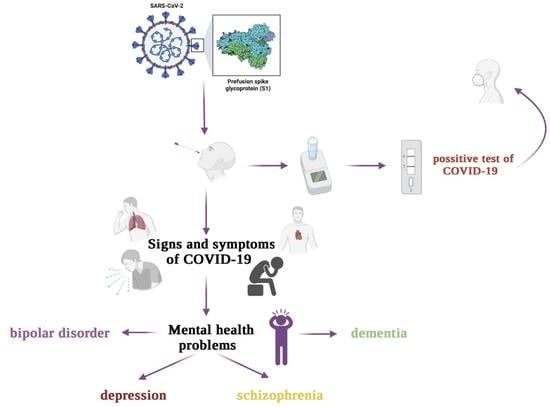
The Relationship between Mental Disorders and the COVID-19 Pandemic—Course, Risk Factors, and Potential Consequences
Abstract
:1. Introduction
2. The Risk Factors of COVID-19 Infection with the Coexistence of Mental Disorders
| Type of Study | Number of People in the Study | Results | Ref. |
|---|---|---|---|
| Survey conducted in China on people over the age of 18; researchers recruited participants through an online questionnaire by sending a link to local groups via the WeChat app | 1593 | 8.3% experienced anxiety and fear related to the presence of COVID-19 14.6% had symptoms of depression | [28] |
| Cross-sectional survey among Chinese students aged 12–18 during the COVID-19 pandemic via an online questionnaire | 8079 | 43.7% had depression 37.4% had anxiety 31.3% had mixed disorders (depression/anxiety) | [29] |
| A survey of undergraduate students in two large cities in southwest China using online versions of the PTSD Checklist Civilian Version and the 9-question Patient Health Questionnaires (PHQ-9) | 2485 | The prevalence of probable PTSD and depression was estimated at 2.7% and 9.0% respectively | [30] |
| Survey in a population of people over the age of 18 living in various provinces of Turkey through an online questionnaire | 343 | 23.6% showed symptoms of depression 45.1% showed symptoms of anxiety | [31] |
| A study conducted as an online survey data from the nationally representative “Understanding America Study” of the U.S. adult population | 6585 | 29% of respondents reported symptoms of depression/anxiety | [32] |
| Survey of undergraduate students in New Jersey through an online survey | 641 | 46% students reported elevated psychological stress 30.3% experienced increased health anxiety | [33] |
| A cross-sectional survey of the Hispanic population via online survey which contained 80 questions | 3480 | 18.7% of respondents showed depressive symptoms 21.6% reported anxiety symptoms 15.8% reported PTSD | [34] |
| A cross-sectional survey of the Italian population via online questionnaire on an online survey platform | 2766 | Depression: (a) 67.3% of respondents had an average level (b) 17% were in the high range (c) 15.4% were in the extremely high range Anxiety: (a) 81.3% of respondents had an average level (b) 7.2% were in the high range (c) 11.5% were in the extremely high range Stress: (a) 72.8% of respondents were in the average range (b) 14.6% were in the high range (c) 12.6% were in the extremely high range | [35] |
| A cross-sectional survey of the Iranian population via an online 15-question questionnaire conducted in 31 provinces of Iran | 10,754 | 50.9% had mild-to-severe anxiety symptoms 31.8% had mild-to-average symptoms 19.1% had severe-to-very severe symptoms | [36] |
| A cross-sectional survey of the Denmark population via online survey which included the five-item WHO-5 well-being scale; the survey also contained six questions regarding the experienced level of anxiety/depression over the past 2 weeks | 2458 | 25.4% showed depressive symptoms | [37] |
3. COVID-19 and Depression
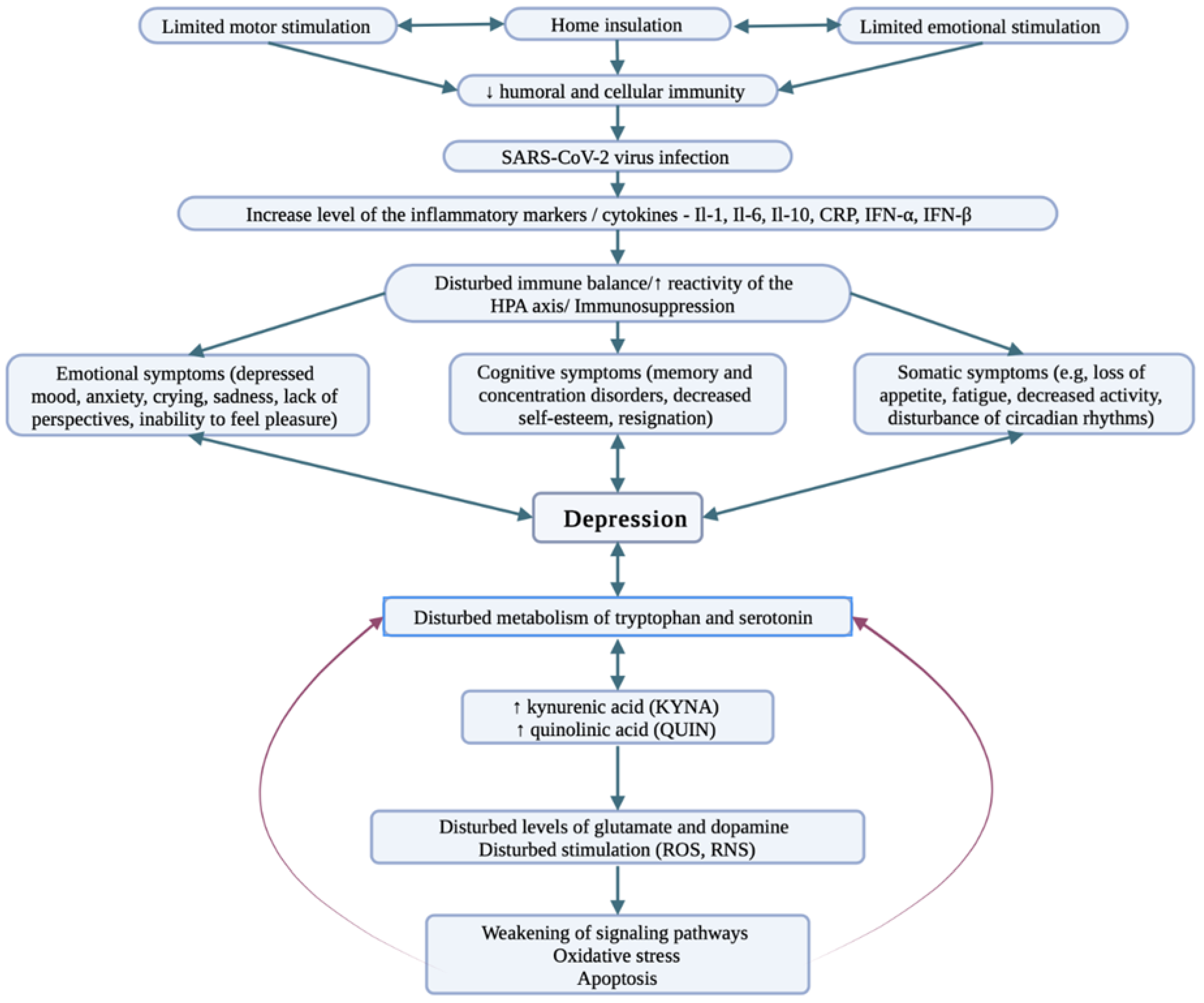
4. COVID-19 and Bipolar Disorder
5. COVID-19 and Schizophrenia
6. Dementia in COVID-19
7. Developmental Disorders in COVID-19
8. COVID-19 Vaccine and Mental Disorders
9. Long-Term Effects of SARS-CoV-2 and Their Potential Impact on the Further Course of Mental Disorders and the Convalescence Process
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Veazie, S.; Lafavor, B.; Vela, K.; Young, S.; Sayer, N.; Carlson, K.; O’Neil, M. Mental health outcomes of adults hospitalized for COVID-19: A systematic review. J. Affect. Disord. Rep. 2022, 8, 100312. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Seo, Y.E.; Sung, H.K.; Park, H.Y.; Han, M.H.; Lee, S.H. Predictors of the development of mental disorders in hospitalized COVID-19 patients without previous psychiatric history: A single-center retrospective study in South Korea. J. Environ. Res. Public Health 2022, 19, 1092. [Google Scholar] [CrossRef]
- Yun-Kuan Thye, A.; Woan-Fei Law, J.; Teng-Hern Tan, L.; Pusparajah, P.; Ser, H.-L.; Thurairajasingam, S.; Letchumanan, V.; Lee, L.-H. Psychological symptoms in COVID-19 patients: Insights into pathophysiology and risk factors of long COVID-19. Biology 2022, 11, 61. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, G.K.; Khedr, E.M.; Hamad, D.A.; Masherf, T.S.; Hashem, M.M.; Aly, M.M. Long term impact of COVID-19 infection on sleep and mental health: A cross-sectional study. Psychiatry Res. 2021, 305, 114243. [Google Scholar] [CrossRef]
- Natoli, S.; Oliveira, V.; Calabresi, P.; Maila, L.F.; Pisani, A. Does SARS-CoV-2 invade in brain? Translational lessons from animal models. Eur. J. Neurol. 2020, 27, 1764–1773. [Google Scholar] [CrossRef] [PubMed]
- Douaud, G.; Lee, S.; Alfaro-Almagro, F.; Arthofer, C.; Wang, C.; McCarthy, P.; Lange, F.; Andersson, J.L.R.; Griffanti, L.; Duff, E.; et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. medRxiv 2022. [Google Scholar] [CrossRef]
- Ahmed, J.O.; Ahmad, S.A.; Hassan, M.N.; Kakamad, F.H.; Salig, R.Q.; Abdulla, B.A.; Fattah, F.H.R.; Mohammed, S.H.; Ali, R.K.; Salig, A.M. Post COVID-19 neurological complications: A meta-analysis. Ann. Med. Surg. 2022, 76, 103440. [Google Scholar] [CrossRef] [PubMed]
- De Girolamo, G.; Cerveri, G.; Clerici, M.; Monzani, E.; Spinogatti, F.; Starace, F.; Tura, G.; Vita, A. Mental health in the coronavirus disease 2019 emergency–the Italian response. JAMA Psychiatry 2020, 77, 974–976. [Google Scholar] [CrossRef] [PubMed]
- Huff, C. Delirium, PTSD, brain fog: The aftermath of surviving COVID-19 effects. Monit. Psychol. 2020, 51. Available online: https://www.apa.org/monitor/2020/09/aftermath-covid-19 (accessed on 1 July 2020).
- Kahl, K.G.; Correll, C.U. Management of patients with severe mental illness during the coronavirus disease 2019 pandemic. JAMA Psychiatry 2020, 77, 977–978. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Z.; Lin, S.; Zhao, P. Perceived accessibility, and mental health consequences of COVID-19 containment policies. J. Transp. Health 2022, 25, 101354. [Google Scholar] [CrossRef]
- Rogers, J.P.; Chesney, E.; Oliver, D.; Pollak, T.A.; McGuire, P.; Fusar-Poli, P.; Zandi, M.S.; Lewis, G.; David, A.S. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: A systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry 2020, 7, 611–627. [Google Scholar] [CrossRef]
- Xie, Y.; Xu, E.; Al-Aly, Z. Risks of mental health outcomes in people with COVID-19: Cohort study. BMJ 2020, 376, e068993. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Qin, Q.; Basta, M.; Chen, B.; Li, Y. Psychological reactions, and insomnia in adults with mental health disorders during the COVID-19 outbreak. BMC Psychiatry 2021, 21, 19. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Coronavirus Disease (COVID-19) Dashboard. Available online: https://covid19.who.int (accessed on 2 April 2022).
- Wang, Q.; Xu, R.; Volkow, N.D. Increased risk of COVID-19 infection and mortality in people with mental disorders: Analysis from electronic health records in the United States. World Psychiatry 2021, 20, 124–130. [Google Scholar] [CrossRef]
- Elharake, J.A.; Akbar, F.; Malik, A.A.; Gilliam, W.; Omer, S.B. Mental health impact of COVID-19 among children and college students: A systematic review. Child Psychiatry Hum. Dev. 2022, 11, 1–13. [Google Scholar] [CrossRef]
- Dai, X.J.; Shao, Y.; Ren, L.; Tao, W.; Wang, Y. Risk factors of COVID-19 in subjects with and without mental disorders. J. Affect. Disord. 2022, 297, 102–111. [Google Scholar] [CrossRef]
- Boden, M.; Zimmerman, L.; Azevedo, K.J.; Ruzek, J.I.; Gala, S.; Abdel Magid, H.S.; Cohen, N.; Walser, R.; Mahtani, N.D.; Hoggatt, K.J.; et al. Addressing the mental health impact of COVID-19 through population health. Clin. Psychol. Rev. 2021, 85, 102006. [Google Scholar] [CrossRef]
- Alzueta, E.; Perrin, P.; Baker, F.C.; Caffarra, S.; Ramos-Usuga, D.; Yuksel, D.; Arango-Lasprilla, J.C. How the COVID-19 pandemic has changed our lives: A study of psychological correlates across 59 countries. J. Clin. Psychol. 2021, 77, 556–570. [Google Scholar] [CrossRef]
- Doerre, A.; Doblhammer, G. The influence of gender on COVID-19 infections and mortality in Germany: Insights from age- and gender-specific modeling of contact rates, infections, and deaths in the early phase of the pandemic. PLoS ONE 2022, 17, e0268119. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Cheshmehzangi, A.; McDonnell, D.; Šegalo, S.; Ahmad, J.; Bennett, B. Gender inequality and health disparity amid COVID-19. Nurs. Outlook 2022, 70, 89–95. [Google Scholar] [CrossRef]
- Glantz, M.D.; Anthony, J.C.; Berglund, P.A.; Degenhardt, L.; Dierker, L.; Kalaydjian, A.; Merikangas, K.R.; Rusico, A.M.; Swendsen, J.; Kessler, R.C. Mental disorders as risk factors for later substance dependence: Estimates of optional prevention and treatment benefits. Psychol. Med. 2009, 39, 1365–1377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vai, B.; Mazza, M.G.; Colli, C.D.; Foiselle, M.; Allen, B.; Benedetti, F.; Borsini, A.; Dias, M.C.; Tamouza, R.; Leboyer, M.; et al. Mental disorders and risk of COVID-10-related mortality, hospitalization, and intensive care unit admission: A systematic review and meta-analysis. Lancet Psychiatry 2021, 8, 797–812. [Google Scholar] [CrossRef]
- Larsen, J.R.; Martin, M.R.; Martin, J.D.; Kuhn, P.; Hicks, J.B. Modeling the onset of symptoms of COVID-19. Front. Public Health 2020, 8, 473. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Baumeister, R.F.; Veilleux, J.C.; Chen, C.; Liu, W.; Yue, Y.; Zhang, S. Rick factors associated with mental illness in hospital discharged patients infected with COVID-19 in Wuhan, China. Psychiatry Res. 2020, 292, 113297. [Google Scholar] [CrossRef] [PubMed]
- Loiwal, M. 20% Increase in Patients with Mental Illness Since Coronavirus Outbreak: Survey. 2020. Available online: https://indiatoday.in (accessed on 31 March 2020).
- Lei, L.; Huang, X.; Zhang, S.; Yang, J.; Yang, L.; Xu, M. Comparison of prevalence and associated factors of anxiety and depression among people affected by versus people unaffected by quarantine during the COVID-19 epidemic in southwestern China. Med. Sci. Monit. 2020, 26, e924609-1–e924609-12. [Google Scholar] [CrossRef] [Green Version]
- Zhou, S.-J.; Zhang, L.-G.; Wang, L.-L.; Guo, Z.-C.; Wang, J.-Q.; Chen, J.-C.; Liu, M.; Chen, X.; Chen, J.-X. Prevalence, and socio-demographic correlates of psychological health problems in Chinese adolescents during the outbreak of COVID-19. Eur. Child. Adolesc. Psychiatry 2020, 29, 749–758. [Google Scholar] [CrossRef]
- Tang, W.; Hu, T.; Hu, B.; Jin, C.; Wang, G.; Xie, C.; Chen, S.; Xu, J. Prevalence and corelates of PTSD and depressive symptoms one month after the outbreak of the COVID-19 epidemic in a sample of home-quarantined Chinese university students. J. Affect. Disord. 2020, 274, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Bayrak, S.; Ozdin, S. Levels and predictors of anxiety, depression, and health anxiety during COVID-19 pandemic in Turkish society: The importance of gender. Int. J. Soc. Psychiatry 2020, 66, 504–511. [Google Scholar] [CrossRef]
- Kampfen, F.; Kohler, I.V.; Ciancio, A.; Bruine de Bruin, W.; Maurer, J.; Kohler, H.-P. Predictors of mental health during the COVID-19 pandemic in the U.S.: Role of economic concerns, health worries and social distancing. PLoS ONE 2020, 15, e0241895. [Google Scholar] [CrossRef]
- Kibbey, M.M.; Fedorenko, E.J.; Farris, S.G. Anxiety, depression, and health anxiety in undergraduate students living in initial US outbreak “hotspot” during COVID-19 pandemic. Cogn. Bahav. Ther. 2021, 50, 409–421. [Google Scholar] [CrossRef] [PubMed]
- Gonzales-Sanguino, C.; Ausin, B.; Castellanos, M.A.; Saiz, J.; Lopez-Gomez, A.; Ugidos, C.; Munoz, M. Mental health consequences during the initial stage of the 2020 Coronavirus pandemic (COVID-19) in Spain. Brain Behav. Immun. 2020, 87, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Mazza, C.; Ricci, E.; Biondi, S.; Colasanti, M.; Ferracuti, S.; Napoli, C.; Roma, P. A nationwide survey of psychological distress among Italian people during the COVID-19 pandemic: Immediate psychological responses and associated factors. Int. J. Environ. Res. Public Health 2020, 17, 3165. [Google Scholar] [CrossRef] [PubMed]
- Moghanibashi-Mansourieh, A. Assessing the anxiety level of Iranian general population during COVID-19 outbreak. Asian J. Psychiatry 2020, 51, 102076. [Google Scholar] [CrossRef]
- Sonderskov, K.M.; Dinesen, P.T.; Santini, Z.I.; Ostergaard, S.D. The depressive state of Denmark during the COVID-19 pandemic. Acta Neuropsuchhiatrica 2020, 32, 226–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cichocki, Ł.; Karbownik, M. Nowe technologie a zdrowie psychiczne–Szanse i trudności. Psychiatria 2020, 17, 41–48. [Google Scholar] [CrossRef]
- Ni, M.Y.; Yang, L.; Leung, C.M.C.; Li, N.; Yao, X.I.; Wang, Y.; Leung, G.M.; Cowling, B.J.; Liao, Q. Mental health, risk factors, and social media use during the COVID-19 epidemic and cordon sanitaire among the community and health professionals in Wuhan, China: Cross-sectional survey. JMIR Ment. Health 2020, 7, e19009. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yang, L.; Zhang, C.; Xiang, Y.T.; Liu, Z.; Hu, S.; Zhang, B. Online mental health services in China during the COVID-19 outbreak. Lancet Psychiatry 2020, 7, e17–e18. [Google Scholar] [CrossRef]
- Depoux, A.; Martin, S.; Karafillakis, E.; Preet, R.; Wilder-Smith, A.; Larson, H. The pandemic of social media panic travel faster than COVID-19 outbreaks. J. Travel Med. 2020, 27, taaa031. [Google Scholar] [CrossRef] [Green Version]
- De Hert, M.; Correll, C.U.; Bobes, J.; Cetkovich-Bakmas, M.; Cohen, D.; Asai, I.; Detraux, J.; Gautam, S.; Moller, H.J.; Ndetei, D.M.; et al. Physical illness in patients with severe mental disorders. I. Prevalence, impact of medications and disparities in health care. World Psychiatry 2011, 10, 52–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- World Health Organization. Depression and Other Common Mental Disorders: Global Health Estimates. 2017. Available online: http://apps.who.int/iris/bitstream/10665/254610/1/WHO-MSD-MER-2017.2-eng.pdf (accessed on 2 April 2022).
- Isometsä, E. Suicidal behavior in mood disorders—Who, when, and why? Can. J. Psychiatry 2014, 59, 120–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiorillo, A.; Carpiniello, B.; De Giorgi, S.; La Pia, S.; Maina, G.; Sampogna, G.; Spina, E.; Tortorella, A.; Vita, A. Assessment and management of cognitive and psychosocial dysfunctions in patients with major depressive disorder: A Clinical Review. Front. Psychiatry 2018, 9, 493. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Depression. Available online: https://www.who.int/news-room/fact-sheets/detail/depression (accessed on 2 April 2022).
- WHO. The Global Burden of Disease: 2004 Update; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Dean, J.; Keshavan, M. The neurobiology of depression: An integrated view. Asian J. Psychiatr. 2017, 27, 101–111. [Google Scholar] [CrossRef]
- Miller, A.H.; Maletic, V.; Raison, C.L. Inflammation, and its discontents: The role of cytokines in the pathophysiology of major depression. Biol. Psychiatry 2009, 65, 732–741. [Google Scholar] [CrossRef] [Green Version]
- Felger, J.C.; Lotrich, F.E. Inflammatory cytokines in depression: Neurobiological mechanisms and therapeutic implications. Neuroscience 2013, 246, 199–229. [Google Scholar] [CrossRef] [Green Version]
- Pasco, J.A.; Nicholson, G.C.; Williams, L.J.; Jacka, F.N.; Henry, M.J.; Kotowicz, M.A.; Schneider, H.G.; Leonard, B.E.; Berk, M. Association of high-sensitivity C-reactive protein with de novo major depression. Br. J. Psychiatry 2010, 197, 372–377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raison, C.L.; Capuron, L.; Miller, A.H. Cytokines sing the blues: Inflammation and the pathogenesis of depression. Trends Immunol. 2006, 27, 24–31. [Google Scholar] [CrossRef] [Green Version]
- Dantzer, R.; O’Connor, J.C.; Freund, G.G.; Johnson, R.W.; Kelley, K.W. From inflammation to sickness and depression: When the immune system subjugates the brain. Nat. Rev. Neurosci. 2008, 9, 46–56. [Google Scholar] [CrossRef] [Green Version]
- Rhie, S.J.; Jung, E.Y.; Shim, I. The role of neuroinflammation on pathogenesis of affective disorders. J. Exerc. Rehabil. 2020, 16, 2–9. [Google Scholar] [CrossRef] [Green Version]
- Besedovsky, H.O.; del Rey, A. Immune-neuro-endocrine interactions: Facts and hypotheses. Endocr. Rev. 1996, 17, 64–102. [Google Scholar] [CrossRef]
- Miller, A.H. Norman Cousins Lecture. Mechanisms of cytokine-induced behavioral changes: Psychoneuroimmunology at the translational interface. Brain Behav. Immun. 2009, 23, 149–158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weiss, G.; Murr, C.; Zoller, H.; Haun, M.; Widner, B.; Ludescher, C.; Fuchs, D. Modulation of neopterin formation and tryptophan degradation by Th1- and Th2-derived cytokines in human monocytic cells. Clin. Exp. Immunol. 1999, 116, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Shao, X.; Wang, Y.; Huang, Y.; Miao, J.; Yang, X.; Zhu, G. An investigation of mental health status of children and adolescents in China during the outbreak of COVID-19. J. Affect. Disord. 2020, 275, 112–118. [Google Scholar] [CrossRef]
- Pérez-Cano, H.J.; Moreno-Murguía, M.B.; Morales-López, O.; Crow-Buchanan, O.; English, J.A.; Lozano-Alcázar, J.; Somilleda-Ventura, S.A. Anxiety, depression, and stress in response to the coronavirus disease-19 pandemic. Cir. Cir. 2020, 88, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Alpert, O.; Begun, L.; Garren, P.; Solhkhah, R. Cytokine storm induced new onset depression in patients with COVID-19. A new look into the association between depression and cytokines–two case reports. Brain Behav. Immun.-Health 2020, 9, 100173. [Google Scholar] [CrossRef] [PubMed]
- da Silva Lopes, L.; Silva, R.O.; de Sousa Lima, G.; de Araújo Costa, A.C.; Barros, D.F.; Silva-Néto, R.P. Is there a common pathophysiological mechanism between COVID-19 and depression? Acta Neurol. Belg. 2021, 121, 1117–1122. [Google Scholar] [CrossRef]
- Santini, Z.I.; Jose, P.E.; York Cornwell, E.; Koyanagi, A.; Nielsen, L.; Hinrichsen, C.; Meilstrup, C.; Madsen, K.R.; Koushede, V. Social disconnectedness, perceived isolation, and symptoms of depression and anxiety among older Americans (NSHAP): A longitudinal mediation analysis. Lancet Public Health 2020, 5, e62–e70. [Google Scholar] [CrossRef] [Green Version]
- Cacioppo, J.T.; Hawkley, L.C.; Crawford, L.E.; Ernst, J.M.; Burleson, M.H.; Kowalewski, R.B.; Malarkey, W.B.; Van Cauter, E.; Berntson, G.G. Loneliness and health: Potential mechanisms. Psychosom. Med. 2002, 64, 407–417. [Google Scholar] [CrossRef]
- Van Rheenen, T.E.; Meyer, D.; Neill, E.; Phillipou, A.; Tan, E.J.; Toh, W.L.; Rossell, S.L. Mental health status of individuals with a mood-disorder during the COVID-19 pandemic in Australia: Initial results from the COLLATE project. J. Affect. Disord. 2020, 275, 69–77. [Google Scholar] [CrossRef]
- Sepúlveda-Loyola, W.; Rodríguez-Sánchex, I.; Pérez-Rodríguez, P.; Ganz, F.; Torralba, R.; Oliveira, D.V.; Rodríguez-Mañas, L. Impact of social isolation due to COVID-19 on health in older people: Mental and physical effects and recommendations. J. Nutr. Health Aging 2020, 24, 938–947. [Google Scholar] [CrossRef]
- Lotfi, M.; Hamblin, M.R.; Rezaei, N. COVID-19: Transmission, prevention, and potential therapeutic opportunities. Clin. Chim. Acta 2020, 508, 254–266. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Zheng, D.; Liu, J.; Hong, Y.; Huan, Z.; Lou, D. Depression, and anxiety among adolescents during COVID-19: A cross-sectional study. Brain Behav. Immun. 2020, 88, 36–38. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Ye, M.; Fu, Y.; Yang, M.; Luo, F.; Yuan, J.; Tao, Q. The psychological impact of the COVID-19 pandemic on teenagers in China. J. Adolesc. Health 2020, 67, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Ellis, W.E.; Dumas, T.M.; Forbes, L.M. Physically isolated but socially connected: Psychological adjustment and stress among adolescents during the initial COVID-19 crisis. Can. J. Behav. Sci. Rev. Can. Sci. Comport. 2020, 52, 177–187. [Google Scholar] [CrossRef]
- Jones, E.A.K.; Mitra, A.K.; Bhuiyan, A.R. Impact of COVID-19 on mental health in adolescents: A systematic review. Int. J. Environ. Res. Public Health 2021, 18, 2470. [Google Scholar] [CrossRef]
- Vogel, E.A.; Rose, J.P.; Roberts, L.R.; Eckles, K. Social comparison, social media, and self-esteem. Psychol. Pop. Media Cult. 2014, 3, 206–222. [Google Scholar] [CrossRef]
- Boers, E.; Afzali, M.H.; Newton, N.; Conrod, P. Association of screen time and depression in adolescence. JAMA Pediatr. 2019, 173, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Turzyńska, D.; Szyndler, J.; Maciejak, P.; Sobolewska, A.; Płaźnik, A. Kwas kynureninowy–neuroprotekcyjna substancja w chorobach ośrodkowego układu nerwowego. Postępy Psychiatr. Neurol. 2010, 19, 61–72. [Google Scholar]
- Steardo, L., Jr.; Steardo, L.; Verkhratsky, A. Psychiatric face of COVID-19. Transl. Psychiatry 2020, 10, 261. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Nabi, G.; Zhang, T.; Yuefeng, W.; Dongming, L. Potential neurochemical and neuroendocrine effects of social distancing amidst the COVID-19 pandemic. Front. Endocrinol. 2020, 11, 582288. [Google Scholar] [CrossRef]
- Anderson, I.M.; Haddad, P.M.; Scott, J. Bipolar disorder. BMJ 2012, 345, e8508. [Google Scholar] [CrossRef] [PubMed]
- Alonso, J.; Petukhova, M.; Vilagut, G.; Chatterji, S.; Herringa, S.; Ustun, T.B.; Alhamzawi, A.O.; Viana, M.C.; Angermeyer, M.; Broment, E.; et al. Days out of role due to common physical and mental conditions: Results from the WHO World Mental Health surveys. Mol. Psychiatry 2011, 16, 1234–1246. [Google Scholar] [CrossRef] [PubMed]
- Bourne, C.; Aydemir, Ö.; Balanzá-Martínez, V.; Bora, E.; Brissos, S.; Cavangh, J.T.O.; Clark, L.; Cubukcuoglu, Z.; Dias, V.V.; Dittmann, S.; et al. Neuropsychological testing of cognitive impairment in euthymic bipolar disorder: An individual patient data meta-analysis. Acta Psychiatr. Scand. 2013, 128, 149–162. [Google Scholar] [CrossRef] [PubMed]
- Kurtz, M.M.; Gerraty, R.T. A meta-analytic investigation of neurocognitive deficits in bipolar illness: Profile and effects of clinical state. Neuropsychology 2009, 23, 551–562. [Google Scholar] [CrossRef]
- Gonda, X.; Pompili, M.; Serafini, G.; Montebovi, F.; Campi, S.; Dome, P.; Duleba, T.; Girardi, P.; Rihmer, Z. Suicidal behavior in bipolar disorder: Epidemiology, characteristics, and major risk factors. J. Affect. Disord. 2012, 143, 16–26. [Google Scholar] [CrossRef]
- Miller, J.N.; Black, D.W. Bipolar disorder and suicide: A Review. Curr. Psychiatry Rep. 2020, 22, 6. [Google Scholar] [CrossRef]
- Bidzińska, E.J. Stress factors in affective diseases. Br. J. Psychiatry 1984, 144, 161–166. [Google Scholar] [CrossRef]
- Ellicott, A.; Hammen, C.; Gitlin, M.; Brown, G.; Jamison, K. Life events and the course of bipolar disorder. Am. J. Psychiatry 1990, 147, 1194–1198. [Google Scholar] [CrossRef] [PubMed]
- Alloy, L.B.; Abramson, L.Y.; Urosevic, S.; Walshaw, P.D.; Nusslock, R.; Neeren, A.M. The psychosocial context of bipolar disorder: Environmental, cognitive, and developmental risk factors. Clin. Psychol. Rev. 2005, 25, 1043–1075. [Google Scholar] [CrossRef] [PubMed]
- Johanson, S.L. Life events in bipolar disorder: Towards more specific models. Clin. Psychol. Rev. 2005, 25, 1008–1027. [Google Scholar] [CrossRef] [Green Version]
- Johson, S.L.; Cueller, A.K.; Ruggero, C.; Winett-Perlman, C.; Goodnick, P.; White, R.; Miller, I. Life events as predictors of mania and depression in bipolar disorder. J. Abnorm. Psychol. 2008, 117, 268–277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, E.Y.; Miklowitz, D.J.; Biuckians, A.; Mullern, K. Life stress and the course of early-onset bipolar disorder. J. Affect. Disord. 2007, 99, 37–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leibenluft, E.; Albert, P.S.; Rosenthal, N.E.; Wehr, T.A. Relationship between sleep and mood in patients with rapid-cycling bipolar disorder. Psychiatry Res. 1996, 63, 161–168. [Google Scholar] [CrossRef]
- Rajkumar, R.P. Bipolar disorder, COVID-19, and the risk of relapse. Bipolar Disord. 2020, 22, 640. [Google Scholar] [CrossRef]
- Crowe, M.; Inder, M.; Swartz, H.A.; Murray, G.; Porter, R. Social rhythm therapy-A potentially translatable psychosocial intervention for bipolar disorder. Bipolar Disord. 2020, 22, 121–127. [Google Scholar] [CrossRef] [Green Version]
- Carta, M.G.; Ouali, U.; Perra, A.; Ben, A.; Ahmed, C.; Boe, L.; Aissa, A.; Lorrai, S.; Cossu, G.; Aresti, A.; et al. Living with bipolar disorder in the time of Covid-19: Biorhythms during the severe lockdown in Caligari, Italy, and the moderate lockdown in Tunis, Tunisia. Front. Psychiatry 2021, 12, 634765. [Google Scholar] [CrossRef]
- Nivoli, A.M.; Pacchiarotti, I.; Rosa, A.R.; Popovic, D.; Murru, A.; Valenti, M.; Bonnin, C.M.; Grande, I.; Sanchez-Moreno, J.; Vieta, E.; et al. Gender differences in a cohort study of 604 bipolar patients: The role of predominant polarity. J. Affect. Disord. 2011, 133, 443–449. [Google Scholar] [CrossRef]
- Arnold, J.G.; Martinez, C.; Zavala, J.; Prihoda, T.J.; Escamilla, M.; Singh, V.; Bazan, M.; Quinones, M.; Bowden, C.L. Investigating symptom domains of bipolar disorder for Spanish-speakers using the Bipolar Inventory of Symptoms Scale. J. Affect. Disord. 2016, 205, 239–244. [Google Scholar] [CrossRef]
- Williams, M.D.; Shah, N.D.; Wagie, A.E.; Wood, D.L.; Frye, M.A. Direct costs of bipolar disorder versus other chronic conditions:Aan employer-based health plan analysis. Psychiatr Serv. 2011, 62, 1073–1078. [Google Scholar] [CrossRef]
- Yocum, A.K.; Zhai, Y.; McInnis, M.G.; Han, P. COVID-19 pandemic, and lockdown impacts: A description in a longitudinal study of bipolar disorder. J. Affect. Disord. 2021, 282, 1226–1233. [Google Scholar] [CrossRef]
- Fellendorf, F.T.; Reininghaus, E.Z.; Ratzenhofer, M.; Lenger, M.; Maget, A.; Platzer, M.; Bengesser, S.A.; Birner, A.; Queissner, R.; Hamm, C.; et al. COVID-19-related fears and information frequency predict sleep behavior in bipolar disorder. Brain Behav. 2021, 11, e02182. [Google Scholar] [CrossRef]
- Sperling, J.D.; Dalkner, N.; Berndt, C.; Fleischmann, E.; Ratzenhofer, M.; Martini, J.; Pfennig, A.; Bauer, M.; Reininghaus, V.; Vinberg, M. Physical health profile and associated behavior during the COVID-19 pandemic in patients with bipolar disorder. Front Psychiatry 2021, 12, 759694. [Google Scholar] [CrossRef] [PubMed]
- Van, O.J.; Kenis, G.; Rutten, B.P.F. The environment and schizophrenia. Nature 2010, 468, 203–212. [Google Scholar] [CrossRef]
- Hajduk, A.; Owsianowska, J.; Robaszkiewicz, R.; Michałowska, Z. Ocena jakości życia pacjentów ze schizofrenią. Probl. Hig. Epidemiol. 2008, 89, 437–440. [Google Scholar]
- Janoutova, J.; Janackova, P.; Sery, O.; Zeman, T.; Ambroz, P.; Kovalova, M.; Varechova, K.; Hosak, L.; Jirik, V.; Janout, V. Epidemiology and risk factors of schizophrenia. Neuroendocrinol. Lett. 2016, 37, 1–8. [Google Scholar]
- Matuszczyk-Krupka, I.; Matuszczyk, M. Psychiatria. Podręcznik dla Studentów Pielęgniarstwa; Śląski Uniwersytet Medyczny: Katowice, Poland, 2007; p. 36. [Google Scholar]
- Tyszkowska, M.; Jarema, M. Między zdrowiem a schizofrenią. Psychiatr. Pol. 2013, 47, 587–597. [Google Scholar]
- Ahmad, T.; Mukhtiar, B.; Hui, J. Coronavirus Disease 2019 (COVID-19) pandemic and economic impact. Pak. J. Med. Sci. 2020, 36, S73–S78. [Google Scholar] [CrossRef]
- Hopper, M.W.; Napoles, A.M.; Perez-Stable, E.J. COVID-19 and racial/ethnic disparities. JAMA 2020, 323, 2466–2467. [Google Scholar] [CrossRef]
- Moreno, C.; Wykes, T.; Galderisi, S.; Nordentoft, M.; Crossley, N.; Jones, N.; Cannon, M.; Correll, C.U.; Byrne, L.; Carr, S.; et al. How mental health care should change as a consequence of the COVID-19 pandemic. Lancet Psychiatry 2020, 7, 813–824. [Google Scholar] [CrossRef]
- Maguire, P.A.; Looi, J.C. Vulnerability of people with schizophrenia to COVID-19. Aust. N. Z. J. Psychiatry 2020, 54, 1044. [Google Scholar] [CrossRef]
- Hjorthoj, C.; Sturup, A.E.; McGrath, J.J.; Nordentoft, M. Years of potential life lost and life expectancy in schizophrenia: A systematic review and meta-analysis. Lancet Psychiatry 2017, 4, 295–301. [Google Scholar] [CrossRef]
- Lehman, A.F. The effects of psychiatric symptoms on quality-of-life assessments among the chronic mentally ill. Eval. Program Plan. 1983, 6, 143–151. [Google Scholar] [CrossRef]
- Badura-Brzoza, K.; Piezga, M.; Błachut, M.; Ścisło, P.; Leksowska, A.; Gorczyca, P. Ocena wpływu wybranych czynników psychicznych i socjodemograficznych na jakość życia pacjentów ze schizofrenią. Psychiatr. Pol. 2012, 6, 975–984. [Google Scholar]
- Palomar-Ciria, N.; Blanco del Valle, P.; Hernandez-Las Heras, M.A.; Martinez-Gallardo, R. Schizophrenia and COVID-19 delirium. Psychiatry Res. 2020, 290, 1131137. [Google Scholar] [CrossRef]
- Fonseca, L.; Diniz, E.; Mendonca, G.; Malinowski, F.; Mari, J.; Gadelha, A. Schizophrenia and COVID-19: Risks and recommendations. Braz. J. Psychiatr. 2020, 42, 236–238. [Google Scholar] [CrossRef] [Green Version]
- Phillips, D.; Paul, G.; Fahy, M.; Dowling-Hetherington, L.; Kroll, T.; Duffy, C.; Fealy, G.; Lafferty, A. The invisible workforce during the COVID-19 pandemic: Family carers at the frontline. HRB Open Res. 2020, 3, 24. [Google Scholar] [CrossRef]
- Taquet, M.; Luciano, S.; Geddes, J.R.; Harrison, P.J. Bidirectional associations between COVID-19 and psychiatric disorder: Retrospective cohort studies of 62 354 COVID-19 cases in the USA. Lancet Psychiatry 2021, 8, 130–140. [Google Scholar] [CrossRef]
- Nemani, K.; Li, C.; Olfson, M.; Blessing, E.M.; Razavian, N.; Chen, J.; Petkova, E.; Goff, D.C. Association of psychiatric disorders with mortality among patients with COVID-19. JAMA Psychiatry 2021, 78, 380–386. [Google Scholar] [CrossRef]
- Bitan, D.T.; Krieger, I.; Kridin, K.; Komantscher, D.; Scheinman, Y.; Weinstein, O.; Cohen, A.D.; Cicurel, A.A.; Feingold, D. COVID-19 prevalence and mortality among schizophrenia patients: A large-scale retrospective cohort study. Schizophr. Bull. 2021, 47, 1211–1217. [Google Scholar] [CrossRef]
- Fond, G.; Pauly, V.; Leone, M.; Llorca, P.-M.; Orleans, V.; Loundou, A.; Lancon, C.; Auquier, P.; Baumstrack, K.; Boyer, L. Disparities in intensive care unit admission and mortality among patients with schizophrenia and COVID-19: A national cohort study. Schizophr. Bull. 2021, 47, 624–634. [Google Scholar] [CrossRef]
- Auquier, P.; Lancon, C.; Rouillon, F.; Lader, M.; Holmes, C. Mortality in schizophrenia. Pharm. Drug Saf. 2006, 15, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Tahira, A.C.; Verjovski-Almeida, S.; Ferreira, S.T. Dementia is an age–independent risk factor for severity and death in COVID-10 inpatients. Alzheimers Dement. 2021, 17, 1818–1831. [Google Scholar] [CrossRef]
- Abreu, W.; Tolson, D.; Jackson, G.A.; Staines, H.; Costa, N. The relationship between frailty, functional dependence, and healthcare needs among community-dwelling people with moderate to severe dementia. Health Soc. Care Community 2019, 27, 642–653. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Li, T.; Barbarino, P.; Gauthier, S.; Brodaty, H.; Molinuevo, J.L.; Xie, H.; Sun, Y.; Yu, E.; Tang, Y.; et al. Dementia care during COVID-19. Lancet 2020, 395, 1190–1191. [Google Scholar] [CrossRef]
- Miocci, M.; Gordon, A.L.; Allen, A.J.; Hocks, T.; Kierkegaard, P.; McLister, A.; Walne, S.; Hayward, G.; Buckle, P. COVID-19 testing in English care homes and implications for staff and residents. Age Ageing 2021, 50, 668–672. [Google Scholar] [CrossRef]
- Rada, A.G. COVID-19: The precarious position of Spain’s nursing homes. BMJ 2020, 369, m1554. [Google Scholar] [CrossRef] [Green Version]
- Bagchi, S.; Mak, J.; Li, Q.; Sheriff, E.; Mungai, E.; Anttila, A.; Soe, M.M.; Edwards, J.R.; Benin, A.L.; Pollock, D.A.; et al. Rates of COVID-19 among residents and staff members in nursing homes–United States. MMWR Morb. Mortal. Wkly. Rep. 2020, 70, 52–55. [Google Scholar] [CrossRef]
- Numbers, K.; Brodaty, H. The effects of the COVID-19 pandemic on people with dementia. Nat. Rev. Neurol. 2021, 17, 69–70. [Google Scholar] [CrossRef]
- Liu, N.; Sun, J.; Wang, X.; Zhao, M.; Huang, Q.; Li, H. The impact of dementia on the clinical outcome of COVID-19: A systematic review and meta-analysis. J. Alzheimers Dis. 2020, 78, 1775–1782. [Google Scholar] [CrossRef]
- Bonafè, M.; Prattichizzo, F.; Giuliani, A.; Storci, G.; Sabbatinelli, J.; Olivieri, F. Inflame-aging: Why older men are the most susceptible to SARS-CoV-2 complicated outcomes. Cytokine Growth Factor Rev. 2020, 53, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; He, W.; Yu, X.; Hu, D.; Bao, M.; Liu, H.; Zhou, J.; Jiang, H. Coronavirus disease 2019 in elderly patients: Characteristics and prognostic factors based on 4-week follow-up. J. Infect. 2020, 80, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Saragih, I.D.; Saragih, I.S.; Batubara, S.O.; Lin, C.-J. Dementia as a mortality predictor among older adults with COVID-19: A systematic review and meta-analysis of observational study. Geriatr. Nurs. 2021, 42, 1230–1239. [Google Scholar] [CrossRef]
- Atkins, J.L.; Masoli, J.A.H.; Delgado, J.; Pilling, L.C.; Kuo, C.L.; Kuchel, G.A.; Mezler, D. Preexisting comorbidities predicting severe COVID-19 in older adults in the UK biobank community cohort. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 2224–2230. [Google Scholar] [CrossRef]
- Perrotta, F.; Corbi, G.; Mazzeo, G.; Boccia, M.; Aronne, L.; D’Agnano, V.; Komici, K.; Mazzerella, G.; Parrella, R.; Bianco, A. COVID-19 and the elderly: Insights into pathogenesis and clinical decision-making. Aging Clin. Exp. Res. 2020, 32, 1599–1608. [Google Scholar] [CrossRef]
- Bauer, K.; Schwarzkopf, L.; Graessel, E.; Holle, R. A claims data-based comparison of comorbidity in individuals with and without dementia. BMC Geriatr. 2014, 14, 10. [Google Scholar] [CrossRef]
- Yamazaki, Y.; Zhao, N.; Caulfield, T.R.; Liu, C.C.; Bu, G. Apolipoprotein E and Alzheimer disease: Pathobiology and targeting strategies. Nat. Rev. Neurol. 2019, 15, 501–518. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Manis, M.; Long, J.; Wang, K.; Sullivan, P.M.; Remolina Serrano, J.; Hoyle, R.; Holtzman, D.M. Microglia drive APOE-dependent neurodegeneration in a tauopathy mouse model. J. Exp. Med. 2019, 216, 2546–2561. [Google Scholar] [CrossRef]
- Kuo, C.L.; Pilling, L.C.; Atkins, J.L.; Masoli, J.A.H.; Delgado, J.; Kuchel, G.A.; Mezler, D. APOE e4 genotype predicts severe COVID-19 in the UK biobank community cohort. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 2231–2232. [Google Scholar] [CrossRef]
- Lim, K.H.; Yang, S.; Kim, S.H.; Joo, J.Y. Elevation of ACE2 as a SARS-CoV-2 entry receptor gene expression in Alzheimer’s disease. J. Inf. Secur. 2020, 81, e33–e34. [Google Scholar] [CrossRef]
- Bacsu, J.D.R.; O’Connel, M.E.; Webster, C.; Poole, L.; Wighton, M.B.; Sivananthan, S. A scoping review of COVID-19 experiences of people living with dementia. Can. J. Public Health 2021, 112, 400–411. [Google Scholar] [CrossRef] [PubMed]
- Smaling, H.J.A.; Tillburgs, B.; Achterberg, W.P.; Visser, M. The impact of social distancing due to the COVID-19 pandemic on people with dementia, family careers and healthcare professionals: A qualitative study. Int. J. Environ. Res. Public Health 2022, 19, 519. [Google Scholar] [CrossRef]
- Lara, B.; Carnes, A.; Dakterzada, F.; Benitez, I.; Piñol-Ripoll, G. Neuropsychiatric symptoms, and quality of life in Spanish patients with Alzheimer’s disease during the COVID-19 lockdown. Eur. J. Neurol. 2020, 27, 1744–1747. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, C. How to support the quality of life of people living with cognitive disorders: A (k)new challenge in the post-COVID-19 world. Eur. J. Neurol. 2020, 27, 1742–1743. [Google Scholar] [CrossRef]
- Boutoleau-Bretonniere, C.; Pouclet, C.H.; Gillet, A.; Bernard, A.; Deruet, A.L.; Gouraud, I.; Mazoue, A.; Lamy, E.; Rocher, L.; Kapogiannis, D.; et al. The effects of confinement on neuropsychiatric symptoms in Alzheimer’s disease during the COVID-19 crisis. J. Alzheimers Dis. 2020, 76, 41–47. [Google Scholar] [CrossRef]
- Goodman-Casanova, J.M.; Dura-Perez, E.; Guzman-Parra, J.; Cuesta-Vargas, A.; Mayoral-Cleries, F. Telehealth home support during COVID-19 confinement for community-dwelling older adults with mild cognitive impairment or mild dementia: Survey study. J. Med. Internet Res. 2020, 22, e19434. [Google Scholar] [CrossRef]
- Ismail, I.I.; Kamel, W.A.; Al-Hashel, J.Y. Association of COVID-19 pandemic and rate of cognitive decline in patients with dementia and mild cognitive impairment: A cross-sectional study. Gerontol. Geriatr. Med. 2021, 7, 23337214211005223. [Google Scholar] [CrossRef]
- Ryoo, N.; Pyun, J.M.; Baek, M.J.; Suh, J.; Kand, M.J.; Wang, M.J.; Youn, Y.C.; Yang, D.W.; Kim, S.Y.; Park, Y.H.; et al. Coping with dementia in the middle of the COVID-19 pandemic. J. Korean Med. Sci. 2020, 35, e383. [Google Scholar] [CrossRef]
- Duggan, M.C.; Van, J.; Ely, E.W. Delirium assessment in critically ill older adults: Considerations during the COVID-19 pandemic. Crit. Care Clin. 2021, 37, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Karpur, A.; Vasudevan, V.; Shih, A.; Fraizer, T. Brief report: Impact of COVID-19 in individuals with autism spectrum disorders: Analysis of national provate claims insurance database. J. Autism Dev. Disord. 2021, 52, 2350–2356. [Google Scholar] [CrossRef]
- Irman, N.; Zashan, M.; Parvaiz, Z. Mental health considerations for children & adolescents in COVID-19 pandemic. Pak. J. Med. Sci. 2020, 36, S67–S72. [Google Scholar] [CrossRef]
- Mulaudzi, M. Developmental disabilities among children younger than 5 years in 195 countries and territories, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Glob. Health 2018, 6, 1100–1121. [Google Scholar] [CrossRef] [Green Version]
- de Bruin, E.I.; Ferdinand, R.F.; Meester, S.; de Nijs, P.F.A.; Verheij, F. High rates of psychiatric co-morbidity in PDD- NOS. J. Autism Dev. Disord. 2007, 37, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Merikangas, K.R.; Calkins, M.E.; Burstein, M.; He, J.-P.; Chiavacci, R.; Lateef, T.l.; Ruparel, K.; Gur, R.C.; Lenher, T.; Hakonarson, H.; et al. Comorbidity of physical and mental disorders in the neurodevelopmental genomics cohort study. Pediatrics 2015, 135, e927–e938. [Google Scholar] [CrossRef] [Green Version]
- Liu, N.; Zhang, F.; Wei, C.; Jia, Y.; Shang, Z.; Sun, L.; Wu, L.; Sun, Z.; Zhou, Y.; Wang, Y.; et al. Prevalence, and predictors of PTSS during COVID-19 outbreak in China hardest-hit areas: Gender differences matter. Psychiatry Res. 2020, 287, 112921. [Google Scholar] [CrossRef] [PubMed]
- Diaz, A.D.; Baweja, R. The role of neurotropism in psychiatric patients with COVID-19. Eur. Arch. Psychiatry Clin. Neurosci. 2020, 271, 385–386. [Google Scholar] [CrossRef]
- Ammerman, B.A.; Burke, T.A.; Jacobucci, R.; McClure, K. Preliminary investigation of the association between COVID-19 and suicidal thoughts and behaviors in the US. J. Psychiatr. Res. 2021, 134, 32–38. [Google Scholar] [CrossRef]
- Baweja, R.; Brown, S.L.; Edwards, E.M.; Murray, M.J. COVID-19 pandemic and impact on patients with autism spectrum disorder. J. Autism Dev. Disord. 2022, 52, 473–482. [Google Scholar] [CrossRef]
- Palgi, Y.; Amit, S.; Lia, R.; Ehud, B.; Sharon, A.; Yoav, B.; Cohen-Fridel, S.; Shoshi, K.; Yaakov, H. The loneliness pandemic: Loneliness and other concomitants of depression, anxiety and their comorbidity during the COVID-19 outbreak. J. Affect. Disord. 2020, 275, 109–111. [Google Scholar] [CrossRef]
- Wallace, G.L.; Kenworthy, L.; Pugliese, C.E.; Popal, H.S.; White, E.I.; Brodsky, E.; Martin, A. Real-world executive functions in adults with autism spectrum disorder: Profiles of impairment and associations with adaptive functioning and co-morbid anxiety and depression. J. Autism Dev. Disord. 2016, 46, 1071–1083. [Google Scholar] [CrossRef]
- Lequia, J.; Machalicek, W.; Rispoli, M.J. Effects of activity schedules on challenging behavior exhibited in children with autism spectrum disorders: A systematic review. Res. Autism Spectr. Disord. 2012, 6, 480–492. [Google Scholar] [CrossRef]
- Kawabe, K.; Hosokawa, R.; Nakachi, K.; Yoshino, A.; Horiuchi, F.; Ueno, S.I. Making brochure of Coronavirus disease (COVID-19) for children with autism spectrum disorder and their family members. Psychiatry Clin. Neurosci. 2020, 74, 498–499. [Google Scholar] [CrossRef]
- Kong, M. What COVID-19 means for non-neurotypical children and their families. Pediatr. Res. 2020, 89, 396–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eshraghi, A.A.; Li, C.; Alessandri, M.; Messingeer, D.S.; Eshraghi, R.S.; Mittal, R.; Armstrong, F.D. COVID-19: Overcoming the challenges faced by individuals with autism and their families. Lancet Psychiatry 2020, 7, 481–483. [Google Scholar] [CrossRef]
- Meral, B.F. Parental views of families of children with autism spectrum disorder and developmental disorders during the COVID-19 pandemic. J. Autism Dev. Disord. 2022, 52, 1712–1724. [Google Scholar] [CrossRef] [PubMed]
- Aishworiya, R.; Kang, Y.Q. Including children with developmental disabilities in the equation during this COVID-19 pandemic. J. Autism Dev. Disord. 2021, 51, 2155–2158. [Google Scholar] [CrossRef]
- Schieve, L.A.; Gonzalez, V.; Boulet, S.L.; Visser, S.N.; Rice, C.E.; Braun, K.V.N.; Boyle, C.A. Concurrent medical conditions and health care use and needs among children with learning and behavioral developmental disabilities, National Health Interview Survey, 2006–2010. Res. Dev. Disabil. 2012, 33, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y. A call for addressing barriers to telemedicine: Health disparities during the COVID-19 pandemic. Psychother. Psychosom. 2020, 90, 64–66. [Google Scholar] [CrossRef]
- Robillard, R.; Daros, A.R.; Phillips, J.L.; Porteous, M.; Saad, M.; Pennestri, M.H.; Kendzerska, T.; Edwards, J.D.; Solomonova, E.; Bhatla, R.; et al. Emerging new psychiatric symptoms and the worsening of pre-existing mental disorders during the COVID-19 pandemic: A Canadian multisite study: Nouveaux symptômes psychiatriques émergents et détérioration des troubles mentaux préexistants durant la pandémie de la COVID-19: Une étude canadienne multisite. Can. J. Psychiatry 2021, 66, 815–826. [Google Scholar] [CrossRef]
- Hao, F.; Tan, W.; Jiang, L.; Zhang, L.; Zhao, X.; Zou, Y.; Hu, Y.; Luo, X.; Jiang, X.; McIntyre, R.S.; et al. Do psychiatric patients experience more psychiatric symptoms during COVID-19 pandemic and lockdown? A case-control study with service and research implications for immunopsychiatry. Brain Behav. Immun. 2020, 87, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Fornaro, M.; De Prisco, M.; Billeci, M.; Ermini, E.; Young, A.H.; Lafer, B.; Soares, J.C.; Vieta, E.; Quevedo, J.; de Bartolomeis, A.; et al. Implications of the COVID-19 pandemic for people with bipolar disorders: A scoping review. J. Affect. Disord. 2021, 295, 740–751. [Google Scholar] [CrossRef]
- Smith, K.; Lambe, S.; Freeman, D.; Cipriani, A. COVID-19 vaccines, hesitancy, and mental health. Evid.-Based Ment. Health 2021, 24, 47–48. [Google Scholar] [CrossRef]
- De Picker, L.J.; Dias, M.C.; Benros, M.E.; Vai, B.; Branchi, I.; Benedetti, F.; Borsini, A.; Leza, J.C.; Kärkkäinen, H.; Männikkö, M.; et al. Severe mental illness, and European COVID-19 vaccination. Lancet Psychiatry 2021, 8, 356–359. [Google Scholar] [CrossRef]
- Stip, E.; Javaid, S.; Amiri, L. People with mental illness should be included in COVID-19 vaccination. Lancet Psychiatry 2021, 8, 275–276. [Google Scholar] [CrossRef]
- Mazereel, V.; Van Assche, K.; Detraux, J.; De Hert, M. COVID-19 vaccination for people with severe mental illness: Why, what, and how? Lancet Psychiatry 2021, 8, 444–450. [Google Scholar] [CrossRef]
- Payberah, E.; Payberah, D.; Sarangi, A.; Gude, J. COVID-19 vaccine hesitancy in patients with mental illness: Strategies to overcome barriers-a review. J. Egypt Public Health Assoc. 2022, 97, 5. [Google Scholar] [CrossRef]
- Pandolfo, G.; Genovese, G.; Iannuzzo, F.; Bruno, A.; Pioggia, G.; Gangemi, S. COVID-19 Vaccination and mental disorders, what has been accomplished and future direction. Brain Sci. 2022, 12, 292. [Google Scholar] [CrossRef]
- Ford, B.N.; Yolken, R.H.; Dickerson, F.B.; Teague, T.K.; Irwin, M.R.; Paulus, M.P.; Savitz, J. Reduced immunity to measles in adults with major depressive disorder. Psychol. Med. 2019, 49, 243–249. [Google Scholar] [CrossRef]
- Irwin, M.R.; Levin, M.J.; Laudenslager, M.L.; Olmstead, R.; Lucko, A.; Lang, N.; Carrillo, C.; Stanley, H.A.; Caulfield, M.J.; Weinberg, A.; et al. Varicella zoster virus-specific immune responses to a herpes zoster vaccine in elderly recipients with major depression and the impact of antidepressant medications. Clin. Infect. Dis. 2013, 56, 1085–1093. [Google Scholar] [CrossRef]
- Segerstrom, S.C.; Hardy, J.K.; Evans, D.R.; Greenberg, R.N. Vulnerability, distress, and immune response to vaccination in older adults. Brain Behav. Immun. 2012, 26, 747–753. [Google Scholar] [CrossRef] [Green Version]
- Russo, R.; Ciminale, M.; Ditommaso, S.; Siliquini, R.; Zotti, C.; Ruggenini, A.M. Hepatitis B vaccination in psychiatric patients. Lancet 1994, 343, 356. [Google Scholar] [CrossRef]
- Więdłocha, M.; Marcinowicz, P.; Krupa, R.; Janoska-Jaździk, M.; Janus, M.; Dębowska, W.; Mosiołek, A.; Waszkiewicz, N.; Szulc, A. Effect of antidepressant treatment on peripheral inflammation markers-A meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 2018, 80, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Curtis, H.J.; Inglesby, P.; Morton, C.E.; MacKenna, B.; Green, A.; Hulme, W.; Walker, A.J.; Morley, J.; Mehrkar, A.; Bacon, S.; et al. Trends and clinical characteristics of COVID-19 vaccine recipients: A federated analysis of 57.9 million patients’ primary care records in situ using OpenSAFELY. Br. J. Gen. Pract. 2022, 72, e51–e62. [Google Scholar] [CrossRef] [PubMed]
- Bitan, D.t. Patients with schizophrenia are under-vaccinated for COVID-19: A report from Israel. World Psychiatry 2021, 20, 300–301. [Google Scholar] [CrossRef] [PubMed]
- Bitan, D.t.; Kridin, K.; Cohen, A.D.; Weinstein, O. COVID-19 hospitalization, mortality, vaccination, and postvaccination trends among people with schizophrenia in Israel: A longitudinal cohort study. Lancet Psychiatry 2021, 8, 901–908. [Google Scholar] [CrossRef]
- Lord, O.; Malone, D.; Mitchell, A.J. Receipt of preventive medical care and medical screening for patients with mental illness: A comparative analysis. Gen. Hosp. Psychiatry 2010, 32, 519–543. [Google Scholar] [CrossRef]
- Miles, L.W.; Williams, N.; Luthy, K.E.; Eden, L. Adult vaccination rates in the mentally Ill population: An outpatient improvement project. J. Am. Psychiatr. Nurses Assoc. 2020, 26, 172–180. [Google Scholar] [CrossRef] [Green Version]
- Hao, F.; Wang, B.; Tan, W.; Husain, S.F.; McIntyre, R.S.; Tang, X.; Zhang, L.; Han, X.; Jiang, L.; Chew, N.W.S.; et al. Attitudes toward COVID-19 vaccination and willingness to pay comparison of people with and without mental disorders in China. BJPsych. Open 2021, 7, e146. [Google Scholar] [CrossRef]
- Jefsen, O.H.; Kølbæk, P.; Gil, Y.; Speed, M.; Dinesen, P.T.; Sønderskov, K.M.; Østergaard, S.D. COVID-19 vaccine willingness amongst patients with mental illness compared with the general population. Acta Neuropsychiatr. 2021, 33, 273–276. [Google Scholar] [CrossRef]
- Ramirez, A.V.; Ojeaga, M.; Espinoza, V.; Hansler, B.; Honrubia, V. Telemedicine in minority and socioeconomically disadvantaged communities amidst COVID-19 pandemic. Otolaryngol. Head Neck Surg. 2021, 164, 91–92. [Google Scholar] [CrossRef]
- Park, H.Y.; Park, W.B.; Lee, S.H.; Kim, J.L.; Lee, J.J.; Lee, H.; Shin, H.S. Posttraumatic stress disorder and depression of survivors 12 months after the outbreak of Middle East respiratory syndrome in South Korea. BMC Public Health 2020, 20, 605. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, N.S.; Chung, C.H.; Chang, C.C.; Chang, H.A.; Kao, Y.C.; Chang, S.Y.; Chien, W.C. What could we learn from SARS when facing the mental health issues related to the COVID-19 outbreak? A nationwide cohort study in Taiwan. Transl. Psychiatry 2020, 10, 339. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lange, K.W. Coronavirus disease 2019 (COVID-19) and global mental health. Glob. Health J. 2021, 5, 31–36. [Google Scholar] [CrossRef]
- Daly, M.; Sutin, A.R.; Robinson, E. Longitudinal changes in mental health and the COVID-19 pandemic: Evidence from the UK Household longitudinal study. Psychol. Med. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.; Ayling, K.; Chalder, T.; Massey, A.; Broadbent, E.; Coupland, C.; Vedhara, K. Mental health in the UK during the COVID-19 pandemic: Cross-sectional analyses from a community cohort study. BMJ Open 2020, 10, e040620. [Google Scholar] [CrossRef] [PubMed]
- Bacaro, V.; Chiabudini, M.; Buonanno, C.; De Bartolo, P.; Riemann, D.; Mancini, F.; Baglioni, C. Insomnia in the Italian population during Covid-19 outbreak: A snapshot on one major risk factor for depression and anxiety. Front. Psychiatry 2020, 11, 579107. [Google Scholar] [CrossRef]
- Paniz-Mondolfi, A.; Bryce, C.; Grimes, Z.; Gordon, R.E.; Reidy, J.; Lednicky, J.; Sordillo, E.M.; Fowkesm, M. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). J. Med. Virol. 2020, 92, 699–702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cespuglio, R.; Strekalova, T.; Spencer, P.S.; Roman, G.C.; Reis, J.; Bouteille, B.; Buguet, A. SARS-CoV-2 infection and sleep disturbances: Nitric oxide involvement and therapeutic opportunity. Sleep 2021, 44, zsab009. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Markiewicz-Gospodarek, A.; Górska, A.; Markiewicz, R.; Chilimoniuk, Z.; Czeczelewski, M.; Baj, J.; Maciejewski, R.; Masiak, J. The Relationship between Mental Disorders and the COVID-19 Pandemic—Course, Risk Factors, and Potential Consequences. Int. J. Environ. Res. Public Health 2022, 19, 9573. https://doi.org/10.3390/ijerph19159573
Markiewicz-Gospodarek A, Górska A, Markiewicz R, Chilimoniuk Z, Czeczelewski M, Baj J, Maciejewski R, Masiak J. The Relationship between Mental Disorders and the COVID-19 Pandemic—Course, Risk Factors, and Potential Consequences. International Journal of Environmental Research and Public Health. 2022; 19(15):9573. https://doi.org/10.3390/ijerph19159573
Chicago/Turabian StyleMarkiewicz-Gospodarek, Agnieszka, Aleksandra Górska, Renata Markiewicz, Zuzanna Chilimoniuk, Marcin Czeczelewski, Jacek Baj, Ryszard Maciejewski, and Jolanta Masiak. 2022. "The Relationship between Mental Disorders and the COVID-19 Pandemic—Course, Risk Factors, and Potential Consequences" International Journal of Environmental Research and Public Health 19, no. 15: 9573. https://doi.org/10.3390/ijerph19159573
APA StyleMarkiewicz-Gospodarek, A., Górska, A., Markiewicz, R., Chilimoniuk, Z., Czeczelewski, M., Baj, J., Maciejewski, R., & Masiak, J. (2022). The Relationship between Mental Disorders and the COVID-19 Pandemic—Course, Risk Factors, and Potential Consequences. International Journal of Environmental Research and Public Health, 19(15), 9573. https://doi.org/10.3390/ijerph19159573