Accelerometer Cut-Points for Physical Activity Assessment in Adults with Mild to Moderate Huntington’s Disease: A Cross-Sectional Multicentre Study
Abstract
1. Introduction
2. Material and Methods
2.1. Design
2.2. Participants
2.3. Ethics
2.4. Procedure
2.4.1. Test Protocol
2.4.2. Assessments
2.4.3. Accelerometers
2.5. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Recommendations on Physical Activity for Health; World Health Organization: Geneva, Switzerland, 2011. Available online: http://apps.who.int/iris/bitstream/handle/10665/44399/9789241599979_eng.pdf;jsessionid=F720BE062F514924A03B872A30E82525?sequence=1 (accessed on 6 June 2022).
- Nuzum, H.; Stickel, A.; Corona, M.; Zeller, M.; Melrose, R.J.; Wilkins, S.S. Potential Benefits of Physical Activity in MCI and Dementia. Behav. Neurol. 2020, 2020, 7807856. [Google Scholar] [CrossRef] [PubMed]
- Do, K.; Laing, B.T.; Landry, T.; Bunner, W.; Mersaud, N.; Matsubara, T.; Li, P.; Yuan, Y.; Lu, Q.; Huang, H. The effects of exercise on hypothalamic neurodegeneration of Alzheimer’s disease mouse model. PLoS ONE 2018, 13, e0190205. [Google Scholar] [CrossRef] [PubMed]
- Bhalsing, K.S.; Abbas, M.M.; Tan, L.C.S. Role of Physical Activity in Parkinson’s Disease. Ann. Indian Acad. Neurol. 2018, 21, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Quinn, N.; Schrag, A. Huntington’s disease and other choreas. J. Neurol. 1998, 245, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Perandones, C.; Radrizzani, M.; Micheli, F. Enfermedad de Huntington. In Movimientos Anormales Clínica y Terapéutica; Médica Panamericana: Madrid, Spain, 2011; pp. 307–333. [Google Scholar]
- Myers, R.H.; Sax, D.S.; Koroshetz, W.J.; Mastromauro, C.; Cupples, L.A.; Kiely, D.K.; Pettengill, F.K.; Bird, E.D. Factors Associated with Slow Progression in Huntington’s Disease. Arch. Neurol. 1991, 48, 800–804. [Google Scholar] [CrossRef]
- Cruickshank, T.; Bartlett, D.; Govus, A.; Hannan, A.; Teo, W.-P.; Mason, S.; Lo, J.; Ziman, M. The relationship between lifestyle and serum neurofilament light protein in Huntington’s disease. Brain Behav. 2020, 10, e01578. [Google Scholar] [CrossRef]
- Strain, T.; Wijndaele, K.; Dempsey, P.C.; Sharp, S.J.; Pearce, M.; Jeon, J.; Lindsay, T.; Wareham, N.; Brage, S. Wearable-device-measured physical activity and future health risk. Nat. Med. 2020, 26, 1385–1391. [Google Scholar] [CrossRef]
- Nero, H.; Wallén, M.B.; Franzén, E.; Ståhle, A.; Hagstromer, M. Accelerometer Cut Points for Physical Activity Assessment of Older Adults with Parkinson’s Disease. PLoS ONE 2015, 10, e0135899. [Google Scholar] [CrossRef]
- Sasaki, J.E.; John, D.; Freedson, P.S. Validation and comparison of ActiGraph activity monitors. J. Sci. Med. Sport 2011, 14, 411–416. [Google Scholar] [CrossRef]
- Duncan, M.J.; Dobell, A.; Noon, M.; Clark, C.C.T.; Roscoe, C.M.P.; Faghy, M.A.; Stodden, D.; Sacko, R.; Eyre, E.L.J.; Dobell, A. Calibration and Cross-Validation of Accelerometery for Estimating Movement Skills in Children Aged 8–12 Years. Sensors 2020, 20, 2776. [Google Scholar] [CrossRef]
- Lee, P.; Tse, C.Y. Calibration of wrist-worn ActiWatch 2 and ActiGraph wGT3X for assessment of physical activity in young adults. Gait Posture 2019, 68, 141–149. [Google Scholar] [CrossRef]
- Nichols, J.F.; Morgan, C.G.; Chabot, L.E.; Sallis, J.F.; Calfas, K.J. Assessment of Physical Activity with the Computer Science and Applications, Inc., Accelerometer: Laboratory versus Field Validation. Res. Q. Exerc. Sport 2000, 71, 36–43. [Google Scholar] [CrossRef]
- Kieburtz, K. Unified Huntington’s disease rating scale: Reliability and consistency. Huntington Study Group. Mov. Disord. 1996, 11, 136–142. [Google Scholar] [CrossRef]
- Martínez-González, M.Á. The SUN cohort study (Seguimiento University of Navarra). Public Health Nutr. 2006, 9, 127–131. [Google Scholar] [CrossRef]
- Kingma, E.M.; van Duijn, E.; Timman, R.; van der Mast, R.C.; Roos, R.A. Behavioural problems in Huntington’s disease using the Problem Behaviours Assessment. Gen. Hosp. Psychiatry 2008, 30, 155–161. [Google Scholar] [CrossRef]
- Ringkøbing, S.P.; Larsen, I.U.; Jørgensen, K.; Vinther-Jensen, T.; Vogel, A. Cognitive Screening Tests in Huntington Gene Mutation Carriers: Examining the Validity of the Mini-Mental State Examination and the Montreal Cognitive Assessment. J. Huntington’s Dis. 2020, 9, 59–68. [Google Scholar] [CrossRef]
- Treuth, M.S.; Schmitz, K.; Catellier, D.J.; McMurray, R.G.; Murray, D.M.; Almeida, M.J.; Going, S.; Norman, J.E.; Pate, R. Defining accelerometer thresholds for activity intensities in adolescent girls. Med. Sci. Sport. Exerc. 2004, 36, 1259–1266. [Google Scholar]
- World Health Organization. Global Database on Body Mass Index [Internet]. Available online: http://www.assessmentpsychology.com/icbmi.htm#:~:text=Global%20Database%20on%20Body%20Mass%20Index%20-%20World%20Health%20Organization&text=Body%20Mass%20Index%20(BMI)%20is%20(kg%2Fm2) (accessed on 1 May 2022).
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef]
- Welk, G.J. Principles of Design and Analyses for the Calibration of Accelerometry-Based Activity Monitors. Med. Sci. Sport. Exerc. 2005, 37 (Suppl. 11), S501–S511. [Google Scholar] [CrossRef]
- Crouter, S.E.; Horton, M.; Bassett, D.R. Validity of ActiGraph Child-Specific Equations during Various Physical Activities. Med. Sci. Sport Exerc. 2013, 45, 1403–1409. [Google Scholar] [CrossRef]
- Diaz, K.M.; Krupka, D.J.; Chang, M.J.; Kronish, I.M.; Moise, N.; Goldsmith, J.; Schwartz, J.E. Wrist-based cut-points for moderate- and vigorous-intensity physical activity for the Actical accelerometer in adults. J. Sport Sci. 2018, 36, 206–212. [Google Scholar] [CrossRef] [PubMed]
- Keadle, S.K.; Shiroma, E.J.; Freedson, P.S.; Lee, I.-M. Impact of accelerometer data processing decisions on the sample size, wear time and physical activity level of a large cohort study. BMC Public Health 2014, 14, 1210. [Google Scholar] [CrossRef] [PubMed]
- Santos-Lozano, A.; Santín-Medeiros, F.; Cardon, G.; Torres-Luque, G.; Bailón, R.; Bergmeir, C.; Ruiz, J.R.; Lucia, A.; Garatachea, N. Actigraph GT3X: Validation and Determination of Physical Activity Intensity Cut Points. Int. J. Sport. Med. 2013, 34, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Jeng, B.; Cederberg, K.L.; Lai, B.; Sasaki, J.E.; Bamman, M.M.; Motl, R.W. Wrist-based accelerometer cut-points for quantifying moderate-to-vigorous intensity physical activity in Parkinson’s disease. Gait Posture 2022, 91, 235–239. [Google Scholar] [CrossRef]
- Mark, A.E.; Prince, S.A.; Reed, J.L.; Reid, R.D. Actigraph GT3X+ cut points in coronary artery disease patients: A pilot study. Health Fit. J. Can. 2015, 8, 13–21. [Google Scholar] [CrossRef]
- O’Brien, C.M.; Duda, J.L.; Kitas, G.D.; Van Zanten, J.J.C.S.V.; Metsios, G.S.; Fenton, S.A.M. Objective measurement of sedentary time and physical activity in people with rheumatoid arthritis: Protocol for an accelerometer and activPALTM validation study. Mediterr. J. Rheumatol. 2019, 30, 125–134. [Google Scholar] [CrossRef]
- Motl, R.W.; Snook, E.M.; Agiovlasitis, S.; Suh, Y. Calibration of Accelerometer Output for Ambulatory Adults With Multiple Sclerosis. Arch. Phys. Med. Rehabil. 2009, 90, 1778–1784. [Google Scholar] [CrossRef]
- Serra, M.C.; Balraj, E.; DiSanzo, B.L.; Ivey, F.M.; Hafer-Macko, C.E.; Treuth, M.S.; Ryan, A.S. Validating accelerometry as a measure of physical activity and energy expenditure in chronic stroke. Top. Stroke Rehabil. 2017, 24, 18–23. [Google Scholar] [CrossRef]
- Trost, S.G.; Mciver, K.L.; Pate, R.R. Conducting Accelerometer-Based Activity Assessments in Field-Based Research. Med. Sci. Sport Exerc. 2005, 37, S531–S543. [Google Scholar] [CrossRef]
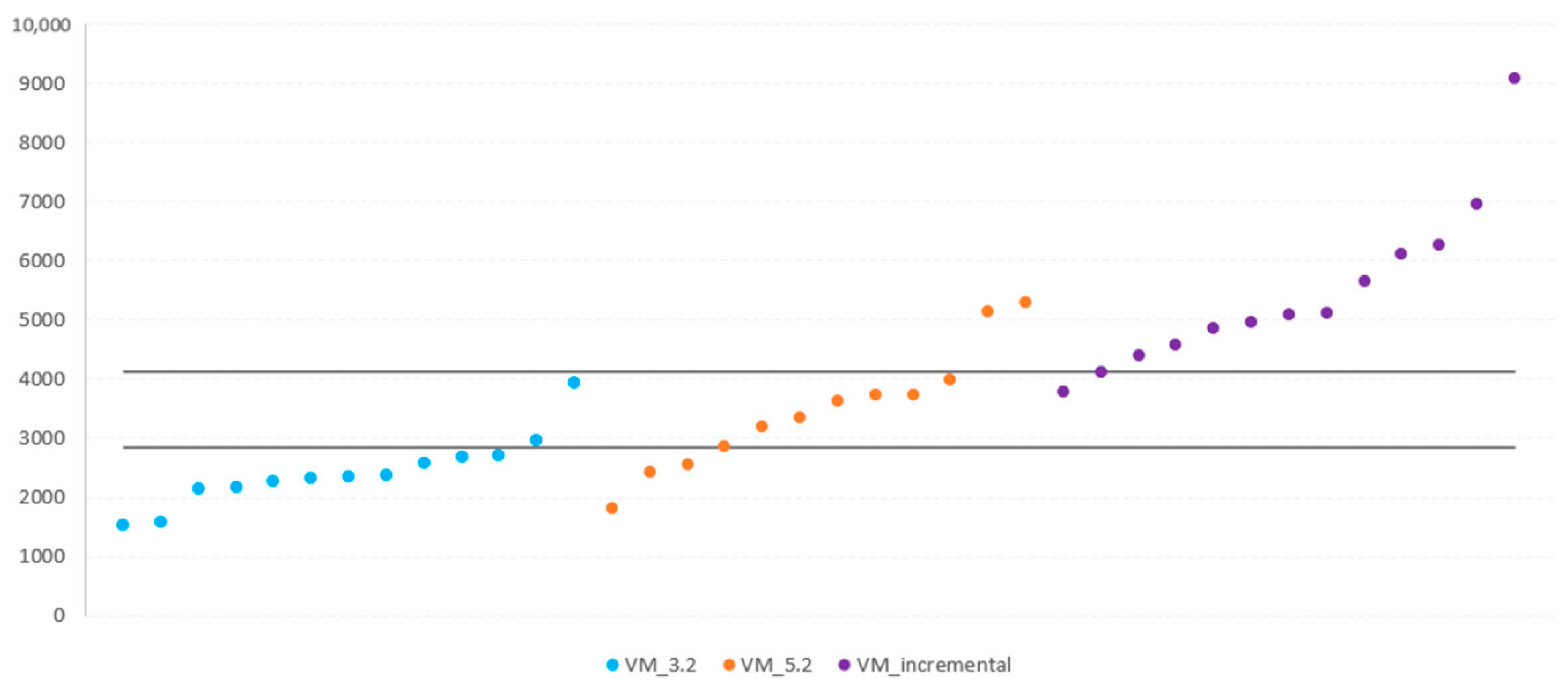
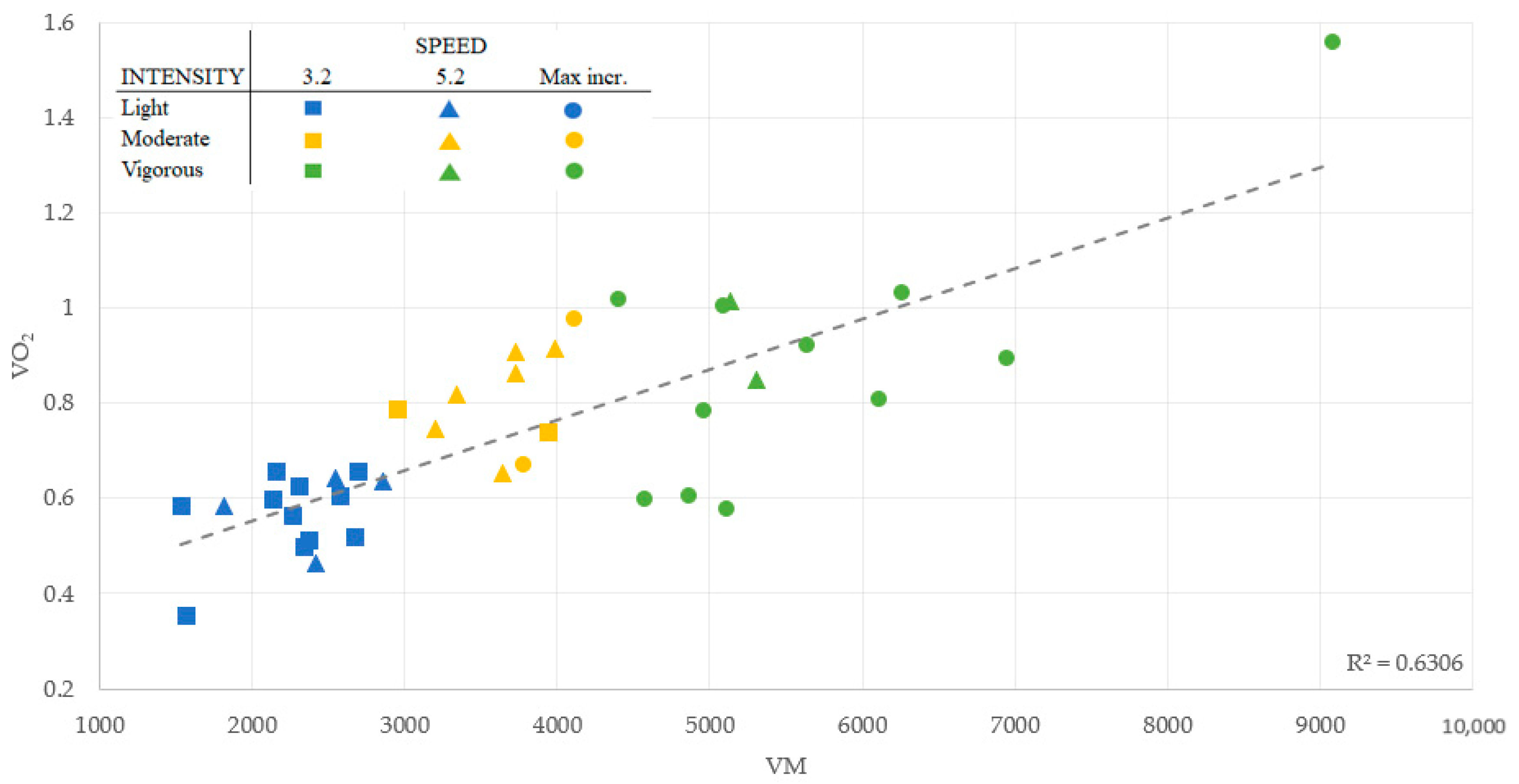

| Axis | Speed a | Sensitivity (%) | Specificity (%) | AUC (95% CI) b | Cut-Point |
|---|---|---|---|---|---|
| VM | 3.2 | 75 | 84.6 | 0.82051 (0.61639–0.94377) | ≤2852 |
| 5.2 | 2852–4117 | ||||
| Incremental test | 92.3 | 83.3 | 0.89103 (0.70174–0.97935) | >4117 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simón-Vicente, L.; Rivadeneyra-Posadas, J.; Soto-Célix, M.; Raya-González, J.; Castillo, D.; Calvo, S.; Collazo, C.; Rodríguez-Fernández, A.; Fahed, V.S.; Mariscal, N.; et al. Accelerometer Cut-Points for Physical Activity Assessment in Adults with Mild to Moderate Huntington’s Disease: A Cross-Sectional Multicentre Study. Int. J. Environ. Res. Public Health 2022, 19, 14834. https://doi.org/10.3390/ijerph192214834
Simón-Vicente L, Rivadeneyra-Posadas J, Soto-Célix M, Raya-González J, Castillo D, Calvo S, Collazo C, Rodríguez-Fernández A, Fahed VS, Mariscal N, et al. Accelerometer Cut-Points for Physical Activity Assessment in Adults with Mild to Moderate Huntington’s Disease: A Cross-Sectional Multicentre Study. International Journal of Environmental Research and Public Health. 2022; 19(22):14834. https://doi.org/10.3390/ijerph192214834
Chicago/Turabian StyleSimón-Vicente, Lucía, Jéssica Rivadeneyra-Posadas, María Soto-Célix, Javier Raya-González, Daniel Castillo, Sara Calvo, Carla Collazo, Alejandro Rodríguez-Fernández, Vitoria S. Fahed, Natividad Mariscal, and et al. 2022. "Accelerometer Cut-Points for Physical Activity Assessment in Adults with Mild to Moderate Huntington’s Disease: A Cross-Sectional Multicentre Study" International Journal of Environmental Research and Public Health 19, no. 22: 14834. https://doi.org/10.3390/ijerph192214834
APA StyleSimón-Vicente, L., Rivadeneyra-Posadas, J., Soto-Célix, M., Raya-González, J., Castillo, D., Calvo, S., Collazo, C., Rodríguez-Fernández, A., Fahed, V. S., Mariscal, N., García-Bustillo, Á., Aguado, L., & Cubo, E. (2022). Accelerometer Cut-Points for Physical Activity Assessment in Adults with Mild to Moderate Huntington’s Disease: A Cross-Sectional Multicentre Study. International Journal of Environmental Research and Public Health, 19(22), 14834. https://doi.org/10.3390/ijerph192214834








