Acute Normobaric Hypoxia Lowers Executive Functions among Young Men despite Increase of BDNF Concentration
Abstract
:1. Introduction
2. Materials and Methods
2.1. Anthropometric Measurements
2.2. Normobaric Hypoxia
2.3. Cognitive Functions
Executive Functions: Stroop Test
2.4. Blood Sampling
2.5. Statistical Analysis
3. Results
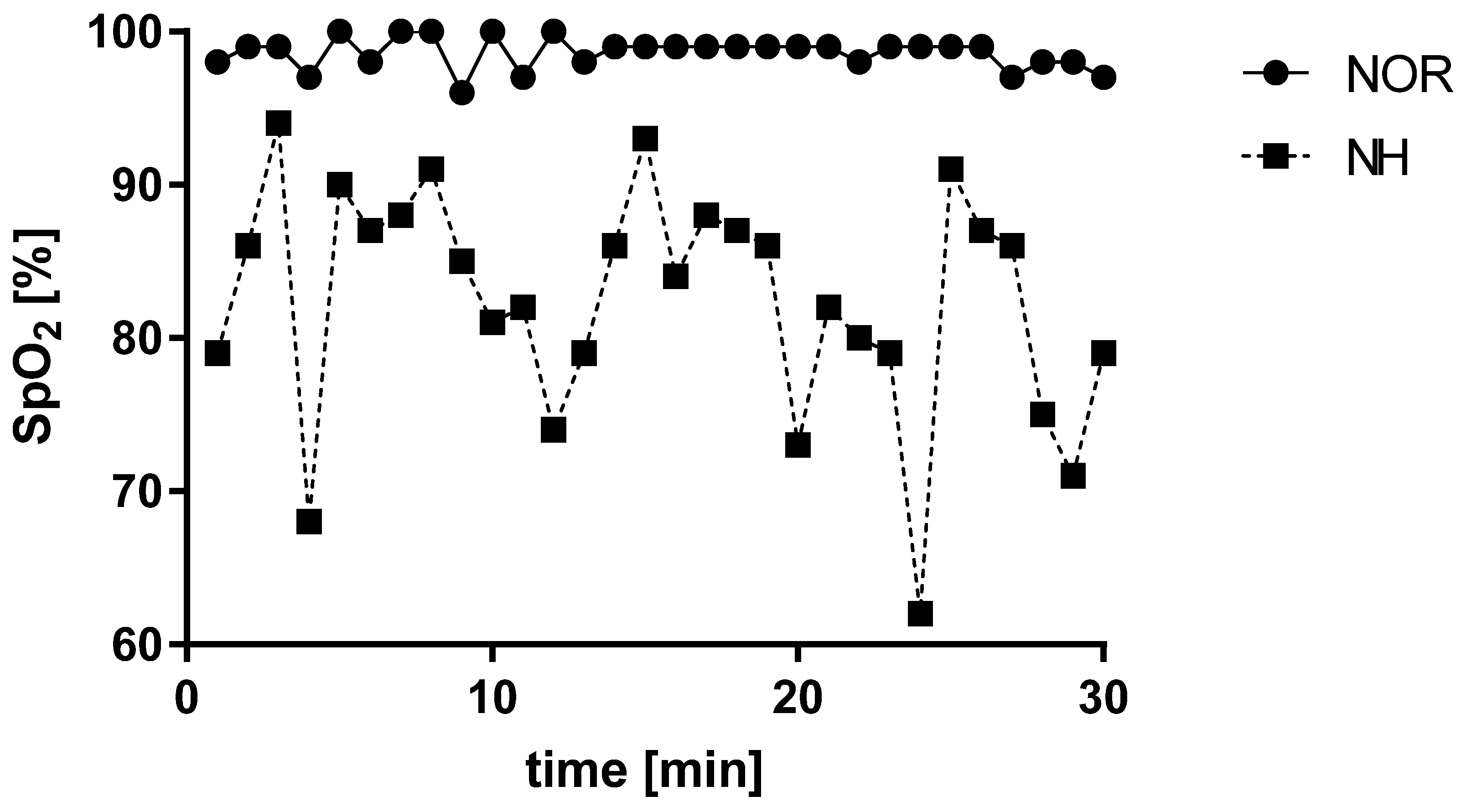
3.1. Blood Saturation under Normoxia and Acute Normobaric Hypoxia Conditions
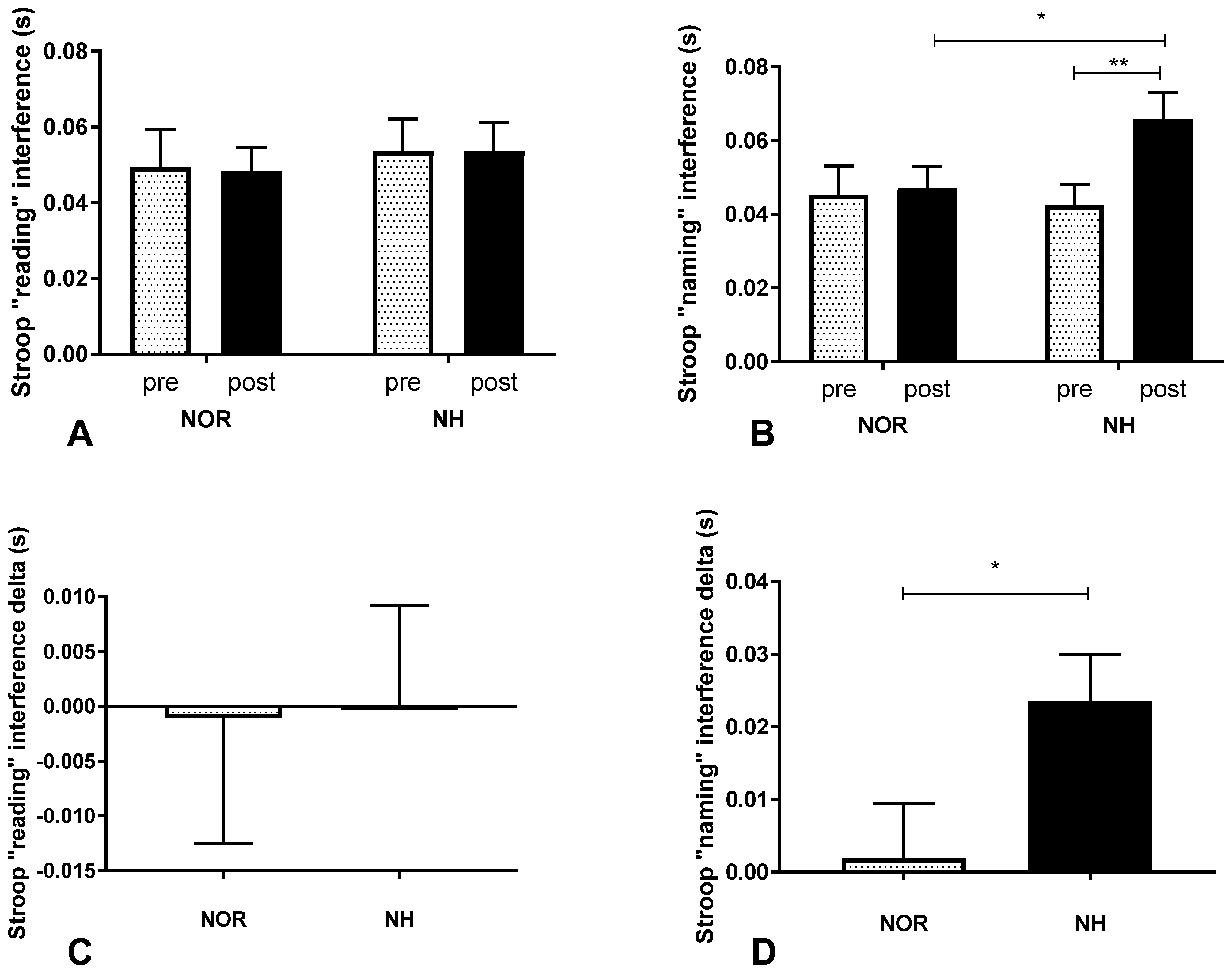
3.2. Stroop Test under Conditions of Normoxia and Acute Normobaric Hypoxia
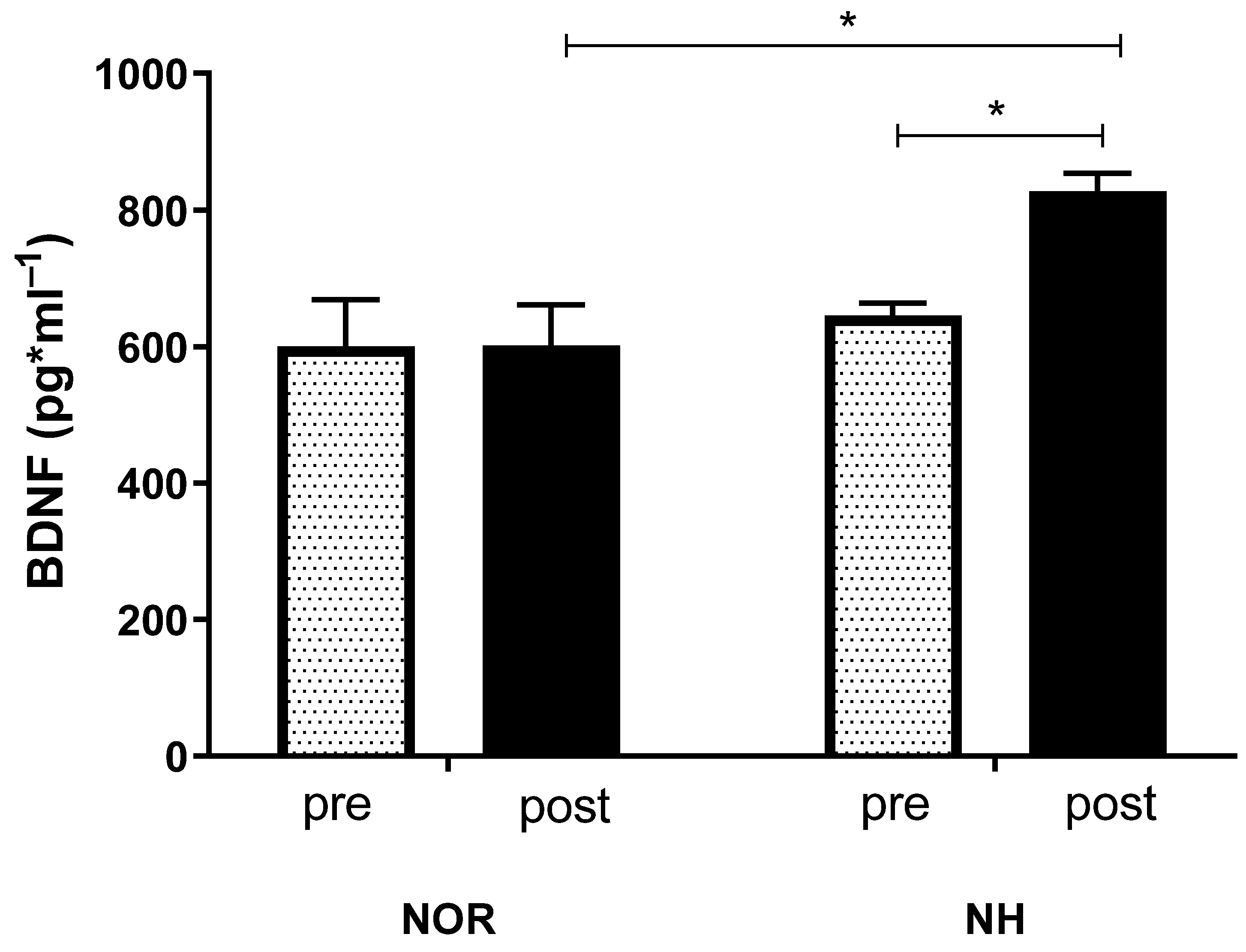
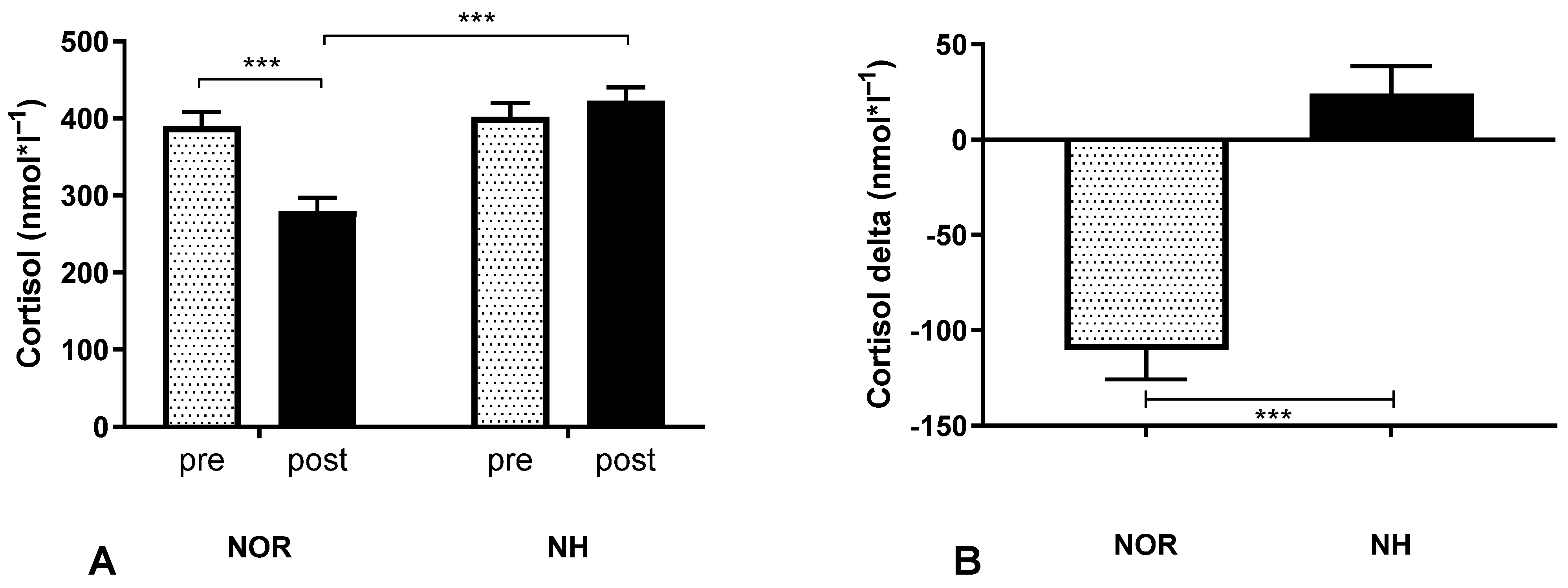
3.3. Blood Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Amann, M.; Kayser, B. Nervous system function during exercise in hypoxia. High Alt. Med. Biol. 2009, 10, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Lieberman, P.; Protopapas, A.; Reed, E.; Youngs, J.W.; Kanki, B.G. Cognitive defects at altitude. Nature 1994, 372, 325. [Google Scholar] [CrossRef]
- Davis, J.M.; Bailey, S.P. Possible mechanisms of central nervous system fatigue during exercise. Med. Sci. Sports Exerc. 1997, 29, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Ratey, J.J.; Loehr, J.E. The positive impact of physical activity on cognition during adulthood: A review of underlying mechanisms, evidence and recommendations. Rev. Neurosci. 2011, 22, 171–185. [Google Scholar] [CrossRef] [PubMed]
- Wrann, C.D.; White, J.P.; Salogiannnis, J.; Laznik-Bogoslavski, D.; Wu, J.; Ma, D.; Lin, J.D.; Greenberg, M.E.; Spiegelman, B.M. Exercise induces hippocampal BDNF through a PGC-1alpha/FNDC5 pathway. Cell Metab. 2013, 18, 649–659. [Google Scholar] [CrossRef]
- Cotman, C.W.; Berchtold, N.C.; Christie, L.A. Exercise builds brain health: Key roles of growth factor cascades and inflammation. Trends Neurosci. 2007, 30, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Wang, D.D.; Zhang, T.Y.; Yu, H.; Wang, Y.; Huang, S.H.; Lee, F.S.; Chen, Z.Y. Region-specific involvement of BDNF secretion and synthesis in conditioned taste aversion memory formation. J. Neurosci. 2011, 31, 2079–2090. [Google Scholar] [CrossRef]
- Mantilla, C.B.; Gransee, H.M.; Zhan, W.Z.; Sieck, G.C. Motoneuron BDNF/TrkB signaling enhances functional recovery after cervical spinal cord injury. Exp. Neurol. 2013, 247, 101–109. [Google Scholar] [CrossRef]
- Vavrek, R.; Girgis, J.; Tetzlaff, W.; Hiebert, G.W.; Fouad, K. BDNF promotes connections of corticospinal neurons onto spared descending interneurons in spinal cord injured rats. Brain 2006, 129, 1534–1545. [Google Scholar] [CrossRef]
- Wheeler, E.F.; Naftel, J.P.; Pan, M.; von Bartheld, C.S.; Byers, M.R. Neurotrophin receptor expression is induced in a subpopulation of trigeminal neurons that label by retrograde transport of NGF or fluoro-gold following tooth injury. Brain Res. Mol. Brain Res. 1998, 61, 23–38. [Google Scholar] [CrossRef]
- Komulainen, P.; Pedersen, M.; Hanninen, T.; Bruunsgaard, H.; Lakka, T.A.; Kivipelto, M.; Hassinen, M.; Rauramaa, T.H.; Pedersen, B.K.; Rauramaa, R. BDNF is a novel marker of cognitive function in ageing women: The DR’s EXTRA Study. Neurobiol. Learn. Mem. 2008, 90, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Griffin, E.W.; Bechara, R.G.; Birch, A.M.; Kelly, A.M. Exercise enhances hippocampal-dependent learning in the rat: Evidence for a BDNF-related mechanism. Hippocampus 2009, 19, 973–980. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.C.; Kim, J.; Kim, S.; Son, Y.H.; Lee, N.; Jung, S.H. The concentrations of serum, plasma and platelet BDNF are all increased by treadmill VO(2)max performance in healthy college men. Neurosci. Lett. 2012, 519, 78–83. [Google Scholar] [CrossRef]
- Knaepen, K.; Goekint, M.; Heyman, E.M.; Meeusen, R. Neuroplasticity—exercise-induced response of peripheral brain-derived neurotrophic factor: A systematic review of experimental studies in human subjects. Sport Med. 2010, 40, 765–801. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.K.; Ha, C.H. The effect of exercise intensity on brain derived neurotrophic factor and memory in adolescents. Environ. Health Prev. Med. 2017, 22, 27. [Google Scholar] [CrossRef]
- Leal, G.; Bramham, C.R.; Duarte, C.B. BDNF and Hippocampal Synaptic Plasticity. Vitam. Horm. 2017, 104, 153–195. [Google Scholar] [CrossRef]
- Liu, P.Z.; Nusslock, R. Exercise-Mediated Neurogenesis in the Hippocampus via BDNF. Front. Neurosci. 2018, 12, 52. [Google Scholar] [CrossRef]
- Dwivedi, Y. Brain-derived neurotrophic factor: Role in depression and suicide. Neuropsychiatr. Dis. Treat. 2009, 5. [Google Scholar] [CrossRef]
- Hellhammer, D.H.; Wüst, S.; Kudielka, B.M. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology 2009, 34, 163–171. [Google Scholar] [CrossRef]
- Russell, E.; Koren, G.; Rieder, M.; Van Uum, S. Hair cortisol as a biological marker of chronic stress: Current status, future directions and unanswered questions. Psychoneuroendocrinology 2012, 37, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Echouffo-Tcheugui, J.B.; Conner, S.C.; Himali, J.J.; Maillard, P.; Decarli, C.S.; Beiser, A.S.; Vasan, R.S.; Seshadri, S. Circulating cortisol and cognitive and structural brain measures. Neurology 2018, 91, e1961–e1970. [Google Scholar] [CrossRef] [PubMed]
- Gaelle, D.; Nadia, H.; Thomas, P.; Vincent, D.; Jean-Louis, G.; Catherine, B.; Nicole, M.; Daniel, B. Sustained corticosterone rise in the prefrontal cortex is a key factor for chronic stress-induced working memory deficits in mice. Neurobiol. Stress 2019, 10, 100161. [Google Scholar] [CrossRef]
- Milani, P.; Piu, P.; Popa, T.; Della Volpe, R.; Bonifazi, M.; Rossi, A.; Mazzocchio, R. Cortisol-induced effects on human cortical excitability. Brain Stimul. 2010, 3, 131–139. [Google Scholar] [CrossRef]
- Woods, D.R.; Davison, A.; Stacey, M.; Smith, C.; Hooper, T.; Neely, D.; Turner, S.; Peaston, R.; Mellor, A. The cortisol response to hypobaric hypoxia at rest and post-exercise. Horm. Metab. Res. 2012, 44, 302–305. [Google Scholar] [CrossRef] [PubMed]
- Pontremolesi, S.; Biselli, R.; Appiani, G.C.; D’Amelio, R.; Simona, S.; Patacchioli, F.R. Acute hypobaric-hypoxia challenge and salivary cortisol and DHEA-S in healthy male subjects. Aviat. Sp. Environ. Med. 2012, 83, 637–642. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.; Cruttenden, R.; Mellor, A.; Lumb, A.; Pattman, S.; Burnett, A.; Boot, C.; Burnip, L.; Boos, C.; O’Hara, J.; et al. A pilot investigation into the effects of acute normobaric hypoxia, high altitude exposure and exercise on serum angiotensin-converting enzyme, aldosterone and cortisol. JRAAS J. Renin-Angiotensin-Aldosterone Syst. 2018, 19, 1470320318782782. [Google Scholar] [CrossRef] [PubMed]
- Benso, A.; Broglio, F.; Aimaretti, G.; Lucatello, B.; Lanfranco, F.; Ghigo, E.; Grottoli, S. Endocrine and metabolic responses to extreme altitude and physical exercise in climbers. Eur. J. Endocrinol. 2007, 157, 733–740. [Google Scholar] [CrossRef]
- Shatilo, V.B.; Korkushko, O.V.; Ischuk, V.A.; Downey, H.F.; Serebrovskaya, T.V. Effects of intermittent hypoxia training on exercise performance, hemodynamics, and ventilation in healthy senior men. High Alt. Med. Biol. 2008, 9, 43–52. [Google Scholar] [CrossRef]
- Griffin, E.W.; Mullally, S.; Foley, C.; Warmington, S.A.; O’Mara, S.M.; Kelly, A.M. Aerobic exercise improves hippocampal function and increases BDNF in the serum of young adult males. Physiol. Behav. 2011, 104, 934–941. [Google Scholar] [CrossRef]
- Chroboczek, M.; Kostrzewa, M.; Micielska, K.; Grzywacz, T.; Laskowski, R. Effect of acute normobaric hypoxia exposure on executive functions among young physically active males. J. Clin. Med. 2021, 10, 1560. [Google Scholar] [CrossRef]
- Brunoni, A.R.; Lopes, M.; Fregni, F. A systematic review and meta-analysis of clinical studies on major depression and BDNF levels: Implications for the role of neuroplasticity in depression. Int. J. Neuropsychopharmacol. 2008, 11, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Maffioletti, E.; Zanardini, R.; Gennarelli, M.; Bocchio-Chiavetto, L. Influence of clotting duration on brain-derived neurotrophic factor (BDNF) dosage in serum. Biotechniques 2014, 57, 111–114. [Google Scholar] [CrossRef] [PubMed]
- Serra-Millas, M. Are the changes in the peripheral brain-derived neurotrophic factor levels due to platelet activation? World J. Psychiatry 2016, 6, 84–101. [Google Scholar] [CrossRef] [PubMed]
- Asmaro, D.; Mayall, J.; Ferguson, S. Cognition at altitude: Impairment in executive and memory processes under hypoxic conditions. Aviat. Sp. Environ. Med. 2013, 84, 1159–1165. [Google Scholar] [CrossRef]
- Virués-Ortega, J.; Buela-Casal, G.; Garrido, E.; Alcázar, B. Neuropsychological functioning associated with high-altitude exposure. Neuropsychol. Rev. 2004, 14, 197–224. [Google Scholar] [CrossRef]
- Li, X.Y.; Wu, X.Y.; Fu, C.; Shen, X.F.; Yang, C.B.; Wu, Y.H. Effects of acute exposure to mild or moderate hypoxia on human psychomotor performance and visual-reaction time. Space Med. Med. Eng. 2000, 13, 235–239. [Google Scholar]
- Guo, X.F.; Zhao, Y.N.; Li, J.M.; Chen, C.X.; Li, S.X. Effect of obstructive sleep apnea hypoxia on learning memory capacity after cerebral ischemia-reperfusion in rats. Zhonghua Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 2016, 51, 282–285. [Google Scholar] [CrossRef]
- Bayer, U.; Likar, R.; Pinter, G.; Stettner, H.; Demschar, S.; Trummer, B.; Neuwersch, S.; Glazachev, O.; Burtscher, M. Intermittent hypoxic–hyperoxic training on cognitive performance in geriatric patients. Alzheimer’s Dement. Transl. Res. Clin. Interv. 2017, 3, 114–122. [Google Scholar] [CrossRef]
- Serebrovska, Z.O.; Serebrovska, T.V.; Kholin, V.A.; Tumanovska, L.V.; Shysh, A.M.; Pashevin, D.A.; Goncharov, S.V.; Stroy, D.; Grib, O.N.; Shatylo, V.B.; et al. Intermittent hypoxia-hyperoxia training improves cognitive function and decreases circulating biomarkers of Alzheimer’s disease in patients with mild cognitive impairment: A pilot study. Int. J. Mol. Sci. 2019, 20, 5405. [Google Scholar] [CrossRef]
- Shimada, Y.; Kobayashi, M.; Yoshida, K.; Terasaki, K.; Fujiwara, S.; Kubo, Y.; Beppu, T.; Ogasawara, K. Reduced Hypoxic Tissue and Cognitive Improvement after Revascularization Surgery for Chronic Cerebral Ischemia. Cerebrovasc. Dis. 2019, 47, 57–64. [Google Scholar] [CrossRef]
- Dreimüller, N.; Schlicht, K.F.; Wagner, S.; Peetz, D.; Borysenko, L.; Hiemke, C.; Lieb, K.; Tadić, A. Early reactions of brain-derived neurotrophic factor in plasma (pBDNF) and outcome to acute antidepressant treatment in patients with Major Depression. Neuropharmacology 2012, 62, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Schölzke, M.N.; Schwaninger, M. Transcriptional regulation of neurogenesis: Potential mechanisms in cerebral ischemia. J. Mol. Med. 2007, 85, 577–588. [Google Scholar] [CrossRef]
- Rybnikova, E.; Vataeva, L.; Tyulkova, E.; Gluschenko, T.; Otellin, V.; Pelto-Huikko, M.; Samoilov, M.O. Mild hypoxia preconditioning prevents impairment of passive avoidance learning and suppression of brain NGFI-A expression induced by severe hypoxia. Behav. Brain Res. 2005, 160, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.H.; Yan, H.C.; Zhang, J.; Qu, H.D.; Qiu, X.S.; Chen, L.; Li, S.J.; Cao, X.; Bean, J.C.; Chen, L.H.; et al. Intermittent hypoxia promotes hippocampal neurogenesis and produces antidepressant-like effects in adult rats. J. Neurosci. 2010, 30, 12653–12663. [Google Scholar] [CrossRef]
- Xie, H.; Leung, K.L.; Chen, L.; Chan, Y.S.; Ng, P.C.; Fok, T.F.; Wing, Y.K.; Ke, Y.; Li, A.M.; Yung, W.H. Brain-derived neurotrophic factor rescues and prevents chronic intermittent hypoxia-induced impairment of hippocampal long-term synaptic plasticity. Neurobiol. Dis. 2010, 40, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.E.; Barker-Collo, S.L.; Connell, C.J.W.; Gant, N. Acute hypoxic gas breathing severely impairs cognition and task learning in humans. Physiol. Behav. 2015, 142, 104–110. [Google Scholar] [CrossRef]
- Eichhorn, L.; Erdfelder, F.; Kessler, F.; Doerner, J.; Thudium, M.O.; Meyer, R.; Ellerkmann, R.K. Evaluation of near-infrared spectroscopy under apnea-dependent hypoxia in humans. J. Clin. Monit. Comput. 2015, 29, 749–757. [Google Scholar] [CrossRef]
- Eichhorn, L.; Erdfelder, F.; Kessler, F.; Dolscheid-Pommerich, R.C.; Zur, B.; Hoffmann, U.; Ellerkmann, R.E.; Meyer, R. Influence of Apnea-induced Hypoxia on Catecholamine Release and Cardiovascular Dynamics. Int. J. Sports Med. 2017, 38, 85–91. [Google Scholar] [CrossRef]
- Steinback, C.D.; Poulin, M.J. Influence of hypoxia on cerebral blood flow regulation in humans. In Advances in Experimental Medicine and Biology; Springer New York LLC: New York, NY, USA, 2016; Volume 903, pp. 131–144. [Google Scholar]
- McMorris, T.; Hale, B.J.; Barwood, M.; Costello, J.; Corbett, J. Corrigendum to “Effect of acute hypoxia on cognition: A systematic review and meta-regression analysis” Neurosci. Biobehav. Rev. 74 (2017) 225–232. Neurosci. Biobehav. Rev. 2019, 98, 333. [Google Scholar] [CrossRef]
- Bugg, J.M.; DeLosh, E.L.; Davalos, D.B.; Davis, H.P. Age differences in Stroop interference: Contributions of general slowing and task-specific deficits. Neuropsychol. Dev. Cogn. B. Aging Neuropsychol. Cogn. 2007, 14, 155–167. [Google Scholar] [CrossRef]
- Ochi, G.; Yamada, Y.; Hyodo, K.; Suwabe, K.; Fukuie, T.; Byun, K.; Dan, I.; Soya, H. Neural basis for reduced executive performance with hypoxic exercise. Neuroimage 2018, 171, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Woods, D.R.; O’Hara, J.P.; Boos, C.J.; Hodkinson, P.D.; Tsakirides, C.; Hill, N.E.; Jose, D.; Hawkins, A.; Phillipson, K.; Hazlerigg, A.; et al. Markers of physiological stress during exercise under conditions of normoxia, normobaric hypoxia, hypobaric hypoxia, and genuine high altitude. Eur. J. Appl. Physiol. 2017, 117, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.B.; Corbett, J.; McMorris, T.; Young, J.S.; Dicks, M.; Ando, S.; Thelwell, R.C.; Tipton, M.J.; Costello, J.T. Cognitive performance is associated with cerebral oxygenation and peripheral oxygen saturation, but not plasma catecholamines, during graded normobaric hypoxia. Exp. Physiol. 2019, 104, 1384–1397. [Google Scholar] [CrossRef] [PubMed]
| N = 32 | X | SD |
|---|---|---|
| Age (years) | 20.4 | 0.6 |
| Height (cm) | 178 | 7.1 |
| Weight (kg) | 76.6 | 11.4 |
| FAT (%) | 18.6 | 4.1 |
| FAT (kg) | 14.4 | 5.0 |
| FFM (kg) | 62.1 | 7.9 |
| BMI (kg∙m−2) | 23.9 | 2.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chroboczek, M.; Kujach, S.; Łuszczyk, M.; Grzywacz, T.; Soya, H.; Laskowski, R. Acute Normobaric Hypoxia Lowers Executive Functions among Young Men despite Increase of BDNF Concentration. Int. J. Environ. Res. Public Health 2022, 19, 10802. https://doi.org/10.3390/ijerph191710802
Chroboczek M, Kujach S, Łuszczyk M, Grzywacz T, Soya H, Laskowski R. Acute Normobaric Hypoxia Lowers Executive Functions among Young Men despite Increase of BDNF Concentration. International Journal of Environmental Research and Public Health. 2022; 19(17):10802. https://doi.org/10.3390/ijerph191710802
Chicago/Turabian StyleChroboczek, Maciej, Sylwester Kujach, Marcin Łuszczyk, Tomasz Grzywacz, Hideaki Soya, and Radosław Laskowski. 2022. "Acute Normobaric Hypoxia Lowers Executive Functions among Young Men despite Increase of BDNF Concentration" International Journal of Environmental Research and Public Health 19, no. 17: 10802. https://doi.org/10.3390/ijerph191710802
APA StyleChroboczek, M., Kujach, S., Łuszczyk, M., Grzywacz, T., Soya, H., & Laskowski, R. (2022). Acute Normobaric Hypoxia Lowers Executive Functions among Young Men despite Increase of BDNF Concentration. International Journal of Environmental Research and Public Health, 19(17), 10802. https://doi.org/10.3390/ijerph191710802







