The Role of Physical Function in the Association between Physical Activity and Gait Speed in Older Adults: A Mediation Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Sample Size
2.3. Participants and Eligibility
2.4. Data Collection
2.4.1. Demographics and Clinical Data
2.4.2. Anthropometry
2.4.3. Cognitive Function
2.4.4. Physical Activity
2.4.5. Physical Function
2.4.6. Gait
2.4.7. Statistical Analysis
3. Results
3.1. Main Characteristics of the Participants
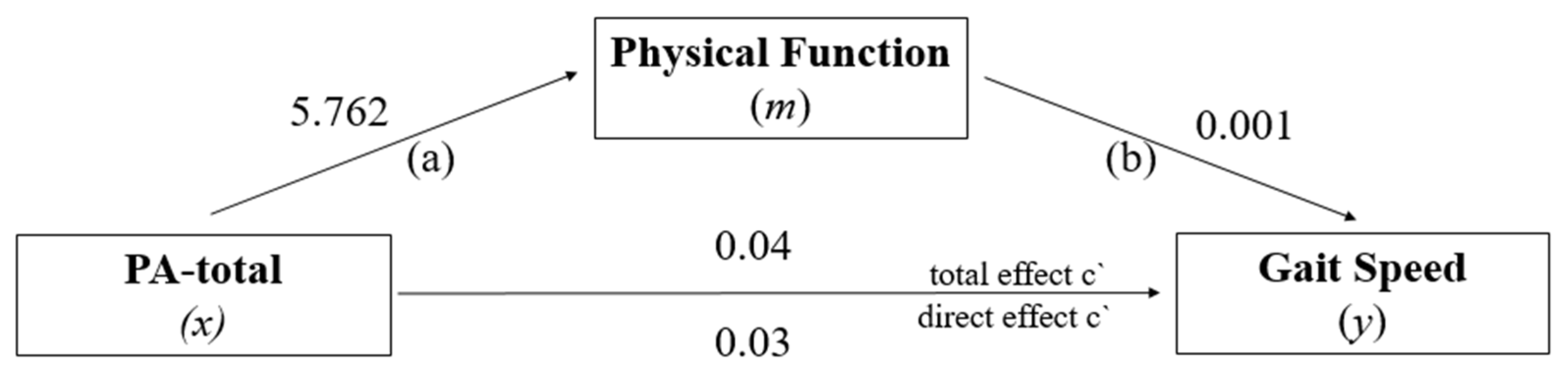
3.2. Mediation Analysis: PF in the Relationship between PA-Total and GS
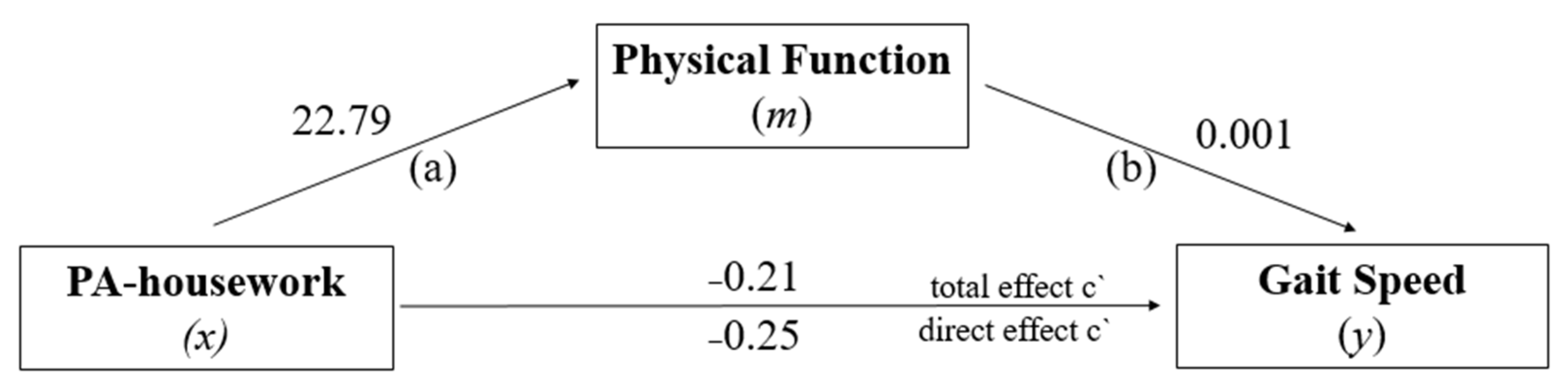
3.3. Mediation Analysis: PF in the Relationship between PA-Housework and GS
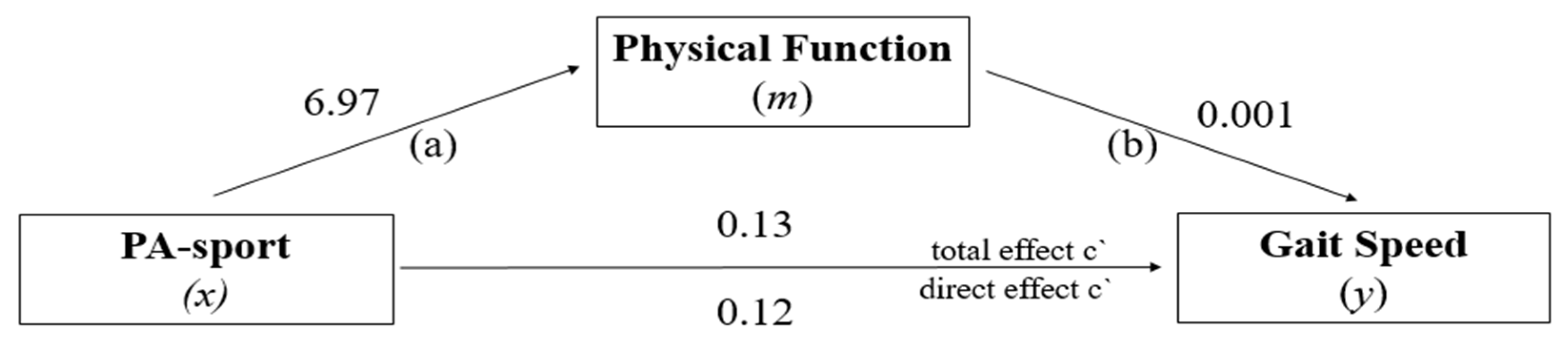
3.4. Mediation Analysis: PF in the Relationship between PA-Sport and GS
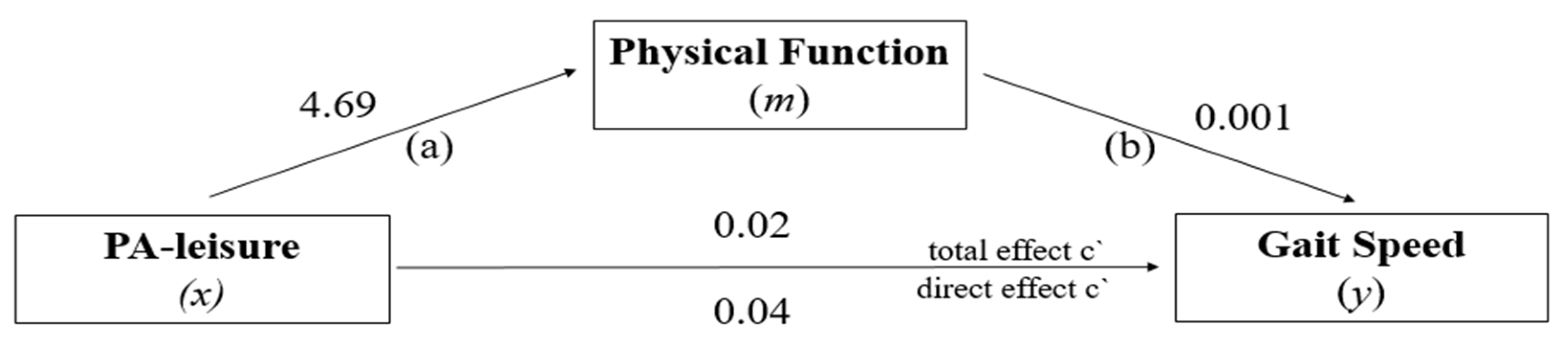
3.5. Mediation Analysis: PF in the Relationship between PA-Leisure and GS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, D.X.M.; Yao, J.; Zirek, Y.; Reijnierse, E.M.; Maier, A.B. Muscle mass, strength, and physical performance predicting activities of daily living: A meta-analysis. J. Cachexia. Sarcopenia Muscle 2020, 11, 3–25. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Shiroy, D.M.; Jones, L.Y.; Clark, D.O. Systematic review of functional training on muscle strength, physical functioning, and activities of daily living in older adults. Eur. Rev. Aging Phys. Act. 2014, 11, 95–106. [Google Scholar] [CrossRef]
- Knapik, A.; Brzęk, A.; Famuła-Wąż, A.; Gallert-Kopyto, W.; Szydłak, D.; Marcisz, C.; Plinta, R. The relationship between physical fitness and health self-assessment in elderly. Medicine 2019, 98, e15984. [Google Scholar] [CrossRef]
- Milanović, Z.; Pantelić, S.; Trajković, N.; Sporiš, G.; Kostić, R.; James, N.Z. Age-related decrease in physical activity and functional fitness among elderly men and women. Clin. Interv. Aging 2013, 8, 549. [Google Scholar] [CrossRef]
- Patrizio, E.; Calvani, R.; Marzetti, E.; Cesari, M. Physical functional assessment in older adults. J. Frailty Aging 2021, 10, 141–149. [Google Scholar] [CrossRef]
- McPhee, J.S.; French, D.P.; Jackson, D.; Nazroo, J.; Pendleton, N.; Degens, H. Physical activity in older age: Perspectives for healthy ageing and frailty. Biogerontology 2016, 17, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Kasović, M.; Štefan, L.; Zvonař, M. Domain-Specific and Total Sedentary Behavior Associated with Gait Velocity in Older Adults: The Mediating Role of Physical Fitness. Int. J. Environ. Res. Public Health 2020, 17, 593. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-H.; Chen, Y.-C.; Tseng, Y.-C.; Tsai, S.; Tseng, Y.-H. Physical activity and successful aging among middle-aged and older adults: A systematic review and meta-analysis of cohort studies. Aging 2020, 12, 7704–7716. [Google Scholar] [CrossRef]
- Sardinha, L.B.; Santos, D.A.; Silva, A.M.; Baptista, F.; Owen, N. Breaking-up Sedentary Time Is Associated with Physical Function in Older Adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2015, 70, 119–124. [Google Scholar] [CrossRef]
- Ebeling, P.R.; Cicuttini, F.; Scott, D.; Jones, G. Promoting mobility and healthy aging in men: A narrative review. Osteoporos. Int. 2019, 30, 1911–1922. [Google Scholar] [CrossRef]
- Musich, S.; Wang, S.S.; Ruiz, J.; Hawkins, K.; Wicker, E. The impact of mobility limitations on health outcomes among older adults. Geriatr. Nurs. 2018, 39, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Hamacher, D.; Liebl, D.; Hödl, C.; Heßler, V.; Kniewasser, C.K.; Thönnessen, T.; Zech, A. Gait Stability and Its Influencing Factors in Older Adults. Front. Physiol. 2019, 9, 1955. [Google Scholar] [CrossRef] [PubMed]
- van Schooten, K.S.; Pijnappels, M.; Lord, S.R.; van Dieën, J.H. Quality of Daily-Life Gait: Novel Outcome for Trials that Focus on Balance, Mobility, and Falls. Sensors 2019, 19, 4388. [Google Scholar] [CrossRef] [PubMed]
- Aboutorabi, A.; Arazpour, M.; Bahramizadeh, M.; Hutchins, S.W.; Fadayevatan, R. The effect of aging on gait parameters in able-bodied older subjects: A literature review. Aging Clin. Exp. Res. 2016, 28, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Herssens, N.; Verbecque, E.; Hallemans, A.; Vereeck, L.; Van Rompaey, V.; Saeys, W. Do spatiotemporal parameters and gait variability differ across the lifespan of healthy adults? A systematic review. Gait Posture 2018, 64, 181–190. [Google Scholar] [CrossRef]
- Steckhan, G.M.A.; Fleig, L.; Schwarzer, R.; Warner, L.M. Perceived Physical Functioning and Gait Speed as Mediators in the Association Between Fear of Falling and Quality of Life in Old Age. J. Appl. Gerontol. 2020, 41, 421–429. [Google Scholar] [CrossRef]
- Florence, C.S.; Bergen, G.; Atherly, A.; Burns, E.; Stevens, J.; Drake, C. Medical Costs of Fatal and Nonfatal Falls in Older Adults. J. Am. Geriatr. Soc. 2018, 66, 693–698. [Google Scholar] [CrossRef]
- Viswanathan, A.; Sudarsky, L. Balance and gait problems in the elderly. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2012; Volume 103, pp. 623–634. [Google Scholar]
- Carter, S.; Hartman, Y.; Holder, S.; Thijssen, D.H.; Hopkins, N.D. Sedentary Behavior and Cardiovascular Disease Risk: Mediating Mechanisms. Exerc. Sport Sci. Rev. 2017, 45, 80–86. [Google Scholar] [CrossRef]
- Slater, L.; Gilbertson, N.M.; Hyngstrom, A.S. Improving gait efficiency to increase movement and physical activity—The impact of abnormal gait patterns and strategies to correct. Prog. Cardiovasc. Dis. 2021, 64, 83–87. [Google Scholar] [CrossRef]
- Ciprandi, D.; Bertozzi, F.; Zago, M.; Ferreira, C.L.P.; Boari, G.; Sforza, C.; Galvani, C. Study of the association between gait variability and physical activity. Eur. Rev. Aging Phys. Act. 2017, 14, 19. [Google Scholar] [CrossRef]
- Jerome, G.J.; Ko, S.; Kauffman, D.; Studenski, S.A.; Ferrucci, L.; Simonsick, E.M. Gait characteristics associated with walking speed decline in older adults: Results from the Baltimore Longitudinal Study of Aging. Arch. Gerontol. Geriatr. 2015, 60, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Huijben, B.; van Schooten, K.S.; van Dieën, J.H.; Pijnappels, M. The effect of walking speed on quality of gait in older adults. Gait Posture 2018, 65, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.M.; Petrica, J.; Serrano, J.; Paulo, R.; Ramalho, A.; Lucas, D.; Ferreira, J.P.; Duarte-Mendes, P. The Sedentary Time and Physical Activity Levels on Physical Fitness in the Elderly: A Comparative Cross Sectional Study. Int. J. Environ. Res. Public Health 2019, 16, 3697. [Google Scholar] [CrossRef]
- Chaabene, H.; Prieske, O.; Herz, M.; Moran, J.; Höhne, J.; Kliegl, R.; Ramirez-Campillo, R.; Behm, D.G.; Hortobágyi, T.; Granacher, U. Home-based exercise programmes improve physical fitness of healthy older adults: A PRISMA-compliant systematic review and meta-analysis with relevance for COVID-19. Ageing Res. Rev. 2021, 67, 101265. [Google Scholar] [CrossRef]
- Cruz-Jimenez, M. Normal Changes in Gait and Mobility Problems in the Elderly. Phys. Med. Rehabil. Clin. N. Am. 2017, 28, 713–725. [Google Scholar] [CrossRef] [PubMed]
- Fukuchi, C.A.; Fukuchi, R.K.; Duarte, M. Effects of walking speed on gait biomechanics in healthy participants: A systematic review and meta-analysis. Syst. Rev. 2019, 8, 153. [Google Scholar] [CrossRef]
- Pau, M.; Leban, B.; Collu, G.; Migliaccio, G.M. Effect of light and vigorous physical activity on balance and gait of older adults. Arch. Gerontol. Geriatr. 2014, 59, 568–573. [Google Scholar] [CrossRef]
- Taraldsen, K.; Helbostad, J.L.; Follestad, T.; Bergh, S.; Selbæk, G.; Saltvedt, I. Gait, physical function, and physical activity in three groups of home-dwelling older adults with different severity of cognitive impairment—A cross-sectional study. BMC Geriatr. 2021, 21, 670. [Google Scholar] [CrossRef]
- Monteiro-Odasso, M.; Schapira, M.; Soriano, E.R.; Varela, M.; Kaplan, R.; Camera, L.A.; Mayorga, L.M. Gait velocity as a single predictor of adverse events in healthy seniors aged 75 years and older. Gerontol. Soc. Am. 2005, 60A, 1304–1309. [Google Scholar] [CrossRef]
- Noce Kirkwood, R.; de Souza Moreira, B.; Mingoti, S.A.; Faria, B.F.; Sampaio, R.F.; Alves Resende, R. The slowing down phenomenon: What is the age of major gait velocity decline? Maturitas 2018, 115, 31–36. [Google Scholar] [CrossRef]
- Fradelos, E.; Papathanasiou, I.; Mitsi, D.; Tsaras, K.; Kleisiaris, C.; Kourkouta, L. Health Based Geographic Information Systems (GIS) and their Applications. Acta Inform. Medica 2014, 22, 402. [Google Scholar] [CrossRef]
- Miranda Goncalves, R.; Moreira Domingos, I. Riverside Population in Amazonas and Inequality in Access to Health. Rev. Estud. Const. Hermenêutica e Teor. Direito 2019, 11, 99–108. [Google Scholar]
- Aracaty, M.L.; de Souza Rojas, S.R. Índice de vulnerabilidade Social vulnerability index (IVS) of the metropolitan regions of Belém do Pará-PA (RMB) and Manaus-AM (RMM). Econ. Desenv 2021, 33, 1–22. [Google Scholar]
- SBGG-Brazilian Society of Geriatrics and Gerontology Mais Idosos Poucos Geriátras [More Older Adult Few Geriatricians]. Available online: http://www.sbgg-sp.com.br/pub/mais-idosos-poucos-geriatras/ (accessed on 26 March 2019).
- Garcia Meneguci, C.A.; Meneguci, J.; Sasaki, J.E.; Tribess, S.; Júnior, J.S.V. Physical activity, sedentary behavior and functionality in older adults: A cross-sectional path analysis. PLoS ONE 2021, 16, e0246275. [Google Scholar] [CrossRef] [PubMed]
- Yatsugi, H.; Chen, T.; Chen, S.; Liu, X.; Kishimoto, H. The Associations between Objectively Measured Physical Activity and Physical Function in Community-Dwelling Older Japanese Men and Women. Int. J. Environ. Res. Public Health 2021, 19, 369. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, C.; O’ Sullivan, R.; Caserotti, P.; Tully, M.A. Consequences of physical inactivity in older adults: A systematic review of reviews and meta-analyses. Scand. J. Med. Sci. Sports 2020, 30, 816–827. [Google Scholar] [CrossRef]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Brucki, S.; Nitrini, R.; Caramelli, P.; Bertolucci, P.H.; Okamoto, I.H. Suggestions for utilization of the mini-mental state examination in Brazil. Arq. Neuropsiquiatr. 2003, 61, 777–781. [Google Scholar] [CrossRef]
- Rose, D.J. Fallproof!: A Comprehensive Balance and Mobility Training Program, 2nd ed.; Human Kinetics: Champaign, IL, USA, 2010; ISBN 978-0-7360-6747-8. [Google Scholar]
- Marfell-Jones, M.; Olds, T.; Stew, A.; Carter, L. International Standards for Anthropometric Assessment; International Society for the Advancement of Kinanthropometry: Wellington, New Zealand, 2018. [Google Scholar]
- Creavin, S.T.; Noel-Storr, A.H.; Smailagic, N.; Giannakou, A.; Ewins, E.; Wisniewski, S.; Cullum, S. Mini-Mental State Examination (MMSE) for the detection of Alzheimer’s dementia and other dementias in asymptomatic and previously clinically unevaluated people aged over 65 years in community and primary care populations. In Cochrane Database of Systematic Reviews; Creavin, S.T., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2016. [Google Scholar]
- Mazo, G.Z.; Mota, J.; Benedetti, T.B.; de Barros, M.V.G. Validade concorrente e reprodutibilidade: Teste-reteste do Questionário de Baecke modificado para idosos. Rev. Bras. Atividade Física Saúde 2001, 6, 5–11. [Google Scholar]
- Voorrips, L.E.; Ravelli, A.C.; Dongelmans, P.C.; Deurenberg, P.A.U.L.; Van Staveren, W.A. A physical activity questionnaire for the elderly. Med. Sci. Sport. Exerc. 1991, 23, 974–979. [Google Scholar] [CrossRef]
- Rikli, R.E.; Jones, C.J. Development and Validation of Criterion-Referenced Clinically Relevant Fitness Standards for Maintaining Physical Independence in Later Years. Gerontologist 2013, 53, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Set correlation and contingency tables. Appl. Psychol. Meas. 1988, 12, 425–434. [Google Scholar] [CrossRef]
- Preacher, K.J.; Rucker, D.D.; Hayes, A.F. Addressing Moderated Mediation Hypotheses: Theory, Methods, and Prescriptions. Multivar. Behav. Res. 2007, 42, 185–227. [Google Scholar] [CrossRef]
- Hayes, A.F. Introduction to Mediation, Moderation, and Conditional Process Analysis: A Regression-Based Approach; Guilford Publications: New York, NY, USA, 2013; ISBN 978-1-60918-230-4. [Google Scholar]
- WHO-World Health Organization Noncommunicable Diseases (NCD). Available online: https://www.who.int/gho/ncd/mortality_morbidity/en/ (accessed on 11 July 2022).
- WHO-World Health Organization. Noncommunicable Diseases Progress Monitor 2022; WHO-World Health Organization: Geneva, Switzerland, 2022. [Google Scholar]
- WHO-World Health Organization. WHO Guidelines on Physical Activity and Sedentary Behaviour; WHO-World Health Organization: Geneva, Switzerland, 2020; ISBN 9781134470006. [Google Scholar]
- Liguori, G.; American College of Sports Medicine. ACSM’s Guidelines for Exercise Testing and Prescription; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2020. [Google Scholar]
- Hörder, H.; Skoog, I.; Frändin, K. Health-related quality of life in relation to walking habits and fitness: A population-based study of 75-year-olds. Qual. Life Res. 2013, 22, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Bruton, A.; Navarrete-Villanueva, D.; Pérez-Gómez, J.; Vila-Maldonado, S.; Gesteiro, E.; Gusi, N.; Villa-Vicente, J.G.; Espino, L.; Gonzalez-Gross, M.; Casajus, J.A.; et al. The Effects of Age, Organized Physical Activity and Sedentarism on Fitness in Older Adults: An 8-Year Longitudinal Study. Int. J. Environ. Res. Public Health 2020, 17, 4312. [Google Scholar] [CrossRef]
- Ferreira, M.L.; Sherrington, C.; Smith, K.; Carswell, P.; Bell, R.; Bell, M.; Nascimento, D.P.; Máximo Pereira, L.S.; Vardon, P. Physical activity improves strength, balance and endurance in adults aged 40–65 years: A systematic review. J. Physiother. 2012, 58, 145–156. [Google Scholar] [CrossRef]
- Spartano, N.L.; Lyass, A.; Larson, M.G.; Tran, T.; Andersson, C.; Blease, S.J.; Esliger, D.W.; Vasan, R.S.; Murabito, J.M. Objective physical activity and physical performance in middle-aged and older adults. Exp. Gerontol. 2019, 119, 203–211. [Google Scholar] [CrossRef]
- Amaral Gomes, E.S.; Ramsey, K.A.; Rojer, A.G.M.; Reijnierse, E.M.; Maier, A.B. The Association of Objectively Measured Physical Activity and Sedentary Behavior with (Instrumental) Activities of Daily Living in Community-Dwelling Older Adults: A Systematic Review. Clin. Interv. Aging 2021, 16, 1877–1915. [Google Scholar] [CrossRef]
- Roberts, C.E.; Phillips, L.H.; Cooper, C.L.; Gray, S.; Allan, J.L. Effect of Different Types of Physical Activity on Activities of Daily Living in Older Adults: Systematic Review and Meta-Analysis. J. Aging Phys. Act. 2017, 25, 653–670. [Google Scholar] [CrossRef]
- Perez-Sousa, M.A.; Venegas-Sanabria, L.C.; Chavarro-Carvajal, D.A.; Cano-Gutierrez, C.A.; Izquierdo, M.; Correa-Bautista, J.E.; Ramírez-Vélez, R. Gait speed as a mediator of the effect of sarcopenia on dependency in activities of daily living. J. Cachexia. Sarcopenia Muscle 2019, 10, 1009–1015. [Google Scholar] [CrossRef]
- Kiselev, J.; Nuritdinow, T.; Spira, D.; Buchmann, N.; Steinhagen-Thiessen, E.; Lederer, C.; Daumer, M.; Demuth, I. Long-term gait measurements in daily life: Results from the Berlin Aging Study II (BASE-II). PLoS ONE 2019, 14, e0225026. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Valentine, R.J.; Evans, E.M.; Sosnoff, J.J. Lower extremity muscle quality and gait variability in older adults. Age Ageing 2012, 41, 595–599. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, A.F.; Paul, G.; Hausdorff, J.M. Risk factors for falls among older adults: A review of the literature. Maturitas 2013, 75, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Allali, G.; Ayers, E.I.; Verghese, J. Multiple modes of assessment of gait are better than one to predict incident falls. Arch. Gerontol. Geriatr. 2015, 60, 389–393. [Google Scholar] [CrossRef]
- Mielke, M.M.; Roberts, R.O.; Savica, R.; Cha, R.; Drubach, D.I.; Christianson, T.; Pankratz, V.S.; Geda, Y.E.; Machulda, M.M.; Ivnik, R.J.; et al. Assessing the Temporal Relationship Between Cognition and Gait: Slow Gait Predicts Cognitive Decline in the Mayo Clinic Study of Aging. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2013, 68, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Desai, P.; Evans, D.; Dhana, K.; Aggarwal, N.T.; Wilson, R.S.; McAninch, E.; Rajan, K.B. Longitudinal Association of Total Tau Concentrations and Physical Activity with Cognitive Decline in a Population Sample. JAMA Netw. Open 2021, 4, e2120398. [Google Scholar] [CrossRef]
- Carvalho, A.; Rea, I.M.; Parimon, T.; Cusack, B.J. Physical activity and cognitive function in individuals over 60 years of age: A systematic review. Clin. Interv. Aging 2014, 9, 661–682. [Google Scholar]
- Studenski, S.; Faulkner, K.; Inzitari, M.; Brach, J.; Chandler, J.; Cawthon, P.; Connor, E.B.; Kritchevsky, S.; Badinelli, S.; Harris, T.; et al. Gait Speed and Survival in Older Adults. JAMA J. Am. Med. Assoc. 2015, 305, 50–58. [Google Scholar] [CrossRef]
- Rasmussen, L.J.H.; Caspi, A.; Ambler, A.; Broadbent, J.M.; Cohen, H.J.; D’Arbeloff, T.; Elliott, M.; Hancox, R.J.; Harrington, H.; Hogan, S.; et al. Association of Neurocognitive and Physical Function with Gait Speed in Midlife. JAMA Netw. Open 2019, 2, e1913123. [Google Scholar] [CrossRef]
- White, D.K.; Neogi, T.; Nevitt, M.C.; Peloquin, C.E.; Zhu, Y.; Boudreau, R.M.; Cauley, J.A.; Ferrucci, L.; Harris, T.B.; Satterfield, S.M.; et al. Trajectories of Gait Speed Predict Mortality in Well-Functioning Older Adults: The Health, Aging and Body Composition Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2013, 68, 456–464. [Google Scholar] [CrossRef]
- Galvão, L.L.; Silva, R.R.; Tribess, S.; Santos, D.A.T.; Junior, J.S.V. Physical activity combined with sedentary behaviour in the risk of mortality in older adults. Rev. Saude Publica 2021, 55, 60. [Google Scholar] [PubMed]
- da Silva Sousa, N.F.; de Paule Barbosa Medina, L.; Bastos, T.F.; Monteiro, C.N.; Lima, M.G.; de Azevedo Barros, M.B. Social inequalities in the prevalence of indicators of active aging in the Brazilian population: National Health Survey, 2013. Rev. Bras. Epidemiol. 2019, 22. [Google Scholar] [CrossRef]
- Ribeiro, A.Q.; Salgado, S.M.L.; Gomes, I.S.; Fogal, A.S.; Martinho, K.O.; Almeida, L.F.F.; de Oliveira, W.C. Prevalence and factors associated with physical inactivity among the elderly: A population-based study. Rev. Bras. Geriatr. Gerontol. 2016, 19, 483–493. [Google Scholar] [CrossRef]
- Dugan, S.A.; Gabriel, K.P.; Lange-Maia, B.S.; Karvonen-Gutierrez, C. Physical Activity and Physical Function. Obstet. Gynecol. Clin. N. Am. 2018, 45, 723–736. [Google Scholar] [CrossRef]
- Manini, T.M.; Pahor, M. Physical activity and maintaining physical function in older adults. Br. J. Sports Med. 2008, 43, 28–31. [Google Scholar] [CrossRef] [PubMed]

| Variable | Low PA Mean (SD) | High PA Mean (SD) | Total Mean (SD) | p-Value |
|---|---|---|---|---|
| (n = 348) | (n = 349) | (n = 697) | ||
| Age (years) | 71.17 ± 7.28 | 69.53 ± 6.31 | 70.35 ± 6.36 | 0.007 |
| Sex (n) (%) | ||||
| women | 196 (56.3) | 234 (67.0) | 430 (61.7) | 0.004 |
| men | 152 (43.7) | 115 (33.0) | 267 (38.3) | |
| BMI (k/m2) | 27.88 ± 5.00 | 25.30 ± 3.86 | 28.21 ± 4.93 | 0.059 |
| Education (years) | 3.65 ± 4.72 | 7.03 ± 7.80 | 5.35 ± 5.55 | <0.001 |
| MMSE (n) | 23.50 ± 4.40 | 25.30 ± 3.86 | 24.41 ± 4.23 | <0.001 |
| Falls n (%) | 121 (34.8) | 106 (30.4) | 227 (32.6) | 0.216 |
| Medication (n) | 1.83 ± 1.83 | 1.95 ± 1.74 | 1.89 ± 1.79 | 0.255 |
| Comorbidities (n) (%) | ||||
| Hypertension | 201 (57.8) | 194 (55.6) | 395 (56.7) | 0.563 |
| Visual impairment | 289 (83.0) | 293 (84.0) | 582 (83.5) | 0.012 |
| Hearing problems | 85 (24.4) | 95 (27.2) | 180 (25.8) | 0.555 |
| Physical activity | ||||
| PA-housework (n) | 2.87 ± 0.44 | 2.78 ± 0.45 | 2.82 ± 0.47 | 0.024 |
| PA-sport (n) | 2.00 ± 0.40 | 2.35 ± 0.62 | 2.18 ± 0.55 | <0.001 |
| PA-leisure (n) | 2.70 ± 0.51 | 2.93 ± 0.55 | 2.71 ± 0.54 | <0.001 |
| PA-total (n) | 2.52 ± 0.45 | 2.68 ± 1.53 | 2.57 ± 0.52 | <0.001 |
| Physical function | ||||
| CST (n) | 11.71 ± 3.41 | 11.74 ± 3.02 | 11.73 ± 3.22 | 0.790 |
| ACT (n) | 13.92 ± 4.41 | 12.36 ± 3.91 | 13.14 ± 4.24 | <0.001 |
| CSAR (cm) | 1.97 ± 8.02 | 3.77 ± 10.70 | 2.87 ± 9.50 | 0.021 |
| BST (cm) | −12.80 ± 12.90 | −6.87 ± 10.61 | −9.84 ± 12.17 | <0.001 |
| FUG (seg.) | 6.71 ± 2.45 | 5.88 ± 1.20 | 6.30 ± 1.98 | <0.001 |
| MWT 6 (m) | 400.47 ± 89.90 | 438.98 ± 80.16 | 419.75 ± 87.25 | <0.001 |
| PF total (score) | 422.75 ± 94.84 | 465.86 ± 86.11 | 444.40 ± 93.04 | <0.001 |
| Gait speed (m/s) | 1.17 ± 0.36 | 1.52 ± 0.49 | 1.35 ± 0.46 | <0.001 |
| Variable | PA-Housework | PA-Sport | PA-Leisure | PA-Total | PF |
|---|---|---|---|---|---|
| PA-sport | 0.245 *** | ||||
| PA-leisure | −0.005 ns | 0.000 ns | |||
| PA-total | 0.003 ns | 0.476 *** | 0.870 *** | ||
| PF | 0.118 ** | 0.139 *** | 0.160 *** | 0.226 *** | |
| Gait speed | −0.215 *** | 0.515 *** | 0.075 * | 0.298 *** | 0.302 *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nascimento, M.d.M.; Gouveia, É.R.; Marques, A.; Gouveia, B.R.; Marconcin, P.; França, C.; Ihle, A. The Role of Physical Function in the Association between Physical Activity and Gait Speed in Older Adults: A Mediation Analysis. Int. J. Environ. Res. Public Health 2022, 19, 12581. https://doi.org/10.3390/ijerph191912581
Nascimento MdM, Gouveia ÉR, Marques A, Gouveia BR, Marconcin P, França C, Ihle A. The Role of Physical Function in the Association between Physical Activity and Gait Speed in Older Adults: A Mediation Analysis. International Journal of Environmental Research and Public Health. 2022; 19(19):12581. https://doi.org/10.3390/ijerph191912581
Chicago/Turabian StyleNascimento, Marcelo de Maio, Élvio Rúbio Gouveia, Adilson Marques, Bruna R. Gouveia, Priscila Marconcin, Cíntia França, and Andreas Ihle. 2022. "The Role of Physical Function in the Association between Physical Activity and Gait Speed in Older Adults: A Mediation Analysis" International Journal of Environmental Research and Public Health 19, no. 19: 12581. https://doi.org/10.3390/ijerph191912581
APA StyleNascimento, M. d. M., Gouveia, É. R., Marques, A., Gouveia, B. R., Marconcin, P., França, C., & Ihle, A. (2022). The Role of Physical Function in the Association between Physical Activity and Gait Speed in Older Adults: A Mediation Analysis. International Journal of Environmental Research and Public Health, 19(19), 12581. https://doi.org/10.3390/ijerph191912581












