- Brasil. Trabalho e Família—Rumo a Novas Formas de Conciliação Com Co-Responsabilidade Social; Brasil: Brasília, Brazil, 2009. [Google Scholar]
- Chen, H.L.; Lee, C.C.; Huang, W.J.; Huang, H.T.; Wu, Y.C.; Hsu, Y.C.; Kao, Y.T. Arsenic Speciation in Rice and Risk Assessment of Inorganic Arsenic in Taiwan Population. Environ. Sci. Pollut. Res. 2016, 23, 4481–4488. [Google Scholar] [CrossRef] [PubMed]
- Meharg, A.A.; Lawgali, Y.Y.; Deacon, C.M.; Williams, P.T. Levels of Arsenic in Rice—Literature Review. Food Stand. Agency Contract CC101045 2007, 1, 1–65. [Google Scholar]
- Meharg, A.A.; Williams, P.N.; Adomako, E.; Lawgali, Y.Y.; Villada, A.; Cambell, R.C.J.; Sun, G.; Zhu, Y.; Raab, A.; Zhao, F.; et al. Geographical Variation in Total and Inorganic Arsenic Content of Polished (White) Rice. Environ. Sci. Technol. 2009, 43, 1612–1617. [Google Scholar] [CrossRef]
- Lange, C.N.; Monteiro, L.R.; Freire, B.M.; Franco, D.F.; de Souza, R.O.; dos Reis Ferreira, C.S.; da Silva, J.J.C.; Batista, B.L. Mineral Profile Exploratory Analysis for Rice Grains Traceability. Food Chem. 2019, 300, 125145. [Google Scholar] [CrossRef]
- Ministry of Health. Guia Alimentar Para a População Brasileira. Ministério Da Saúde; Ministry of Health: Chiyoda, Tokyo, 2014; ISBN 9788533421769. [Google Scholar]
- Jo, G.; Todorov, T.I. Distribution of Nutrient and Toxic Elements in Brown and Polished Rice. Food Chem. 2019, 289, 299–307. [Google Scholar] [CrossRef]
- Meharg, A.A.; Raab, A. Getting to the Bottom of Arsenic Standards and Guidelines. Environ. Sci. Technol. 2010, 44, 4395–4399. [Google Scholar] [CrossRef]
- Yim, S.R.; Park, G.Y.; Lee, K.W.; Chung, M.S.; Shim, S.M. Determination of Total Arsenic Content and Arsenic Speciation in Different Types of Rice. Food Sci. Biotechnol. 2017, 26, 293–298. [Google Scholar] [CrossRef]
- Upadhyay, M.K.; Majumdar, A.; Barla, A.; Bose, S.; Srivastava, S. An Assessment of Arsenic Hazard in Groundwater–Soil–Rice System in Two Villages of Nadia District, West Bengal, India. Environ. Geochem. Health 2019, 41, 2381–2395. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Lu, S.; Wang, J.; Yang, Y. The Arsenic Contamination of Rice in Guangdong Province, the Most Economically Dynamic Provinces of China: Arsenic Speciation and Its Potential Health Risk. Environ. Geochem. Health 2015, 37, 353–361. [Google Scholar] [CrossRef]
- Cano-Lamadrid, M.; Munera-Picazo, S.; Burló, F.; Hojjati, M.; Carbonell-Barrachina, Á.A. Total and Inorganic Arsenic in Iranian Rice. J. Food Sci. 2015, 80, T1129–T1135. [Google Scholar] [CrossRef] [PubMed]
- Cerveira, C.; Pozebon, D.; De Moraes, D.P.; Silva De Fraga, J.C. Speciation of Inorganic Arsenic in Rice Using Hydride Generation Atomic Absorption Spectrometry (HG-AAS). Anal. Methods 2015, 7, 4528–4534. [Google Scholar] [CrossRef]
- Corguinha, A.P.B.; de Souza, G.A.; Gonçalves, V.C.; de Andrade Carvalho, C.; de Lima, W.E.A.; Martins, F.A.D.; Yamanaka, C.H.; Francisco, E.A.B.; Guilherme, L.R.G. Assessing Arsenic, Cadmium, and Lead Contents in Major Crops in Brazil for Food Safety Purposes. J. Food Compos. Anal. 2015, 37, 143–150. [Google Scholar] [CrossRef]
- Fu, Y.; Chen, M.; Bi, X.; He, Y.; Ren, L.; Xiang, W.; Qiao, S.; Yan, S.; Li, Z.; Ma, Z. Occurrence of Arsenic in Brown Rice and Its Relationship to Soil Properties from Hainan Island, China. Environ. Pollut. 2011, 159, 1757–1762. [Google Scholar] [CrossRef]
- Dittmar, J.; Voegelin, A.; Maurer, F.; Roberts, L.C.; Hug, S.J.; Saha, G.C.; Ali, M.A.; Badruzzaman, A.B.M.; Kretzschmar, R. Arsenic in Soil and Irrigation Water Affects Arsenic Uptake by Rice: Complementary Insights from Field and Pot Studies. Environ. Sci. Technol. 2010, 44, 8842–8848. [Google Scholar] [CrossRef]
- Moreno-Jiménez, E.; Meharg, A.A.; Smolders, E.; Manzano, R.; Becerra, D.; Sánchez-Llerena, J.; Albarrán, Á.; López-Piñero, A. Sprinkler Irrigation of Rice Fields Reduces Grain Arsenic but Enhances Cadmium. Sci. Total Environ. 2014, 485–486, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Duan, G.; Shao, G.; Tang, Z.; Chen, H.; Wang, B.; Tang, Z.; Yang, Y.; Liu, Y.; Zhao, F.-J. Genotypic and Environmental Variations in Grain Cadmium and Arsenic Concentrations Among a Panel of High Yielding Rice Cultivars. Rice 2017, 10, 9. [Google Scholar] [CrossRef]
- Sommella, A.; Deacon, C.; Norton, G.; Pigna, M.; Violante, A.; Meharg, A.A. Total Arsenic, Inorganic Arsenic, and Other Elements Concentrations in Italian Rice Grain Varies with Origin and Type. Environ. Pollut. 2013, 181, 38–43. [Google Scholar] [CrossRef]
- Monteiro, L.R.; Lange, C.N.; Freire, B.M.; Pedron, T.; da Silva, J.J.C.; de Magalhães, A.M.; Pegoraro, C.; Busanello, C.; Batista, B.L. Inter- and Intra-Variability in the Mineral Content of Rice Varieties Grown in Various Microclimatic Regions of Southern Brazil. J. Food Compos. Anal. 2020, 92, 103535. [Google Scholar] [CrossRef]
- Avelar, A.C.; Ferreira, W.M.; Pemberthy, D.; Abad, E.; Amaral, M.A. Dioxins, Furans, Biphenyls, Arsenic, Thorium and Uranium in Natural and Anthropogenic Sources of Phosphorus and Calcium Used in Agriculture. Sci. Total Environ. 2016, 551–552, 695–698. [Google Scholar] [CrossRef]
- Ministry of Agriculture. Adubação e Calagem Para o Arroz Irrigado No Rio Grande Do Sul; Ministry of Agriculture: Pelotas, Brazil, 2007. [Google Scholar]
- da Silva, F.B.V.; do Nascimento, C.W.A.; Araújo, P.R.M. Environmental Risk of Trace Elements in P-Containing Fertilizers Marketed in Brazil. J. Soil Sci. Plant Nutr. 2017, 17, 635–647. [Google Scholar] [CrossRef]
- Campos, V. Arsenic in Groundwater Affected by Phosphate Fertilizers at São Paulo, Brazil. Environ. Geol. 2002, 42, 83–87. [Google Scholar] [CrossRef]
- Mirlean, N.; Andrus, V.E.; Baisch, P.; Griep, G.; Casartelli, M.R. Arsenic Pollution in Patos Lagoon Estuarine Sediments, Brazil. Mar. Pollut. Bull. 2003, 46, 1480–1484. [Google Scholar] [CrossRef] [PubMed]
- Alves, R.I.S.; Sampaio, C.F.; Nadal, M.; Schuhmacher, M.; Domingo, J.L.; Segura-Muñoz, S.I. Metal Concentrations in Surface Water and Sediments from Pardo River, Brazil: Human Health Risks. Environ. Res. 2014, 133, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Cagnin, R.C.; Quaresma, V.S.; Chaillou, G.; Franco, T.; Bastos, A.C. Arsenic Enrichment in Sediment on the Eastern Continental Shelf of Brazil. Sci. Total Environ. 2017, 607–608, 304–316. [Google Scholar] [CrossRef] [PubMed]
- Espinoza-Quiñones, F.R.; Módenes, A.N.; De Pauli, A.R.; Palácio, S.M. Analysis of Trace Elements in Groundwater Using ICP-OES and TXRF Techniques and Its Compliance with Brazilian Protection Standards. Water. Air. Soil Pollut. 2015, 226, 1–12. [Google Scholar] [CrossRef]
- Mirlean, N.; Baisch, P.; Diniz, D. Arsenic in Groundwater of the Paraiba Do Sul Delta, Brazil: An Atmospheric Source? Sci. Total Environ. 2014, 482–483, 148–156. [Google Scholar] [CrossRef]
- Joint FAO/WHO. Evaluation of Certain Contaminants in Food; Joint FAO/WHO: Geneva, Switzerland, 2011. [Google Scholar]
- Sengupta, M.K.; Hossain, M.A.; Mukherjee, A.; Ahamed, S.; Das, B.; Nayak, B.; Pal, A.; Chakraborti, D. Arsenic Burden of Cooked Rice: Traditional and Modern Methods. Food Chem. Toxicol. 2006, 44, 1823–1829. [Google Scholar] [CrossRef]
- Naito, S.; Matsumoto, E.; Shindoh, K.; Nishimura, T. Effects of Polishing, Cooking, and Storing on Total Arsenic and Arsenic Species Concentrations in Rice Cultivated in Japan. Food Chem. 2015, 168, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Gray, P.J.; Conklin, S.D.; Todorov, T.I.; Kasko, S.M. Cooking Rice in Excess Water Reduces Both Arsenic and Enriched Vitamins in the Cooked Grain. Food Addit. Contam. Part A-Chem. Anal. Control Expo. Risk Assess. 2016, 33, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Nachman, K.E.; Punshon, T.; Rardin, L.; Signes-Pastor, A.J.; Murray, C.J.; Jackson, B.P.; Guerinot, M.L.; Burke, T.A.; Chen, C.Y.; Ahsan, H.; et al. Opportunities and Challenges for Dietary Arsenic Intervention. Environ. Health Perspect. 2018, 126, 6–11. [Google Scholar] [CrossRef] [PubMed]
- United Nations. Transforming Our World: The 2030 Agenda for Sustainable Development; United Nations: San Francisco, CA, USA, 2015. [Google Scholar]
- Pedron, T.; Segura, F.R.; Paniz, F.P.; de Moura Souza, F.; dos Santos, M.C.; de Magalhães Júnior, A.M.; Batista, B.L. Mitigation of Arsenic in Rice Grains by Polishing and Washing: Evidencing the Benefit and the Cost. J. Cereal Sci. 2019, 87, 52–58. [Google Scholar] [CrossRef]
- Segura, F.R.; Paulelli, A.C.C.; Braga, G.Ú.L.; Dos Reis Pedreira Filho, W.; Silva, F.F.; Batista, B.L. Promising Filamentous Native Fungi Isolated from Paddy Soils for Arsenic Mitigation in Rice Grains Cultivated under Flooded Conditions. J. Environ. Chem. Eng. 2018, 6, 3926–3932. [Google Scholar] [CrossRef]
- UNICEF-WHO-UNESCO. Facts for Life, 4th ed.; UNICEF-WHO-UNESCO: New York, NY, USA, 2010. [Google Scholar]
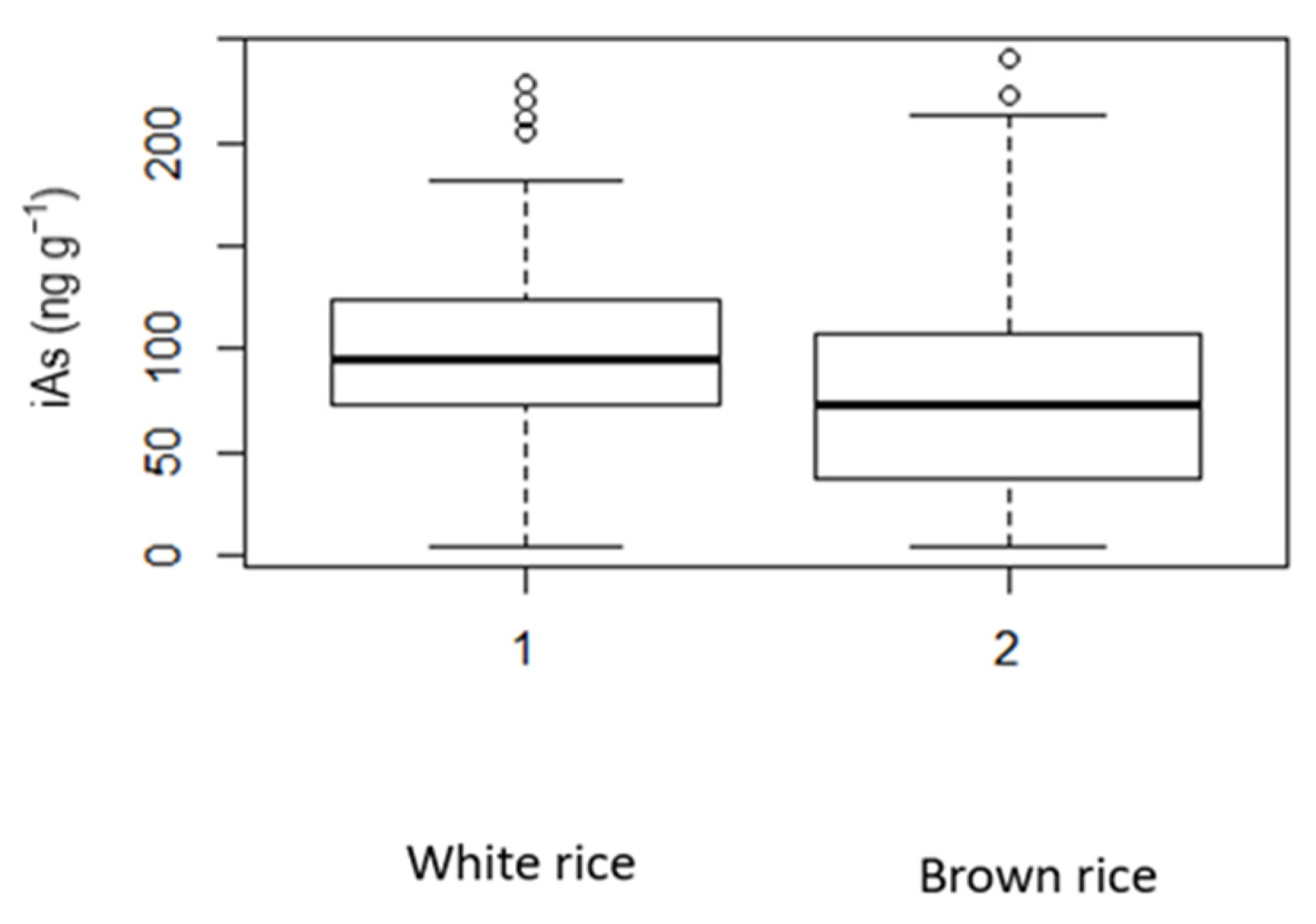

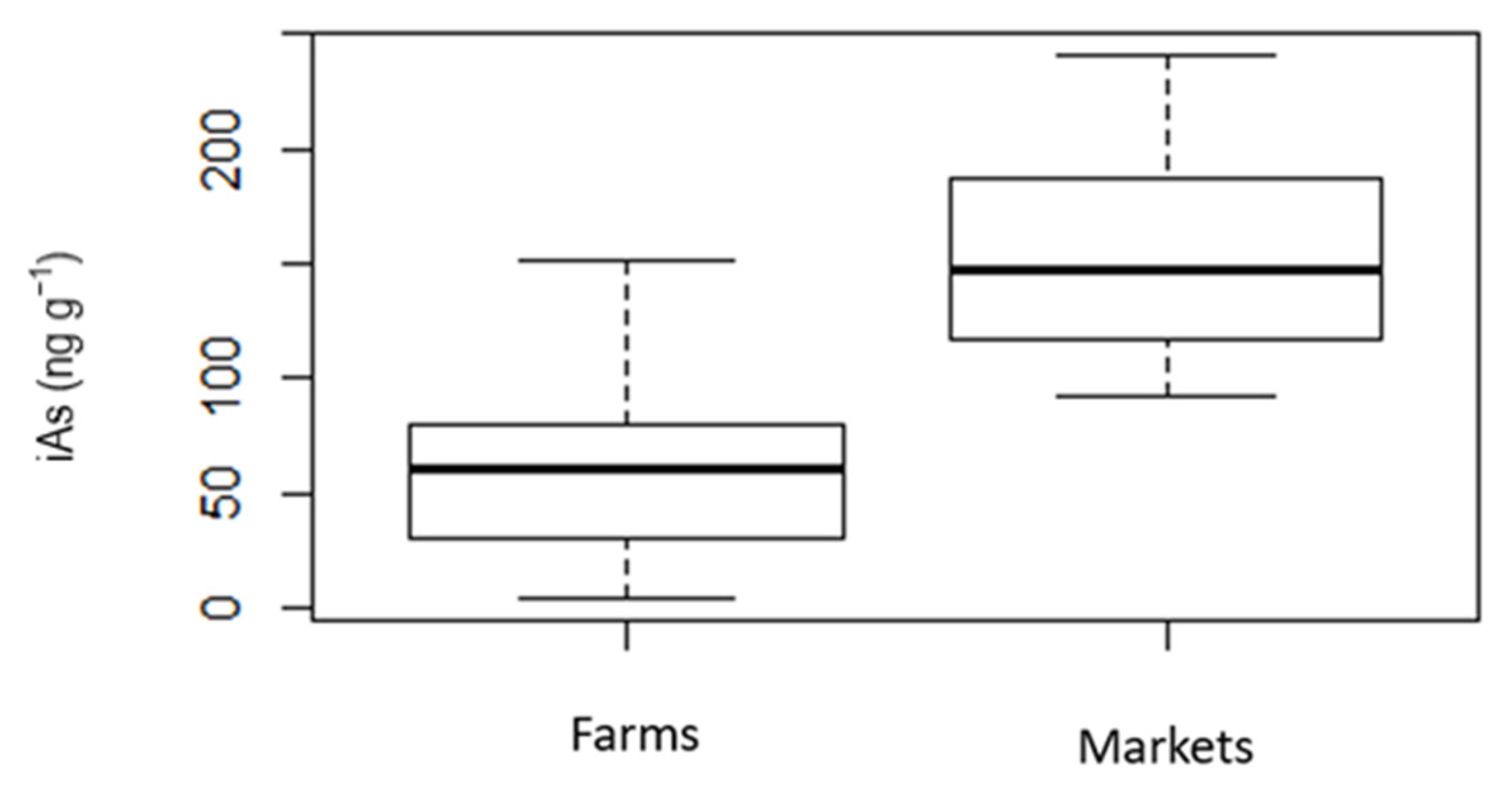
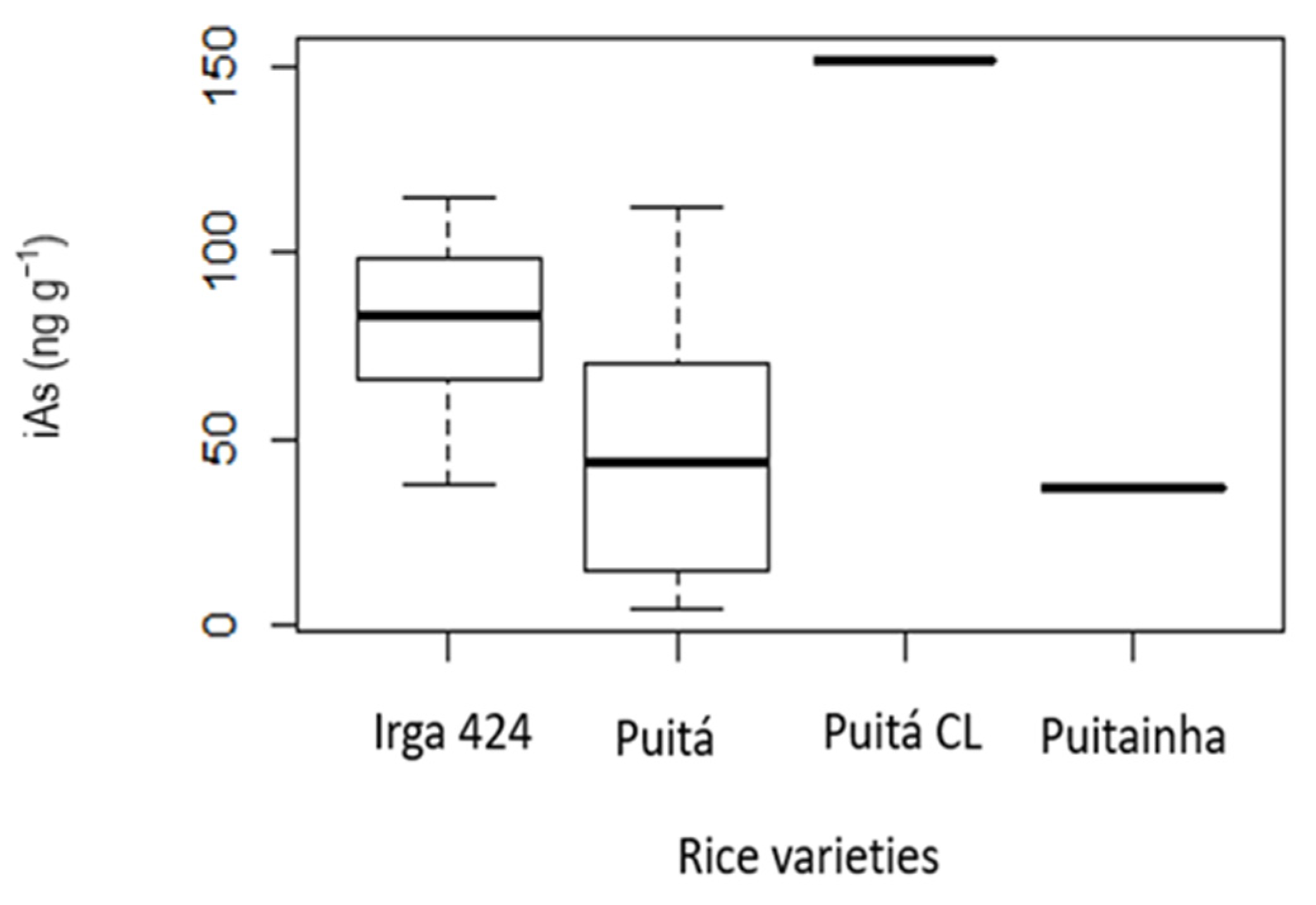
| Age (Years) | White Rice Cancer Risk and ILCR | Brown Rice Cancer Risk and ILCR | ||
|---|---|---|---|---|
| Mean | 95th Percentile | Mean | 95th Percentile | |
| <1 | 1.62 × 10−6 | 2.79 × 10−6 | 6.47 × 10−8 | 9.08 × 10−8 |
| 1 | 9.19 × 10−6 | 1.58 × 10−05 | 3.66 × 10−7 | 5.14 × 10−7 |
| 2 | 7.39 × 10−6 | 1.27 × 10−05 | 2.95 × 10−7 | 4.14 × 10−7 |
| 3 | 7.23 × 10−6 | 1.24 × 10−05 | 2.88 × 10−7 | 4.05 × 10−7 |
| 4 | 7.02 × 10−6 | 1.21 × 10−05 | 2.80 × 10−7 | 3.93 × 10−7 |
| 5 | 5.52 × 10−6 | 9.48 × 10−6 | 2.20 × 10−7 | 3.09 × 10−7 |
| 6 | 5.09 × 10−6 | 8.75 × 10−6 | 2.03 × 10−7 | 2.85 × 10−7 |
| 7 | 4.52 × 10−6 | 7.76 × 10−6 | 1.80 × 10−7 | 2.53 × 10−7 |
| 8 | 4.07 × 10−6 | 7.00 × 10−6 | 1.62 × 10−7 | 2.28 × 10−7 |
| 9 | 3.56 × 10−6 | 6.12 × 10−6 | 1.42 × 10−7 | 1.99 × 10−7 |
| 10 | 3.12 × 10−6 | 5.36 × 10−6 | 1.24 × 10−7 | 1.74 × 10−7 |
| 11 | 2.77 × 10−6 | 4.75 × 10−6 | 1.10 × 10−7 | 1.55 × 10−7 |
| 12 | 2.45 × 10−6 | 4.21 × 10−6 | 9.76 × 10−08 | 1.37 × 10−7 |
| 13 | 2.21 × 10−6 | 3.80 × 10−6 | 8.82 × 10−08 | 1.24 × 10−7 |
| 14 | 2.06 × 10−6 | 3.54 × 10−6 | 8.21 × 10−08 | 1.15 × 10−7 |
| 15 | 1.92 × 10−6 | 3.30 × 10−6 | 7.66 × 10−08 | 1.07 × 10−7 |
| 16 | 1.85 × 10−6 | 3.19 × 10−6 | 7.40 × 10−08 | 1.04 × 10−7 |
| 17 | 1.79 × 10−6 | 3.08 × 10−6 | 7.15 × 10−08 | 1.00 × 10−7 |
| 18 | 1.75 × 10−6 | 3.00 × 10−6 | 6.96 × 10−08 | 9.77 × 10−08 |
| 19 | 1.79 × 10−6 | 3.07 × 10−6 | 7.13 × 10−08 | 1.00 × 10−7 |
| 20 to <25 | 8.58 × 10−6 | 1.47 × 10−05 | 3.42 × 10−7 | 4.80 × 10−7 |
| 25 to <30 | 8.20 × 10−6 | 1.41 × 10−05 | 3.27 × 10−7 | 4.59 × 10−7 |
| 30 to <35 | 8.05 × 10−6 | 1.38 × 10−05 | 3.21 × 10−7 | 4.50 × 10−7 |
| 35 to <45 | 1.59 × 10−05 | 2.73 × 10−05 | 6.32 × 10−7 | 8.88 × 10−7 |
| 45 to <55 | 1.57 × 10−05 | 2.70 × 10−05 | 6.27 × 10−7 | 8.80 × 10−7 |
| 55 to <65 | 1.46 × 10−05 | 2.51 × 10−05 | 5.82 × 10−7 | 8.18 × 10−7 |
| 65 to <70 | 3.46 × 10−6 | 5.94 × 10−6 | 1.38 × 10−7 | 1.94 × 10−7 |
| ILCR | 1.51 × 10−4 | 2.60 × 10−4 | 6.04 × 10−6 | 8.47 × 10−6 |
| Age (Years) | White Rice | Brown Rice | ||
|---|---|---|---|---|
| Hazard Quotient | Hazard Quotient | |||
| Mean | 95th Percentile | Mean | 95th Percentile | |
| <1 | 3.61 × 10−3 | 6.20 × 10−3 | 1.44 × 10−4 | 2.04 × 10−4 |
| 1 | 2.04 × 10−2 | 3.51 × 10−2 | 8.15 × 10−4 | 1.15 × 10−3 |
| 2 | 1.64 × 10−2 | 2.82 × 10−2 | 6.55 × 10−4 | 9.27 × 10−4 |
| 3 | 1.61 × 10−2 | 2.76 × 10−2 | 6.41 × 10−4 | 9.07 × 10−4 |
| 4 | 1.56 × 10−2 | 2.68 × 10−2 | 6.23 × 10−4 | 8.81 × 10−4 |
| 5 | 1.23 × 10−2 | 2.11 × 10−2 | 4.89 × 10−4 | 6.92 × 10−4 |
| 6 | 1.13 × 10−2 | 1.94 × 10−2 | 4.51 × 10−4 | 6.38 × 10−4 |
| 7 | 1.00 × 10−2 | 1.73 × 10−2 | 4.00 × 10−4 | 5.67 × 10−4 |
| 8 | 9.06 × 10−3 | 1.56 × 10−2 | 3.61 × 10−4 | 5.11 × 10−4 |
| 9 | 7.93 × 10−3 | 1.36 × 10−2 | 3.16 × 10−4 | 4.47 × 10−4 |
| 10 | 6.93 × 10−3 | 1.19 × 10−2 | 2.76 × 10−4 | 3.91 × 10−4 |
| 11 | 6.15 × 10−3 | 1.06 × 10−2 | 2.45 × 10−4 | 3.47 × 10−4 |
| 12 | 5.45 × 10−3 | 9.35 × 10−3 | 2.17 × 10−4 | 3.07 × 10−4 |
| 13 | 4.92 × 10−3 | 8.45 × 10−3 | 1.96 × 10−4 | 2.77 × 10−4 |
| 14 | 4.58 × 10−3 | 7.86 × 10−3 | 1.82 × 10−4 | 2.58 × 10−4 |
| 15 | 4.27 × 10−3 | 7.34 × 10−3 | 1.70 × 10−4 | 2.41 × 10−4 |
| 16 | 4.13 × 10−3 | 7.09 × 10−3 | 1.64 × 10−4 | 2.33 × 10−4 |
| 17 | 3.99 × 10−3 | 6.85 × 10−3 | 1.59 × 10−4 | 2.25 × 10−4 |
| 18 | 3.88 × 10−3 | 6.67 × 10−3 | 1.55 × 10−4 | 2.19 × 10−4 |
| 19 | 3.98 × 10−3 | 6.83 × 10−3 | 1.59 × 10−4 | 2.24 × 10−4 |
| 20 to <25 | 1.91 × 10−2 | 3.28 × 10−2 | 7.60 × 10−4 | 1.08 × 10−3 |
| 25 to <30 | 1.82 × 10−2 | 3.13 × 10−2 | 7.27 × 10−4 | 1.03 × 10−3 |
| 30 to <35 | 1.79 × 10−2 | 3.07 × 10−2 | 7.14 × 10−4 | 1.01 × 10−3 |
| 35 to <45 | 3.53 × 10−2 | 6.06 × 10−2 | 1.41 × 10−3 | 1.99 × 10−3 |
| 45 to <55 | 3.50 × 10−2 | 6.01 × 10−2 | 1.39 × 10−3 | 1.97 × 10−3 |
| 55 to <65 | 3.25 × 10−2 | 5.58 × 10−2 | 1.30 × 10−3 | 1.83 × 10−3 |
| 65 to <75 | 7.69 × 10−3 | 1.32 × 10−2 | 3.07 × 10−4 | 4.34 × 10−4 |
| 0 to <70 | 3.37 × 10−01 | 5.78 × 10−01 | 1.34 × 10−2 | 1.90 × 10−2 |
| Scenario | iAs Limit | N | iAs Concentration | Distribution (Mean; SD or μ; δ) | ILCR | Reduction in ILCR | |
|---|---|---|---|---|---|---|---|
| (ng g−1) | |||||||
| (ng g−1) | Mean; SD | Mean | 95th Percentile | (%) | |||
| 1. Consumption of brown rice from farms with low iAs levels | 151.9 | 69 | 57.36; 34.36 | Log-normal (3.77; 0.87) | 5.73 × 10−6 | 8.13 × 10−6 | 5.03 |
| 2. Consumption of brown rice of the Puitá variety | 112 | 48 | 46.58; 31.68 | Log-normal (3.51; 0.91) | 5.38 × 10−6 | 7.95 × 10−6 | 10.85 |
| 3. Imposition of a white rice MCL of 100 ng g−1 | 100 | 37 | 71.94; 22.47 | Normal (71.94; 22.47) | 1.10 × 10−4 | 1.66 × 10−4 | 27.29 |
| 4. Imposition of a white rice MCL of 75 ng g−1) | 75 | 17 | 53.74; 21.15 | Normal (53.74; 21.15) | 8.27 × 10−05 | 1.35 × 10−4 | 45.39 |
| 5. Imposition of a white rice MCL of 50 ng g−1) | 50 | 6 | 30.55; 16.85 | Normal (30.55; 16.85) | 4.88 × 10−05 | 8.95 × 10−05 | 67.79 |
| 6. No consumption of white rice ≤ 6 years of age | 200 | 64 | 100.17; 44.62 | Normal (100.17; 44.62) | 1.17 × 10−4 | 1.99 × 10−4 | 22.67 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toledo, M.C.; Lee, J.S.; Batista, B.L.; Olympio, K.P.K.; Nardocci, A.C. Exposure to Inorganic Arsenic in Rice in Brazil: A Human Health Risk Assessment. Int. J. Environ. Res. Public Health 2022, 19, 16460. https://doi.org/10.3390/ijerph192416460
Toledo MC, Lee JS, Batista BL, Olympio KPK, Nardocci AC. Exposure to Inorganic Arsenic in Rice in Brazil: A Human Health Risk Assessment. International Journal of Environmental Research and Public Health. 2022; 19(24):16460. https://doi.org/10.3390/ijerph192416460
Chicago/Turabian StyleToledo, Michele C., Janice S. Lee, Bruno L. Batista, Kelly P. K. Olympio, and Adelaide C. Nardocci. 2022. "Exposure to Inorganic Arsenic in Rice in Brazil: A Human Health Risk Assessment" International Journal of Environmental Research and Public Health 19, no. 24: 16460. https://doi.org/10.3390/ijerph192416460
APA StyleToledo, M. C., Lee, J. S., Batista, B. L., Olympio, K. P. K., & Nardocci, A. C. (2022). Exposure to Inorganic Arsenic in Rice in Brazil: A Human Health Risk Assessment. International Journal of Environmental Research and Public Health, 19(24), 16460. https://doi.org/10.3390/ijerph192416460







