MRSA Femoral Osteomyelitis from Superinfected Scabies Lesions: A Pediatric Case Report
Abstract
:1. Introduction
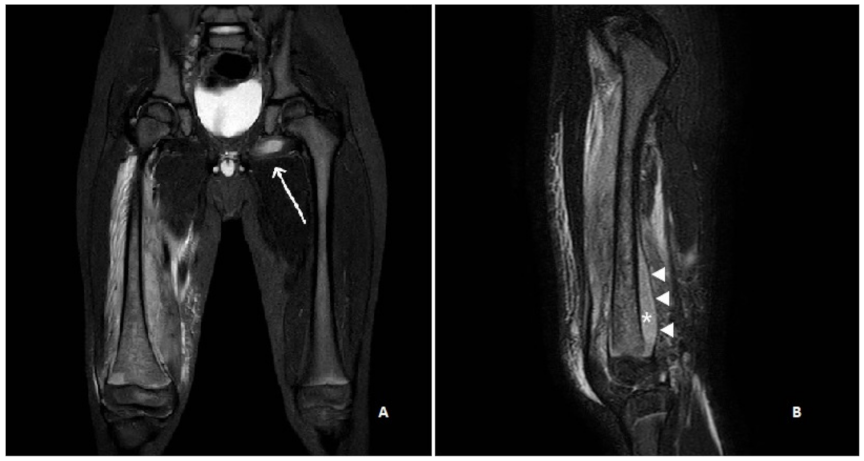
2. Case Report
2.1. Diagnosis
2.2. Treatment Plan
2.3. Treatment Progress
3. Discussion
Strenghts and Limitations
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hay, R.J.; Steer, A.C.; Engelman, D.; Walton, S. Scabies in the developing world--its prevalence, complications, and management. Clin. Microbiol. Infect. 2012, 18, 313–323. [Google Scholar] [CrossRef] [Green Version]
- Scabies and Other Ectoparasites. Available online: https://www.who.int/neglected_diseases/diseases/scabies-and-other-ectoparasites/en/ (accessed on 19 November 2021).
- Heukelbach, J.; Mazigo, H.D.; Ugbomoiko, U.S. Impact of scabies in resource-poor communities. Curr. Opin. Infect. Dis. 2013, 26, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Johnston, G.; Sladden, M. Scabies: Diagnosis and treatment. BMJ (Clin. Res. Ed.) 2005, 331, 619–622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thompson, R.; Westbury, S.; Slape, D. Paediatrics: How to manage scabies. Drugs Context 2021, 10, 1–13. [Google Scholar] [CrossRef]
- Karthikeyan, K. Scabies in children. Arch. Dis. Child. Educ. Pract. Ed. 2007, 92, ep65–ep69. [Google Scholar] [CrossRef]
- Walton, S.F.; Currie, B.J. Problems in diagnosing scabies, a global disease in human and animal populations. Clin. Microbiol. Rev. 2007, 20, 268–279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chosidow, O. Clinical practices. Scabies. N. Engl. J. Med. 2006, 354, 1718–1727. [Google Scholar] [CrossRef]
- Arlian, L.G.; Morgan, M.S. A review of Sarcoptes scabiei: Past, present and future. Parasites Vectors 2017, 10, 297. [Google Scholar] [CrossRef]
- Yeoh, D.K.; Bowen, A.C.; Carapetis, J.R. Impetigo and scabies—Disease burden and modern treatment strategies. J. Infect. 2016, 72, S61–S67. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.K.C.; Lam, J.M.; Leong, K.F. Scabies: A Neglected Global Disease. Curr. Pediatric Rev. 2020, 16, 33–42. [Google Scholar] [CrossRef]
- Jouret, G.; Bounemeur, R.; Presle, A.; Takin, R. Annales De Dermatologie Et De Venereologie. Elsevier Mass 2016, 143, 251–256. [Google Scholar] [CrossRef]
- Abdel-Latif, A.A.; Elshahed, A.R.; Salama, O.A.; Elsaie, M.L. Comparing the diagnostic properties of skin scraping, adhesive tape, and dermoscopy in diagnosing scabies. Acta Dermatovenerol. Alp. Pannonica Adriat. 2018, 27, 75–78. [Google Scholar] [CrossRef] [PubMed]
- Ogbuefi, N.; Kenner-Bell, B. Common pediatric infestations: Update on diagnosis and treatment of scabies, head lice, and bed bugs. Curr. Opin. Pediatrics 2021, 33, 410–415. [Google Scholar] [CrossRef] [PubMed]
- Rosumeck, S.; Nast, A.; Dressler, C. Ivermectin and permethrin for treating scabies. Cochrane Database Syst. Rev. 2018, 4, Cd012994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levy, M.; Martin, L.; Bursztejn, A.C.; Chiaverini, C.; Miquel, J.; Mahé, E.; Maruani, A.; Boralevi, F. Ivermectin safety in infants and children under 15 kg treated for scabies: A multicentric observational study. Br. J. Dermatol. 2020, 182, 1003–1006. [Google Scholar] [CrossRef]
- Bernigaud, C.; Fischer, K.; Chosidow, O. The Management of Scabies in the 21st Century: Past, Advances and Potentials. Acta Derm.-Venereol. 2020, 100, adv00112. [Google Scholar] [CrossRef]
- Romani, L.; Steer, A.C.; Whitfeld, M.J.; Kaldor, J.M. Prevalence of scabies and impetigo worldwide: A systematic review. Lancet. Infect. Dis. 2015, 15, 960–967. [Google Scholar] [CrossRef]
- Steer, A.C.; Jenney, A.W.; Kado, J.; Batzloff, M.R.; La Vincente, S.; Waqatakirewa, L.; Mulholland, E.K.; Carapetis, J.R. High burden of impetigo and scabies in a tropical country. PLoS Negl. Trop. Dis. 2009, 3, e467. [Google Scholar] [CrossRef]
- Chandler, D.J.; Fuller, L.C. A Review of Scabies: An Infestation More than Skin Deep. Dermatology 2019, 235, 79–90. [Google Scholar] [CrossRef]
- Heukelbach, J.; Feldmeier, H. Scabies. Lancet 2006, 367, 1767–1774. [Google Scholar] [CrossRef]
- Esposito, L.; Veraldi, S. Skin bacterial colonizations and superinfections in immunocompetent patients with scabies. Int. J. Dermatol. 2018, 57, 1218–1220. [Google Scholar] [CrossRef]
- Lowy, F.D. Staphylococcus aureus infections. N. Engl. J. Med. 1998, 339, 520–532. [Google Scholar] [CrossRef] [PubMed]
- Mallo-García, S.; Taborga-Díaz, E.; Menéndez-Cuervo, S.; Santos-Juanes-Jiménez, J. Staphylococcus aureus sepsis as a complication of scabies. Actas Dermo-Sifiliogr. 2008, 99, 425–426. [Google Scholar] [CrossRef]
- Gaida, G.; Töllner, U.; Krüpe, H.; Haas, J.P. Acute hematogenic osteomyelitis after impetiginized scabies. Klin. Padiatr. 1985, 197, 489–491. [Google Scholar] [CrossRef]
- Parlet, C.P.; Brown, M.M.; Horswill, A.R. Commensal Staphylococci Influence Staphylococcus aureus Skin Colonization and Disease. Trends Microbiol. 2019, 27, 497–507. [Google Scholar] [CrossRef]
- Salavastru, C.M.; Chosidow, O.; Boffa, M.J.; Janier, M.; Tiplica, G.S. European guideline for the management of scabies. J. Eur. Acad. Dermatol. Venereol. JEADV 2017, 31, 1248–1253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scott, G.R.; Chosidow, O. European guideline for the management of scabies, 2010. Int. J. STD AIDS 2011, 22, 301–303. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.E.; Al-Jahdali, H.; Jradi, H.; BI, A.L.; AlBuraikan, D.A.; Albaijan, M.A.; Ali, Y.Z.; Al Shehri, A.M. Recurrence rate of scabies in patients 14 years or older in Saudi Arabia. Saudi Med. J. 2019, 40, 1267–1271. [Google Scholar] [CrossRef] [PubMed]
- Makigami, K.; Ohtaki, N.; Ishii, N.; Tamashiro, T.; Yoshida, S.; Yasumura, S. Risk factors for recurrence of scabies: A retrospective study of scabies patients in a long-term care hospital. J. Dermatol. 2011, 38, 874–879. [Google Scholar] [CrossRef]
- Schmidt-Guerre, A.R.; Aranda-Hulin, B.; Maumy-Bertrand, M.; Aubin, F. Diagnosis and treatment of scabies by general practitioners: A survey of practices in France. Ann. Dermatol. Venereol. 2018, 145, 89–94. [Google Scholar] [CrossRef]
- Hicks, M.I.; Elston, D.M. Scabies. Dermatol. Ther. 2009, 22, 279–292. [Google Scholar] [CrossRef] [PubMed]
- Chambliss, M.L. Treating asymptomatic bodily contacts of patients with scabies. Arch. Fam. Med. 2000, 9, 473–474. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sartorio, M.U.A.; Munari, A.M.; Carlucci, P.; Erba, P.; Calcaterra, V.; Fabiano, V. MRSA Femoral Osteomyelitis from Superinfected Scabies Lesions: A Pediatric Case Report. Int. J. Environ. Res. Public Health 2022, 19, 1007. https://doi.org/10.3390/ijerph19021007
Sartorio MUA, Munari AM, Carlucci P, Erba P, Calcaterra V, Fabiano V. MRSA Femoral Osteomyelitis from Superinfected Scabies Lesions: A Pediatric Case Report. International Journal of Environmental Research and Public Health. 2022; 19(2):1007. https://doi.org/10.3390/ijerph19021007
Chicago/Turabian StyleSartorio, Marco Ugo Andrea, Alice Marianna Munari, Patrizia Carlucci, Paola Erba, Valeria Calcaterra, and Valentina Fabiano. 2022. "MRSA Femoral Osteomyelitis from Superinfected Scabies Lesions: A Pediatric Case Report" International Journal of Environmental Research and Public Health 19, no. 2: 1007. https://doi.org/10.3390/ijerph19021007
APA StyleSartorio, M. U. A., Munari, A. M., Carlucci, P., Erba, P., Calcaterra, V., & Fabiano, V. (2022). MRSA Femoral Osteomyelitis from Superinfected Scabies Lesions: A Pediatric Case Report. International Journal of Environmental Research and Public Health, 19(2), 1007. https://doi.org/10.3390/ijerph19021007







