Anti-Müllerian Hormone Levels among Female Firefighters
Abstract
:1. Introduction
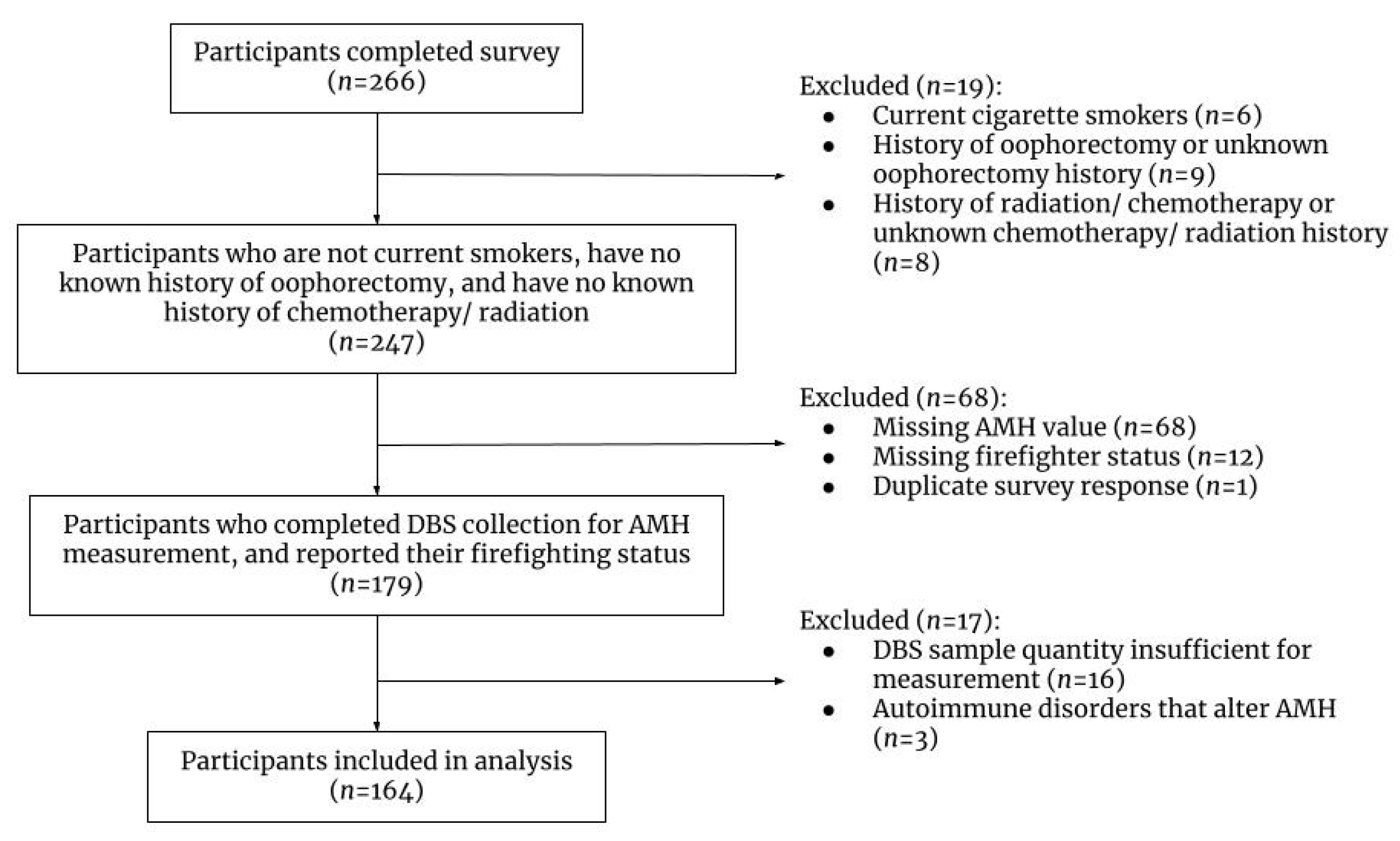
2. Materials and Methods
2.1. Study Design and Recruitment
2.2. Measures
2.2.1. Exposure
2.2.2. Outcome
2.2.3. Other Covariates
2.3. Statistical Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mendola, P.; Sundaram, R.; Louis, G.M.B.; Sun, L.; Wallace, M.E.; Smarr, M.M.; Sherman, S.; Zhu, Y.; Ying, Q.; Liu, D. Proximity to major roadways and prospectively-measured time-to-pregnancy and infertility. Sci. Total Environ. 2017, 576, 172–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whitcomb, B.W.; Purdue-Smithe, A.; Szegda, K.L.; Boutot, M.E.; Hankinson, S.E.; Manson, J.E.; Rosner, B.; Willett, W.C.; Eliassen, A.H.; Bertone-Johnson, E.R. Cigarette Smoking and Risk of Early Natural Menopause. Am. J. Epidemiol. 2017, 187, 696–704. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, A.J.; Keller, J.P.; Zhao, S.; Carroll, R.; Kaufman, J.D.; Sandler, D.P. Air Pollution, Clustering of Particulate Matter Components, and Breast Cancer in the Sister Study: A U.S.-Wide Cohort. Environ. Health Perspect. 2019, 127, 107002. [Google Scholar] [CrossRef] [PubMed]
- Jahnke, S.A.; Poston, W.S.C.; Jitnarin, N.; Haddock, C.K. Maternal and Child Health Among Female Firefighters in the U.S. Matern. Child Health J. 2018, 22, 922–931. [Google Scholar] [CrossRef] [PubMed]
- McDiarmid, M.A.; Lees, P.S.; Agnew, J.; Midzenski, M.; Duffy, R. Reproductive hazards of fire fighting. II. Chemical hazards. Am. J. Ind. Med. 1991, 19, 447–472. [Google Scholar] [CrossRef] [PubMed]
- Agnew, J.; McDiarmid, M.A.; Lees, P.S.; Duffy, R. Reproductive hazards of fire fighting. I. Non-chemical hazards. Am. J. Ind. Med. 1991, 19, 433–445. [Google Scholar] [CrossRef]
- Amani, R.; Gill, T. Shiftworking, nutrition and obesity: Implications for workforce health- a systematic review. Asia Pac. J. Clin. Nutr. 2013, 22, 505–515. [Google Scholar] [CrossRef]
- Mahoney, M.M. Shift work, jet lag, and female reproduction. Int. J. Endocrinol. 2010, 2010, 813764. [Google Scholar] [CrossRef]
- Nurminen, T. Shift work and reproductive health. Scand. J. Work Environ. Health 1998, 24 (Suppl. S3), 28–34. [Google Scholar]
- Puttonen, S.; Harma, M.; Hublin, C. Shift work and cardiovascular disease—Pathways from circadian stress to morbidity. Scand. J. Work Environ. Health 2010, 36, 96–108. [Google Scholar] [CrossRef] [Green Version]
- Jung, A.M.; Jahnke, S.A.; Dennis, L.K.; Bell, M.L.; Burgess, J.L.; Jitnarin, N.; Kaipust, C.M.; Farland, L.V. Occupational factors and miscarriages in the US fire service: A cross-sectional analysis of women firefighters. Environ. Health 2021, 20, 116. [Google Scholar] [CrossRef] [PubMed]
- Weenen, C.; Laven, J.S.; Von Bergh, A.R.; Cranfield, M.; Groome, N.P.; Visser, J.A.; Kramer, P.; Fauser, B.C.J.M.; Themmen, A.P.N. Anti-Mullerian hormone expression pattern in the human ovary: Potential implications for initial and cyclic follicle recruitment. Mol. Hum. Reprod. 2004, 10, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Lie Fong, S.; Visser, J.A.; Welt, C.K.; de Rijke, Y.B.; Eijkemans, M.J.; Broekmans, F.J.; Roes, E.M.; Peters, W.H.M.; Hokken-Koelega, A.C.S.; Fauser, B.C.J.M.; et al. Serum anti-mullerian hormone levels in healthy females: A nomogram ranging from infancy to adulthood. J. Clin. Endocrinol. Metab. 2012, 97, 4650–4655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seifer, D.B.; Baker, V.L.; Leader, B. Age-specific serum anti-Müllerian hormone values for 17,120 women presenting to fertility centers within the United States. Fertil. Steril. 2011, 95, 747–750. [Google Scholar] [CrossRef] [PubMed]
- de Kat, A.C.; van der Schouw, Y.T.; Eijkemans, M.J.; Herber-Gast, G.C.; Visser, J.A.; Verschuren, W.M.; Broekmans, F.J.M. Back to the basics of ovarian aging: A population-based study on longitudinal anti-Mullerian hormone decline. BMC Med. 2016, 14, 151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baird, D.D.; Steiner, A.Z. Anti-Mullerian Hormone: A Potential New Tool in Epidemiologic Studies of Female Fecundability. Am. J. Epidemiol. 2012, 175, 245–249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Plante, B.J.; Cooper, G.S.; Baird, D.; Steiner, A.Z. The impact of smoking on antimüllerian hormone levels in women aged 38 to 50 years. Menopause 2010, 17, 571–576. [Google Scholar] [CrossRef] [Green Version]
- White, A.J.; Sandler, D.P.; D’Aloisio, A.A.; Stanczyk, F.; Whitworth, K.W.; Baird, D.D.; Nichols, H.B. Antimüllerian hormone in relation to tobacco and marijuana use and sources of indoor heating/cooking. Fertil. Steril. 2016, 106, 723–730. [Google Scholar] [CrossRef] [Green Version]
- Whitworth, K.W.; Baird, D.D.; Steiner, A.Z.; Bornman, R.M.S.; Travlos, G.S.; Wilson, R.E.; Longnecker, M.P. Anti-Müllerian Hormone and Lifestyle, Reproductive, and Environmental Factors Among Women in Rural South Africa. Epidemiology 2015, 26, 429–435. [Google Scholar] [CrossRef] [Green Version]
- Sadeu, J.; Foster, W. Effect of in vitro exposure to benzo[a]pyrene, a component of cigarette smoke, on folliculogenesis, steroidogenesis and oocyte nuclear maturation. Reprod. Toxicol. 2011, 31, 402–408. [Google Scholar] [CrossRef]
- Gai, H.-F.; An, J.-X.; Qian, X.-Y.; Wei, Y.-J.; Williams, J.P.; Gao, G.-L. Ovarian Damages Produced by Aerosolized Fine Particulate Matter (PM2.5) Pollution in Mice. Chin. Med. J. 2017, 130, 1400–1410. [Google Scholar] [CrossRef] [PubMed]
- Kales, S.N.; Soteriades, E.S.; Christophi, C.A.; Christiani, D.C. Emergency duties and deaths from heart disease among firefighters in the United States. N. Engl. J. Med. 2007, 356, 1207–1215. [Google Scholar] [CrossRef] [PubMed]
- Burgess, J.L.; Nanson, C.J.; Bolstad-Johnson, D.M.; Gerkin, R.; Hysong, T.A.; Lantz, R.C.; Sherrill, D.L.; Crutchfield, C.D.; Quan, S.F.; Bernard, A.M.; et al. Adverse Respiratory Effects Following Overhaul in Firefighters. J. Occup. Environ. Med. 2001, 43, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Daniels, R.D.; Kubale, T.L.; Yiin, J.H.; Dahm, M.M.; Hales, T.R.; Baris, D.; Zahm, S.H.; Beaumont, J.J.; Waters, K.M.; Pinkerton, L.E. Mortality and cancer incidence in a pooled cohort of US firefighters from San Francisco, Chicago and Philadelphia (1950–2009). Occup. Environ. Med. 2014, 71, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.; Koru-Sengul, T.; Hernandez, M.N.; Caban-Martinez, A.J.; McClure, L.A.; MacKinnon, J.A.; Kobetz, E.N. Cancer risk among career male and female Florida firefighters: Evidence from the Florida Firefighter Cancer Registry (1981-2014). Am. J. Ind. Med. 2020, 63, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.U.; Hansen, J.; Ebbehoej, N.E.; Bonde, J.P. Infertility in a Cohort of Male Danish Firefighters: A Register-Based Study. Am. J. Epidemiol. 2018, 188, 339–346. [Google Scholar] [CrossRef]
- Hollerbach, B.S.; Kaipust, C.M.; Poston, W.S.; Haddock, C.K.; Heinrich, K.M.; Jahnke, S.A. Injury Correlates Among a National Sample of Women in the US Fire Service. J. Occup. Environ. Med. 2020, 62, 634–640. [Google Scholar] [CrossRef]
- Dewailly, D.; Andersen, C.Y.; Balen, A.; Broekmans, F.; Dilaver, N.; Fanchin, R.; Griesinger, G.; Kelsey, T.W.; la Marca, A.; Lambalk, C.; et al. The physiology and clinical utility of anti-Mullerian hormone in women. Hum. Reprod. Update 2014, 20, 370–385. [Google Scholar] [CrossRef] [Green Version]
- Wakimoto, Y.; Pors, S.E.; Cadenas, J.; Colmorn, L.; Ernst, E.; Dueholm, M.; Fedder, J.; Mamsen, L.S.; Kristensen, S.G.; Andersen, C.Y. The precise ovarian volume is significantly associated with serum concentrations of antimullerian hormone, the luteinizing hormone/follicle-stimulating hormone ratio, and total testosterone. Fertil. Steril. 2020, 113, 453–459. [Google Scholar] [CrossRef]
- Rasool, S.; Shah, D. Fertility with early reduction of ovarian reserve: The last straw that breaks the Camel’s back. Fertil. Res. Pr. 2017, 3, 15. [Google Scholar] [CrossRef] [Green Version]
- Szeliga, A.; Calik-Ksepka, A.; Maciejewska-Jeske, M.; Grymowicz, M.; Smolarczyk, K.; Kostrzak, A.; Smolarczyk, R.; Rudnicka, E.; Meczekalski, B. Autoimmune Diseases in Patients with Premature Ovarian Insufficiency—Our Current State of Knowledge. Int. J. Mol. Sci. 2021, 22, 2594. [Google Scholar] [CrossRef] [PubMed]
- La Marca, A.; Brozzetti, A.; Sighinolfi, G.; Marzotti, S.; Volpe, A.; Falorni, A. Primary ovarian insufficiency: Autoimmune causes. Curr. Opin. Obstet. Gynecol. 2010, 22, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Warren, B.D.; Kinsey, W.K.; McGinnis, L.K.; Christenson, L.K.; Jasti, S.; Stevens, A.M.; Petroff, B.K.; Petroff, M.G. Ovarian autoimmune disease: Clinical concepts and animal models. Cell. Mol. Immunol. 2014, 11, 510–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Komorowska, B. Autoimmune premature ovarian failure. Menopausal Rev. 2016, 4, 210–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirshenbaum, M.; Orvieto, R. Premature ovarian insufficiency (POI) and autoimmunity-an update appraisal. J. Assist. Reprod. Genet. 2019, 36, 2207–2215. [Google Scholar] [CrossRef]
- Garnett, E.R.; Jariwala, P.; Rector, K.; Gibbons, W.E.; Zarutskie, P.W.; Devaraj, S. Validation of the picoAMH assay on the Dynex DS2 platform. Pr. Lab. Med. 2019, 17, e00140. [Google Scholar] [CrossRef]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef]
- Verma, A.K.; Rajbhar, S.; Mishra, J.; Gupta, M.; Sharma, M.; Deshmukh, G.; Ali, W. Anti-Mullerian Hormone: A Marker of Ovarian Reserve and its Association with Polycystic Ovarian Syndrome. J. Clin. Diagn. Res. 2016, 10, QC10–QC12. [Google Scholar] [CrossRef]
- Garg, D.; Tal, R. The role of AMH in the pathophysiology of polycystic ovarian syndrome. Reprod. Biomed. Online 2016, 33, 15–28. [Google Scholar] [CrossRef] [Green Version]
- Song, D.K.; Oh, J.-Y.; Lee, H.; Sung, Y.-A. Differentiation between polycystic ovary syndrome and polycystic ovarian morphology by means of an anti-Müllerian hormone cutoff value. Korean J. Intern. Med. 2017, 32, 690–698. [Google Scholar] [CrossRef] [Green Version]
- Weghofer, A.; Kim, A.; Barad, D.H.; Gleicher, N. Age at menarche: A predictor of diminished ovarian function? Fertil. Steril. 2013, 100, 1039–1043. [Google Scholar] [CrossRef] [PubMed]
- McQuerry, M. Effect of structural turnout suit fit on female versus male firefighter range of motion. Appl. Ergon. 2020, 82, 102974. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.-Z.; Zhou, F.-J.; Sun, Y.-P. Psychological stress is related to a decrease of serum anti-müllerian hormone level in infertile women. Reprod. Biol. Endocrinol. 2017, 15, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jahnke, S.A.; Poston, W.S.C.; Haddock, C.K.; Murphy, B. Firefighting and mental health: Experiences of repeated exposure to trauma. Work 2016, 53, 737–744. [Google Scholar] [CrossRef]
- Jahnke, S.A.; Poston, W.S.C.; Haddock, C.K.; Jitnarin, N.; Hyder, M.L.; Horvath, C. The health of women in the US fire service. BMC Women’s Health 2012, 12, 39. [Google Scholar] [CrossRef] [Green Version]
- Jahnke, S.A.; Poston, W.S.C.; Jitnarin, N.; Haddock, C.K. Health Concerns of the U.S. Fire Service: Perspectives from the Firehouse. Am. J. Health Promot. 2012, 27, 111–118. [Google Scholar] [CrossRef] [Green Version]
- Allen, A.M.; Lundeen, K.; Eberly, L.E.; Allen, S.S.; Al’Absi, M.; Muramoto, M.; Hatsukami, D. Hormonal contraceptive use in smokers: Prevalence of use and associations with smoking motives. Addict. Behav. 2017, 77, 187–192. [Google Scholar] [CrossRef] [Green Version]
- Allen, A.M.; Lundeen, K.; Murphy, S.E.; Spector, L.; Harlow, B.L. Web-Delivered Multimedia Training Materials for the Self-Collection of Dried Blood Spots: A Formative Project. JMIR Form. Res. 2018, 2, e11025. [Google Scholar] [CrossRef] [Green Version]
- McDade, T.W.; Woodruff, T.; Huang, Y.-Y.; Funk, W.E.; Prewitt, M.; Kondapalli, L.; Gracia, C.R. Quantification of anti-Mullerian hormone (AMH) in dried blood spots: Validation of a minimally invasive method for assessing ovarian reserve. Hum. Reprod. 2012, 27, 2503–2508. [Google Scholar] [CrossRef]
- Hehenkamp, W.J.; Looman, C.W.; Themmen, A.P.; de Jong, F.H.; Te Velde, E.R.; Broekmans, F.J. Anti-Mullerian hormone levels in the spontaneous menstrual cycle do not show substantial fluctuation. J. Clin. Endocrinol. Metab. 2006, 91, 4057–4063. [Google Scholar] [CrossRef] [Green Version]
- Moran, L.J.; Harrison, C.L.; Hutchison, S.K.; Stepto, N.K.; Strauss, B.J.; Teede, H.J. Exercise Decreases Anti-Müllerian Hormone in Anovulatory Overweight Women with Polycystic Ovary Syndrome—A Pilot Study. Horm. Metab. Res. 2011, 43, 977–979. [Google Scholar] [CrossRef] [PubMed]
- Foroozanfard, F.; Rafiei, H.; Samimi, M.; Gilasi, H.R.; Gorjizadeh, R.; Heidar, Z.; Asemi, Z. The effects of dietary approaches to stop hypertension diet on weight loss, anti-Müllerian hormone and metabolic profiles in women with polycystic ovary syndrome: A randomized clinical trial. Clin. Endocrinol. 2017, 87, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Vosnakis, C.; Georgopoulos, N.A.; Rousso, D.; Mavromatidis, G.; Katsikis, I.; Roupas, N.D.; Mamali, I.; Panidis, D. Diet, physical exercise and Orlistat administration increase serum Anti-Müllerian Hormone (AMH) levels in women with polycystic ovary syndrome (PCOS). J. Gynaecol. Endocrinol. 2013, 29, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Nilsson-Condori, E.; Hedenbro, J.L.; Thurin-Kjellberg, A.; Giwercman, A.; Friberg, B. Impact of diet and bariatric surgery on anti-Müllerian hormone levels. Hum. Reprod. 2018, 33, 690–693. [Google Scholar] [CrossRef] [PubMed]
- Farland, L.V.; Collier, A.Y.; Correia, K.F.; Grodstein, F.; Chavarro, J.E.; Rich-Edwards, J.; Missmer, S.A. Who receives a medical evaluation for infertility in the United States? Fertil. Steril. 2016, 105, 1274–1280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristic | Firefighting Status, n (%) | p-Value | |||
|---|---|---|---|---|---|
| Firefighter, | Non-Firefighter, | ||||
| 106 (64.6) | 58 (35.4) | ||||
| Mean ± SD, [Range] | |||||
| Age, years | 38 ± 7.2, [20, 55] | 35.3 ± 6.6, [20, 54] | 0.02 | ||
| Perceived stress a | 15.6 ± 6.9, [3, 34] | 13.4 ± 6.1, [0, 27] | 0.04 | ||
| BMI, kg/m2 | 26.3 ± 4.5, [17.7, 42.9] | 25.8 ± 5.2, [17.5, 38.7] | 0.55 | ||
| Number of known pregnancies | 1.2 ± 1.5, [0, 6] | 1.6 ± 1.8, [0, 6] | 0.18 | ||
| Number of biological children | 0.8 ± 1.0, [0, 5] | 1.2 ± 1.3, [0, 5] | 0.15 | ||
| nb (%) | |||||
| BMI, kg/m2 | 0.13 | ||||
| BMI < 18.5 | <5 | <5 | |||
| 18.5 ≤ BMI < 25 | 41 | (38.7) | 30 | (51.7) | |
| 25 ≤ BMI < 30 | 47 | (44.3) | 15 | (25.9) | |
| BMI ≥ 30 | 16 | (15.1) | 11 | (19.0) | |
| Race | 0.17 | ||||
| White | 101 | (95.3) | 56 | (96.6) | |
| Ethnicity | 0.88 | ||||
| Hispanic | 8 | (7.5) | <5 | ||
| Annual household income | 0.24 | ||||
| USD 50,000 or less | 11 | (10.4) | 8 | (13.8) | |
| USD 50,001–75,000 | 20 | (18.9) | 5 | (8.8) | |
| USD 75,001–100,000 | 19 | (17.9) | 12 | (21.1) | |
| More than USD 100,000 | 56 | (52.8) | 33 | (56.1) | |
| Highest educational attainment | 0.003 | ||||
| High school graduate or GED, some college, or technical school | 46 | (43.4) | 11 | (19.0) | |
| College graduate (4-year degree) | 43 | (40.6) | 27 | (46.6) | |
| Advanced degree (graduate or professional) | 17 | (16.0) | 20 | (34.5) | |
| Marital status | 0.30 | ||||
| Married or in a registered domestic partnership or civil union | 59 | (55.7) | 41 | (66.7) | |
| Never married | 27 | (25.5) | 12 | (21.1) | |
| Divorced or separated | 19 | (17.9) | 5 | (7.0) | |
| Current exercise habits | 0.001 | ||||
| 1: Sedentary | <5 | <5 | |||
| 2 | <5 | 6 | (10.3) | ||
| 3 | 31 | (29.2) | 27 | (39.7) | |
| 4 | 45 | (42.5) | 19 | (32.8) | |
| 5: Very strenuous | 27 | (25.5) | <5 | ||
| Age at menarche | 0.41 | ||||
| ≤11 | 26 | (24.5) | 8 | (13.8) | |
| 12 | 28 | (26.4) | 17 | (29.3) | |
| 13 | 21 | (19.8) | 17 | (29.3) | |
| ≥14 | 31 | (29.2) | 16 | (27.6) | |
| History of infertility | 19 | (17.9) | <5 | 0.05 | |
| Ever used fertility treatment | 13 | (12.3) | 7 | (12.1) | 0.97 |
| Ever used hormonal contraceptives | |||||
| Oral contraceptives | 85 | (80.2) | 48 | (82.8) | 0.69 |
| Other hormonal contraceptive (implant, injectable, patch, ring) | 32 | (30.2) | 15 | (25.9) | 0.56 |
| Currently using hormonal contraceptives | |||||
| Oral contraceptives | 13 | (12.3) | 6 | (10.3) | 0.71 |
| Other hormonal contraceptive (implant, injectable, patch, ring) | <5 | <5 | 0.83 | ||
| Endometriosis history | 8 | (7.5) | <5 | 0.58 | |
| History of polycystic ovary syndrome | 6 | (5.6) | <5 | 0.53 | |
| Characteristic | Firefighters | |
|---|---|---|
| (n = 106) | ||
| mean ± SD, [range] | ||
| Years in the fire service | 13.3 ± 6.8, [2, 34] | |
| Calls responded to in a typical month | 61.7 ± 55.4, [0, 350] | |
| Live fires responded to in a typical month | 2.3 ± 5.1, [0, 50] | |
| nc (%) | ||
| Current or past firefighter | ||
| Current | 97 | (91.5) |
| Past | 9 | (8.5) |
| Current rank a | ||
| Firefighter, Driver, or Operator | 39 | (36.8) |
| Firefighter/Paramedic | 29 | (27.4) |
| Company Officer (Lieutenant, Captain) | 29 | (27.4) |
| Chief (Battalion, Deputy, Other) | 6 | (5.7) |
| Other | <5 | |
| Participation in wildland firefighting | 37 | (34.9) |
| Poor fit of at least one part of personal protective equipment b | 55 | (51.9) |
| Poor fit of self-contained breathing apparatus (SCBA) | 26 | (24.5) |
| Firefighting Status | AMH (ng/mL), Mean ± SD, [Range] | Model 1 % Difference (95% CI) | Model 2 % Difference (95% CI) |
|---|---|---|---|
| Non-firefighters (n = 58) | 4.37 ± 4.50, [0.16, 17.30] | 0.00 (Ref.) | 0.00 (Ref.) |
| Firefighters (n = 106) | 2.93 ± 3.83, [0.02, 17.30] | −57.49 (−75.06, −27.54) | −33.38 (−54.97, −1.43) |
| Firefighting-Related Exposure | AMH (ng/mL), Mean ± SD, [Range] | Model 1 % Difference (95% CI) | Model 2 % Difference (95% CI) |
|---|---|---|---|
| Years in the fire service continuous (per 5-year increase) | −54.60 (−63.39, −43.69) | 0.70 (−25.11, 35.43) | |
| Live fires responded to in a typical month | 1.00 (−5.88, 8.37) | 1.42 (−3.40, 6.48) | |
| Poor fit of any personal protective equipment | |||
| No (n = 51) | 3.29 ± 4.54, [0.02, 17.3] | 0.00 (Ref.) | 0.00 (Ref.) |
| Yes (n = 55) | 2.60 ± 3.05, [0.02, 15.1] | 9.61 (−46.23, 123.46) | 46.66 (−10.10, 139.25) |
| Poor fit of self-contained breathing apparatus (SCBA) | |||
| No (n = 80) | 2.99 ± 4.09, [0.02, 17.3] | 0.00 (Ref.) | 0.00 (Ref.) |
| Yes (n = 26) | 2.75 ± 2.96, [0.02, 11.3] | 22.88 (−46.22, 180.80) | 56.13 (−13.03, 180.29) |
| Wildland firefighting | |||
| No (n = 68) | 2.99 ± 3.78, [0.02, 17.3] | 0.00 (Ref.) | 0.00 (Ref.) |
| Yes (n = 37) | 2.79 ± 4.02, [0.02, 16.7] | 8.96 (−48.53, 130.65) | −7.90 (−45.05, 54.35) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Davidson, S.; Jahnke, S.; Jung, A.M.; Burgess, J.L.; Jacobs, E.T.; Billheimer, D.; Farland, L.V. Anti-Müllerian Hormone Levels among Female Firefighters. Int. J. Environ. Res. Public Health 2022, 19, 5981. https://doi.org/10.3390/ijerph19105981
Davidson S, Jahnke S, Jung AM, Burgess JL, Jacobs ET, Billheimer D, Farland LV. Anti-Müllerian Hormone Levels among Female Firefighters. International Journal of Environmental Research and Public Health. 2022; 19(10):5981. https://doi.org/10.3390/ijerph19105981
Chicago/Turabian StyleDavidson, Samantha, Sara Jahnke, Alesia M. Jung, Jefferey L. Burgess, Elizabeth T. Jacobs, Dean Billheimer, and Leslie V. Farland. 2022. "Anti-Müllerian Hormone Levels among Female Firefighters" International Journal of Environmental Research and Public Health 19, no. 10: 5981. https://doi.org/10.3390/ijerph19105981
APA StyleDavidson, S., Jahnke, S., Jung, A. M., Burgess, J. L., Jacobs, E. T., Billheimer, D., & Farland, L. V. (2022). Anti-Müllerian Hormone Levels among Female Firefighters. International Journal of Environmental Research and Public Health, 19(10), 5981. https://doi.org/10.3390/ijerph19105981






