Does Diabetes Mellitus Increase the Risk of Avascular Osteonecrosis? A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol and Registration
2.2. Search Strategy
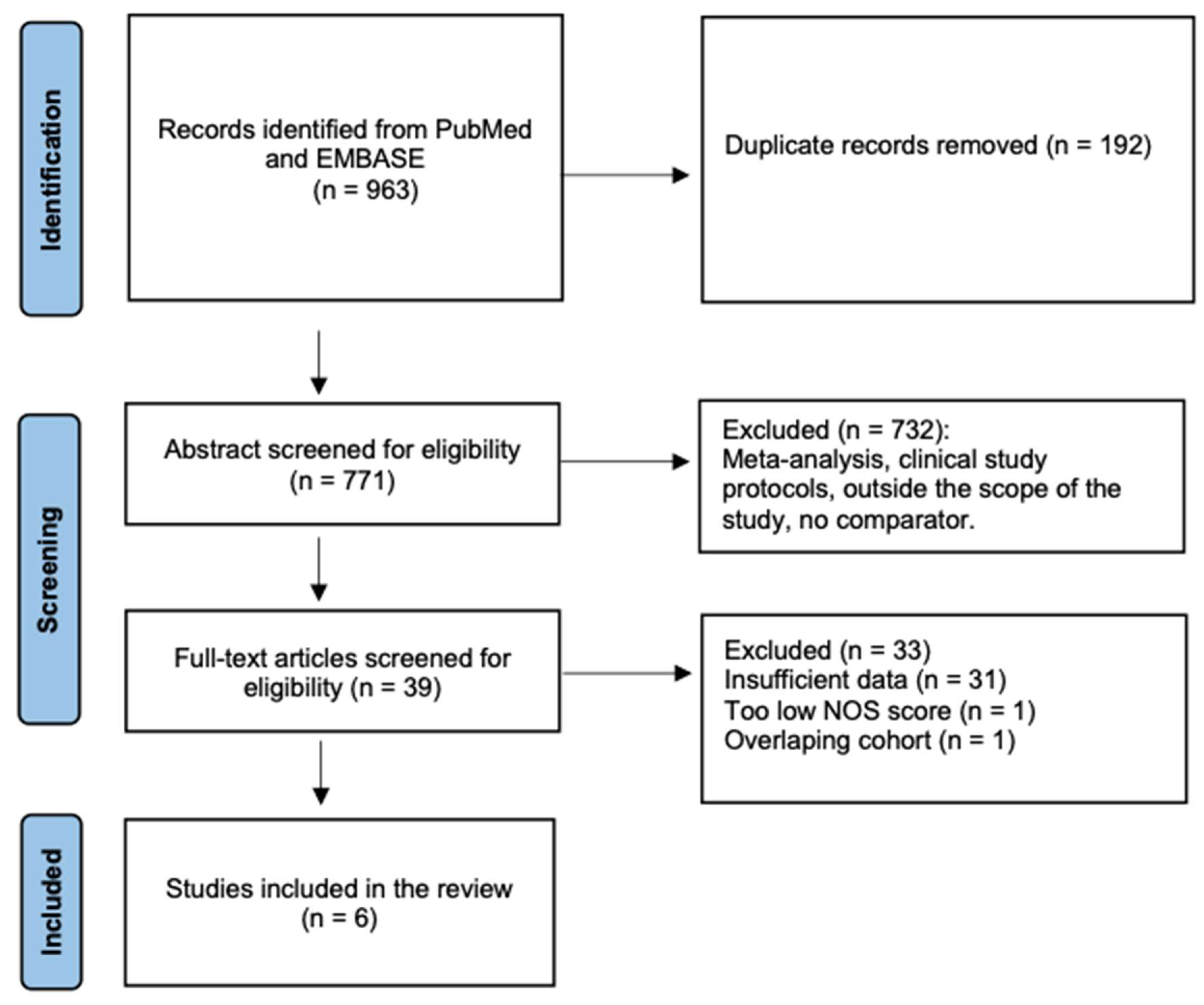
2.3. Study Selection
2.4. Data Extraction and Quality Assessment
2.5. Data Synthesis and Analysis
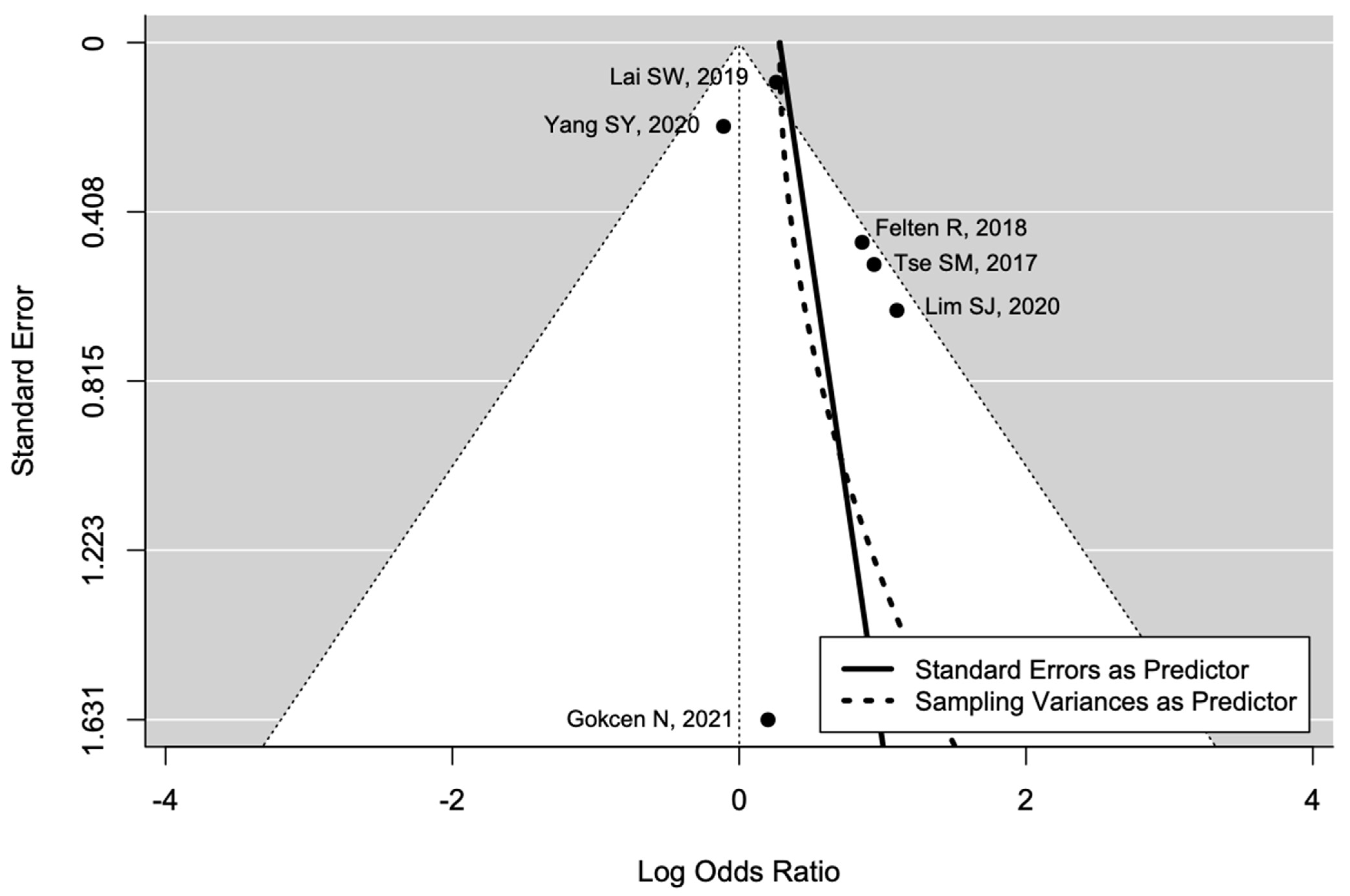
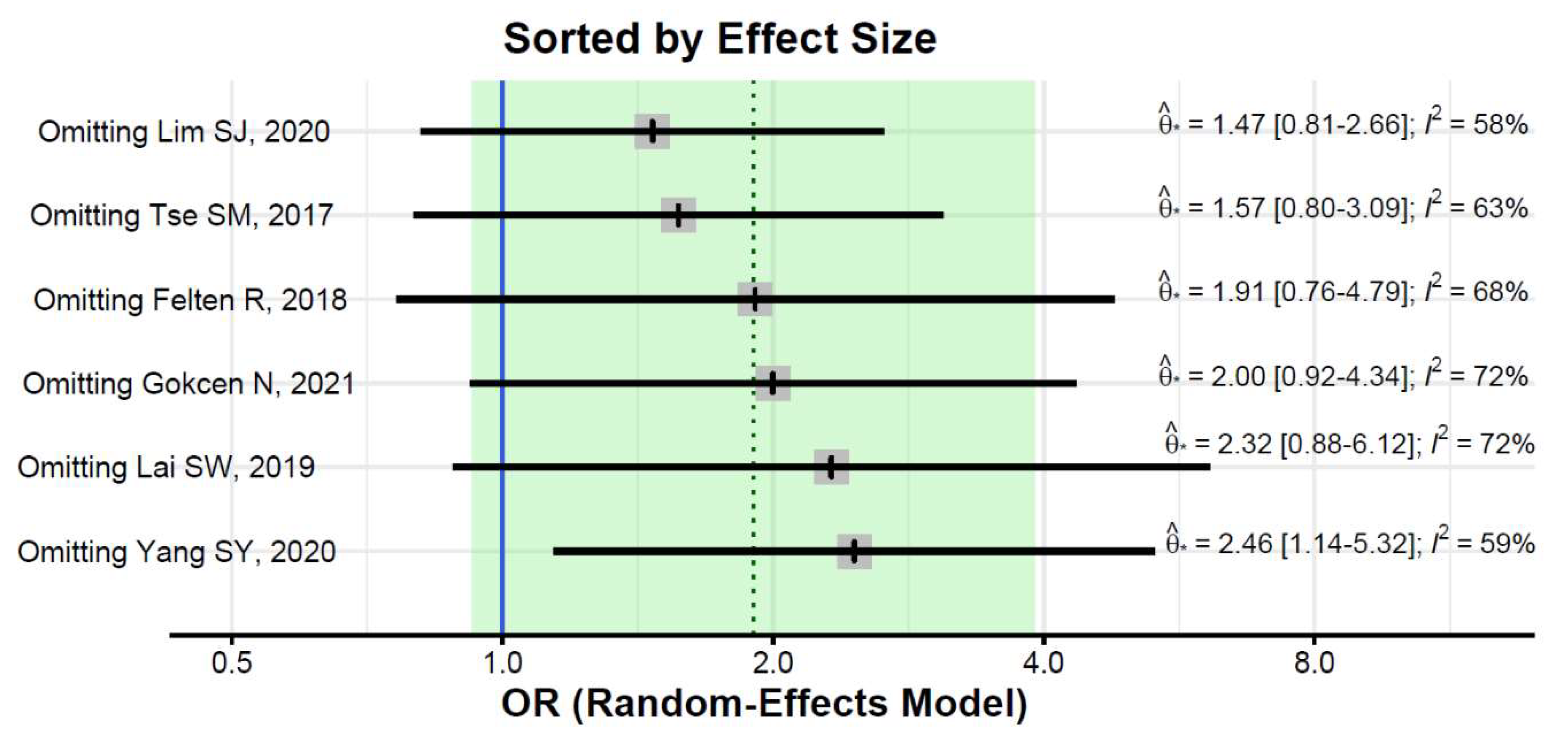
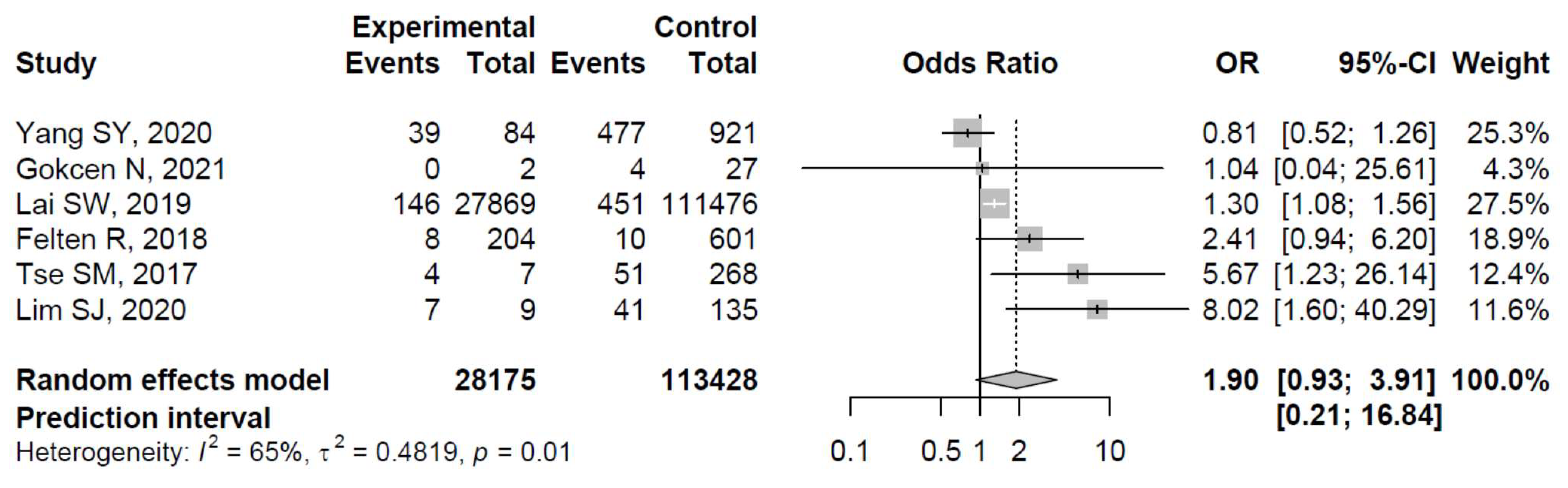
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Barney, J.; Piuzzi, N.S.; Akhondi, H. Femoral Head Avascular Necrosis; StatPearls: Treasure Island, FL, USA, 2022.
- Narayanan, A.; Khanchandani, P.; Borkar, R.M.; Ambati, C.R.; Roy, A.; Han, X.; Bhoskar, R.N.; Ragampeta, S.; Gannon, F.; Mysorekar, V.; et al. Avascular Necrosis of Femoral Head: A Metabolomic, Biophysical, Biochemical, Electron Microscopic and Histopathological Characterization. Sci. Rep. 2017, 7, 10721. [Google Scholar] [CrossRef] [PubMed]
- Petek, D.; Hannouche, D.; Suva, D. Osteonecrosis of the femoral head: Pathophysiology and current concepts of treatment. EFORT Open Rev. 2019, 4, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Shah, K.N.; Racine, J.; Jones, L.C.; Aaron, R.K. Pathophysiology and risk factors for osteonecrosis. Curr. Rev. Musculoskelet. Med. 2015, 8, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Konarski, W.; Poboży, T.; Hordowicz, M.; Śliwczyński, A.; Kotela, I.; Krakowiak, J.; Kotela, A. Bone Infarcts and Tumorigenesis—Is There a Connection? A Mini-Mapping Review. Int. J. Environ. Res. Public Health 2022, 19, 9282. [Google Scholar] [CrossRef]
- Matthews, A.H.; Davis, D.D.; Fish, M.J.; Stitson, D. Avascular Necrosis; StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Chen, S.; Kavanagh, A.; Zarick, C. Steroid-Induced Avascular Necrosis in the Foot and Ankle—Pathophysiology, Surgical, and Nonsurgical Therapies: Case Study and Literature Review. Foot Ankle Spec. 2021, 193864002110173. [Google Scholar] [CrossRef]
- Large, T.M.; Adams, M.R.; Loeffler, B.J.; Gardner, M.J. Posttraumatic Avascular Necrosis after Proximal Femur, Proximal Humerus, Talar Neck, and Scaphoid Fractures. J. Am. Acad. Orthop. Surg. 2019, 27, 794–805. [Google Scholar] [CrossRef]
- Lavernia, C.J.; Sierra, R.J.; Grieco, F.R. Osteonecrosis of the Femoral Head. J. Am. Acad. Orthop. Surg. 1999, 7, 250–261. [Google Scholar] [CrossRef]
- Mont, M.A.; Salem, H.S.; Piuzzi, N.S.; Goodman, S.B.; Jones, L.C. Nontraumatic Osteonecrosis of the Femoral Head: Where Do We Stand Today? J. Bone Jt. Surg. 2020, 102, 1084–1099. [Google Scholar] [CrossRef]
- Zhao, D.; Zhang, F.; Wang, B.; Liu, B.; Li, L.; Kim, S.-Y.; Goodman, S.B.; Hernigou, P.; Cui, Q.; Lineaweaver, W.C.; et al. Guidelines for clinical diagnosis and treatment of osteonecrosis of the femoral head in adults (2019 version). J. Orthop. Transl. 2020, 21, 100–110. [Google Scholar] [CrossRef]
- Malizos, K.N.; Karantanas, A.H.; Varitimidis, S.E.; Dailiana, Z.H.; Bargiotas, K.; Maris, T. Osteonecrosis of the femoral head: Etiology, imaging and treatment. Eur. J. Radiol. 2007, 63, 16–28. [Google Scholar] [CrossRef]
- Coskun Benlidayi, I.; Guzel, R. Oral Bisphosphonate Related Osteonecrosis of the Jaw: A Challenging Adverse Effect. ISRN Rheumatol. 2013, 2013, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, Z.; Camacho, P.M. Rare adverse effects of bisphosphonate therapy. Curr. Opin. Endocrinol. Diabetes Obes. 2019, 26, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Konarski, W.; Poboży, T.; Śliwczyński, A.; Kotela, I.; Krakowiak, J.; Hordowicz, M.; Kotela, A. Avascular Necrosis of Femoral Head—Overview and Current State of the Art. Int. J. Environ. Res. Public Health 2022, 19, 7348. [Google Scholar] [CrossRef] [PubMed]
- Quaranta, M.; Miranda, L.; Oliva, F.; Aletto, C.; Maffulli, N. Osteotomies for avascular necrosis of the femoral head. Br. Med. Bull. 2021, 137, 98–111. [Google Scholar] [CrossRef]
- Lamb, J.N.; Holton, C.; O’Connor, P.; Giannoudis, P.V. Avascular necrosis of the hip. BMJ 2019, l2178. [Google Scholar] [CrossRef]
- Konarski, W.; Poboży, T.; Kotela, A.; Śliwczyński, A.; Kotela, I.; Hordowicz, M.; Krakowiak, J. The Risk of Avascular Necrosis Following the Stabilization of Femoral Neck Fractures: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 19, 10050. [Google Scholar] [CrossRef]
- Grond, S.E.; Little, R.E.; Campbell, D.A.; Loehrl, T.A.; Poetker, D.M. Oral corticosteroid use and the risk of developing avascular necrosis: A large retrospective review. Int. Forum Allergy Rhinol. 2022, 12, 903–909. [Google Scholar] [CrossRef]
- Yoon, B.-H.; Jones, L.C.; Chen, C.-H.; Cheng, E.Y.; Cui, Q.; Drescher, W.; Fukushima, W.; Gangji, V.; Goodman, S.B.; Ha, Y.-C.; et al. Etiologic Classification Criteria of ARCO on Femoral Head Osteonecrosis Part 2: Alcohol-Associated Osteonecrosis. J. Arthroplasty 2019, 34, 169–174.e1. [Google Scholar] [CrossRef]
- Jeong, H.-J.; Kim, D.; Cho, S.-K.; Kim, Y.; Bae, S.-C.; Sung, Y.-K. Clinical characteristics of multifocal osteonecrosis in Korean patients with rheumatic disease. Int. J. Rheum. Dis. 2018, 21, 1301–1308. [Google Scholar] [CrossRef]
- Li, X.; Brazauskas, R.; Wang, Z.; Al-Seraihy, A.; Baker, K.S.; Cahn, J.-Y.; Frangoul, H.A.; Gajewski, J.L.; Hale, G.A.; Hsu, J.W.; et al. Avascular Necrosis of Bone after Allogeneic Hematopoietic Cell Transplantation in Children and Adolescents. Biol. Blood Marrow Transplant. 2014, 20, 587–592. [Google Scholar] [CrossRef]
- Bayard, C.; Ledergerber, B.; Flepp, M.; Lecompte, T.; Moulin, E.; Hoffmann, M.; Weber, R.; Staehelin, C.; Di Benedetto, C.; Fux, C.A.; et al. Associations Between Antiretroviral Treatment and Avascular Bone Necrosis: The Swiss HIV Cohort Study. Open Forum Infect. Dis. 2017, 4, ofx177. [Google Scholar] [CrossRef]
- Paul, S.; Ali, A.; Katare, R. Molecular complexities underlying the vascular complications of diabetes mellitus—A comprehensive review. J. Diabetes Complicat. 2020, 34, 107613. [Google Scholar] [CrossRef] [PubMed]
- Chawla, A.; Chawla, R.; Jaggi, S. Microvasular and macrovascular complications in diabetes mellitus: Distinct or continuum? Indian J. Endocrinol. Metab. 2016, 20, 546. [Google Scholar] [CrossRef]
- Huang, D.; Refaat, M.; Mohammedi, K.; Jayyousi, A.; Al Suwaidi, J.; Abi Khalil, C. Macrovascular Complications in Patients with Diabetes and Prediabetes. Biomed Res. Int. 2017, 2017, 7839101. [Google Scholar] [CrossRef]
- Khalil, H. Diabetes microvascular complications—A clinical update. Diabetes Metab. Syndr. Clin. Res. Rev. 2017, 11, S133–S139. [Google Scholar] [CrossRef] [PubMed]
- Peer, A.; Khamaisi, M. Diabetes as a Risk Factor for Medication-Related Osteonecrosis of the Jaw. J. Dent. Res. 2015, 94, 252–260. [Google Scholar] [CrossRef]
- Rahimi-Nedjat, R.; Sagheb, K.; Pabst, A.; Olk, L.; Walter, C. Diabetes Mellitus and Its Association to the Occurrence of Medication-Related Osteonecrosis of the Jaw. Dent. J. 2016, 4, 17. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Yang, S.Y.; Zeng, L.Y.; Li, C.; Yan, H. Correlation between an ABO Blood Group and Primary Femoral Head Necrosis: A Case–Control Study. Orthop. Surg. 2020, 12, 450–456. [Google Scholar] [CrossRef]
- Gokcen, N.; Komac, A.; Tuncer, F.; Kocak Buyuksutcu, G.; Ozdemir Isik, O.; Yazici, A.; Cefle, A. Risk factors of avascular necrosis in Takayasu arteritis: A cross sectional study. Rheumatol. Int. 2022, 42, 529–534. [Google Scholar] [CrossRef]
- Lai, S.W.; Lin, C.L.; Liao, K.F. Real-world database examining the association between avascular necrosis of the femoral head and diabetes in Taiwan. Diabetes Care 2019, 42, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Felten, R.; Perrin, P.; Caillard, S.; Moulin, B.; Javier, R.M. Avascular osteonecrosis in kidney transplant recipients: Risk factors in a recent cohort study and evaluation of the role of secondary hyperparathyroidism. PLoS ONE 2019, 14, e0212931. [Google Scholar] [CrossRef] [PubMed]
- Tse, S.M.; Mok, C.C. Time trend and risk factors of avascular bone necrosis in patients with systemic lupus erythematosus. Lupus 2017, 26, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.J.; Yeo, I.; Park, C.W.; Lee, H.; Park, Y.S.; Lee, J., II. Risk factors for osteonecrosis of the femoral head in brain tumor patients receiving corticosteroid after surgery. PLoS ONE 2020, 15, e0238368. [Google Scholar] [CrossRef]
- Vannala, V.; Palaian, S.; Shankar, P.R. Therapeutic Dimensions of Bisphosphonates: A Clinical Update. Int. J. Prev. Med. 2020, 11, 166. [Google Scholar] [CrossRef]
- Martins, L.H.I.; Ferreira, D.C.; Silva, M.T.; Motta, R.H.L.; Franquez, R.T.; de Cássia Bergamaschi Bergamaschi, C. Frequency of osteonecrosis in bisphosphonate users submitted to dental procedures: A systematic review. Oral Dis. 2021. [Google Scholar] [CrossRef]
- Payne, K.F.; Goodson, A.M.; Tahim, A.S.; Rafi, I.; Brennan, P.A. Why worry about bisphosphonate-related osteonecrosis of the jaw? A guide to diagnosis, initial management, and referral of patients. Br. J. Gen. Pract. 2017, 67, 330–331. [Google Scholar] [CrossRef]
- Wilkinson, G.S.; Kuo, Y.-F.; Freeman, J.L.; Goodwin, J.S. Intravenous Bisphosphonate Therapy and Inflammatory Conditions or Surgery of the Jaw: A Population-Based Analysis. JNCI J. Natl. Cancer Inst. 2007, 99, 1016–1024. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]

| Study | Population | Study Design | AVN Sites | Mean Age in AVN Cases | Male % in AVN Cases | Corticosteroid Use in AVN Cases | NOS |
|---|---|---|---|---|---|---|---|
| Yang SY, 2020 [31] | Primary and secondary femoral head AVN vs. extremity factures | Retrospective, case-control | Femoral head | 57 | 52% | 6 | |
| Gokcen N, 2021 [32] | Takayasu arteritis | Retrospective cross sectional | Femoral head | 38 | 100% | Mean duration of corticosteroid use of 7.5 years | 6 |
| Lai SW, 2019 [33] | General population (diabetes vs. non-diabetes) | Retrospective cohort study | Femoral head | - | - | - | 9 |
| Felten R, 2018 [34] | Kidney transplant recipient | Retrospective cohort study | Femoral head (15); femoral condyle (2); and ankle (1) | 55 | 22% | Median total dose of prednisolone 3 month after transplant was 2594 mg | 8 |
| Tse SM, 2017 [35] | Systemic lupus erythematosus | Retrospective cohort study | Hip (82%); femoral condyle (9%); and humeral head (5%) | 34 | 13% | All used corticosteroids | 8 |
| Lim SJ, 2020 [36] | Primary brain tumors after surgery | Retrospective cohort study | Femoral head | 36 | 50% | All used corticosteroids | 9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konarski, W.; Poboży, T.; Kotela, A.; Śliwczyński, A.; Kotela, I.; Hordowicz, M.; Krakowiak, J. Does Diabetes Mellitus Increase the Risk of Avascular Osteonecrosis? A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2022, 19, 15219. https://doi.org/10.3390/ijerph192215219
Konarski W, Poboży T, Kotela A, Śliwczyński A, Kotela I, Hordowicz M, Krakowiak J. Does Diabetes Mellitus Increase the Risk of Avascular Osteonecrosis? A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2022; 19(22):15219. https://doi.org/10.3390/ijerph192215219
Chicago/Turabian StyleKonarski, Wojciech, Tomasz Poboży, Andrzej Kotela, Andrzej Śliwczyński, Ireneusz Kotela, Martyna Hordowicz, and Jan Krakowiak. 2022. "Does Diabetes Mellitus Increase the Risk of Avascular Osteonecrosis? A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 19, no. 22: 15219. https://doi.org/10.3390/ijerph192215219
APA StyleKonarski, W., Poboży, T., Kotela, A., Śliwczyński, A., Kotela, I., Hordowicz, M., & Krakowiak, J. (2022). Does Diabetes Mellitus Increase the Risk of Avascular Osteonecrosis? A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 19(22), 15219. https://doi.org/10.3390/ijerph192215219