Association of Dietary Pattern, Lifestyle and Chronotype with Metabolic Syndrome in Elderly—Lessons from the Population-Based Hamburg City Health Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Outcome Variable—MetS
- Raised triglycerides ≥ 150 mg/dL or specific medication for this lipid abnormality.
- Reduced HDL cholesterol (men < 40 mg/dL, women < 50 mg/dL) or specific medication for this lipid abnormality/
- Raised blood pressure exceeds ≥ 130 mm Hg systolic or ≥ 85 mm Hg diastolic or medication of the previously diagnosed hypertension.
- Raised fasting plasma glucose ≥ 100 mg/dL or previously diagnosed type 2 diabetes mellitus.
2.3. Dietary Scores
2.4. Chronotype
2.5. Covariates
2.6. Statistical Analyses
3. Results
3.1. Study Sample
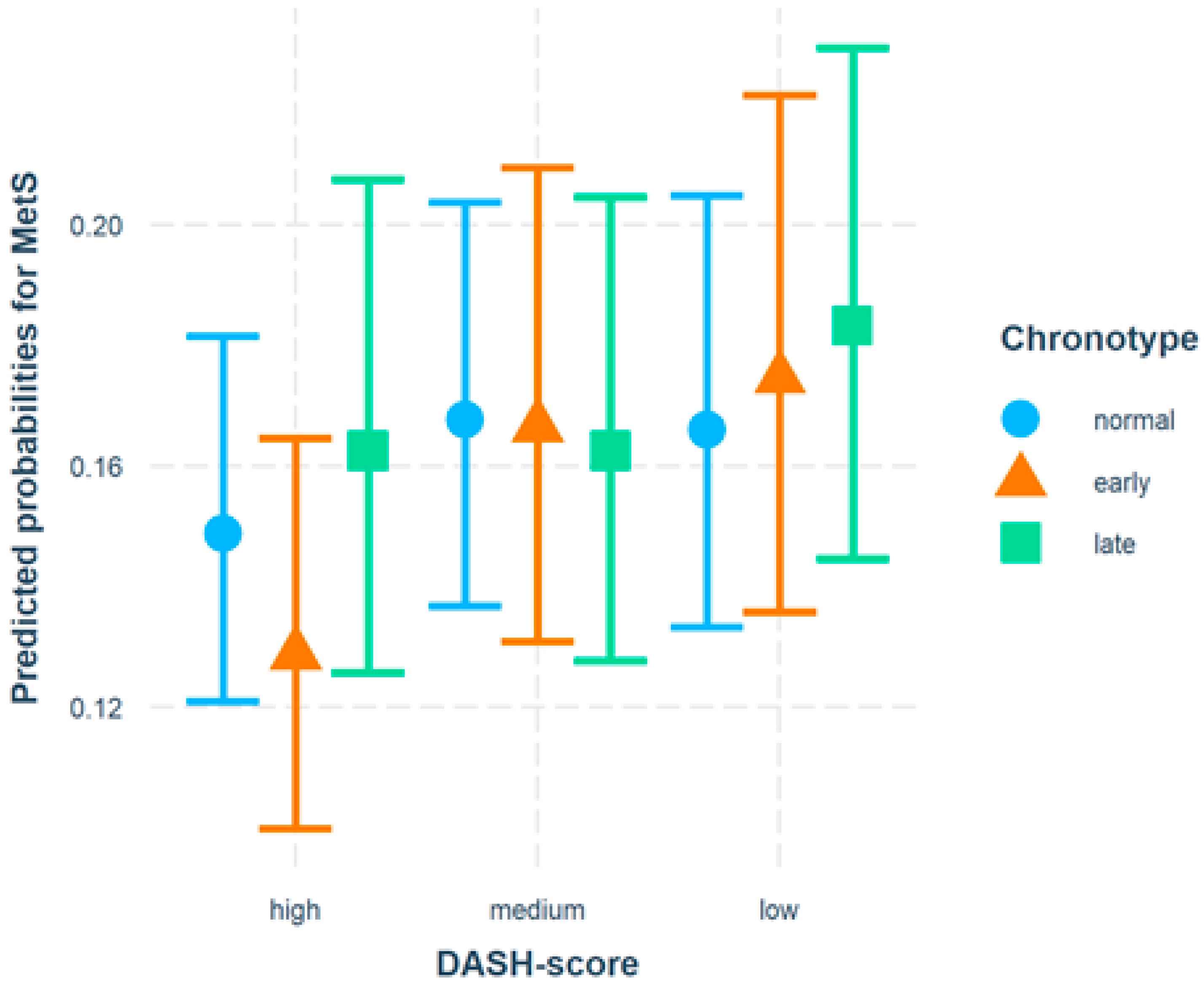
3.2. Results of Logistic Regression Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Score Item | MEDAS Question | Data Recorded by FFQ 1 Point Given, if … |
|---|---|---|
| 1 | Do you use olive oil as the principal source of fat for cooking? | use of olive oil for the preparation of at least 2 of the following groceries: salad, vegetable, meat/fish |
| 2 | How much olive oil do you consume per day (including that used in frying, salads, meals eaten away from home, etc.)? | based on FFQ calculation, if >48 g vegetable oil per day |
| 3 | How many servings of vegetables do you consume per day? | based on FFQ calculation, if ≥2 portions of vegetables per day (including raw and cooked vegetables, salad, olives, mushrooms except potatoes and legumes) |
| 4 | How many pieces of fruit (including fresh-squeezed juice) do you consume per day? | based on FFQ calculation, if ≥3 portions of fruit (including fruit, mixed fruit, fruit salad, mixed stewed fruit and fruit juices excluding sweetened beverages) |
| 5 | How many servings of red meat, hamburger, or sausages do you consume per day? | based on FFQ calculation, if <100 g red meat (eg beef, veal, pork, lamb) and processed meat products |
| 6 | How many servings (12 g) of butter, margarine, or cream do you consume per day? | based on FFQ calculation, if <1 portion butter, margarine and cream and other animal fat |
| 7 | How many carbonated and/or sugar-sweetened beverages do you consume per day? | based on FFQ calculation, sugar-sweetened beverages <1 portion per day (including lemonade and colas) |
| 8 | Do you drink wine? How much do you consume per week? | based on FFQ calculation, if ≥7 portions wine (red and white wine; 1 portion = 0.251) |
| 9 | How many servings of pulses do you consume per week? | ≥3 portions pulses (e.g., beans, lentils, peas, chickpeas) |
| 10 | How many servings of fish/seafood do you consume per week? | based on FFQ calculation, if ≥3 portions fish, fish products and seafood per week |
| 11 | How many times do you consume commercial (not homemade)pastry such as cookies or cake per week? | based on FFQ calculation, if <3 portions cakes, chocolate, cookies, sweets with and without chocolate per week |
| 12 | How many times do you consume nuts per week? | based on FFQ calculation, if ≥3 portions nuts per week |
| 13 | Do you prefer to eat chicken, turkey or rabbit instead of beef, pork, hamburgers, or sausages? | Based on FFQ calculation, if g white meat (e.g., chicken, hen and other poultry) > g red meat (e.g., beef, veal, pork, lamb and processed meat products) |
| 14 | How many times per week do you consume boiled vegetables, pasta, rice, or other dishes with a sauce of tomato, garlic, onion, or leeks sautéed in olive oil? | >1–2 times a week tomato sauce |
References
- Alberti, K.G.M.M.; Zimmet, P.; Shaw, J.; IDF Epidemiology Task Force Consensus Group. The metabolic syndrome—A new worldwide definition. Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef]
- Sundström, J.; Lind, L.; Lampa, E.; Angerås, O.; Bachus, E.; Bergström, G.; Carlberg, B.; Engström, G.; Engvall, J.E.; Eriksson, M.; et al. Weight gain and blood pressure. J. Hypertens. 2020, 38, 387–394. [Google Scholar] [CrossRef]
- Schipf, S.; Alte, D.; Völzke, H.; Friedrich, N.; Haring, R.; Lohmann, T.; Rathmann, W.; Nauck, M.; Felix, S.; Hoffmann, W.; et al. Prävalenz des Metabolischen Syndroms in Deutschland: Ergebnisse der Study of Health in Pomerania (SHIP). Diabetol. Stoffwechs. 2010, 5, 161–168. [Google Scholar] [CrossRef]
- Heidemann, C.; Kuhnert, R.; Born, S.; Scheidt-Nave, C. 12-Monats-Prävalenz des bekannten Diabetes mellitus in Deutschland. J. Health Monit. 2017, 2, 48–56. [Google Scholar] [CrossRef]
- Neuhauser, H.; Kuhnert, R.; Born, S. 12-Monats-Prävalenz von Bluthochdruck in Deutschland. J. Health Monit. 2017, 2, 57–63. [Google Scholar] [CrossRef]
- Myers, J.; Kokkinos, P.; Nyelin, E. Physical Activity, Cardiorespiratory Fitness, and the Metabolic Syndrome. Nutrients 2019, 11, 1652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castro-Barquero, S.; Ruiz-León, A.M.; Sierra-Pérez, M.; Estruch, R.; Casas, R. Dietary Strategies for Metabolic Syndrome: A Comprehensive Review. Nutrients 2020, 12, 2983. [Google Scholar] [CrossRef] [PubMed]
- Almoosawi, S.; Vingeliene, S.; Gachon, F.; Voortman, T.; Palla, L.; Johnston, J.; Van Dam, R.M.; Darimont, C.; Karagounis, L.G. Chronotype: Implications for Epidemiologic Studies on Chrono-Nutrition and Cardiometabolic Health. Adv. Nutr. 2018, 10, 30–42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suh, S.; Yang, H.-C.; Kim, N.; Yu, J.H.; Choi, S.; Yun, C.-H.; Shin, C. Chronotype Differences in Health Behaviors and Health-Related Quality of Life: A Population-Based Study Among Aged and Older Adults. Behav. Sleep Med. 2016, 15, 361–376. [Google Scholar] [CrossRef] [PubMed]
- Maukonen, M.; Kanerva, N.; Partonen, T.; Kronholm, E.; Konttinen, H.; Wennman, H.; Männistö, S. The associations between chronotype, a healthy diet and obesity. Chronobiol. Int. 2016, 33, 972–981. [Google Scholar] [CrossRef]
- Kanerva, N.; Kronholm, E.; Partonen, T.; Ovaskainen, M.-L.; Kaartinen, N.E.; Konttinen, H.; Broms, U.; Männistö, S. Tendency toward Eveningness Is Associated with Unhealthy Dietary Habits. Chronobiol. Int. 2012, 29, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Patterson, F.; Malone, S.K.; Lozano, A.; Grandner, M.; Hanlon, A.L. Smoking, Screen-Based Sedentary Behavior, and Diet Associated with Habitual Sleep Duration and Chronotype: Data from the UK Biobank. Ann. Behav. Med. 2016, 50, 715–726. [Google Scholar] [CrossRef] [Green Version]
- Mazri, F.H.; Manaf, Z.A.; Shahar, S.; Ludin, A.F.M. The Association between Chronotype and Dietary Pattern among Adults: A Scoping Review. Int. J. Environ. Res. Public Health 2019, 17, 68. [Google Scholar] [CrossRef] [Green Version]
- Roenneberg, T.; Allebrandt, K.V.; Merrow, M.; Vetter, C. Social Jetlag and Obesity. Curr. Biol. 2012, 22, 939–943. [Google Scholar] [CrossRef] [Green Version]
- Jagodzinski, A.; Johansen, C.; Koch-Gromus, U.; Aarabi, G.; Adam, G.; Anders, S.; Augustin, M.; Der Kellen, R.B.; Beikler, T.; Behrendt, C.-A.; et al. Rationale and Design of the Hamburg City Health Study. Eur. J. Epidemiol. 2019, 35, 169–181. [Google Scholar] [CrossRef] [Green Version]
- Nöthlings, U.; Hoffmann, K.; Bergmann, M.M.; Boeing, H. Fitting Portion Sizes in a Self-Administered Food Frequency Questionnaire. J. Nutr. 2007, 137, 2781–2786. [Google Scholar] [CrossRef]
- Folsom, A.R.; Parker, E.; Harnack, L.J. Degree of Concordance With DASH Diet Guidelines and Incidence of Hypertension and Fatal Cardiovascular Disease. Am. J. Hypertens. 2007, 20, 225–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Epstein, D.E.; Sherwood, A.; Smith, P.J.; Craighead, L.; Caccia, C.; Lin, P.-H.; Babyak, M.A.; Johnson, J.J.; Hinderliter, A.; Blumenthal, J.A. Determinants and Consequences of Adherence to the Dietary Approaches to Stop Hypertension Diet in African-American and White Adults with High Blood Pressure: Results from the ENCORE Trial. J. Acad. Nutr. Diet. 2012, 112, 1763–1773. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hebestreit, K.; Yahiaoui-Doktor, M.; Engel, C.; Vetter, W.; Siniatchkin, M.; Erickson, N.; Halle, M.; Kiechle, M.; Bischoff, S.C. Validation of the German version of the Mediterranean Diet Adherence Screener (MEDAS) questionnaire. BMC Cancer 2017, 17, 341. [Google Scholar] [CrossRef]
- Roenneberg, T.; Wirz-Justice, A.; Merrow, M. Life between Clocks: Daily Temporal Patterns of Human Chronotypes. J. Biol. Rhythm. 2003, 18, 80–90. [Google Scholar] [CrossRef] [Green Version]
- Wareham, N.J.; Jakes, R.W.; Rennie, K.L.; Mitchell, J.; Hennings, S.; Day, N.E. Validity and repeatability of the EPIC-Norfolk Physical Activity Questionnaire. Int. J. Epidemiol. 2002, 31, 168–174. [Google Scholar] [CrossRef] [Green Version]
- Altun, E.; Walther, C.; Borof, K.; Petersen, E.; Lieske, B.; Kasapoudis, D.; Jalilvand, N.; Beikler, T.; Jagemann, B.; Zyriax, B.-C.; et al. Association between Dietary Pattern and Periodontitis—A Cross-Sectional Study. Nutrients 2021, 13, 4167. [Google Scholar] [CrossRef] [PubMed]
- Munshi, A.; Desai, S.; Munshi, D. Gender bias in cardiovascular disease prevention, detection, and management, with specific reference to coronary artery disease. J. Mid-Life Health 2021, 12, 8–15. [Google Scholar] [CrossRef]
- De Amicis, R.; Galasso, L.; Leone, A.; Vignati, L.; De Carlo, G.; Foppiani, A.; Montaruli, A.; Roveda, E.; Ce, E.; Esposito, F.; et al. Is Abdominal Fat Distribution Associated with Chronotype in Adults Independently of Lifestyle Factors? Nutrients 2020, 12, 592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zyriax, B.C.; Lau, K.; Klahn, T.; Boeing, H.; Volzke, H.; Windler, E. Association between alcohol consumption and carotid intima-media thickness in a healthy population: Data of the STRATEGY study (Stress, Atherosclerosis and ECG Study). Eur. J. Clin. Nutr. 2010, 64, 1199–1206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zyriax, B.-C.; Dransfeld, K.; Windler, E. Carotid intima–media thickness and cardiovascular risk factors in healthy volunteers. Ultrasound J. 2021, 13, 17. [Google Scholar] [CrossRef]
- Pot, G.K.; Teams, T.N.S.A.D.C.; Prynne, C.J.; Almoosawi, S.; Kuh, D.; Stephen, A.M. Trends in food consumption over 30 years: Evidence from a British birth cohort. Eur. J. Clin. Nutr. 2014, 69, 817–823. [Google Scholar] [CrossRef] [Green Version]
- Randler, C.; Engelke, J. Gender differences in chronotype diminish with age: A meta-analysis based on morningness/chronotype questionnaires. Chronobiol. Int. 2019, 36, 888–905. [Google Scholar] [CrossRef]
- Muscogiuri, G.; Barrea, L.; Aprano, S.; Framondi, L.; Di Matteo, R.; Altieri, B.; Laudisio, D.; Pugliese, G.; Savastano, S.; Colao, A. Chronotype and cardio metabolic health in obesity: Does nutrition matter? Int. J. Food Sci. Nutr. 2021, 72, 892–900. [Google Scholar] [CrossRef]
- Pérez-Martínez, P.; Mikhailidis, D.P.; Athyros, V.G.; Bullo, M.; Couture, P.; Covas, M.I.; de Koning, L.; Delgado-Lista, J.; Díaz-López, A.; Drevon, C.A.; et al. Lifestyle recommendations for the prevention and management of metabolic syndrome: An international panel recommendation. Nutr. Rev. 2017, 75, 307–326. [Google Scholar] [CrossRef] [Green Version]
- Filippou, C.D.; Tsioufis, C.P.; Thomopoulos, C.G.; Mihas, C.C.; Dimitriadis, K.S.; Sotiropoulou, L.I.; Chrysochoou, C.A.; Nihoyannopoulos, P.I.; Tousoulis, D.M. Dietary Approaches to Stop Hypertension (DASH) Diet and Blood Pressure Reduction in Adults with and without Hypertension: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2020, 11, 1150–1160. [Google Scholar] [CrossRef] [PubMed]
- Mensink, G.B.; Schienkiewitz, A.; Haftenberger, M.; Lampert, T.; Ziese, T.; Scheidt-Nave, C. Overweight and obesity in Germany: Results of the German Health Interview and Examination Survey for Adults (DEGS1). Bundesgesundheitsblatt -Gesundh. -Gesundh. 2013, 56, 786–794. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Score Items | DASH Component | Scoring |
|---|---|---|
| 1 | Total Grain | |
| ≥7 servings/day | 1 | |
| 5–6 servings/day | 0.5 | |
| <5 servings/day | 0 | |
| 2 | Vegetables | |
| ≥4 servings/day | 1 | |
| 2–3 servings/day | 0.5 | |
| <2 servings/day | 0 | |
| 3 | Fruits | |
| ≥4 servings/day | 1 | |
| 2–3 servings/day | 0.5 | |
| <2 servings/day | 0 | |
| 4 | Total dairy | |
| ≥2 servings/day | 1 | |
| 1 servings/day | 0.5 | |
| <1 serving/day | 0 | |
| 5 | Meat, poultry, and fish | |
| ≤2 servings/day | 1 | |
| 3 servings/day | 0.5 | |
| ≥4 serving/day | 0 | |
| 6 | Nuts, seeds, and legumes | |
| ≥4 servings/day | 1 | |
| 2–3 servings/day | 0.5 | |
| <2 servings/day | 0 | |
| 7 | % kcal from fat | |
| ≤27% | 1 | |
| ≥28 ≤29% | 0.5 | |
| ≥30% | 0 | |
| 8 | % kcal from saturated fat | |
| ≤6% | 1 | |
| ≤7 ≥8% | 0.5 | |
| ≥9% | 0 | |
| 9 | Sweets | |
| ≤5 servings/week | 1 | |
| 6–7 servings/week | 0.5 | |
| ≥8 serving/week | 0 | |
| 10 | Sodium | |
| ≤2400 mg/day | 1 | |
| 2400–3000 mg/day | 0.5 | |
| >3000 mg/day | 0 |
| Women | Men | Total | |
|---|---|---|---|
| n (%) | 2070 (47.8%) | 2260 (52.2%) | 4330 (100%) |
| Diagnosis heart attack, n (%) | 19 (0.9%) | 101 (4.5%) | 120 (2.8%) |
| Diagnosis stroke, n (%) | 42 (2.0%) | 71 (3.1%) | 113 (2.6%) |
| Diagnosis coronary heart disease, n (%) | 32 (1.6%) | 178 (8.0%) | 210 (4.9%) |
| At least one with diagnosis and thus excluded | 81 (100.0%) | 277 (100.0%) | 358 (100.0%) |
| Also excluded by age | |||
| 45–49 years | 153 (7.7%) | 137 (6.9%) | 290 (7.3%) |
| 75+ years | 64 (3.2%) | 105 (5.3%) | 169 (4.3%) |
| After exclusion due to prior history of relevant illnesses and age | |||
| n (%) | 1772 (50.4%) | 1741 (49.6%) | 3513 (100%) |
| Age years, mean (SD) | 61.1 (6.707) | 61.799 (6.8) | 61.4 (6.777) |
| Age categories, n (%) | |||
| 50–54 | 368 (20.8%) | 326 (18.7%) | 694 (19.8%) |
| 55–59 | 424 (23.9%) | 358 (20.6%) | 782 (22.3%) |
| 60–64 | 401 (22.6%) | 388 (22.3%) | 789 (22.5%) |
| 65–69 | 341 (19.2%) | 399 (22.9%) | 740 (21.1%) |
| 70–74 | 238 (13.4%) | 270 (15.5%) | 508 (14.5%) |
| Age ≥ 65 years, n (%) | 579 (32.7%) | 669 (38.4%) | 1248 (35.5%) |
| Sport ≥ 1 h/week, n (%) | 1326 (74.8%) | 1165 (66.9%) | 2491 (70.9%) |
| School diploma, n (%) | |||
| low | 331 (18.7%) | 315 (18.1%) | 646 (18.4%) |
| medium | 586 (33.1%) | 408 (23.4%) | 994 (28.3%) |
| high | 855 (48.3%) | 1018 (58.5%) | 1873 (53.3%) |
| Smoking status, n (%) | |||
| Never-smoker | 711 (40.1%) | 587 (33.7%) | 1298 (36.9%) |
| Ex-smoker | 735 (41.5%) | 820 (47.1%) | 1555 (44.3%) |
| Smoker | 326 (18.4%) | 334 (19.2%) | 660 (18.8%) |
| BMI, mean (SD) | 26.0 (4.8) | 27.0 (4.2) | 26.5 (4.5) |
| BMI category, n (%) | |||
| Normalweight (BMI 18.5–24.9) | 841 (32.0%) | 557 (32.0%) | 1398 (39.8%) |
| Undeweight (BMI < 18.5) | 30 (1.7%) | 5 (0.3%) | 35 (1.0%) |
| Overweight BMI (25–29.9) | 599 (33.8%) | 843 (48.4%) | 1442 (41.0%) |
| Adipose (BMI ≥ 30) | 302 (17.0%) | 336 (19.3%) | 638 (18.2%) |
| Sleeptime in h, n (%) | |||
| 6–8 | 999 (56.4%) | 1024 (58.8%) | 2023 (57.6%) |
| <6 | 141 (8.0%) | 115 (6.6%) | 256 (7.3%) |
| >8 | 632 (35.7%) | 602 (34.6%) | 1234 (35.1%) |
| Chorotype, n (%) | |||
| “normal” | 808 (45.6%) | 740 (42.5%) | 1548 (44.1%) |
| early | 514 (29.0%) | 496 (28.5%) | 1010 (28.8%) |
| late | 450 (25.4%) | 505 (29.0%) | 955 (27.2%) |
| Energy intake kcal/day, mean (SD) | 1843.4 (572.5) | 2518.9 (808.5) | 2178.2 (776.7) |
| DASH score (max. possible 10), mean (SD) | 4.82 (0.9) | 4.13 (1.0) | 4.48 (1.0) |
| DASH score in tertiles, n (%) | |||
| low | 366 (20.7%) | 797 (45.8%) | 1163 (33.1%) |
| medium | 620 (35.0%) | 586 (33.7%) | 1206 (34.3%) |
| high | 786 (44.4%) | 358 (20.6%) | 1144 (32.6%) |
| MEDAS score (max. possible 14), mean (SD) | 5.16 (1.7) | 3.91 (1.7) | 4.54 (1.8) |
| MEDAS score in tertiles, n (%) | |||
| low | 357 (20.1%) | 827 (47.5%) | 1184 (33.7%) |
| medium | 606 (34.2%) | 563 (32.3%) | 1169 (33.3%) |
| high | 809 (45.7%) | 351 (20.2%) | 1160 (33.0%) |
| “Normal” (n = 1548) | Early (n = 1010) | Late (n = 995) | Total (n = 3513) | p-Value “ Normal” vs. Early | p-Value “ Normal” vs. Late | |
|---|---|---|---|---|---|---|
| Energy intake, kcal/day, mean (SD) | 2179.5 (784.1) | 2101.0 (719.8) | 2257.8 (814.2) | 2178.2 (776.7) | 0.024 (1) | 0.009 (1) |
| DASH score, mean (SD) | 4.53 (1.1) | 4.50 (1.0) | 4.37 (1.1) | 4.48 (1.0) | 0.552 (1) | <0.001 (1) |
| DASH score in tertiles, n (%) | 0.900 (2) | 0.004 (2) | ||||
| - low | 485 (31.3%) | 325 (32.2%) | 353 (37.0%) | 1163 (33.1%) | ||
| - medium | 533 (34.4%) | 345 (34.2%) | 328 (34.3%) | 1206 (34.3%) | ||
| - high | 530 (34.2%) | 340 (33.7%) | 274 (28.7%) | 1144 (32.6%) | ||
| MEDAS score, mean (SD) | 4.56 (1.9) | 4.52 (1.8) | 4.53 (1.8) | 4.54 (1.8) | 0.412 (1) | 0.492 (1) |
| MEDAS score in tertiles, n (%) | 0.248 (2) | 0.475 (2) | ||||
| - low | 500 (32.3%) | 356 (35.2%) | 328 (34.3%) | 1184 (33.7%) | ||
| - medium | 517 (33.4%) | 333 (33.0%) | 319 (33.4%) | 1169 (33.3%) | ||
| - high | 531 (34.3%) | 321 (31.8%) | 308 (32.3%) | 1160 (33.0%) |
| Women (n = 1772) | Men (n = 1741) | Total (n = 3513) | p-Value | |
|---|---|---|---|---|
| Waist circumference (>80 cm women, >94 cm men), n (%) | 1298 (73.9%) | 1198 (69.2%) | 2496 (71.6%) | 0.002 (1) |
| Triglycerides > 150 mg, n (%) | 300 (17.0%) | 476 (27.5%) | 776 (22.2%) | <0.001 (1) |
| HDL (<50 mg/dL women, <40 men), n (%) | 187 (10.6%) | 200 (11.5%) | 387 (11.1%) | 0.369 (1) |
| Blood pressure (systolic > 130 mmHg or diastolic > 85 mmHg), n (%) | 924 (53.4%) | 1145 (67.3%) | 2069 (60.3%) | <0.001 (1) |
| Fasting plasma glucose (>100 mg/dL) or diabetes mellitus, n (%) | 305 (18.4%) | 541 (34.1%) | 846 (26.1%) | <0.001 (1) |
| Metabolic syndrome, n (%) | 500 (28.2%) | 770 (44.2%) | 1270 (36.2%) | <0.001 (1) |
| Risk Factor | Category | Reference | OR | 95% CI | p-Value |
|---|---|---|---|---|---|
| Age | ≥65 | <65 | 1.54 | (1.32, 1.79) | <0.001 |
| Sex | men | women | 1.93 | (1.65, 2.25) | <0.001 |
| School diploma | medium | high | 1.48 | (1.24, 1.75) | <0.001 |
| low | 2.04 | (1.68, 2.49) | <0.001 | ||
| Smoking status | ex-smoker | never-smoker | 1.29 | (1.10, 1.52) | 0.002 |
| current smoker | 1.47 | (1.20, 1.81) | <0.001 | ||
| Sport/physical training | <1 h/week | ≥1 h/week | 1.55 | (1.32, 1.81) | <0.001 |
| Chronotype | early | “normal” | 0.97 | (0.81, 1.15) | 0.698 |
| late | 1.06 | (0.89, 1.26) | 0.522 | ||
| DASH score (tertiles) | medium | high | 1.16 | (0.97, 1.40) | 0.100 |
| low | 1.23 | (1.01, 1.48) | 0.035 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Terschüren, C.; Damerau, L.; Petersen, E.L.; Harth, V.; Augustin, M.; Zyriax, B.-C. Association of Dietary Pattern, Lifestyle and Chronotype with Metabolic Syndrome in Elderly—Lessons from the Population-Based Hamburg City Health Study. Int. J. Environ. Res. Public Health 2022, 19, 377. https://doi.org/10.3390/ijerph19010377
Terschüren C, Damerau L, Petersen EL, Harth V, Augustin M, Zyriax B-C. Association of Dietary Pattern, Lifestyle and Chronotype with Metabolic Syndrome in Elderly—Lessons from the Population-Based Hamburg City Health Study. International Journal of Environmental Research and Public Health. 2022; 19(1):377. https://doi.org/10.3390/ijerph19010377
Chicago/Turabian StyleTerschüren, Claudia, Lukas Damerau, Elina Larissa Petersen, Volker Harth, Matthias Augustin, and Birgit-Christiane Zyriax. 2022. "Association of Dietary Pattern, Lifestyle and Chronotype with Metabolic Syndrome in Elderly—Lessons from the Population-Based Hamburg City Health Study" International Journal of Environmental Research and Public Health 19, no. 1: 377. https://doi.org/10.3390/ijerph19010377
APA StyleTerschüren, C., Damerau, L., Petersen, E. L., Harth, V., Augustin, M., & Zyriax, B.-C. (2022). Association of Dietary Pattern, Lifestyle and Chronotype with Metabolic Syndrome in Elderly—Lessons from the Population-Based Hamburg City Health Study. International Journal of Environmental Research and Public Health, 19(1), 377. https://doi.org/10.3390/ijerph19010377






