Phase Angle Is Related to 10 m and 30 m Sprint Time and Repeated-Sprint Ability in Young Male Soccer Players
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Procedures
2.3. Body Composition (Covariates)
2.4. Phase Angle (Independent Variable)
2.5. Physical Performance Indices (Dependent Variables)
2.6. Statistical Analysis
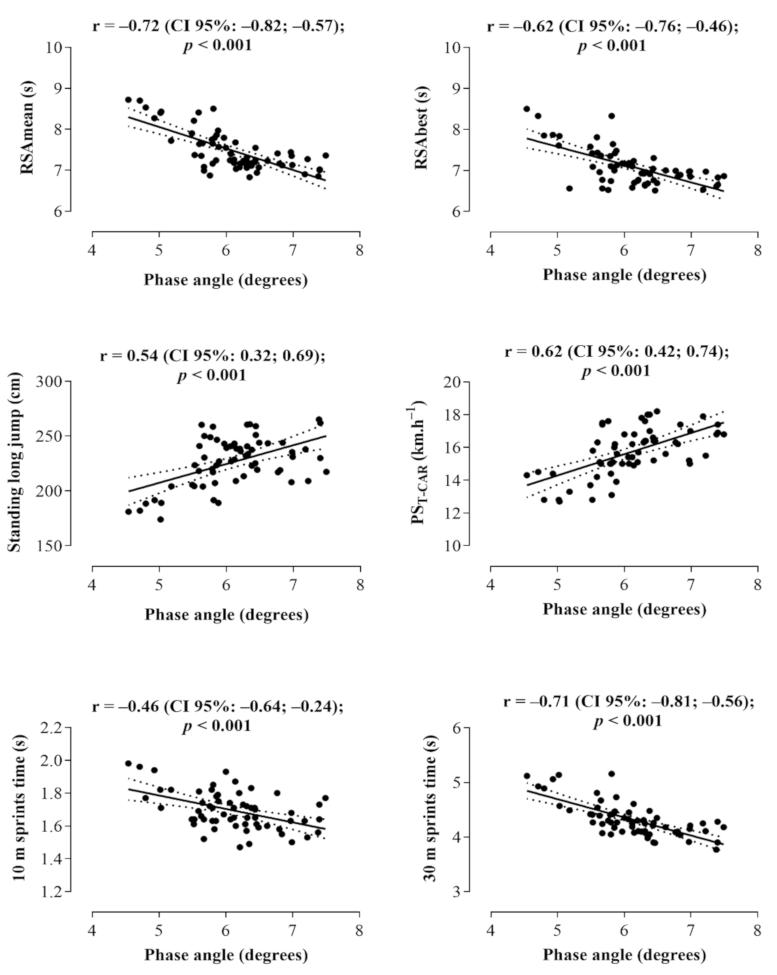
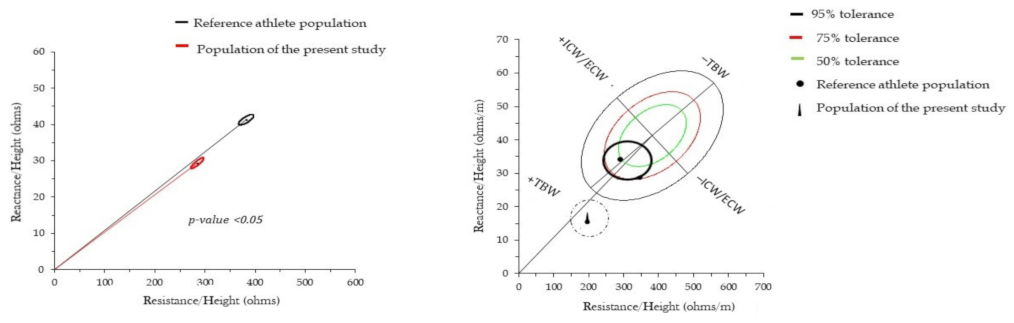
3. Results
4. Discussion
5. Conclusions
6. What Does This Article Add?
- Physical assessments requiring the application of maximum efforts from players are not always possible to be implemented over the season in order to monitor the players’ physical readiness state. From our findings, PhA emerge as a non-invasive cellular health marker obtained at rest condition to be considered in the context of screening tools used in adolescent athletes due to its relationship with crucial soccer-specific anaerobic running activities such as sprinting speed and RSA.
- Our results contribute to the body of knowledge produced to date on this topic, addressing the practical application of this measurement (i.e., PhA) within youth sports settings.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Disclosure Statement
References
- Stølen, T.; Chamari, K.; Castagna, C.; Wisløff, U. Physiology of Soccer: An update. Sports Med. 2005, 35, 501–536. [Google Scholar] [CrossRef]
- Vieira, L.H.P.; Aquino, R.; Moura, F.A.; De Barros, R.M.; Arpini, V.M.; Oliveira, L.P.; Bedo, B.L.; Santiago, P.R. Team dynamics, running, and skill-related performances of Brazilian U11 to professional soccer players during official matches. J. Strength Cond. Res. 2019, 33, 2202–2216. [Google Scholar] [CrossRef] [PubMed]
- Rebelo, A.; Brito, J.; Seabra, A.; Oliveira, J.; Krustrup, P. Physical match performance of youth football players in relation to physical capacity. Eur. J. Sport Sci. 2014, 14, S148–S156. [Google Scholar] [CrossRef]
- Baldi, M.; DASilva, J.F.; Buzzachera, C.F.; Castagna, C.; Guglielmo, L.G. Repeated sprint ability in soccer players: Associations with physiological and neuromuscular factors. J. Sports Med. Phys. Fit. 2017, 57, 26–32. [Google Scholar] [CrossRef]
- Buchheit, M.; Samozino, P.; Glynn, J.A.; Michael, B.S.; Al Haddad, H.; Mendez-Villanueva, A.; Morin, J.B. Mechanical determinants of acceleration and maximal sprinting speed in highly trained young soccer players. J. Sports Sci. 2014, 32, 1906–1913. [Google Scholar] [CrossRef] [PubMed]
- Campa, F.; Semprini, G.; Júdice, P.B.; Messina, G.; Toselli, S. Anthropometry, Physical and Movement Features, and Repeated-sprint Ability in Soccer Players. Int. J. Sports Med. 2019, 40, 100–109. [Google Scholar] [CrossRef]
- Deprez, D.; Valente-Dos-Santos, J.; Coelho-E-Silva, M.J.; Lenoir, M.; Philippaerts, R.; Vaeyens, R. Longitudinal Development of Explosive Leg Power from Childhood to Adulthood in Soccer Players. Int. J. Sports Med. 2015, 36, 672–679. [Google Scholar] [CrossRef] [PubMed]
- Deprez, D.; Valente-dos-Santos, J.; Coelho-e-Silva, M.J.; Lenoir, M.; Philippaerts, R.; Vaeyens, R. Multilevel Development Models of Explosive Leg Power in High-Level Soccer Players. Med. Sci. Sports Exerc. 2015, 47, 1408–1415. [Google Scholar] [CrossRef]
- Deprez, D.; Valente-dos-santos, J.; Coelho, M.; Lenoir, M.; Philippaerts, R.M.; Vaeyens, R. Modeling Developmental Changes in the Yo-Yo Intermittent Recovery Test Level 1 in Elite Pubertal Soccer Players. Int. J. Sports Physiol. Perform. 2014, 9, 1006–1012. [Google Scholar] [CrossRef] [PubMed]
- Valente-Dos-Santos, J.; Coelho-e-silva, M.J.; Duarte, J.; Figueiredo, A.J.; Liparotti, J.R.; Sherar, L.B.; Elferink-Gemser, M.T.; Malina, R.M. Longitudinal predictors of aerobic performance in adolescent soccer players. Medicina 2012, 48, 410–416. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23128461 (accessed on 3 March 2021).
- Marini, E.; Campa, F.; Buffa, R.; Stagi, S.; Matias, C.N.; Toselli, S.; Sardinha, L.B.; Silva, A.M. Phase angle and bioelectrical impedance vector analysis in the evaluation of body composition in athletes. Clin. Nutr. 2020, 39, 447–454. [Google Scholar] [CrossRef]
- Sardinha, L.B. Physiology of exercise and phase angle: Another look at BIA. Eur. J. Clin. Nutr. 2018, 72, 1323–1327. [Google Scholar] [CrossRef] [PubMed]
- Mascherini, G.; Gatterer, H.; Lukaski, H.; Burtscher, M.; Galanti, G. Changes in hydration, body-cell mass and endurance performance of professional soccer players through a competitive season. J. Sports Med. Phys. Fit. 2015, 55, 749–755. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25303072 (accessed on 3 March 2021).
- Koury, J.C.; Trugo, N.M.F.; Torres, A.G. Phase angle and bioelectrical impedance vectors in adolescent and adult male athletes. Int. J. Sports Physiol. Perform. 2014, 9, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Campa, F.; Silva, A.M.; Iannuzzi, V.; Mascherini, G.; Benedetti, L.; Toselli, S. The role of somatic maturation on bioimpedance patterns and body composition in male elite youth soccer players. Int. J. Environ. Res. Public Health 2019, 16, 4711. [Google Scholar] [CrossRef]
- Micheli, M.L.; Pagani, L.; Marella, M.; Gulisano, M.; Piccoli, A.; Angelini, F.; Burtscher, M.; Gatterer, H. Bioimpedance and im-pedance vector patterns as predictors of league level in male soccer players. Int. J. Sports Physiol. Perform. 2014, 9, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Nabuco, H.C.G.; Silva, A.M.; Sardinha, L.B.; Rodrigues, F.B.; Tomeleri, C.M.; Ravagnani, F.C.P.; Cyrino, E.S.; Ravagnani, C.F.C. Phase Angle is Moderately Associated with Short-term Maximal Intensity Efforts in Soccer Players. Int. J. Sports Med. 2019, 40, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Impellizzeri, F.M.; Rampinini, E.; Castagna, C.; Bishop, D.; Bravo, D.F.; Tibaudi, A.; Wisloff, U. Validity of a re-peated-sprint test for football. Int. J. Sports Med. 2008, 29, 899–905. [Google Scholar] [CrossRef]
- Melchiorri, G.; Viero, V.; Sorge, R.; Triossi, T.; Campagna, A.; Volpe, S.L.; Lecis, D.; Tancredi, V.; Andreoli, A. Body composition analysis to study long-term training effects in elite male water polo athletes. J. Sports Med. Phys. Fit. 2017, 58, 1269–1274. [Google Scholar] [CrossRef]
- Andreoli, A.; Melchiorri, G.; Brozzi, M.; Di Marco, A.; Volpe, S.L.; Garofano, P.; Di Daniele, N.; De Lorenzo, A. Effect of different sports on body cell mass in highly trained athletes. Acta Diabetol. 2003, 40, s122–s125. [Google Scholar] [CrossRef]
- Campa, F.; Matias, C.N.M.; Teixeira, F.J.; Reis, J.F.; Valamatos, M.J.; Toselli, S.; Monteiro, C.P. Leucine metabolites do not induce changes in phase angle, bioimpedance vector analysis patterns, and strength in resistance trained men. Appl. Physiol. Nutr. Metab. 2020. [Google Scholar] [CrossRef] [PubMed]
- Francisco, R.; Matias, C.N.; Santos, D.A.; Campa, F.; Minderico, C.S.; Rocha, P.; Heymsfield, S.B.; Lukaski, H.; Sardinha, L.B.; Silva, A.M. The Predictive Role of Raw Bioelectrical Impedance Parameters in Water Compartments and Fluid Distribution Assessed by Dilution Techniques in Athletes. Int. J. Environ. Res. Public Health 2020, 17, 759. [Google Scholar] [CrossRef]
- Crabtree, N.J.; Arabi, A.; Bachrach, L.K.; Fewtrell, M.; Fuleihan, G.E.-H.; Kecskemethy, H.H.; Jaworski, M.; Gordon, C.M. Dual-Energy X-Ray Absorptiometry Interpretation and Reporting in Children and Adolescents: The Revised 2013 ISCD Pediatric Official Positions. J. Clin. Densitom. 2014, 17, 225–242. [Google Scholar] [CrossRef]
- Crabtree, N.; Kibirige, M.; Fordham, J.; Banks, L.; Muntoni, F.; Chinn, D.; Boivin, C.; Shaw, N. The relationship between lean body mass and bone mineral content in paediatric health and disease. Bone 2004, 35, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.; Stobäus, N.; Pirlich, M.; Bosy-Westphal, A. Bioelectrical phase angle and impedance vector analysis—Clinical relevance and applicability of impedance parameters. Clin. Nutr. 2012, 31, 854–861. [Google Scholar] [CrossRef]
- De Castro, J.A.C.; De Lima, L.R.A.; Silva, D.A.S. Accuracy of octa-polar bioelectrical impedance analysis for the assessment of total and appendicular body composition in children and adolescents with HIV: Comparison with dual ener-gy X-ray absorptiometry and air displacement plethysmography. J. Hum. Nutr. Diet. 2018, 31, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Kyle, U.G. Bioelectrical impedance analysis?part I: Review of principles and methods. Clin. Nutr. 2004, 23, 1226–1243. [Google Scholar] [CrossRef] [PubMed]
- Malina, R.M.; Bouchard, C. Growth, Maturation, and Physical Activity. Med. Sci. Sports Exerc. 1992, 24, 841. [Google Scholar] [CrossRef]
- Moya-Ramon, M.; Nakamura, F.Y.; Teixeira, A.S.; Granacher, U.; Santos-Rosa, F.J.; Sanz-Rivas, D.; Fernandez-Fernandez, J. Effects of Resisted vs. Conventional Sprint Training on Physical Fitness in Young Elite Tennis Players. J. Hum. Kinet. 2020, 73, 181–192. [Google Scholar] [CrossRef]
- Teixeira, A.S.; da Silva, J.F.; Carminatti, L.J.; Dittrich, N.; Castagna, C.; Guglielmo, L.G. Reliability and Validity of the Carminatti’s Test for Aerobic Fitness in Youth Soccer Players. J. Strength Cond. Res. 2014, 28, 3264–3273. [Google Scholar] [CrossRef] [PubMed]
- Haugen, T.; Buchheit, M. Sprint Running Performance Monitoring: Methodological and Practical Considerations. Sports Med. 2015, 46, 641–656. [Google Scholar] [CrossRef] [PubMed]
- Altmann, S.; Spielmann, M.; Engel, F.A.; Neumann, R.; Ringhof, S.; Oriwol, D.; Haertel, S. Validity of Single-Beam Timing Lights at Different Heights. J. Strength Cond. Res. 2017, 31, 1994–1999. [Google Scholar] [CrossRef]
- Hopkins, W.G.; Marshall, S.W.; Batterham, A.M.; Hanin, J. Progressive Statistics for Studies in Sports Medicine and Exercise Science. Med. Sci. Sports Exerc. 2009, 41, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Toselli, S.; Marini, E.; Latessa, P.M.; Benedetti, L.; Campa, F. Maturity Related Differences in Body Composition Assessed by Classic and Specific Bioimpedance Vector Analysis among Male Elite Youth Soccer Players. Int. J. Environ. Res. Public Health 2020, 17, 729. [Google Scholar] [CrossRef] [PubMed]
- Hotelling, H. Multivariate Quality Control. Techniques of Statistical Analysis; McGraw-Hill: New York, NY, USA, 1947. [Google Scholar]
- Mathias-Genovez, M.G.; Oliveira, C.C.; Camelo, J.S.; Del Ciampo, L.A.; Monteiro, J.P. Bioelectrical Impedance of Vectorial Analysis and Phase Angle in Adolescents. J. Am. Coll. Nutr. 2015, 35, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Koury, J.C.; De Oliveira-Junior, A.V.; Portugal, M.R.C.; Oliveira, K.D.J.F.D.; Donangelo, C.M. Bioimpedance parameters in adolescent athletes in relation to bone maturity and biochemical zinc indices. J. Trace Elements Med. Biol. 2018, 46, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Faude, O.; Koch, T.; Meyer, T. Straight sprinting is the most frequent action in goal situations in professional foot-ball. J. Sports Sci. 2012, 30, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Pollastri, L.; Lanfranconi, F.; Tredici, G.; Burtscher, M.; Gatterer, H. Body Water Status and Short-term Maximal Power Output during a Multistage Road Bicycle Race (Giro d’Italia 2014). Int. J. Sports Med. 2015, 37, 329–333. [Google Scholar] [CrossRef]
- De Lorenzo, A.; Andreoli, A.; Matthie, J.; Withers, P. Predicting body cell mass with bioimpedance by using theoretical methods: A technological review. J. Appl. Physiol. 1997, 82, 1542–1558. [Google Scholar] [CrossRef]
- Silva, A.; Fields, D.A.; Heymsfield, S.B.; Sardinha, L.B. Body Composition and Power Changes in Elite Judo Athletes. Int. J. Sports Med. 2010, 31, 737–741. [Google Scholar] [CrossRef]
- Matias, C.N.; Monteiro, C.P.; Santos, D.A.; Martins, F.; Silva, A.M.; Laires, M.J.; Sardinha, L.B. Magnesium and phase angle: A prognostic tool for monitoring cellular integrity in judo athletes. Magnes. Res. 2015, 28, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.M.; Matias, C.N.; Nunes, C.L.; Santos, D.A.; Marini, E.; Lukaski, H.C.; Sardinha, L.B. Lack of agreement of in vivo raw bioimpedance measurements obtained from two single and multi-frequency bioelectrical impedance devic-es. Eur. J. Clin. Nutr. 2019, 73, 1077–1083. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 62) | U-13 [1] (n = 14) | U-15 [2] (n = 25) | U-17 [3] (n = 23) | ANOVA | Effect Size (95%CI) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | F | p | 3 vs. 1 | 3 vs. 2 | 2 vs. 1 | |
| Age (years) | 15.0 (1.4) | 12.7 (0.2) | 14.9 (0.2) | 16.5 (0.5) | - | - | - | - | - |
| Body mass (kg) | 62.7 (11.2) | 45.5 (6.3) * † | 65.8 (6.4) § | 69.7 (5.2) | 77.2 | <0.01 | 4.30 (3.04; 5.37) Very large | 0.67 (0.07; 1.24) Moderate | 3.19 (2.17; 4.08) Very large |
| Height (cm) | 172.8 (11.6) | 155.6 (6.6) * † | 175.9 (7.2) | 179.8 (6.0) | 61.8 | <0.01 | 3.80 (2.71; 4.89) Very large | 0.59 (0.00; 1.15) Small | 2.90 (1.93; 3.75) Very large |
| Body fat (kg) | 7.7 (2.7) | 7.5 (1.6) | 8.2 (3.0) | 7.2 (2.8) | 0.8 | 0.20 | −0.12 (−0.79; 0.54) Trivial | −0.34 (−0.91; 0.23) Small | 0.27 (−0.39; 0.92) Small |
| LSTM (kg) | 52.6 (10.1) | 38.2 (8.5) † | 54.9 (4.3) | 58.9 (6.5) | 49.9 | 0.01 | 2.83 (1.86; 3.69) Very large | 0.73 (0.14; 1.30) Moderate | 2.73 (1.79; 3.56) Very large |
| Resistance (ohms) | 485.6 (47.1) | 518.0 (54.3) * | 471.6 (40.7) | 481.0 (41.1) | 5.1 | 0.03 | −0.80 (−1.47; −0.09) Moderate | 0.23 (−0.34; 0.79) Small | −1.01 (−1.68; −0.30) Moderate |
| Reactance (ohms) | 51.4 (5.4) | 47.4 (3.0) † | 50.9 (4.1) § | 54.6 (6.0) | 10.0 | <0.01 | 1.41 (0.65; 2.12) Large | 0.73 (0.13; 1.30) Moderate | 0.93 (0.23; 1.60) Moderate |
| Phase angle (degrees) | 6.1 (0.6) | 5.2 (0.4) * † | 6.2 (0.4) | 6.5 (0.6) | 26.8 | <0.01 | 2.43 (1.52; 3.24) Very Large | 0.59 (0.01; 1.16) Small | 2.50 (1.60; 3.30) Very large |
| Standing long jump (cm) | 226.9 (22.6) | 194.4 (11.7) * † | 233.2 (14.6) | 239.4 (15.2) | 52.7 | <0.01 | 3.21 (2.17; 4.12) Very Large | 0.42 (−0.16; 0.98) Small | 2.84 (1.88; 3.68) Very large |
| 10 m sprint time (s) | 1.7 (0.1) | 1.8 (0.1) * † | 1.7 (0.1) | 1.6 (0.1) | 5.4 | <0.01 | −2.00 (−2.76; −1.16) Large | −1.00 (−1.58; −0.38) Moderate | −1.00 (−1.67; −0.29) Moderate |
| 30 m sprint time (s) | 4.3 (0.3) | 4.8 (0.3) * † | 4.2 (0.2) | 4.1 (0.2) | 45.1 | <0.01 | −2.89 (−3.76; −1.90) Very large | −0.50 (−1.07; 0.08) Small | −2.50 (−3.30; −1.60) Very large |
| PST-CAR (km·h−1) | 15.7 (1.4) | 13.7 (0.8) * † | 15.6 (0.7) § | 17.0 (0.6) | 88.3 | <0.01 | 4.84 (3.47; 6.01) Very large | 2.14 (1.40; 2.81) Very large | 2.58 (1.66; 3.39) Very large |
| RSAmean (s) | 7.4 (0.5) | 8.2 (0.3) *† | 7.3 (0.2) § | 7.1 (0.2) | 83.6 | <0.01 | −4.55 (−5.66; −3.24) Very large | −1.00 (−1.58; −0.38) Moderate | −3.75 (−4.72; −2.63) Very large |
| RSAbest (s) | 7.0 (0.4) | 7.7 (0.4) *† | 7.0 (0.1) § | 6.7 (0.2) | 50.9 | <0.01 | −3.44 (−4.38; −2.35) Very large | −1.92 (−2.57; −1.21) Large | −2.80 (−3.63; −1.85) Very large |
| β (95%CI) | β Standardized | Adjusted R2 | p | |
|---|---|---|---|---|
| Standing long jump | ||||
| Phase angle ** | 17.717 (10.464; 24.970) | 0.533 | 0.272 | <0.001 |
| Model 1 | 5.485 (4.778; 19.227) | 0.165 | 0.463 | 0.181 |
| Model 2 | 5.093 (−2.983; 13.171) | 0.153 | 0.470 | 0.212 |
| Model 3 | 6.217 (−1.418; 13.853) | 0.187 | 0.531 | 0.109 |
| 10 m sprint time | ||||
| Phase angle ** | −0.799 (−0.119; −0.404) | −0.466 | 0.204 | <0.001 |
| Model 1 * | −0.060 (−0.110; −0.009) | −0.351 | 0.211 | 0.021 |
| Model 2 * | −0.059 (−0.111; −0.008) | −0.349 | 0.201 | 0.023 |
| Model 3 * | −0.065 (−0.115; −0.014) | −0.379 | 0.240 | 0.012 |
| 30 m sprint time | ||||
| Phase angle ** | −0.331 (−0.416; −0.247) | −0.712 | 0.499 | <0.001 |
| Model 1 ** | −0.206 (−0.305; −0.108) | −0.443 | 0.598 | <0.001 |
| Model 2 ** | −0.200 (−0.296; −0.103) | −0.429 | 0.615 | <0.001 |
| Model 3 ** | −0.204 (−0.301; −0.107) | −0.438 | 0.614 | <0.001 |
| PST-CAR | ||||
| Phase angle ** | 1.295 (0.859; 1730) | 0.609 | 0.360 | <0.001 |
| Model 1 ** | 0.263 (−0.123; 0.650) | 0.124 | 0.701 | <0.001 |
| Model 2 | 0.248 (−0.139; 0.635) | 0.116 | 0.702 | 0.205 |
| Model 3 | 0.270 (0.118; 0.658) | 0.116 | 0.703 | 0.169 |
| RSAmean | ||||
| Phase angle ** | −0.523 (−0.654; −0.392) | −0.718 | 0.508 | <0.001 |
| Model 1 ** | −0.236 (−0.362; −0.110) | −0.324 | 0.731 | <0.001 |
| Model 2 ** | −0.229 (−0.354; −0.104) | −0.314 | 0.738 | 0.001 |
| Model 3 ** | −0.238 (−0.362; −0.113) | −0.362 | 0.742 | <0.001 |
| RSAbest | ||||
| Phase angle ** | −0.434 (−0.568; −0.300) | −0.641 | 0.401 | <0.001 |
| Model 1 * | −0.154 (−0.288; −0.205) | −0.228 | 0.651 | 0.025 |
| Model 2 * | −0.154 (−0.289; −0.018) | −0.227 | 0.640 | 0.027 |
| Model 3 * | −0.162 (−0.298; −0.026) | −0.239 | 0.644 | 0.020 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, P.C.; Teixeira, A.S.; Guglielmo, L.G.A.; Francisco, J.S.; Silva, D.A.S.; Nakamura, F.Y.; Lima, L.R.A.d. Phase Angle Is Related to 10 m and 30 m Sprint Time and Repeated-Sprint Ability in Young Male Soccer Players. Int. J. Environ. Res. Public Health 2021, 18, 4405. https://doi.org/10.3390/ijerph18094405
Martins PC, Teixeira AS, Guglielmo LGA, Francisco JS, Silva DAS, Nakamura FY, Lima LRAd. Phase Angle Is Related to 10 m and 30 m Sprint Time and Repeated-Sprint Ability in Young Male Soccer Players. International Journal of Environmental Research and Public Health. 2021; 18(9):4405. https://doi.org/10.3390/ijerph18094405
Chicago/Turabian StyleMartins, Priscila Custódio, Anderson Santiago Teixeira, Luiz Guilherme ANTONACCI Guglielmo, Juliana Sabino Francisco, Diego Augusto Santos Silva, Fábio Yuzo Nakamura, and Luiz Rodrigo Augustemak de Lima. 2021. "Phase Angle Is Related to 10 m and 30 m Sprint Time and Repeated-Sprint Ability in Young Male Soccer Players" International Journal of Environmental Research and Public Health 18, no. 9: 4405. https://doi.org/10.3390/ijerph18094405
APA StyleMartins, P. C., Teixeira, A. S., Guglielmo, L. G. A., Francisco, J. S., Silva, D. A. S., Nakamura, F. Y., & Lima, L. R. A. d. (2021). Phase Angle Is Related to 10 m and 30 m Sprint Time and Repeated-Sprint Ability in Young Male Soccer Players. International Journal of Environmental Research and Public Health, 18(9), 4405. https://doi.org/10.3390/ijerph18094405







