Effects of Interval Walking Training on Oral Health Status in Middle-Aged and Older Adults: A Case-Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Study Protocol
2.2.1. Physical Exercise Intervention Program (Interval Walking Training)
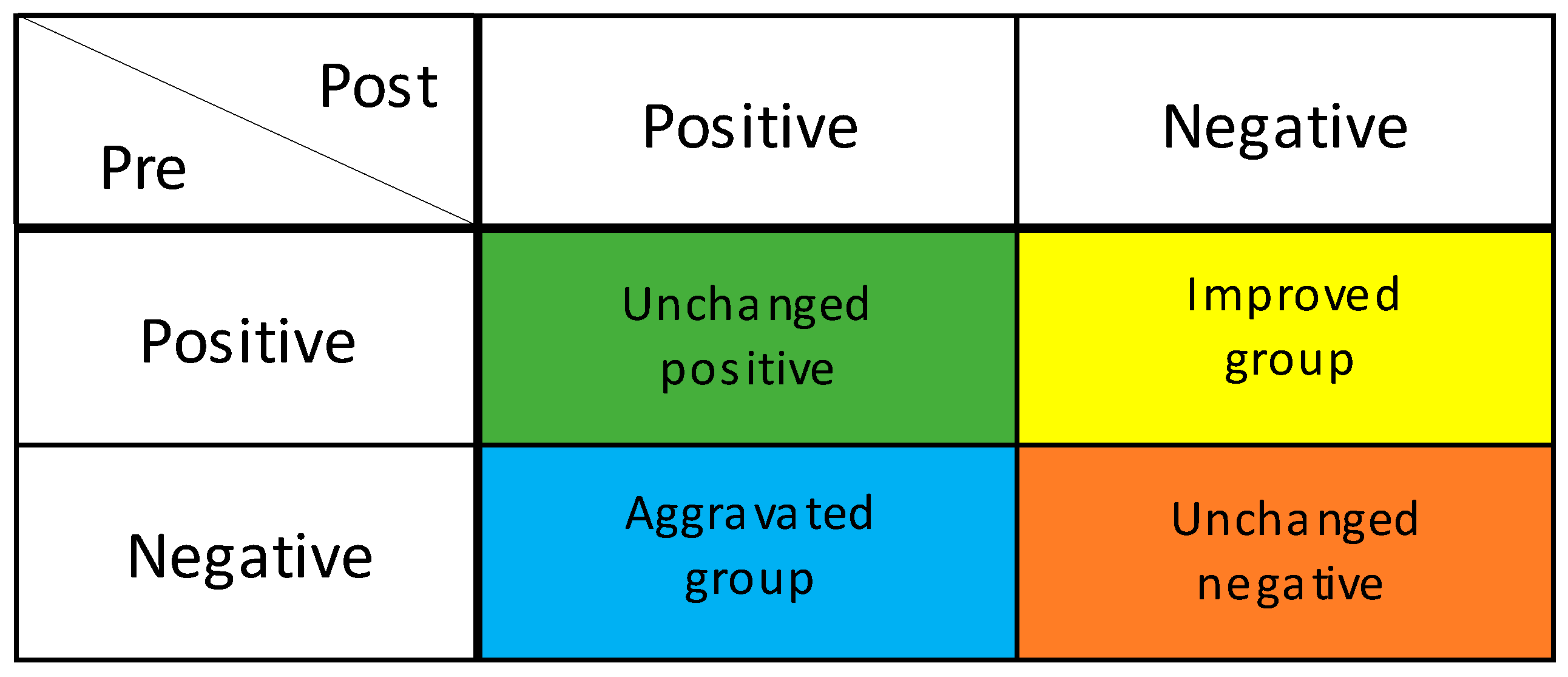
2.2.2. Oral Health Status Assessment
Number of Teeth
Periodontal Health Status
Tongue Pressure
Masticatory Performance
Occlusal Force
2.3. Statistical Analysis
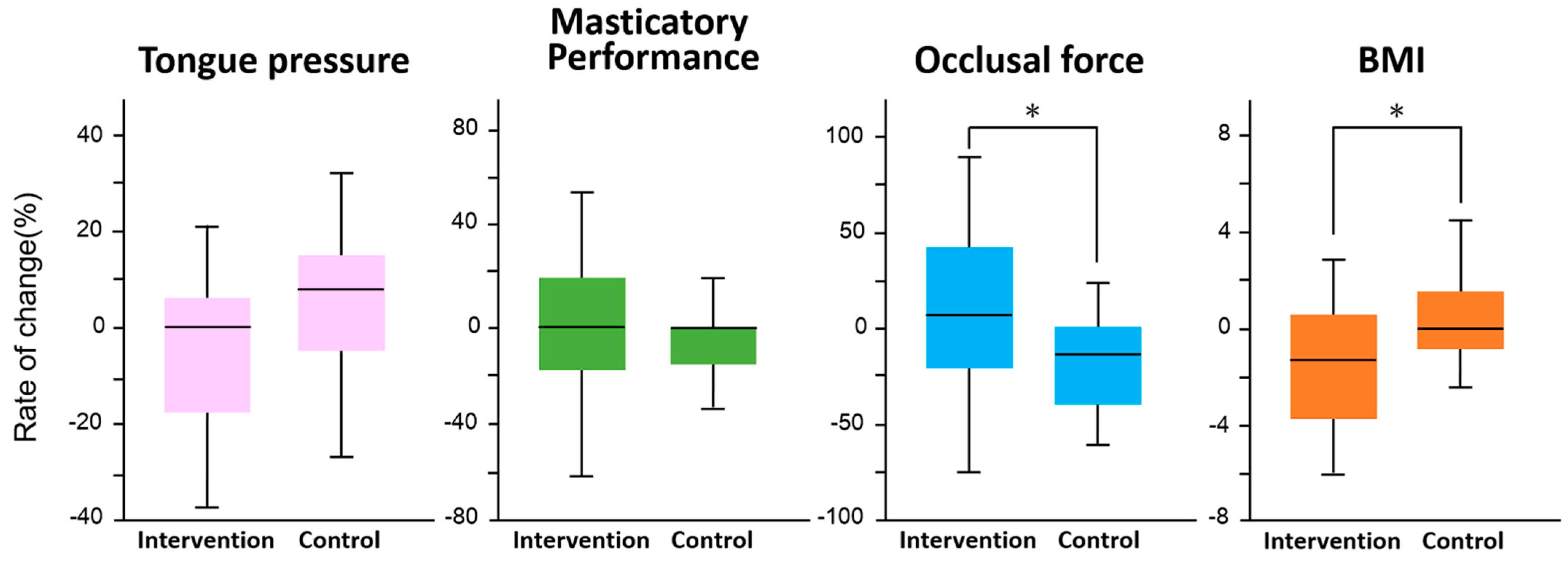
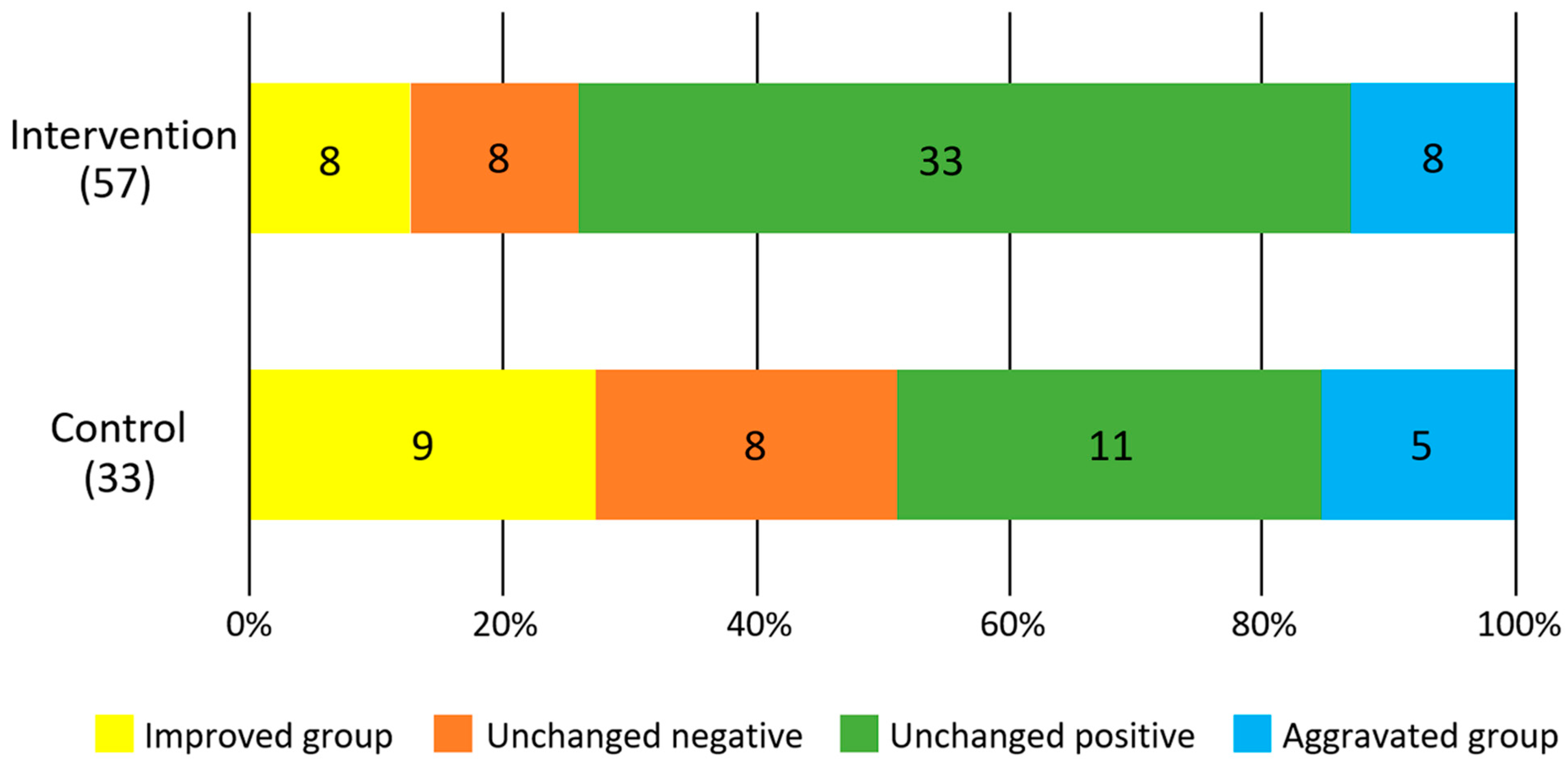
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Evans, W.J.; Campbell, W.W. Sarcopenia and age-related changes in body composition and functional capacity. J. Nutr. 1993, 123, 465–468. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, A.; Seino, S.; Abe, T. Sarcopenia: Prevalence, associated factors, and the risk of mortality and disability in Japanese older adults. J. Cachexia Sarcopenia Muscle 2021, 12, 30–38. [Google Scholar] [CrossRef]
- Song, P.; Tang, W. The community-based integrated care system in Japan: Health care and nursing care challenges posed by super-aged society. Biosci. Trends 2019, 13, 279–281. [Google Scholar] [CrossRef] [PubMed]
- Health and Welfare Bureau for the Elderly. Ministry of Health, Labour and Welfare. Long-Term Care Insurance System of Japan. 2016. Available online: https://www.mhlw.go.jp/english/policy/care-welfare/care-welfare-elderly/dl/ltcisj_e.pdf (accessed on 10 May 2022).
- Beckwée, D.; Delaere, A.; Aelbrecht, S.; Baert, V.; Beaudart, C.; Bruyere, O.; de Saint-Hubert, M.; Bautmans, I. Exercise Interventions for the Prevention and Treatment of Sarcopenia. A Systematic Umbrella Review. J. Nutr. Health Aging 2019, 23, 494–502. [Google Scholar] [CrossRef]
- Alcantar González, B.V.; Trujillo, X.; Huerta, M. Efecto de un protocolo de ejercicio por cuatro semanas sobre el peso corporal y la glucemia en rata Wistar. Sport. Sci. J. Sch. Sport Phys. Educ. Psychomot. 2017, 3, 471–485. [Google Scholar] [CrossRef][Green Version]
- Minakuchi, S.; Tsuga, K.; Ikebe, K.; Ueda, T.; Tamura, F.; Nagao, K.; Furuya, J.; Matsuo, K.; Yamamoto, K.; Kanazawa, M.; et al. Oral hypofunction in the older population: Position paper of the Japanese Society of Gerodontology in 2016. Gerodontology 2018, 35, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Gerritsen, A.E.; Allen, P.F.; Witter, D.J.; Bronkhorst, E.M.; Creugers, N.H.J. Tooth loss and oral health-r elated quality of life: A systematic review and meta-analysis. Health Qual Life Outcomes 2010, 8, 126. [Google Scholar] [CrossRef]
- Iyota, K.M.; Mizutani, S.; Oku, S. A cross-sectional study of age-related changes in oral function in healthy Japanese individuals. Int. J. Environ. Res. Public Health 2020, 17, 1376. [Google Scholar] [CrossRef]
- Tanaka, T.; Takahashi, K.; Hirano, H.; Kikutani, T.; Watanabe, Y.; Ohara, Y.; Furuya, H.; Tsuji, T.; Akishita, M.; Iijima, K. Oral Frailty as a Risk Factor for Physical Frailty and Mortality in Community-Dwelling Elderly. J. Gerontol. Biol. Sci. Med. Sci. 2018, 73, 1661–1667. [Google Scholar] [CrossRef]
- Iwasaki, M.; Yoshihara, A.; Ogawa, H.; Sato, M.; Muramatsu, K.; Watanabe, R.; Ansai, T.; Miyazaki, H. Longitudinal association of dentition status with dietary intake in Japanese adults aged 75 to 80 years. J. Oral Rehabil. 2016, 43, 737–744. [Google Scholar] [CrossRef]
- Inomata, C.; Ikebe, K.; Kagawa, R.; Okubo, H.; Sasaki, S.; Okada, T.; Takeshita, H.; Tada, S.; Matsuda, K.; Kurushima, Y. Significance of occlusal force for dietary fibre and vitamin intakes in independently living 70-year-old Japanese: From SONIC Study. J. Dent. 2014, 42, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Nose, H. Interval Walking Training for Middle-Aged and Older People: Methods and Evidence. IEICE Trans. Commun. 2014, 3, 534–539. [Google Scholar] [CrossRef]
- Nemoto, K.; Gen-no, H.; Masuki, S.; Okazaki, K.; Nose, H. Effects of High-Intensity Interval Walking Training on Physical Fitness and Blood Pressure in Middle-Aged and Older People. Mayo Clin. Proc. 2007, 82, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, M.; Okazaki, K.; Masuki, S.; Kamijo, Y.; Nose, H. Physical fitness and indices of lifestyle-related diseases before and after interval walking training in middle-aged and older males and females. Br. J. Sport. Med. 2011, 45, 216–224. [Google Scholar] [CrossRef]
- Handschin, C.; Spiegelman, B.M. The role of exercise and PGC1alpha in inflammation and chronic disease. Nature 2008, 454, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, T.; Hasegawa, Y.; Salazar, S.; Kikuchi, S.; Hori, K.; Ono, T. Factors Affecting Masticatory Satisfaction in Patients with Removable Partial Dentures. Int. J. Environ. Res. Public Health 2021, 18, 6620. [Google Scholar] [CrossRef] [PubMed]
- Masuki, S.; Morikawa, M.; Nose, H. High-Intensity Walking Time Is a Key Determinant to Increase Physical Fitness and Improve Health Outcomes After Interval Walking Training in Middle-Aged and Older People. Mayo Clin. Proc. 2019, 94, 2415–2426. [Google Scholar] [CrossRef]
- Ohshima, M.; Fujikawa, K.; Kumagai, K.; Idesawa, M.; Ezawa, S.; Ito, K.; Otsuka, K. Evaluation of a Novel Test Paper Strip Method for Detecting Occult Blood in Saliva as a Screening Test for Periodontal Disease. J. Jpn. Soc. Periodontol. 2001, 43, 416–423. [Google Scholar] [CrossRef][Green Version]
- Hayashi, R.; Tsuga, K.; Hosokawa, R. A novel handy probe for tongue pressure measurement. Int. J. Prosthodont. 2002, 15, 385–388. [Google Scholar]
- Nokubi, T.; Yoshimuta, Y.; Nokubi, F.; Yasui, S.; Kusunoki, C.; Ono, T.; Maeda, Y.; Yokota, K. Validity and reliability of a visual scoring method for masticatory ability using test gummy jelly. Gerodontology 2013, 30, 76–82. [Google Scholar] [CrossRef]
- Horibe, Y.; Matsuo, K.; Ikebe, K.; Minakuchi, S.; Sato, Y.; Sakurai, K.; Ueda, T. Relationship between two pressure-sensitive films for testing reduced occlusal force in diagnostic criteria for oral hypofunction. Gerodontology 2022, 39, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Ichikawa, T. Relationship between declines of physical performances and consciousness in elderly: Occlusal force, grip strength, and walking speed. J. Jpn. Acad. Occulusion Health 2019, 25, 39–43. [Google Scholar]
- Miyabara, H.; Nishi, M.; Hagi, Y. Relationships between Independence and Motor Functions, Activities of Daily Living, and Social Attributes of Community-Dwelling Elderly. JPTS 2010, 25, 217–222. [Google Scholar] [CrossRef]
- Yamashita, Y.; Kogo, N.; Kawaguchi, N.; Mizota, N. The relationship between the occlusal force and physical function in the frail elderly. Jpn. J. Health Promot. Phys. Ther. 2015, 5, 129–133. [Google Scholar] [CrossRef][Green Version]
- Kono, R. Relationship between occlusal force and preventive factors for disability among community-dwelling elderly persons. Nihon Ronen Igakkai Zasshi 2009, 46, 55–62. [Google Scholar] [CrossRef]
- Matake, A.; Sato, H.; Fukuba, Y.; Sato, H. The effect of the difference of the motion experience to biting force and face form. Bull. Sch. Nurs. 2001, 5, 31–37. [Google Scholar]
- Tsurui, C.; Arai, Y.; Taniguchi, H. The electromyographic evaluation of head and neck coordinated movements during jaw movements. J. Jpn. Soc. Stomatognath. Funct. 2010, 16, 112–123. [Google Scholar] [CrossRef][Green Version]
- Tsuchida, Y. Influences of Head Positions on Masticatory Muscle Activities during Voluntary Contractions. J. Jpn. Prosthodont. Soc. 1991, 35, 89–102. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Matsumoto, T.; Ishigami, K.; Hirai, T. The Relationship between Occlusion and the Body Function. J. Jpn. Prosthodont. Soc. 1996, 40, 1–23. [Google Scholar] [CrossRef]
- Sasaki, K.; Hannam, A.G.; Wood, W.W. Relationships between the size, position, and angulation of the human jaw muscles unilateral first molar bite force. J. Dent. Res. 1989, 68, 499–553. [Google Scholar] [CrossRef]
- Sasaki, Y. Effect of teeth clenching on isometric and isokinetic strength of ankle plantar flexion. J. Med. Dent. Sci. 1998, 45, 29–37. [Google Scholar] [PubMed]
- Sayar, F.; Fallah, S.; Akhondi, N.; Jamshidi, S. Association of serum lipid indices and statin consumption with periodontal status. Oral Dis. 2016, 22, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Masuki, S.; Nishida, K.; Hashimoto, S.; Morikawa, M.; Takasugi, S.; Nagata, M.; Taniguchi, S.; Rokutan, K.; Nose, H. Effects of milk product intake on thigh muscle strength and NFKB gene methylation during home-based interval walking training in older women: A randomized, controlled pilot study. PLoS ONE 2017, 12, e0176757. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hashimoto, S.; Fujii, C.; Hida, S.; Ito, K.; Matsumura, T.; Sakaizawa, T.; Morikawa, M.; Masuki, S.; Nose, H.; et al. NFκB2 Gene as a Novel Candidate that Epigenetically Responds to Interval Walking Training. Int. J. Sport. Med. 2015, 36, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Bunte, K.; Kuhn, C.; Walther, C.; Peters, U.; Aarabi, G.; Smeets, R.; Beikler, T. Clinical significance of ragA, ragB, and PG0982 genes in Porphyromonas gingivalis isolates from periodontitis patients. Eur. J. Oral Sci. 2021, 129, e12776. [Google Scholar] [CrossRef]
- Londhe, P.; Guttridge, D.C. Inflammation induced loss of skeletal muscle. Bone 2015, 80, 131–142. [Google Scholar] [CrossRef]
- Hayashi, K.; Takeuchi, Y.; Shimizu, S.; Tanabe, G.; Churei, H.; Kobayashi, H.; Ueno, T. Continuous Oral Administration of Sonicated P. gingivalis Delays Rat Skeletal Muscle Healing Post-Treadmill Training. Int. J. Environ. Res. Public Health 2022, 19, 13046. [Google Scholar] [CrossRef]
- Sasajima, M.; Yoshihara, A.; Odajima, A. Effects of Oral Function Training and Oral Health Status on Physical Performance in Potentially Dependent Older Adults. Int. J. Environ. Res. Public Health 2021, 18, 11348. [Google Scholar] [CrossRef]
- Robinder, J.S.; Dhillon, M.D.M.; Sarfaraz Hasni, M. Pathogenesis and management of Sarcopenia. Clin. Geriatr. Med. 2017, 33, 17–36. [Google Scholar]

| Intervention (n = 57) | Control (n = 33) | p | ||
|---|---|---|---|---|
| Age(years) * | 66.7 ± 0.8 | 74.5 ± 1.1 | <0.001 | |
| Gender | Male (%) | 18(31.6) | 14(42.4) | 0.36 |
| Female (%) | 39(68.4) | 19(57.6) | ||
| Number of teeth | 25.3 ± 0.7 | 25.2 ± 0.6 | 0.37 | |
| Gum bleeding | Positive (%) | 18(72) | 15(45.5) | 0.48 |
| Negative (%) | 7(28) | 18(54.5) | ||
| Tongue pressure(kPa) | 36.2 ± 1.0 | 34.4 ± 0.9 | 0.42 | |
| Masticatory Performance (Score) | 5.6 ± 0.3 | 5.9 ± 0.3 | 0.58 | |
| Occlusal force(N) | 379.4 ± 27.0 | 468.9 ± 41.9 | 0.08 | |
| BMI * | 24.0 ± 0.4 | 21.4 ± 0.5 | <0.001 |
| Change in BMI | p | Change in VO2peak | p | ||||
|---|---|---|---|---|---|---|---|
| Decrease | Increase | Decrease | Increase | ||||
| GUM bleeding | Improved group (%) | 7(17.9) | 3(16.7) | 0.06 | 1(6.3) | 7(21.2) | 0.47 |
| Unchanged negative (%) | 7(17.9) | 0(0) | 2(12.5) | 4(12.1) | |||
| Unchanged positive (%) | 22(56.5) | 11(61.1) | 10(62.5) | 19(57.6) | |||
| Aggravated group (%) | 3(7.7) | 4(22.2) | 3(18.7) | 3(9.1) | |||
| Rate of change in Tongue pressure | −1.2 ± 3.3 | 5.3 ± 6.6 | 0.32 | −1.2 ± 5.1 | 0.2 ± 4.3 | 0.86 | |
| Rate of change in Masticatory Performance | 16.7 ± 10.2 | 5.5 ± 8.5 | 0.47 | 24.7 ± 15.8 | 10.0 ± 9.9 | 0.42 | |
| Rate of change in Occlusal force | 12.6 ± 8.7 | 6.0 ± 10.3 | 0.68 | 6.9 ± 17.3 | 6.8 ± 8.2 | 0.99 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoshimoto, T.; Hasegawa, Y.; Furihata, M.; Yoshihara, A.; Shiramizu, M.; Sta. Maria, M.T.; Hori, S.; Morikawa, M.; Marito, P.; Kaneko, N.; et al. Effects of Interval Walking Training on Oral Health Status in Middle-Aged and Older Adults: A Case-Control Study. Int. J. Environ. Res. Public Health 2022, 19, 14465. https://doi.org/10.3390/ijerph192114465
Yoshimoto T, Hasegawa Y, Furihata M, Yoshihara A, Shiramizu M, Sta. Maria MT, Hori S, Morikawa M, Marito P, Kaneko N, et al. Effects of Interval Walking Training on Oral Health Status in Middle-Aged and Older Adults: A Case-Control Study. International Journal of Environmental Research and Public Health. 2022; 19(21):14465. https://doi.org/10.3390/ijerph192114465
Chicago/Turabian StyleYoshimoto, Tasuku, Yoko Hasegawa, Mayuka Furihata, Akihiro Yoshihara, Masako Shiramizu, Ma. Therese Sta. Maria, Shoko Hori, Mayuko Morikawa, Pinta Marito, Noboru Kaneko, and et al. 2022. "Effects of Interval Walking Training on Oral Health Status in Middle-Aged and Older Adults: A Case-Control Study" International Journal of Environmental Research and Public Health 19, no. 21: 14465. https://doi.org/10.3390/ijerph192114465
APA StyleYoshimoto, T., Hasegawa, Y., Furihata, M., Yoshihara, A., Shiramizu, M., Sta. Maria, M. T., Hori, S., Morikawa, M., Marito, P., Kaneko, N., Nohno, K., Nose, H., Masuki, S., & Ono, T. (2022). Effects of Interval Walking Training on Oral Health Status in Middle-Aged and Older Adults: A Case-Control Study. International Journal of Environmental Research and Public Health, 19(21), 14465. https://doi.org/10.3390/ijerph192114465







