Mental Health during the COVID-19 Lockdown over the Christmas Period in Austria and the Effects of Sociodemographic and Lifestyle Factors
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Sample
2.3. Measures
2.4. Quality of Life (WHO-QOL BREF)
2.5. Well-Being (WHO-5)
2.6. Perceived Stress (PSS-10)
2.7. Depressive Symptoms (PHQ-9)
2.8. Anxiety (GAD-7)
2.9. Sleep Quality (ISI)
2.10. Other Variables
2.11. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nussbaumer-Streit, B.; Mayr, V.; Dobrescu, A.I.; Chapman, A.; Persad, E.; Klerings, I.; Wagner, G.; Siebert, U.; Christof, C.; Zachariah, C.; et al. Quarantine alone or in combination with other public health measures to control COVID-19: A rapid review. Cochrane Database Syst. Rev. 2020, 4, CD013574. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO/Europe Coronavirus Disease (COVID-19) Outbreak; WHO/Europe COVID-19; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Pieh, C.; Budimir, S.; Delgadillo, J.; Barkham, M.; Fontaine, J.R.J.; Probst, T. Mental health during COVID-19 lockdown in the United Kingdom. Psychosom. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.K.; Webster, R.K.; Smith, L.E.; Woodland, L.; Wessely, S.; Greenberg, N.; Rubin, G.J. The psychological impact of quarantine and how to reduce it: Rapid review of the evidence. Lancet 2020, 395, 912–920. [Google Scholar] [CrossRef]
- Shigemura, J.; Ursano, R.J.; Morganstein, J.C.; Kurosawa, M.; Benedek, D.M. Public responses to the novel 2019 coronavirus (2019-nCoV) in Japan: Mental health consequences and target populations. Psychiatry Clin. Neurosci. 2020, 74, 281–282. [Google Scholar] [CrossRef]
- Pieh, C.; Budimir, S.; Probst, T. The effect of age, gender, income, work, and physical activity on mental health during coronavirus disease (COVID-19) lockdown in Austria. J. Psychosom. Res. 2020, 136, 110186. [Google Scholar] [CrossRef] [PubMed]
- Ammar, A.; Mueller, P.; Trabelsi, K.; Chtourou, H.; Boukhris, O.; Masmoudi, L.; Bouaziz, B.; Brach, M.; Schmicker, M.; Bentlage, E.; et al. Psychological consequences of COVID-19 home confinement: The ECLB-COVID19 multicenter study. PLoS ONE 2020, 15, e0240204. [Google Scholar] [CrossRef]
- Kola, L.; Kohrt, B.A.; Hanlon, C.; Naslund, J.A.; Sikander, S.; Balaji, M.; Benjet, C.; Cheung, E.Y.L.; Eaton, J.; Gonsalves, P.; et al. COVID-19 mental health impact and responses in low-income and middle-income countries: Reimagining global mental health. Lancet Psychiatry 2021, 366, 1–16. [Google Scholar] [CrossRef]
- Salari, N.; Hosseinian-Far, A.; Jalali, R.; Vaisi-Raygani, A.; Rasoulpoor, S.; Mohammadi, M.; Rasoulpoor, S.; Khaledi-Paveh, B. Prevalence of stress, anxiety, depression among the general population during the COVID-19 pandemic: A systematic review and meta-analysis. Glob. Health 2020, 16, 1–11. [Google Scholar] [CrossRef]
- Usher, K.; Durkin, J.; Bhullar, N. The COVID-19 pandemic and mental health impacts. Int. J. Ment. Health Nurs. 2020, 29, 315–318. [Google Scholar] [CrossRef]
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The socio-economic implications of the coronavirus pandemic (COVID-19): A review. Int. J. Surg. 2020, 78, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Galea, S.; Merchant, R.M.; Lurie, N. The Mental Health Consequences of COVID-19 and Physical Distancing: The need for prevention and early intervention. JAMA Intern. Med. 2020, 180, 817–818. [Google Scholar] [CrossRef]
- Rossi, R.; Socci, V.; Talevi, D.; Mensi, S.; Niolu, C.; Pacitti, F.; Di Marco, A.; Rossi, A.; Siracusano, A.; Di Lorenzo, G. COVID-19 Pandemic and Lockdown Measures Impact on Mental Health Among the General Population in Italy. Front. Psychiatry 2020, 11, 790. [Google Scholar] [CrossRef]
- Ueda, M.; Stickley, A.; Sueki, H.; Matsubayashi, T. Mental Health Status of the General Population during the COVID-19 Pandemic: A Cross-sectional National Survey in Japan. medRxiv 2020. [Google Scholar] [CrossRef]
- Ozamiz-Etxebarria, N.; Dosil-Santamaria, M.; Picaza-Gorrochategui, M.; Idoiaga-Mondragon, N. Níveis de estresse, ansiedade e depressão na primeira fase do surto de {COVID}-19 em uma amostra no norte da {Espanha}. Cad. Saúde Pública 2020, 36, e00054020. [Google Scholar] [CrossRef]
- Kazmi, S.S.H.; Hasan, K.; Talib, S.; Saxena, S. COVID-19 and Lockdown: A Study on the Impact on Mental Health. SSRN Electron. J. 2020. [Google Scholar] [CrossRef]
- Moghanibashi-Mansourieh, A. Assessing the anxiety level of Iranian general population during COVID-19 outbreak. Asian J. Psychiatry 2020, 51, 102076. [Google Scholar] [CrossRef]
- Wang, Y.; Di, Y.; Ye, J.; Wei, W. Study on the public psychological states and its related factors during the outbreak of coronavirus disease 2019 (COVID-19) in some regions of China. Psychol. Health Med. 2021, 26, 13–22. [Google Scholar] [CrossRef]
- Moreira, P.S.; Ferreira, S.; Couto, B.; Machado-Sousa, M.; Fernández, M.; Raposo-Lima, C.; Sousa, N.; Picó-Pérez, M.; Morgado, P. Protective Elements of Mental Health Status during the COVID-19 Outbreak in the Portuguese Population. Int. J. Environ. Res. Public Health 2021, 18, 1910. [Google Scholar] [CrossRef]
- Probst, T.; Budimir, S.; Pieh, C. Depression in and after COVID-19 lockdown in Austria and the role of stress and loneliness in lockdown: A longitudinal study. J. Affect. Disord. 2020, 277, 962–963. [Google Scholar] [CrossRef]
- Pieh, C.; Probst, T.; Budimir, S.; Humer, E. Diminished well-being persists beyond the end of the COVID-19 lockdown. Gen. Hosp. Psychiatry 2021, 2020–2022. [Google Scholar] [CrossRef]
- Pieh, C.; Budimir, S.; Humer, E.; Probst, T. Comparing Mental Health during COVID-19 Lockdown and Six Months Later in Austria: A Longitudinal Study. SSRN Electron. J. 2020, 1–2. [Google Scholar] [CrossRef]
- Pettigrew, S.; Roberts, M. Addressing loneliness in later life. Aging Ment. Health 2008, 12, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Paez, D.; Bilbao, M.Á.; Bobowik, M.; Campos, M.; Basabe, N. Merry Christmas and Happy New Year! The impact of Christmas rituals on subjective well-being and family’s emotional climate. Int. J. Soc. Psychol. 2011, 26, 373–386. [Google Scholar] [CrossRef]
- Beauchamp, G.A.; Ho, M.L.; Yin, S. Variation in Suicide Occurrence by Day and during Major American Holidays. J. Emerg. Med. 2014, 46, 776–781. [Google Scholar] [CrossRef]
- Plöderl, M.; Fartacek, C.; Kunrath, S.; Pichler, E.-M.; Fartacek, R.; Datz, C.; Niederseer, D. Nothing like Christmas--suicides during Christmas and other holidays in Austria. Eur. J. Public Health 2014, 25, 410–413. [Google Scholar] [CrossRef] [PubMed]
- Mutz, M. Christmas and Subjective Well-Being: A Research Note. Appl. Res. Qual. Life 2016, 11, 1341–1356. [Google Scholar] [CrossRef]
- Sansone, R.A.; Sansone, L.A. The Christmas Effect on Psychopathology. Innov. Clin. Neurosci. 2011, 8, 10–13. [Google Scholar]
- Velamoor, V.R.; Voruganti, L.P.; Nadkarni, N.K. Feelings about Christmas, as Reported by Psychiatric Emergency Patients. Soc. Behav. Pers. Int. J. 1999, 27, 303–308. [Google Scholar] [CrossRef]
- Statistik Austria. Österreichische Gesundheitsbefragung 2019: Hauptergebnisse des Austrian Health Interview Survey (ATHIS) und Methodische Dokumentation; Statistik Austria: Vienna, Austria, 2020. [Google Scholar]
- Qualtrics. Qualtrics P.U.; Qualtrics: Provo, UT, USA, 2019. [Google Scholar]
- Statistik Austria Statistics Austria. Available online: https://www.statistik.at/web_en/statistics/PeopleSociety/population/population_censuses_register_based_census_register_based_labour_market_statistics/totaL_population/index.html (accessed on 28 March 2021).
- World Health Organization. Programme on Mental Health WHOQOL User Manual, 2012 Revis; WHO: Geneva, Switzerland, 1998. [Google Scholar]
- Skevington, S.; Lotfy, M.; O’Connell, K. The World Health Organization’s WHOQOL-BREF quality of life assessment: Psychometric properties and results of the international field trial. A Report from the WHOQOL Group. Qual. Life Res. 2004, 13, 299–310. [Google Scholar] [CrossRef]
- Hawthorne, G.; Herrman, H.; Murphy, B. Interpreting the WHOQOL-Brèf: Preliminary Population Norms and Effect Sizes. Soc. Indic. Res. 2006, 77, 37–59. [Google Scholar] [CrossRef]
- WHO. WHO Wellbeing Measures in Primary Health Care/The Depcare Project; WHO Regional Office for Europe: Copenhagen, Denmark, 1998. [Google Scholar]
- Brähler, E.; Mühlan, H.; Albani, C.; Schmidt, S. Teststatistische Prüfung und Normierung der deutschen Versionen des EUROHIS-QOL Lebensqualität-Index und des WHO-5 Wohlbefindens-Index. Diagnostica 2007, 53, 83–96. [Google Scholar] [CrossRef]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.; Janet, B.W. Validation and utility of a self-report version of PRIME-MD: The PHQ primary care study. JAMA 1999, 282, 1737–1744. [Google Scholar] [CrossRef]
- Kroenke, K.; Spitzer, R.L. The PHQ-9: A New Depression Diagnostic and Severity Measure. Psychiatr. Ann. 2002, 32, 509–515. [Google Scholar] [CrossRef]
- Spitzer, R.L.; Kroenke, K.; Williams, J.B.W.; Löwe, B. A Brief Measure for Assessing Generalized Anxiety Disorder. Arch. Intern. Med. 2006, 166, 1092–1097. [Google Scholar] [CrossRef]
- Löwe, B.; Decker, O.; Müller, S.; Brähler, E.; Schellberg, D.; Herzog, W.; Herzberg, P.Y. Validation and Standardization of the Generalized Anxiety Disorder Screener (GAD-7) in the General Population. Med. Care 2008, 46, 266–274. [Google Scholar] [CrossRef]
- IBM. IBM SPSS Statistics for Windows; Version 27.0.; IBM: Armonk, NY, USA, 2020. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2015. [Google Scholar]
- Fritz, C.O.; Morris, P.E.; Richler, J.J. Effect size estimates: Current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012, 141, 2–18. [Google Scholar] [CrossRef]
- Kocalevent, R.-D.; Hinz, A.; Brähler, E. Standardization of the depression screener Patient Health Questionnaire (PHQ-9) in the general population. Gen. Hosp. Psychiatry 2013, 35, 551–555. [Google Scholar] [CrossRef]
- Huang, Y.; Zhao, N. Generalized anxiety disorder, depressive symptoms and sleep quality during COVID-19 outbreak in China: A web-based cross-sectional survey. Psychiatry Res. 2020, 288, 112954. [Google Scholar] [CrossRef]
- Hinz, A.; Klein, A.M.; Brähler, E.; Glaesmer, H.; Luck, T.; Riedel-Heller, S.G.; Wirkner, K.; Hilbert, A. Psychometric evaluation of the Generalized Anxiety Disorder Screener GAD-7, based on a large German general population sample. J. Affect. Disord. 2017, 210, 338–344. [Google Scholar] [CrossRef]
- Limcaoco, R.S.G.; Mateos, E.M.; Fernandez, J.M.; Roncero, C. Anxiety, worry and perceived stress in the world due to the COVID-19 pandemic, March 2020. Preliminary results. Psychiatry Clin. Psychol. 2020. [Google Scholar] [CrossRef]
- Riecher-Rössler, A. Sex and gender differences in mental disorders. Lancet Psychiatry 2017, 4, 8–9. [Google Scholar] [CrossRef]
- Connor, J.; Madhavan, S.; Mokashi, M.; Amanuel, H.; Johnson, N.R.; Pace, L.E.; Bartz, D. Health risks and outcomes that disproportionately affect women during the Covid-19 pandemic: A review. Soc. Sci. Med. 2020, 266, 113364. [Google Scholar] [CrossRef]
- Klein, E.M.; Brähler, E.; Dreier, M.; Reinecke, L.; Müller, K.W.; Schmutzer, G.; Wölfling, K.; Beutel, M.E. The German version of the Perceived Stress Scale—Psychometric characteristics in a representative German community sample. BMC Psychiatry 2016, 16, 1–10. [Google Scholar] [CrossRef]
- Almeida, M.; Shrestha, A.D.; Stojanac, D.; Miller, L.J. The impact of the COVID-19 pandemic on women’s mental health. Arch. Women’s Ment. Health 2020, 23, 741–748. [Google Scholar] [CrossRef]
- Smith, J. Overcoming the ‘tyranny of the urgent’: Integrating gender into disease outbreak preparedness and response. Gend. Dev. 2019, 27, 355–369. [Google Scholar] [CrossRef]
- Nwachukwu, I.; Nkire, N.; Shalaby, R.; Hrabok, M.; Vuong, W.; Gusnowski, A.; Surood, S.; Urichuk, L.; Greenshaw, A.J.; Agyapong, V.I. COVID-19 Pandemic: Age-Related Differences in Measures of Stress, Anxiety and Depression in Canada. Int. J. Environ. Res. Public Health 2020, 17, 6366. [Google Scholar] [CrossRef]
- Ekelund, U.; Steene-Johannessen, J.; Brown, W.J.; Fagerland, M.W.; Owen, N.; Powell, K.E.; Bauman, A.; Lee, I.-M. Does physical activity attenuate, or even eliminate, the detrimental association of sitting time with mortality? A harmonised meta-analysis of data from more than 1 million men and women. Lancet 2016, 388, 1302–1310. [Google Scholar] [CrossRef]
- Devaux, M.; Sassi, F. Social disparities in hazardous alcohol use: Self-report bias may lead to incorrect estimates. Eur. J. Public Health 2016, 26, 129–134. [Google Scholar] [CrossRef]
| Variable | N | % |
|---|---|---|
| Total | 1505 | |
| Gender | ||
| Women | 764 | 49.24 |
| Men | 741 | 50.76 |
| Other | 0 | 0 |
| Age | ||
| 18–24 | 153 | 10.17 |
| 25–34 | 279 | 18.54 |
| 35–44 | 289 | 19.2 |
| 45–54 | 326 | 21.66 |
| 55–64 | 272 | 18.07 |
| 65+ | 186 | 12.36 |
| Region | ||
| Burgenland | 58 | 3.85 |
| Lower Austria | 305 | 20.27 |
| Vienna | 346 | 22.99 |
| Carinthia | 103 | 6.84 |
| Styria | 221 | 14.68 |
| Upper Austria | 227 | 15.08 |
| Salzburg | 79 | 5.25 |
| Tyrol | 114 | 7.57 |
| Vorarlberg | 52 | 3.46 |
| Education | ||
| Low | 40 | 2.66 |
| Middle | 809 | 53.75 |
| High | 656 | 43.59 |
| Gender | |||||
|---|---|---|---|---|---|
| Male | Female | Total | Statistic | ||
| PHQ-9 score N (%) | <10 | 585 (78.95) | 523 (68.46) | 1108 (73.62) | χ2 (1) = 21.32, p < 0.001 |
| ≥10 | 156 (21.05) | 241 (31.54) | 397 (26.38) | ||
| GAD-7 score N (%) | <10 | 616 (83.13) | 540 (70.68) | 1156 (76.81) | χ2 (1) = 32.74, p < 0.001 |
| ≥10 | 125 (16.87) | 224 (29.32) | 349 (23.19) | ||
| ISI score N (%) | <15 | 620 (83.67) | 604 (79.06) | 1224 (83.33) | χ2(1) = 5.27, p < 0.05 |
| ≥15 | 121 (16.33) | 160 (20.94) | 281 (18.67) | ||
| Total (N) | 741 | 764 | 1505 | ||
| PHQ-9 | Mean (SD) | 5.87 (5.41) | 7.69 (6.11) | 6.8 (5.84) | t (1503) = −6.11, p < 0.001, g = 0.32 |
| GAD-7 | Mean (SD) | 5.34 (4.73) | 7.12 (5.24) | 6.25 (5.07) | t (1503) = −7.0, p < 0.001, g = 0.36 |
| ISI | Mean (SD) | 8.42 (6.0) | 9.5 (6.15) | 8.97 (6.1) | t (1503) = −3.46, p < 0.001, g = 0.18 |
| WHOQOL BREF (psychological domain) | Mean (SD) | 71.87 (17.64) | 66.0 (19.16) | 68.89 (18.66) | t (1503) = −6.19, p < 0.001, g = 0.32 |
| WHO-5 | Mean (SD) | 15.09 (5.56) | 13.6 (5.92) | 14.34 (5.79) | t (1503) = 5.03, p < 0.001, g = 0.26 |
| PSS-10 | Mean (SD) | 15.03 (7.21) | 17.78 (7.73) | 16.42 (7.6) | t (1503) = −7.14, p < 0.001, g = 0.37 |
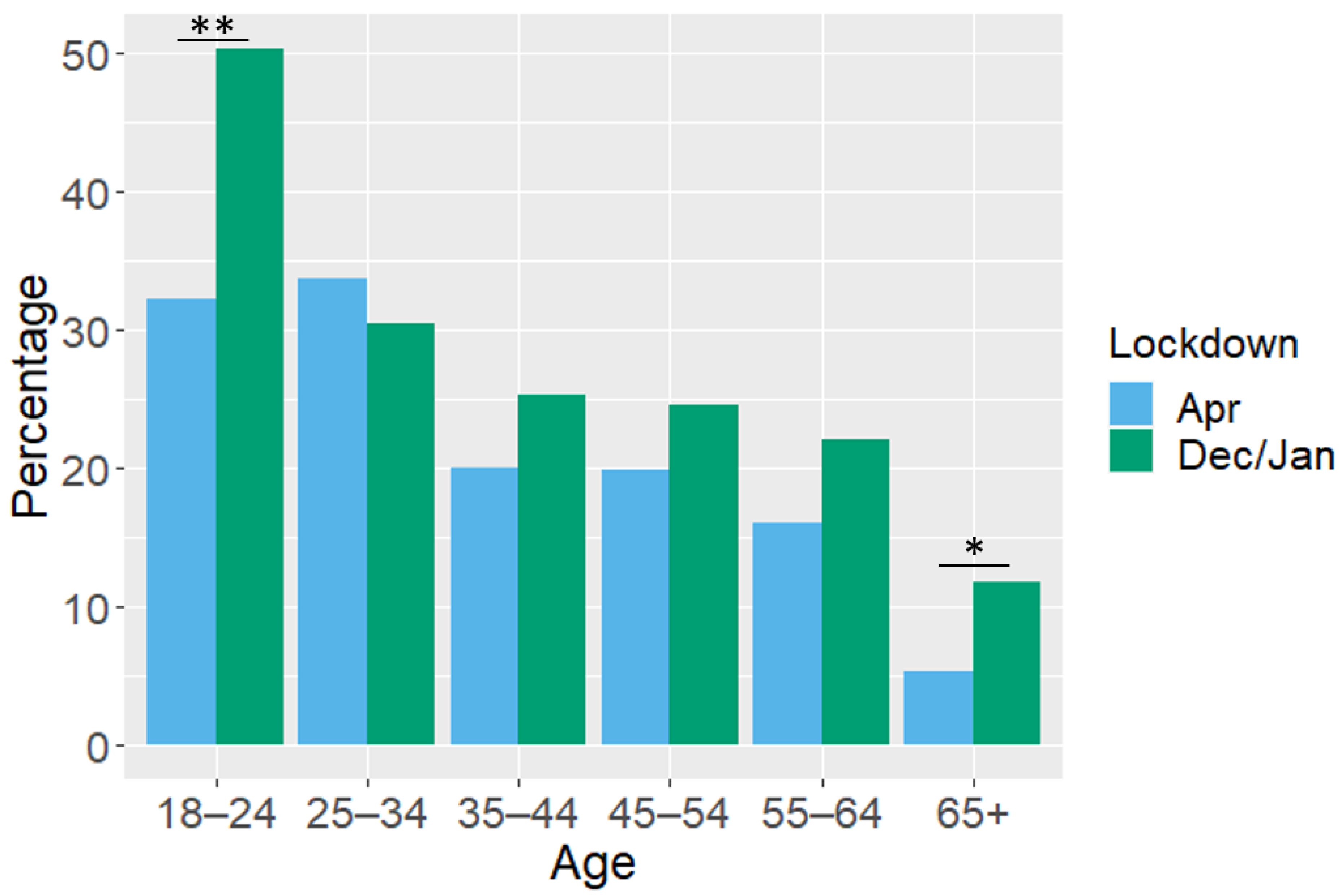
| Age (years) | ||||||||
|---|---|---|---|---|---|---|---|---|
| 18–24 | 25–34 | 35–44 | 45–54 | 55–64 | 65+ | Statistic | ||
| PHQ-9 score N (%) | <10 | 76 (49.67) | 194 (69.53) | 216 (74.74) | 246 (75.46) | 212 (77.94) | 164 (88.17) | χ2 (5) = 71.23, p < 0.001 |
| ≥10 | 77 (50.33) | 85 (30.47) | 73 (25.26) | 80 (24.54) | 60 (22.06) | 22 (11.83) | ||
| GAD-7 score N (%) | <10 | 100 (65.36) | 200 (71.68) | 222 (76.82) | 246 (75.15) | 221 (81.25) | 167 (89.78) | χ2 (5) = 36.3, p < 0.001 |
| ≥10 | 53 (34.64) | 79 (28.32) | 67 (23.18) | 80 (24.85) | 51 (18.75) | 19 (10.22) | ||
| ISI score N (%) | <15 | 114 (74.51) | 228 (81.72) | 229 (79.24) | 257 (78.83) | 231 (84.93) | 165 (88.71) | χ2 (5) = 15.87, p < 0.01 |
| ≥15 | 39 (25.49) | 51 (18.28) | 60 (20.76) | 69 (21.17) | 41 (15.07) | 21 (11.29) | ||
| Total (N) | 153 | 279 | 289 | 326 | 272 | 186 | ||
| PHQ-9 | Mean (SD) | 9.92 (5.98) | 7.89 (5.88) | 6.91 (5.71) | 6.6 (6.04) | 5.87 (5.6) | 4.12 (4.18) | F (5, 1499) = 21.26, p < 0.001, η2 = 0.027 |
| GAD-7 | Mean (SD) | 8.42 (5.27) | 7.08 (4.72) | 6.4 (4.86) | 6.31 (5.44) | 5.5 (5.08) | 3.95 (3.89) | F (5, 1499) = 16.85, p < 0.001, η2 = 0.053 |
| ISI | Mean (SD) | 10.42 (5.95) | 9.54 (5.83) | 9.37 (6.07) | 9.28 (6.48) | 8.21 (6.1) | 6.85 (5.34) | F (5, 1499) = 8.15, p < 0.001, η2 = 0.026 |
| WHOQOL BREF (psychological domain) | Mean (SD) | 63.13 (17.96) | 66.02 (19.51) | 69.26 (16.63) | 68.92 (19.96) | 70.88 (19.38) | 74.4 (15.36) | F (5, 1499) = 8.31, p < 0.001, η2 = 0.027 |
| WHO-5 | Mean (SD) | 12.33 (5.29) | 13.79 (5.32) | 14.04 (5.76) | 14.08 (6.19) | 15.06 (6.09) | 16.66 (4.86) | F (5, 1499) = 11.69, p < 0.001, η2 = 0.038 |
| PSS-10 | Mean (SD) | 20.01 (7.1) | 17.98 (7.1) | 17.03 (7.01) | 16.14 (8.11) | 15.0 (7.84) | 12.77 (6.26) | F (5, 1499) = 21.5, p < 0.001, η2 = 0.067 |
| Marital Status | ||||||||
|---|---|---|---|---|---|---|---|---|
| Single | Separated | Divorced | Cohabiting | Married | Widowed | Statistic | ||
| PHQ-9 score N (%) | <10 | 283 (64.91) | 23 (53.49) | 73 (69.52) | 245 (75.15) | 457 (81.46) | 27 (79.41) | χ2 (5) = 45.67, p < 0.001 |
| ≥10 | 153 (35.09) | 20 (46.51) | 32 (30.48) | 81 (24.85) | 104 (18.54) | 7 (20.59) | ||
| GAD-7 score N (%) | <10 | 318 (72.94) | 28 (65.12) | 80 (76.19) | 247 (75.77) | 455 (81.11) | 28 (82.35) | χ2 (5) = 13.59, p < 0.05 |
| ≥10 | 118 (27.06) | 15 (34.88) | 25 (23.81) | 79 (24.23) | 106 (18.89) | 6 (17.65) | ||
| ISI score N (%) | <15 | 346 (79.36) | 27 (62.79) | 83 (79.05) | 271 (83.13) | 468 (83.42) | 29 (85.29) | χ2 (5) = 13.87, p < 0.05 |
| ≥15 | 90 (20.64) | 16 (37.21) | 22 (20.95) | 55 (16.87) | 93 (16.58) | 5 (14.71) | ||
| Total (N) | 436 | 43 | 105 | 326 | 561 | 34 | ||
| PHQ-9 | Mean (SD) | 8.09 (6.29) | 8.86 (6.46) | 6.87 (5.96) | 6.86 (5.36) | 5.66 (5.44) | 5.44 (5.92) | F (5, 1499) = 10.27, p < 0.001, η2 = 0.033 |
| GAD-7 | Mean (SD) | 6.9 (5.25) | 7.98 (5.0) | 6.2 (5.34) | 6.52 (4.87) | 5.54 (4.91) | 4.76 (4.86) | F (5, 1499) = 5.44, p < 0.001, η2 = 0.018 |
| ISI | Mean (SD) | 9.37 (6.06) | 11.44 (6.28) | 9.43 (6.65) | 8.83 (5.81) | 8.52 (6.14) | 7.97 (5.75) | F (5, 1499) = 2.76, p < 0.05, η2 = 0.009 |
| WHOQOL BREF (psychological domain) | Mean (SD) | 64.57 (19.71) | 61.82 (19.35) | 68.45 (17.92) | 68.9 (18.05) | 72.55 (17.55) | 74.02 (15.89) | F (5, 1499) = 11.1, p < 0.001, η2 = 0.036 |
| WHO-5 | Mean (SD) | 13.27 (6.05) | 12.26 (6.23) | 14.22 (6.18) | 14.24 (5.41) | 15.27 (5.55) | 16.53 (5.06) | F (5, 1499) = 8.19, p < 0.001, η2 = 0.027 |
| PSS-10 | Mean (SD) | 17.5 (7.89) | 20.49 (7.67) | 15.13 (7.8) | 17.01 (7.27) | 15.36 (7.24) | 13.32 (7.8) | F (5, 1499) = 8.74, p < 0.001, η2 = 0.028 |
| Income (Monthly, €) | |||||||
|---|---|---|---|---|---|---|---|
| <1000 | 1000–2000 | 2000–3000 | 3000–4000 | >4000 | Statistic | ||
| PHQ-9 score N (%) | <10 | 73 (56.59) | 212 (66.04) | 300 (73.89) | 244 (76.01) | 279 (85.06) | χ2 (4) = 51.82, p < 0.001 |
| ≥10 | 56 (43.41) | 109 (33.96) | 106 (26.11) | 77 (23.99) | 49 (14.94) | ||
| GAD-7 score N (%) | <10 | 81 (62.79) | 230 (71.65) | 312 (76.85) | 250 (77.88) | 283 (86.28) | χ2 (4) = 35.75, p < 0.001 |
| ≥10 | 48 (37.21) | 91 (28.35) | 94 (23.15) | 71 (22.12) | 45 (13.72) | ||
| ISI score N (%) | <15 | 90 (69.77) | 250 (77.88) | 333 (82.02) | 259 (80.69) | 292 (89.02 | χ2 (4) = 26.87, p < 0.01 |
| ≥15 | 39 (30.23) | 71 (22.12) | 73 (17.98) | 62 (19.31) | 36 (10.98) | ||
| Total (N) | 129 | 321 | 406 | 321 | 328 | ||
| PHQ-9 | Mean (SD) | 9.49 (7.28) | 7.73 (6.33) | 6.83 (5.46) | 6.36 (5.59) | 5.2 (4.82) | F (4, 1500) = 16.03, p < 0.001, η2 = 0.041 |
| GAD-7 | Mean (SD) | 8.02 (5.84) | 6.89 (5.39) | 6.29 (4.78) | 6.07 (4.99) | 5.03 (4.54) | F (4, 1500) = 10.3, p < 0.001, η2 = 0.027 |
| ISI | Mean (SD) | 10.67 (6.49) | 9.86 (6.19) | 9.14 (6.0) | 8.92 (6.17) | 7.27 (5.51) | F (4, 1500) = 10.93, p < 0.001, η2 = 0.028 |
| WHOQOL BREF (psychological domain) | Mean (SD) | 59.95 (21.61) | 65.1 (19.37) | 68.08 (17.65) | 70.9 (17.82) | 75.15 (16.26) | F (4, 1500) = 22.28, p < 0.001, η2 = 0.056 |
| WHO-5 | Mean (SD) | 12.32 (6.86) | 13.56 (6.0) | 14.31 (5.48) | 14.57 (5.56) | 15.69 (5.39) | F (4, 1500) = 11.69, p < 0.001, η2 = 0.038 |
| PSS-10 | Mean (SD) | 18.95 (9.05) | 17.65 (7.6) | 16.53 (7.24) | 15.91 (7.45) | 14.6 (7.1) | F (4, 1500) = 11.05, p < 0.001, η2 = 0.029 |
| Physical Activity (Days/Week) | |||||||
|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | >4 | Statistic | ||
| PHQ-9 score N (%) | <10 | 135 (61.36) | 135 (72.58) | 197 (71.64) | 205 (75.65) | 436 (78.84) | χ2 (4) = 26.02, p < 0.001 |
| ≥10 | 85 (38.64) | 51 (27.42) | 78 (28.36) | 66 (24.35) | 117 (21.16) | ||
| GAD-7 score N (%) | <10 | 151 (68.64) | 143 (76.88) | 209 (76) | 222 (81.92) | 431 (77.94) | χ2 (4) = 12.72, p < 0.05 |
| ≥10 | 69 (31.36) | 43 (23.12) | 66 (24) | 49 (18.08) | 122 (22.06) | ||
| ISI score N (%) | <15 | 162 (73.64) | 146 (78.49) | 230 (83.64) | 231 (85.24) | 455 (82.28) | χ2 (4) = 13.58, p < 0.01 |
| ≥15 | 58 (26.36) | 40 (21.51) | 45 (16.36) | 40 (14.76) | 98 (17.72) | ||
| Total (N) | 220 | 186 | 275 | 271 | 553 | ||
| PHQ-9 | Mean (SD) | 8.66 (6.39) | 7.21 (6.0) | 7.16 (5.4) | 6.48 (5.38) | 5.87 (5.81) | F (4, 1500) = 10.08, p < 0.001, η2 = 0.026 |
| GAD-7 | Mean (SD) | 7.43 (5.65) | 6.56 (5.3) | 6.29 (4.53) | 5.92 (4.63) | 5.81 (5.15) | F(4, 1500) = 4.52, p < 0.001, η2 = 0.012 |
| ISI | Mean (SD) | 10.1 (6.01) | 8.89 (6.22) | 9.09 (5.54) | 8.64 (5.86) | 8.64 (6.42) | F(4, 1500) = 2.53, p < 0.05, η2 = 0.007 |
| WHOQOL BREF (psychological domain) | Mean (SD) | 60.64 (20.5) | 67.29 (19.27) | 68.32 (15.83) | 69.74 (17.23) | 72.57 (18.59) | F(4, 1500) = 17.41, p < 0.001, η2 = 0.044 |
| WHO-5 | Mean (SD) | 11.66 (6.13) | 13.54 (5.6) | 14.11 (5.11) | 14.77 (5.5) | 15.57 (5.79) | F(4, 1500) = 20.35, p < 0.001, η2 = 0.051 |
| PSS-10 | Mean (SD) | 19.04 (7.96) | 17.05 (7.82) | 16.67 (6.67) | 15.8 (7.24) | 15.36 (7.73) | F(4, 1500) = 10.3, p < 0.001, η2 = 0.027 |
| April Lockdown | December/January Lockdown | Statistic | ||||
|---|---|---|---|---|---|---|
| Male | Female | Male | Female | |||
| PHQ-9 score (%) | ≥10 | 16.4 | 25.1 | 21.1 | 31.5 | Lockdown: t (2506) = 1.85, p = 0.06 Lockdown * gender: t (2506) = 0.53, p > 0.05 |
| GAD-7 score (%) | ≥10 | 14.1 | 23.4 | 16.9 | 29.3 | Lockdown: t(2506) = 1.16, p > 0.05 Lockdown * gender: t (2506) = 0.95, p > 0.05 |
| ISI score (%) | ≥15 | 14.3 | 17.0 | 16.3 | 20.9 | Lockdown: t(2506) = 0.9, p > 0.05 Lockdown * gender: t (2506) = 0.63, p = 0.6 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dale, R.; Budimir, S.; Probst, T.; Stippl, P.; Pieh, C. Mental Health during the COVID-19 Lockdown over the Christmas Period in Austria and the Effects of Sociodemographic and Lifestyle Factors. Int. J. Environ. Res. Public Health 2021, 18, 3679. https://doi.org/10.3390/ijerph18073679
Dale R, Budimir S, Probst T, Stippl P, Pieh C. Mental Health during the COVID-19 Lockdown over the Christmas Period in Austria and the Effects of Sociodemographic and Lifestyle Factors. International Journal of Environmental Research and Public Health. 2021; 18(7):3679. https://doi.org/10.3390/ijerph18073679
Chicago/Turabian StyleDale, Rachel, Sanja Budimir, Thomas Probst, Peter Stippl, and Christoph Pieh. 2021. "Mental Health during the COVID-19 Lockdown over the Christmas Period in Austria and the Effects of Sociodemographic and Lifestyle Factors" International Journal of Environmental Research and Public Health 18, no. 7: 3679. https://doi.org/10.3390/ijerph18073679
APA StyleDale, R., Budimir, S., Probst, T., Stippl, P., & Pieh, C. (2021). Mental Health during the COVID-19 Lockdown over the Christmas Period in Austria and the Effects of Sociodemographic and Lifestyle Factors. International Journal of Environmental Research and Public Health, 18(7), 3679. https://doi.org/10.3390/ijerph18073679







