Histomorphometric Comparison between Two Types of Acellular Dermal Matrix Grafts: A Mini Pig Animal Model Study
Abstract
1. Introduction
2. Materials and Methods
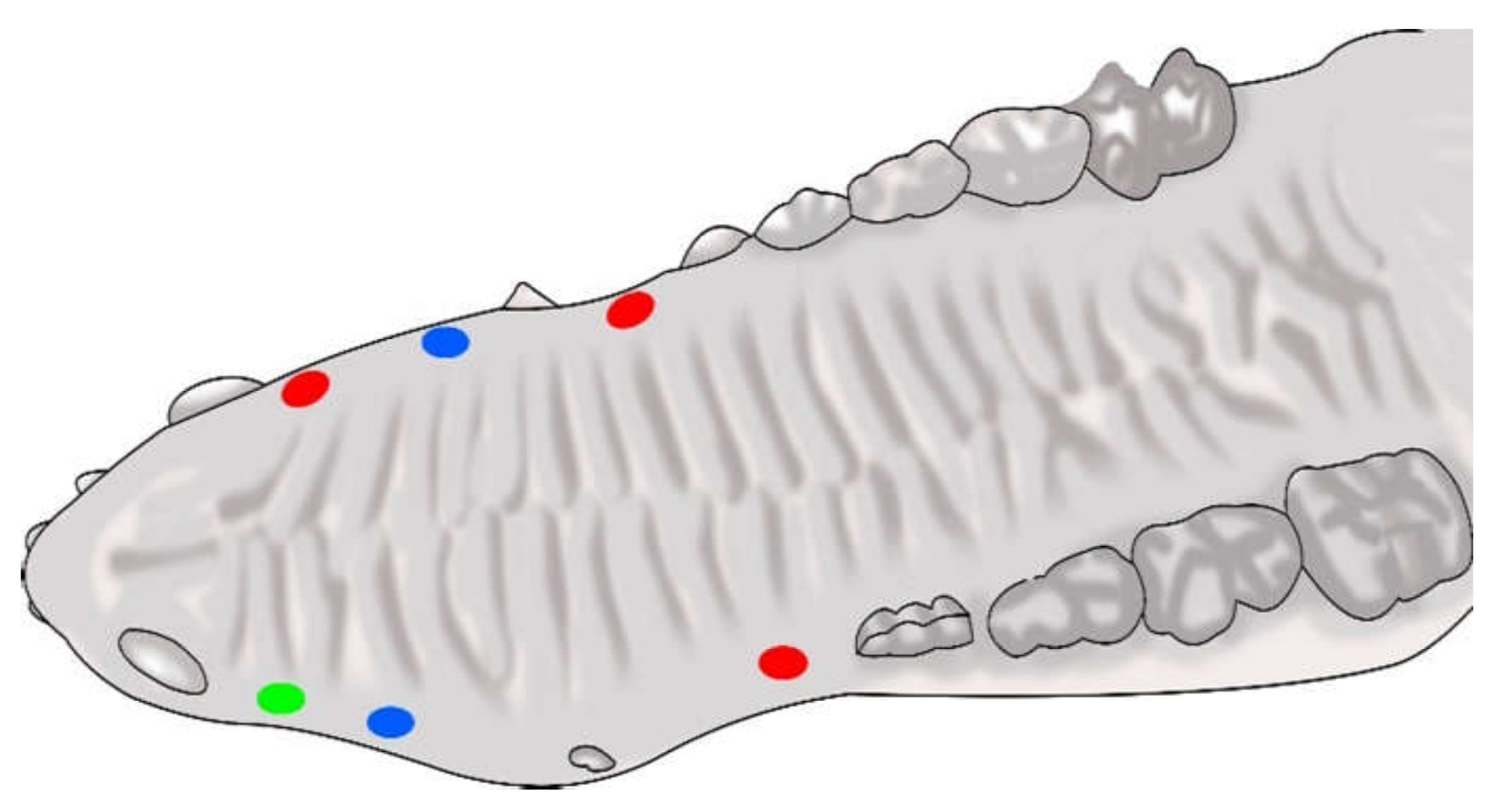
2.1. Pre-Surgical Procedure
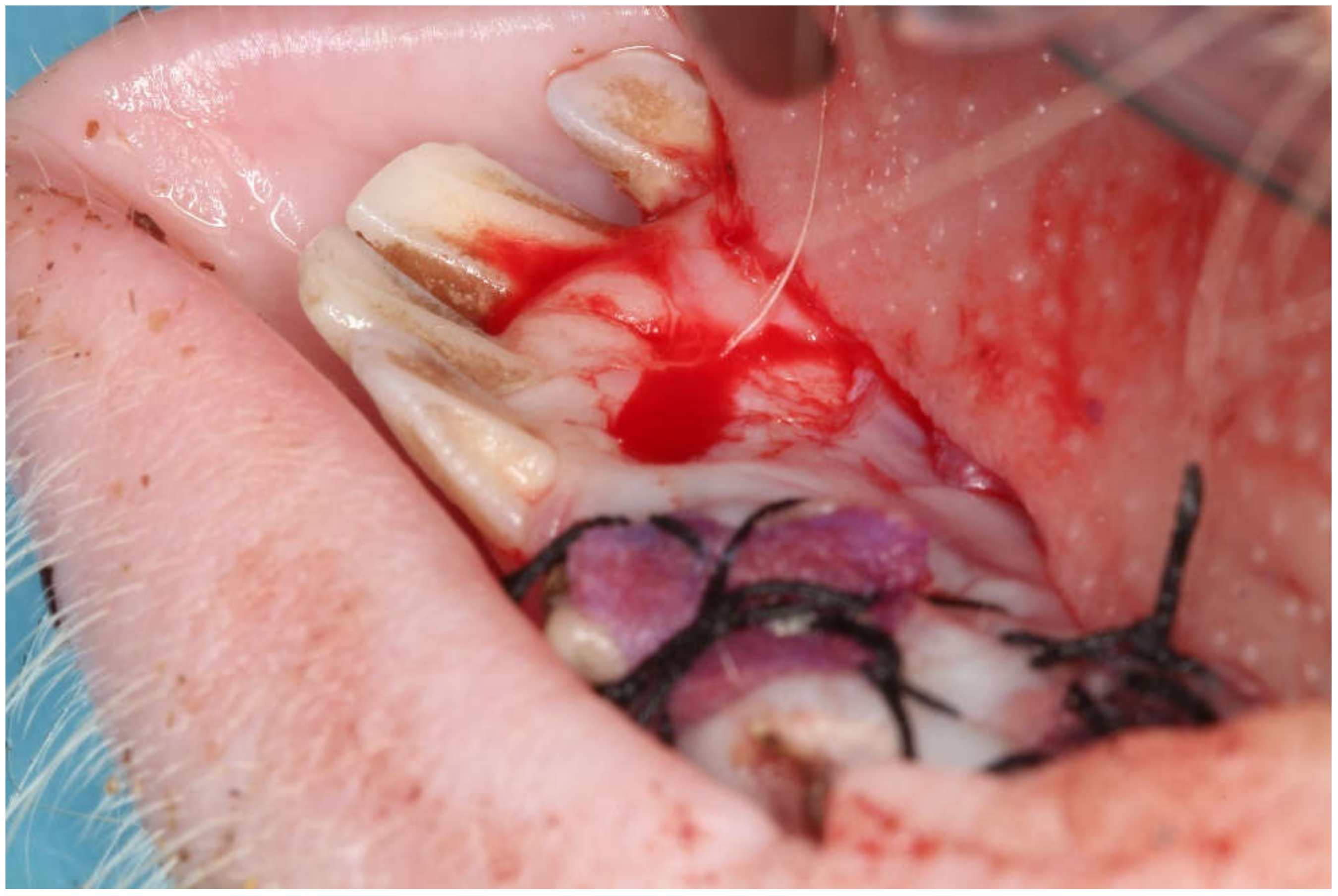
2.2. Surgical Intervention
2.3. Surgical Biopsy
2.4. Euthanasia
2.5. Histological Preparation
2.6. Laboratory Process
2.6.1. Matrix Staining
2.6.2. Histomorphometric Analysis
2.7. Statistical Analysis
3. Results
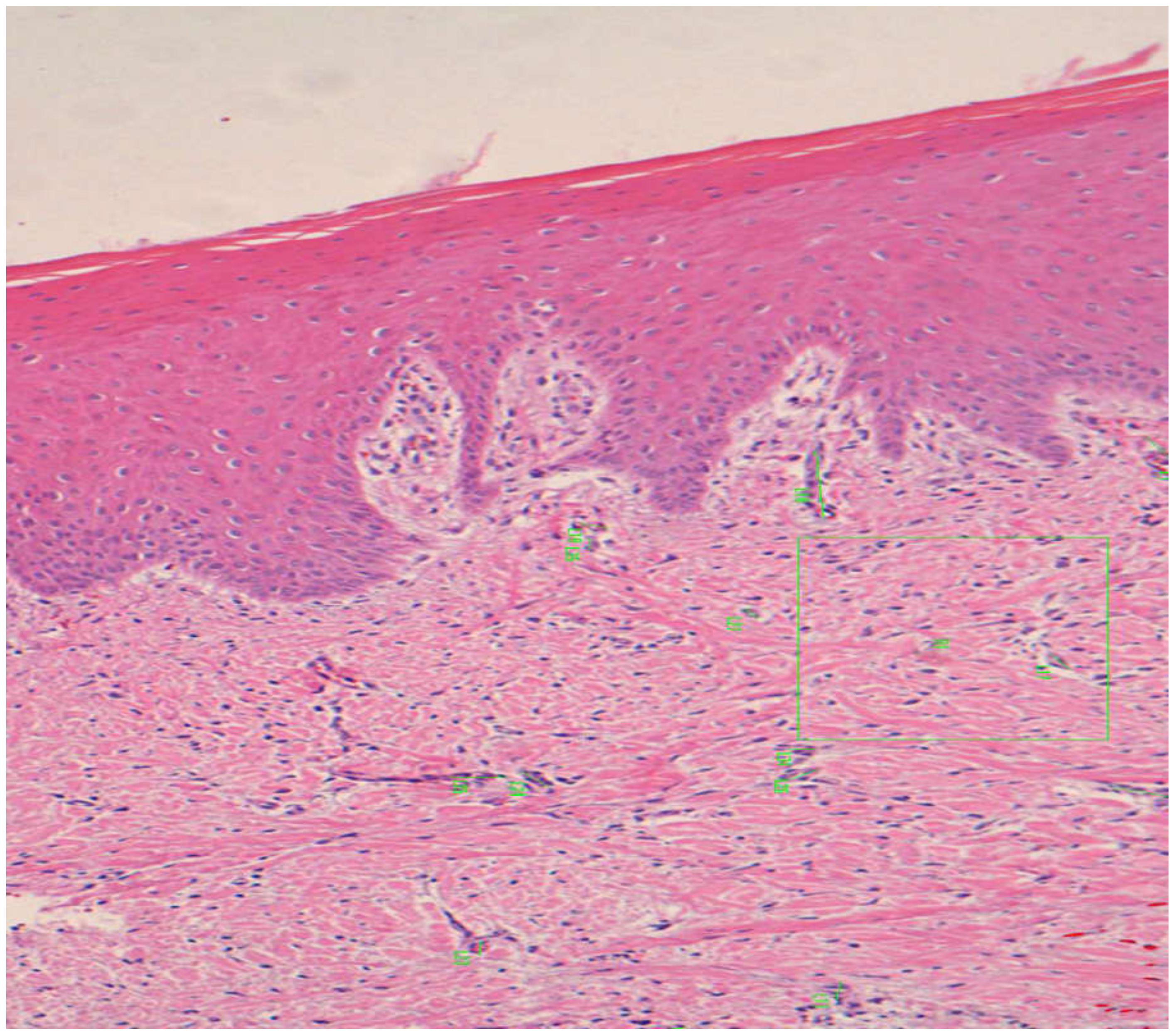
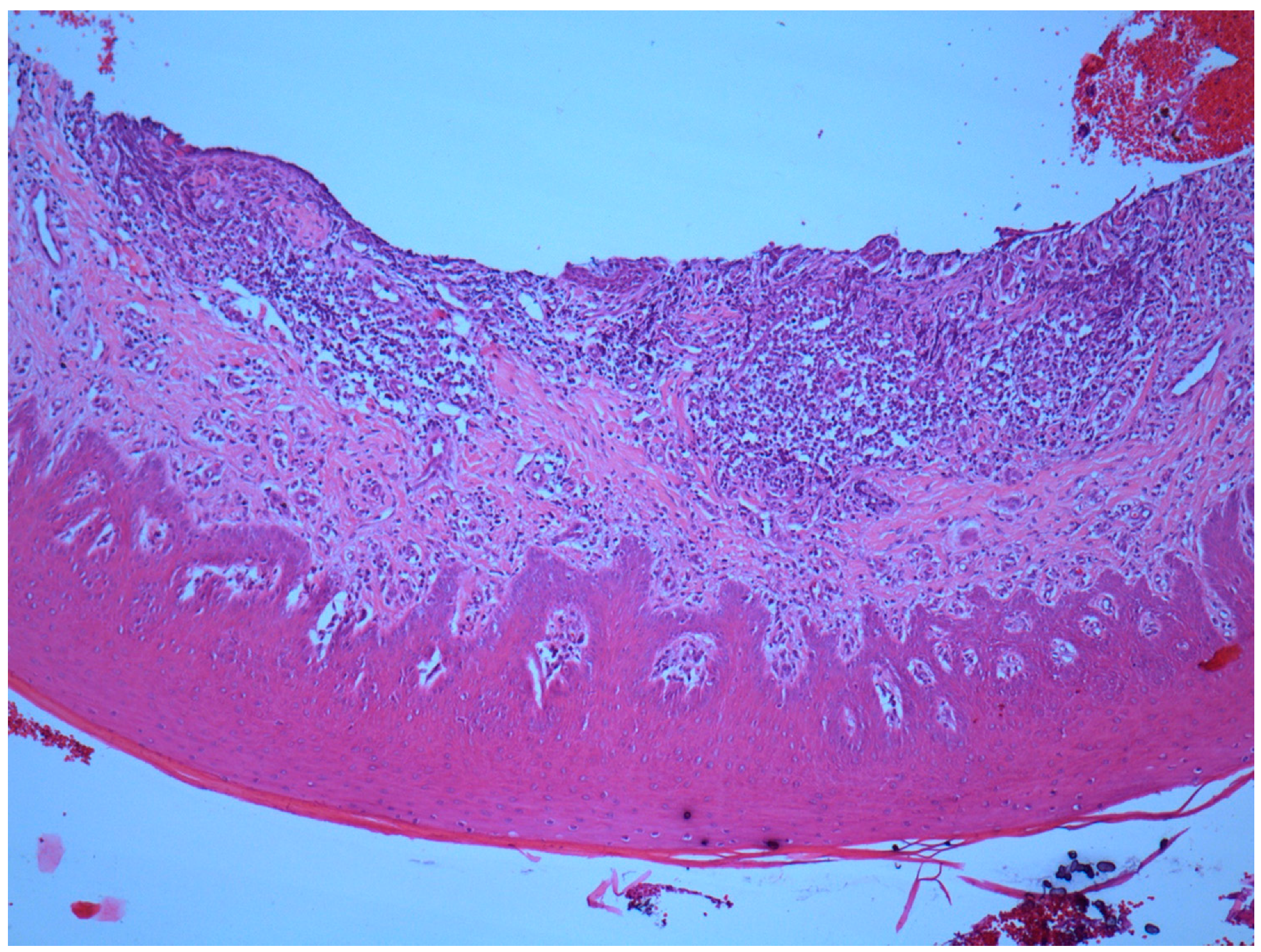
3.1. Epithelial Thickness
3.2. Angiogenesis
3.3. Keratinized Layer Thickness
3.4. Cellularity
3.5. Inflammatory Infiltrate
3.6. Matrix Resorption
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Mehta, P.; Lim, L.P. The width of the attached gingiva—Much ado about nothing? J. Dent. 2010, 38, 517–525. [Google Scholar] [CrossRef]
- Haeri, A.; Serio, F.G. Mucogingival surgical procedures: A review of the literature. Quintessence Int. (Berlin Ger. 1985) 1999, 30, 475–483. [Google Scholar]
- Maynard, J.G.; Wilson, R.D.K. Physiologic Dimensions of the Periodontium Significant to the Restorative Dentist. J. Periodontol. 1979, 50, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Consensus Report Mucogingival Therapy. Ann Periodontol. 1996, 1, 702–706. [CrossRef]
- Lang, N.P.; Löe, H. The Relationship Between the Width of Keratinized Gingiva and Gingival Health. J. Periodontol. 1972, 43, 623–627. [Google Scholar] [CrossRef]
- Wennstrom, J.L. Lack of association between width of attached gingiva and development of soft tissue recession. A 5-year longitudinal study. J. Clin. Periodontol. 1987, 14, 181–184. [Google Scholar] [CrossRef]
- Wennstrom, J.; Lindhe, J.; Nyman, S. Role of keratinized gingiva for gingival health. Clinical and histologic study of normal and regenerated gingival tissue in dogs. J. Clin. Periodontol. 1981, 8, 311–328. [Google Scholar] [CrossRef]
- Han, T.J.; Klokkevold, P.R.; Takei, H.H. Strip gingival autograft used to correct mucogingival problems around implants. Int. J. Periodontics Restor. Dent. 1995, 15, 404–411. [Google Scholar]
- Adell, R.; Lekholm, U.; Rockler, B.; Brånemark, P.-I.; Lindhe, J.; Eriksson, B.; Sbordone, L. Marginal tissue reactions at osseointegrated titanium fixtures. Int. J. Oral Maxillofac. Surg. 1986, 15, 39–52. [Google Scholar] [CrossRef]
- Bower, R.C.; Radny, N.R.; Wall, C.D.; Henry, P.J. Clinical and microscopic findings in edentulous patients 3 years after incorporation of osseointegrated implant-supported bridgework. J. Clin. Periodontol. 1989, 16, 580–587. [Google Scholar] [CrossRef] [PubMed]
- Wennström, J.L.; Bengazi, F.; Lekholm, U. The influence of the masticatory mucosa on the peri-implant soft tissue condition: Soft tissue conditions at oral implants. Clin. Oral Implant. Res. 1994, 5, 1–8. [Google Scholar] [CrossRef]
- Batenhorst, K.F.; Bowers, G.M.; Williams, J.E. Tissue Changes Resulting from Facial Tipping and Extrusion of Incisors in Monkeys. J. Periodontol. 1974, 45, 660–668. [Google Scholar] [CrossRef] [PubMed]
- Foushee, D.G.; Moriarty, J.D.; Simpson, D.M. Effects of Mandibular Orthognathic Treatment on Mucogingival Tissues. J. Periodontol. 1985, 56, 727–733. [Google Scholar] [CrossRef]
- Coatoam, G.W.; Behrents, R.G.; Bissada, N.F. The Width of Keratinized Gingiva During Orthodontic Treatment: Its Significance and Impact on Periodontal Status. J. Periodontol. 1981, 52, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Scarano, A.; Barros, R.R.M.; Iezzi, G.; Piattelli, A.; Novaes, A.B., Jr. Acellular Dermal Matrix Graft for Gingival Augmentation: A Preliminary Clinical, Histologic, and Ultrastructural Evaluation. J. Periodontol. 2009, 80, 253–259. [Google Scholar] [CrossRef]
- Langer, B.; Langer, L. Subepithelial Connective Tissue Graft Technique for Root Coverage. J. Periodontol. 1985, 56, 715–720. [Google Scholar] [CrossRef] [PubMed]
- Felipe, M.E.M.; Andrade, P.F.; Grisi, M.F.; Souza, S.L.; Taba, M.; Palioto, D.B.; Novaes, A.B.; Mario, J.T. Comparison of Two Surgical Procedures for Use of the Acellular Dermal Matrix Graft in the Treatment of Gingival Recessions: A Randomized Controlled Clinical Study. J. Periodontol. 2007, 78, 1209–1217. [Google Scholar] [CrossRef]
- Harris, R.J. Root Coverage with a Connective Tissue with Partial Thickness Double Pedicle Graft and an Acellular Dermal Matrix Graft: A Clinical and Histological Evaluation of a Case Report. J. Periodontol. 1998, 69, 1305–1311. [Google Scholar] [CrossRef]
- Klingsberg, J. Preserved Sclera in Periodontal Surgery. J. Periodontol. 1972, 43, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Bartolucci, E.G. A Clinical Evaluation of Freeze-Dried Homologous Dura Mater as a Periodontal Free Graft Material: Study in Humans. J. Periodontol. 1981, 52, 354–361. [Google Scholar] [CrossRef]
- Harris, R.J. A Comparative Study of Root Coverage Obtained with Guided Tissue Regeneration Utilizing a Bioabsorbable Membrane Versus the Connective Tissue with Partial-Thickness Double Pedicle Graft. J. Periodontol. 1997, 68, 779–790. [Google Scholar] [CrossRef] [PubMed]
- Aichelmann-Reidy, M.E.; Yukna, R.A.; Evans, G.H.; Nasr, H.F.; Mayer, E.T. Clinical Evaluation of Acellular Allograft Dermis for the Treatment of Human Gingival Recession. J. Periodontol. 2001, 72, 998–1005. [Google Scholar] [CrossRef]
- Gapski, R.; Parks, C.A.; Wang, H.-L. Acellular Dermal Matrix for Mucogingival Surgery: A Meta-Analysis. J. Periodontol. 2005, 76, 1814–1822. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, D. Use of an acellular allograft dermal matrix (AlloDerm) in the management of full-thickness burns. Burns 1995, 21, 243–248. [Google Scholar] [CrossRef]
- Cevallos, C.A.R.; de Resende, D.R.B.; Damante, C.A.; Sant’Ana, A.C.P.; de Rezende, M.L.R.; Greghi, S.L.A.; Zangrando, M.S.R. Free gingival graft and acellular dermal matrix for gingival augmentation: A 15-year clinical study. Clin. Oral Investig. 2020, 24, 1197–1203. [Google Scholar] [CrossRef]
- Agarwal, C.; Tarun Kumar, A.; Mehta, D. Comparative evaluation of free gingival graft and AlloDerm® in enhancing the width of attached gingival: A clinical study. Contemp. Clin. Dent. 2015, 6, 483. [Google Scholar] [CrossRef] [PubMed]
- Henderson, R.D.; Greenwell, H.; Drisko, C.; Regennitter, F.J.; Lamb, J.W.; Mehlbauer, M.J.; Goldsmith, L.J.; Rebitski, G. Predictable Multiple Site Root Coverage Using an Acellular Dermal Matrix Allograft. J. Periodontol. 2001, 72, 571–582. [Google Scholar] [CrossRef]
- Hart, C.E.; Loewen-Rodriguez, A.; Lessem, J. Dermagraft: Use in the Treatment of Chronic Wounds. Adv. Wound Care 2012, 1, 138–141. [Google Scholar] [CrossRef] [PubMed]
- Gentzkow, G.D.; Iwasaki, S.D.; Hershon, K.S.; Mengel, M.; Prendergast, J.J.; Ricotta, J.J.; Steed, D.P.; Lipkin, S. Use of Dermagraft, a Cultured Human Dermis, to Treat Diabetic Foot Ulcers. Diabetes Care 1996, 19, 350–354. [Google Scholar] [CrossRef]
- Macadam, S.A.; Lennox, P.A. Acellular dermal matrices: Use in reconstructive and aesthetic breast surgery. Can. J. Plast. Surg. 2012, 20, 75–89. [Google Scholar] [CrossRef]
- Rothamel, D.; Benner, M.; Fienitz, T.; Happe, A.; Kreppel, M.; Nickenig, H.-J.; Zöller, J.E. Biodegradation pattern and tissue integration of native and cross-linked porcine collagen soft tissue augmentation matrices—An experimental study in the rat. Head Face Med. 2014, 10, 10. [Google Scholar] [CrossRef]
- Eeckhout, C.; Bouckaert, E.; Verleyen, D.; De Bruyckere, T.; Cosyn, J. A 3-Year Prospective Study on a Porcine-Derived Acellular Collagen Matrix to Re-Establish Convexity at the Buccal Aspect of Single Implants in the Molar Area: A Volumetric Analysis. J. Clin. Med. 2020, 9, 1568. [Google Scholar] [CrossRef]
- Puišys, A.; Jonaitis, A.; Vindašiūtė, E.; Žukauskas, S.; Linkevičius, T. The use of a porcine-derived collagen matrix for vertical soft tissue augmentation. A case report. Stomatologija 2019, 21, 125–128. [Google Scholar]
- Badylak, S.F.; Freytes, D.O.; Gilbert, T.W. Extracellular matrix as a biological scaffold material: Structure and function. Acta Biomater. 2009, 5, 1–13. [Google Scholar] [CrossRef]
- Luczyszyn, S.M.; Grisi, M.F.M.; Novaes, A.B.; Palioto, D.B.; Souza, S.L.S.; Taba, M. Histologic analysis of the acellular dermal matrix graft incorporation process: A pilot study in dogs. Int. J. Periodontics Restor. Dent. 2007, 27, 341–347. [Google Scholar]
- Artzi, Z.; Wasersprung, N.; Weinreb, M.; Steigmann, M.; Prasad, H.S.; Tsesis, I. Effect of Guided Tissue Regeneration on Newly Formed Bone and Cementum in Periapical Tissue Healing after Endodontic Surgery: An In Vivo Study in the Cat. J. Endod. 2012, 38, 163–169. [Google Scholar] [CrossRef]
- Moyer, H.R.; Hart, A.M.; Yeager, R.J.; Losken, A. A Histological Comparison of Two Human Acellular Dermal Matrix Products in Prosthetic-Based Breast Reconstruction. Plast. Reconstr. Surg. Glob. Open 2017, 5, e1576. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.J. Cellular dermal matrix used for root coverage: 18-month follow-up observation. Int. J. Periodontics Restor. Dent. 2002, 22, 156–163. [Google Scholar]
- Nunez, J.; Caffesse, R.; Vignoletti, F.; Guerra, F.; Roman, F.S.; Sanz, M. Clinical and histological evaluation of an acellular dermal matrix allograft in combination with the coronally advanced flap in the treatment of miller class I recession defects: An experimental study in the mini-pig. J. Clin. Periodontol. 2009, 36, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Cummings, L.C.; Kaldahl, W.B.; Allen, E.P. Histologic Evaluation of Autogenous Connective Tissue and Acellular Dermal Matrix Grafts in Humans. J. Periodontol. 2005, 76, 178–186. [Google Scholar] [CrossRef]
- Sallum, E.A.; Nogueira-Filho, G.R.; Casati, M.Z.; Pimentel, S.P.; Saldanha, J.B.; Nociti, F.H. Coronally positioned flap with or without acellular dermal matrix graft in gingival recessions: A histometric study. Am. J. Dent. 2006, 19, 128–132. [Google Scholar]
- Schmitt, C.M.; Schlegel, K.A.; Gammel, L.; Moest, T. Gingiva thickening with a porcine collagen matrix in a preclinical dog model: Histological outcomes. J. Clin. Periodontol. 2019, 46, 1273–1281. [Google Scholar] [CrossRef]
- Kantarci, A.; Hasturk, H.; Van Dyke, T.E. Animal models for periodontal regeneration and peri-implant responses. Periodontol. 2000 2015, 68, 66–82. [Google Scholar] [CrossRef]
- Wei, P.-C.; Laurell, L.; Lingen, M.W.; Geivelis, M. Acellular Dermal Matrix Allografts to Achieve Increased Attached Gingiva. Part 2. A Histological Comparative Study. J. Periodontol. 2002, 73, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Del Amo, F.S.-L.; Rodriguez, J.C.; Asa’Ad, F.; Wang, H.-L. Comparison of two soft tissue substitutes for the treatment of gingival recession defects: An animal histological study. J. Appl. Oral Sci. 2019, 27, e20180584. [Google Scholar] [CrossRef] [PubMed]
- Blatt, S.; Burkhardt, V.; Kämmerer, P.W.; Pabst, A.M.; Sagheb, K.; Heller, M.; Al-Nawas, B.; Schiegnitz, E. Biofunctionalization of porcine-derived collagen matrices with platelet rich fibrin: Influence on angiogenesis in vitro and in vivo. Clin. Oral Investig. 2020, 24, 3425–3436. [Google Scholar] [CrossRef]
- Nica, C.; Lin, Z.; Sculean, A.; Asparuhova, M.B. Adsorption and Release of Growth Factors from Four Different Porcine-Derived Collagen Matrices. Materials 2020, 13, 2635. [Google Scholar] [CrossRef] [PubMed]
- Mak, K.; Manji, A.; Gallant-Behm, C.; Wiebe, C.; Hart, D.A.; Larjava, H.; Häkkinen, L. Scarless healing of oral mucosa is characterized by faster resolution of inflammation and control of myofibroblast action compared to skin wounds in the red Duroc pig model. J. Dermatol. Sci. 2009, 56, 168–180. [Google Scholar] [CrossRef] [PubMed]




| Variables | dADMG | mADMG | Control | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 15d (n = 39) | 45d (n = 20) | 90d (n = 25) | 15d (n = 29 | 45d (n = 18) | 90d (n = 20) | 15d (n = 5) | 45d (n = 8) | 90d (n = 10) | |
| Epithelial Thickness (μm) | 438.28 ± 159.41 | 270.75 ± 75.19 | 281.49 ± 63.66 | 330.10 ± 139.71 | 347.15 ± 65.69 | 279.56 ± 72.27 | 267.74 ± 63.26 | 489.10 ± 148.89 | 236.70 ± 82.94 |
| Angiogenesis (Vessel Size) | 34.15 ± 18.76 | 27.37 ± 15.27 | 33.45 ± 21.72 | 30.15 ± 17.35 | 40.41 ± 25.14 | 31.33 ± 22.05 | 24.64 ± 10.24 | 39.76 ± 24.72 | 38.34 ± 28.91 |
| Keratinized layer thickness (μm) | 25.27 ± 11.04 | 23.60 ± 6.13 | 19.46 ± 4.28 | 18.01 ± 6.19 | 20.07 ± 6.58 | 21.34 ± 7.95 | 19.57 ± 4.60 | 23.95 ± 6.64 | 17.25 ± 8.71 |
| Cellularity | 76.85 ± 20.74 | 96.95 ± 36.33 | 58.42 ± 35.77 | 73.77 ± 22.88 | 80.52 ± 33.62 | 69.65 ± 28.95 | 94.40 ± 12.99 | 58.62 ± 15.04 | 73.20 ± 27.86 |
| Inflammatory Infiltrate | 40.00 ± 14.14 | 10.50 ± 6.13 | 10.25 ± 16.50 | 51.66 ± 31.75 | 17.66 ± 14.64 | 17.66 ± 19.85 | 1.00 ± 0.01 | 5.00 ± 0.001 | 20.00 ± 21.21 |
| Matrix resorption | 71.25 | 52 | 12 | 81.66 | 63.75 | 0 | - | - | - |
| Outcome Variables | Groups | ANOVA F Value | p-Value | ||
|---|---|---|---|---|---|
| 15d | 45d | 90d | |||
| Epithelial Thickness | dADMG | 147.149 | 0.001 ** | ||
| mADMG | 23.193 | 0.001 ** | |||
| Control | 139.392 | 0.001 ** | |||
| Angiogenesis | dADMG | 19.66 | 0.001 ** | ||
| mADMG | 32.77 | 0.001 ** | |||
| Control | 12.89 | 0.001 ** | |||
| Keratinized layer Thickness | dADMG | 37.42 | 0.001 ** | ||
| mADMG | 5.43 | 0.005 * | |||
| Control | 24.81 | 0.001 ** | |||
| Cellularity | dADMG | 18.51 | 0.001 ** | ||
| mADMG | 1.46 | 0.23 | |||
| Control | 8.61 | 0.001 ** | |||
| Inflammatory infiltrate | dADMG | 6.88 | 0.015 * | ||
| mADMG | 2.14 | 0.19 | |||
| Control | 1.33 | 0.38 | |||
| Outcome Variables | Time Period | ANOVA F Value | p-Value | ||
|---|---|---|---|---|---|
| dADMG | mADMG | Control | |||
| Epithelial Thickness | 15 days | 44.07 | 0.001 ** | ||
| 45 days | 112.41 | 0.001 ** | |||
| 90 days | 16.33 | 0.001 ** | |||
| Angiogenesis | 15 days | 16.31 | 0.001 ** | ||
| 45 days | 47.34 | 0.001 ** | |||
| 90 days | 7.30 | 0.001 ** | |||
| Keratinized layer Thickness | 15 days | 21.65 | 0.001 ** | ||
| 45 days | 9.82 | 0.001 ** | |||
| 90 days | 14.67 | 0.001 ** | |||
| Cellularity | 15 days | 4.02 | 0.02 * | ||
| 45 days | 8.21 | 0.001 ** | |||
| 90 days | 2.12 | 0.12 | |||
| Inflammatory infiltrate | 15 days | 3.72 | 0.08 | ||
| 45 days | 1.12 | 0.38 | |||
| 90 days | 0.23 | 0.79 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aragoneses, J.; Suárez, A.; Rodríguez, C.; Aragoneses, J.M. Histomorphometric Comparison between Two Types of Acellular Dermal Matrix Grafts: A Mini Pig Animal Model Study. Int. J. Environ. Res. Public Health 2021, 18, 3881. https://doi.org/10.3390/ijerph18083881
Aragoneses J, Suárez A, Rodríguez C, Aragoneses JM. Histomorphometric Comparison between Two Types of Acellular Dermal Matrix Grafts: A Mini Pig Animal Model Study. International Journal of Environmental Research and Public Health. 2021; 18(8):3881. https://doi.org/10.3390/ijerph18083881
Chicago/Turabian StyleAragoneses, Javier, Ana Suárez, Cinthia Rodríguez, and Juan Manuel Aragoneses. 2021. "Histomorphometric Comparison between Two Types of Acellular Dermal Matrix Grafts: A Mini Pig Animal Model Study" International Journal of Environmental Research and Public Health 18, no. 8: 3881. https://doi.org/10.3390/ijerph18083881
APA StyleAragoneses, J., Suárez, A., Rodríguez, C., & Aragoneses, J. M. (2021). Histomorphometric Comparison between Two Types of Acellular Dermal Matrix Grafts: A Mini Pig Animal Model Study. International Journal of Environmental Research and Public Health, 18(8), 3881. https://doi.org/10.3390/ijerph18083881








