Ambient Air Pollution and Risk of Admission Due to Asthma in the Three Largest Urban Agglomerations in Poland: A Time-Stratified, Case-Crossover Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Setting
2.2. Definition and Measurements
2.3. Statistical Analysis
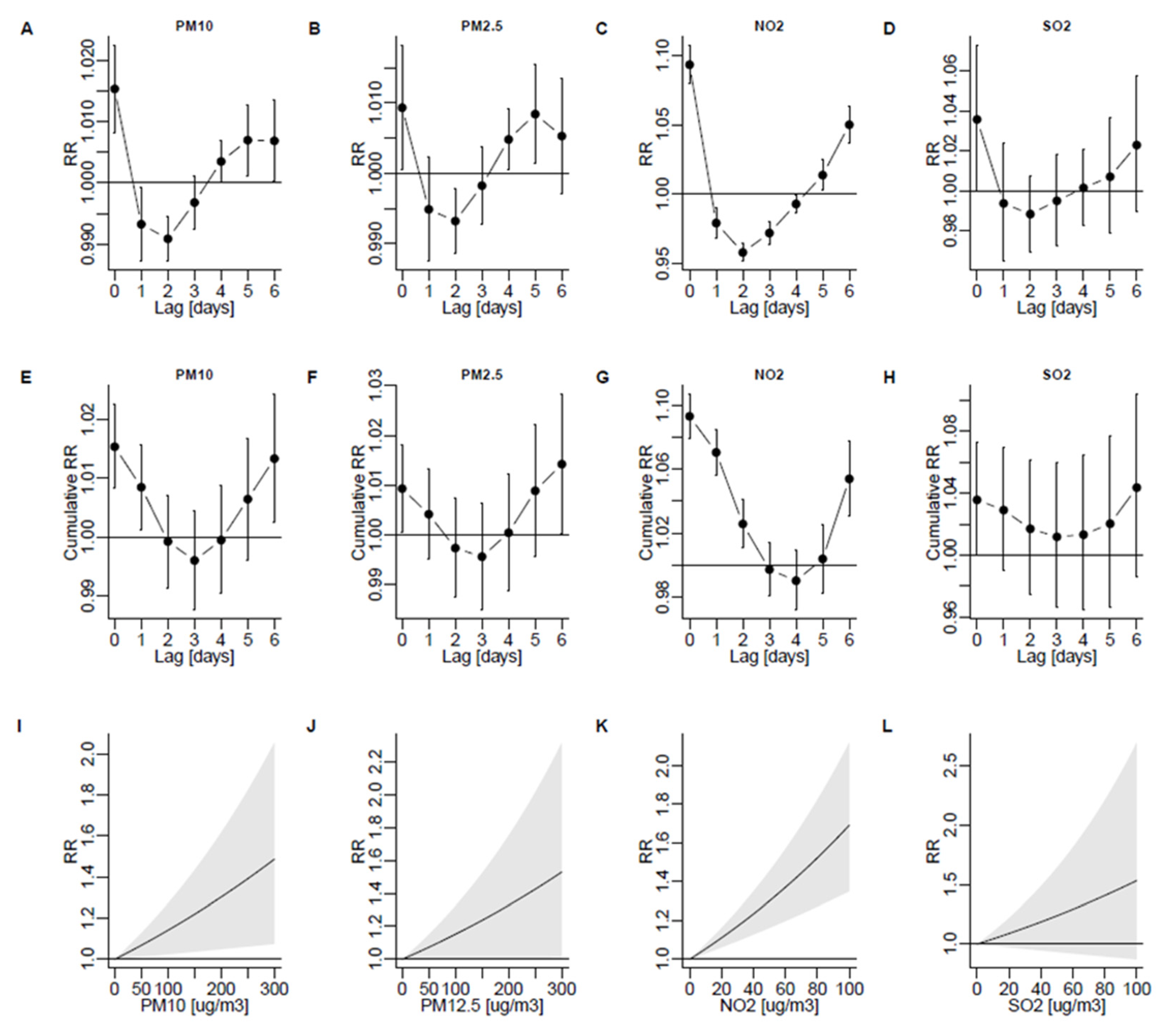
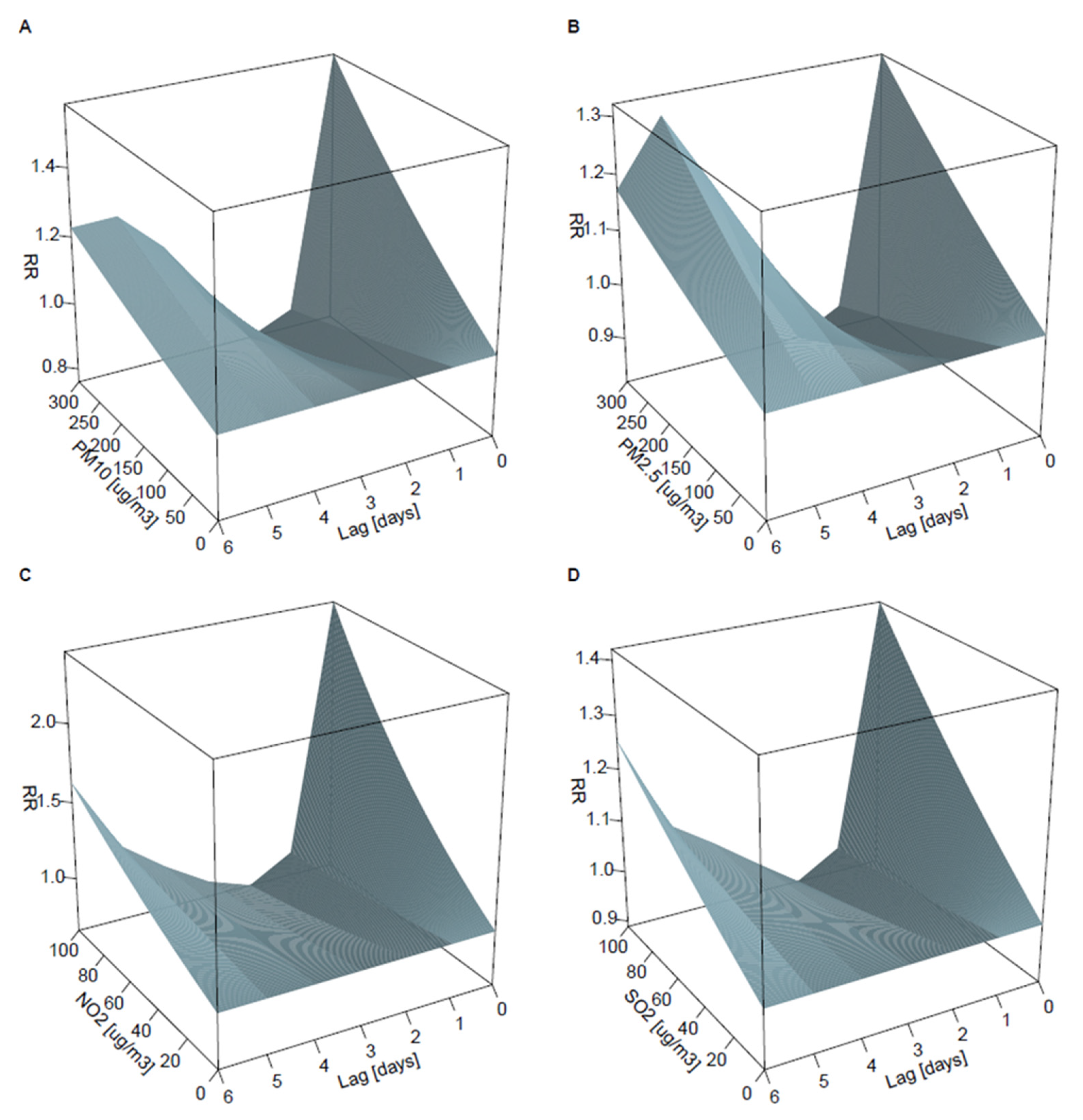
3. Results
3.1. Air Pollution and Asthma Hospitalizations
3.2. Lagged Associations between PM10, PM2.5, NO2, and SO2 and the Risk of Admission
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vos, T.; Allen, C.; Arora, M.; Barber, R.M.; Bhutta, Z.A.; Brown, A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef] [Green Version]
- The Global Asthma Report 2018; Global Asthma Network: Auckland, New Zealand, 2018; ISBN 978-0-473-46524-7.
- Papi, A.; Brightling, C.; Pedersen, S.E.; Reddel, H.K. Asthma. Lancet 2018, 391, 783–800. [Google Scholar] [CrossRef]
- Yang, J.F.; Chaudhuri, R.; Thomson, N.C.; Ramparsad, N.; O’Pray, H.; Barclay, S.; MacBride-Stewart, S.; McCallum, C.; Sharma, V.; McSharry, C.; et al. Insights into frequent asthma exacerbations from a primary care perspective and the implications of UK National Review of Asthma Deaths recommendations. NPJ Prim. Care Respir. Med. 2018, 28, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DiMango, E.; Rogers, L.; Reibman, J.; Gerald, L.B.; Brown, M.; Sugar, E.A.; Henderson, R.; Holbrook, J.T. Risk Factors for Asthma Exacerbation and Treatment Failure in Adults and Adolescents with Well-controlled Asthma during Continuation and Step-Down Therapy. Ann. Am. Thorac. Soc. 2018, 15, 955–961. [Google Scholar] [CrossRef]
- Suruki, R.Y.; Daugherty, J.B.; Boudiaf, N.; Albers, F.C. The frequency of asthma exacerbations and healthcare utilization in patients with asthma from the UK and USA. BMC Pulm. Med. 2017, 17, 74. [Google Scholar] [CrossRef] [Green Version]
- Guarnieri, M.; Balmes, J.R. Outdoor air pollution and asthma. Lancet 2014, 383, 1581–1592. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Yang, X.; Fan, F.; Hu, Y.; Wang, X.; Zhu, S.; Ren, G.; Wang, G. Outdoor air pollution and the risk of asthma exacerbations in single lag0 and lag1 exposure patterns: A systematic review and meta-analysis. J. Asthma 2021, 28, 1–18. [Google Scholar] [CrossRef]
- World Bank Poland Air Quality Management—Poland Final Report. Available online: https://openknowledge.worldbank.org/bitstream/handle/10986/31531/Air-Quality-Management-in-Poland.pdf (accessed on 28 April 2022).
- Badyda, A.J. Zrodla zanieczyszczen powietrza. Najważniejsze szkodliwe substancje spotykane w atmosferze. In Smog. Konsekwencje Zdrowotne Zanieczyszczeń Powietrza; Mazurek, H., Badyda, A.J., Eds.; PZWL Wydawnictwo Lekarskie: Warsaw, Poland, 2018; pp. 17–30. ISBN 978-83-200-5575-7. [Google Scholar]
- Kowalska, M.; Skrzypek, M.; Kowalski, M.; Cyrys, J. Effect of NOx and NO2 Concentration Increase in Ambient Air to Daily Bronchitis and Asthma Exacerbation, Silesian Voivodeship in Poland. Int. J. Environ. Res. Public Health 2020, 17, 754. [Google Scholar] [CrossRef] [Green Version]
- Nazar, W.; Niedoszytko, M. Air Pollution in Poland: A 2022 Narrative Review with Focus on Respiratory Diseases. Int. J. Environ. Res. Public Health 2022, 19, 895. [Google Scholar] [CrossRef]
- Armstrong, B.G.; Gasparrini, A.; Tobias, A. Conditional Poisson models: A flexible alternative to conditional logistic case cross-over analysis. BMC Med. Res. Methodol. 2014, 14, 122. [Google Scholar] [CrossRef] [Green Version]
- Ragettli, M.S.; Vicedo-Cabrera, A.M.; Schindler, C.; Röösli, M. Exploring the association between heat and mortality in Switzerland between 1995 and 2013. Environ. Res. 2017, 158, 703–709. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.; Peng, J.; Liu, P.; Duan, Y.; Huang, S.; Wen, Y.; Liao, Y.; Li, H.; Yan, S.; Cheng, J.; et al. Association between short-term exposure to air pollution and ischemic stroke onset: A time-stratified case-crossover analysis using a distributed lag nonlinear model in Shenzhen, China. Environ. Health 2020, 19, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasparrini, A. Modeling exposure–lag–response associations with distributed lag non-linear models. Stat. Med. 2014, 33, 881–899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasparrini, A. Distributed Lag Linear and Non-Linear Models in R: The Package dlnm. J. Stat. Softw. 2011, 43, 1–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gasparrini, A.; Leone, M. Attributable risk from distributed lag models. BMC Med. Res. Methodol. 2014, 14, 55. [Google Scholar] [CrossRef] [Green Version]
- Lu, P.; Zhang, Y.; Lin, J.; Xia, G.; Zhang, W.; Knibbs, L.D.; Morgan, G.G.; Jalaludin, B.; Marks, G.; Abramson, M.; et al. Multi-city study on air pollution and hospital outpatient visits for asthma in China. Environ. Pollut. 2020, 257, 113638. [Google Scholar] [CrossRef]
- Ko, F.W.S.; Tam, W.; Wong, T.W.; Lai, C.K.W.; Wong, G.W.K.; Leung, T.-F.; Ng, S.S.S.; Hui, D.S.C. Effects of air pollution on asthma hospitalization rates in different age groups in Hong Kong. Clin. Exp. Allergy 2007, 37, 1312–1319. [Google Scholar] [CrossRef]
- Liu, F.; Qu, F.; Zhang, H.; Chao, L.; Li, R.; Yu, F.; Guan, J.; Yan, X. The effect and burden modification of heating on adult asthma hospitalizations in Shijiazhuang: A time-series analysis. Respir. Res. 2019, 20, 122. [Google Scholar] [CrossRef] [Green Version]
- Sunyer, J.; Spix, C.; Quenel, P.; Ponce-de-Leon, A.; Ponka, A.; Barumandzadeh, T.; Touloumi, G.; Bacharova, L.; Wojtyniak, B.; Vonk, J.; et al. Urban air pollution and emergency admissions for asthma in four European cities: The APHEA Project. Thorax 1997, 52, 760–765. [Google Scholar] [CrossRef] [Green Version]
- Zheng, X.; Ding, H.; Jiang, L.; Chen, S.; Zheng, J.; Qiu, M.; Zhou, Y.; Chen, Q.; Guan, W. Association between Air Pollutants and Asthma Emergency Room Visits and Hospital Admissions in Time Series Studies: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0138146. [Google Scholar] [CrossRef]
- Cai, J.; Zhao, A.; Zhao, J.; Chen, R.; Wang, W.; Ha, S.; Xu, X.; Kan, H. Acute effects of air pollution on asthma hospitalization in Shanghai, China. Environ. Pollut. 2014, 191, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, Z.; Darvishi Boloorani, A.; Khalifeh, R.; Teymouri, P.; Mesdaghinia, A.; Griffin, D.W. Air pollution and respiratory hospital admissions in Shiraz, Iran, 2009 to 2015. Atmos. Environ. 2019, 209, 233–239. [Google Scholar] [CrossRef]
- Krasnov, H.; Katra, I.; Koutrakis, P.; Friger, M.D. Contribution of dust storms to PM 10 levels in an urban arid environment. J. Air Waste Manag. Assoc. 2014, 64, 89–94. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galán, I.; Tobías, A.; Banegas, J.R.; Aránguez, E. Short-term effects of air pollution on daily asthma emergency room admissions. Eur. Respir. J. 2003, 22, 802–808. [Google Scholar] [CrossRef]
- Villeneuve, P.J.; Chen, L.; Rowe, B.H.; Coates, F. Outdoor air pollution and emergency department visits for asthma among children and adults: A case-crossover study in northern Alberta, Canada. Environ. Health 2007, 6, 40. [Google Scholar] [CrossRef] [Green Version]
- Basagaña, X.; Barrera-Gómez, J. Reflection on modern methods: Visualizing the effects of collinearity in distributed lag models. Int. J. Epidemiol. 2021, 51, 334–344. [Google Scholar] [CrossRef]
- Rabl, A. Air Pollution Mortality: Harvesting And Loss Of Life Expectancy. J. Toxicol. Environ. Health Part A 2005, 68, 1175–1180. [Google Scholar] [CrossRef]
- Guo, H.; Chen, M. Short-term effect of air pollution on asthma patient visits in Shanghai area and assessment of economic costs. Ecotoxicol. Environ. Saf. 2018, 161, 184–189. [Google Scholar] [CrossRef]
- Nadali, A.; Leili, M.; Karami, M.; Bahrami, A.; Afkhami, A. The short-term association between air pollution and asthma hospitalization: A time-series analysis. Air Qual. Atmos. Health 2021, 1–15. [Google Scholar] [CrossRef]
- AlBalawi, S.M.; Namdeo, A.; Hodgson, S.; Pless-Mulloli, T.; McNally, R.J.Q. Short-term effects of air pollution on daily asthma-related emergency department visits in an industrial city. J. Public Health 2021, 43, e45–e53. [Google Scholar] [CrossRef]
- Çapraz, Ö.; Deniz, A. Assessment of hospitalizations from asthma, chronic obstructive pulmonary disease and acute bronchitis in relation to air pollution in İstanbul, Turkey. Sustain. Cities Soc. 2021, 72, 103040. [Google Scholar] [CrossRef]
- Lee, S.W.; Yon, D.K.; James, C.C.; Lee, S.; Koh, H.Y.; Sheen, Y.H.; Oh, J.-W.; Han, M.Y.; Sugihara, G. Short-term effects of multiple outdoor environmental factors on risk of asthma exacerbations: Age-stratified time-series analysis. J. Allergy Clin. Immunol. 2019, 144, 1542–1550.e1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ratajczak, A.; Badyda, A.; Czechowski, P.O.; Czarnecki, A.; Dubrawski, M.; Feleszko, W. Air Pollution Increases the Incidence of Upper Respiratory Tract Symptoms among Polish Children. J. Clin. Med. 2021, 10, 2150. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowiecki, P.; Adamkiewicz, Ł.; Mucha, D.; Czechowski, P.O.; Soliński, M.; Chciałowski, A.; Badyda, A. Impact of Air Pollution on Lung Function among Preadolescent Children in Two Cities in Poland. J. Clin. Med. 2021, 10, 2375. [Google Scholar] [CrossRef]
- Institute of Environmental Protection Sources of Emissions of SO2, NOX, CO, NH3, NMVOCs, Particulate Matter, Heavy Metals, and POPs. Available online: https://www.kobize.pl/uploads/materialy/Inwentaryzacje_krajowe/2015/Bilansemisji—raportpodstawowy_2013.pdf (accessed on 19 January 2022).
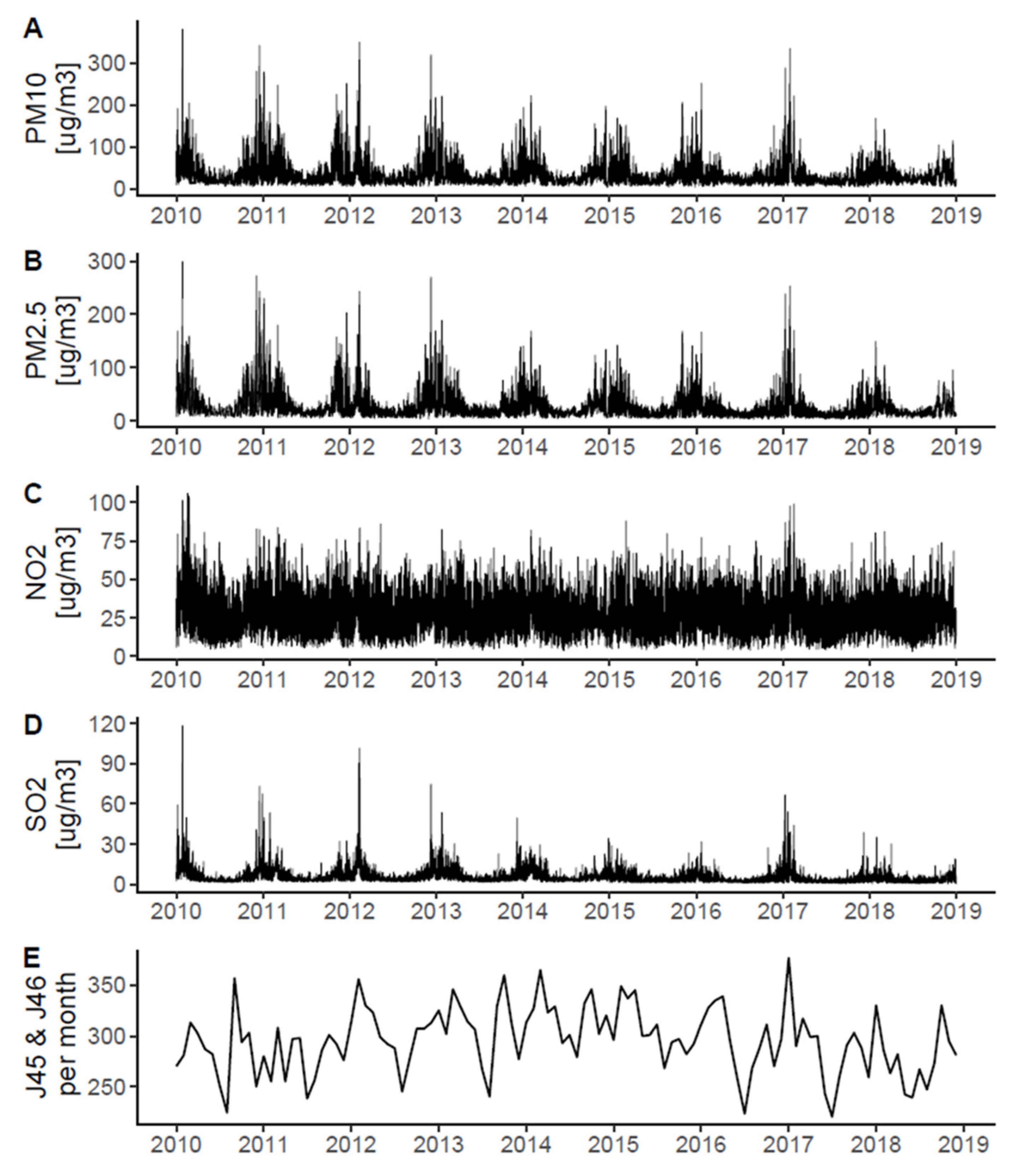
| Warsaw | Cracow | Tricity | |
|---|---|---|---|
| PM10, μg/m3, median (IQR) | 28.4 (20.7–40.3) | 36.3 (24.4–60.0) | 15.5 (11.2–22.6) |
| PM2.5, μg/m3, median (IQR) | 20.6 (14.5–31.9) | 26.0 (17.1–46.5) | 11.5 (7.87–17.6) |
| NO2, μg/m3, median (IQR) | 33.6 (26.1–41.4) | 40.8 (33.6–49.0) | 13.8 (9.69–19.1) |
| SO2, μg/m3, median (IQR) | 4.56 (2.94–7.12) | 5.63 (4.02) | 2.61 (1.84–3.92) |
| Temperature, °C, median (IQR) | 9.58 (2.90–16.9) | 9.90 (2.95–16.6) | 9.29 (3.71–5.9) |
| Relative humidity, %, median (IQR) | 77.5 (66.1–87.1) | 78.2 (68.6–86.2) | 74.9 (67.6–80.7) |
| Atmospheric pressure, hPa, median (IQR) | 1003 (998–1008) | 988 (983–992) | 1007 (1001–1012) |
| J45 and J46 admissions, n | 17,015 | 8062 | 6842 |
| J45 and J46 admissions per million, n | 9452 | 10,470 | 9122 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dąbrowiecki, P.; Chciałowski, A.; Dąbrowiecka, A.; Badyda, A. Ambient Air Pollution and Risk of Admission Due to Asthma in the Three Largest Urban Agglomerations in Poland: A Time-Stratified, Case-Crossover Study. Int. J. Environ. Res. Public Health 2022, 19, 5988. https://doi.org/10.3390/ijerph19105988
Dąbrowiecki P, Chciałowski A, Dąbrowiecka A, Badyda A. Ambient Air Pollution and Risk of Admission Due to Asthma in the Three Largest Urban Agglomerations in Poland: A Time-Stratified, Case-Crossover Study. International Journal of Environmental Research and Public Health. 2022; 19(10):5988. https://doi.org/10.3390/ijerph19105988
Chicago/Turabian StyleDąbrowiecki, Piotr, Andrzej Chciałowski, Agata Dąbrowiecka, and Artur Badyda. 2022. "Ambient Air Pollution and Risk of Admission Due to Asthma in the Three Largest Urban Agglomerations in Poland: A Time-Stratified, Case-Crossover Study" International Journal of Environmental Research and Public Health 19, no. 10: 5988. https://doi.org/10.3390/ijerph19105988
APA StyleDąbrowiecki, P., Chciałowski, A., Dąbrowiecka, A., & Badyda, A. (2022). Ambient Air Pollution and Risk of Admission Due to Asthma in the Three Largest Urban Agglomerations in Poland: A Time-Stratified, Case-Crossover Study. International Journal of Environmental Research and Public Health, 19(10), 5988. https://doi.org/10.3390/ijerph19105988







