Mild to Severe Neurological Manifestations of COVID-19: Cases Reports
Abstract
1. Introduction
2. Materials and Methods
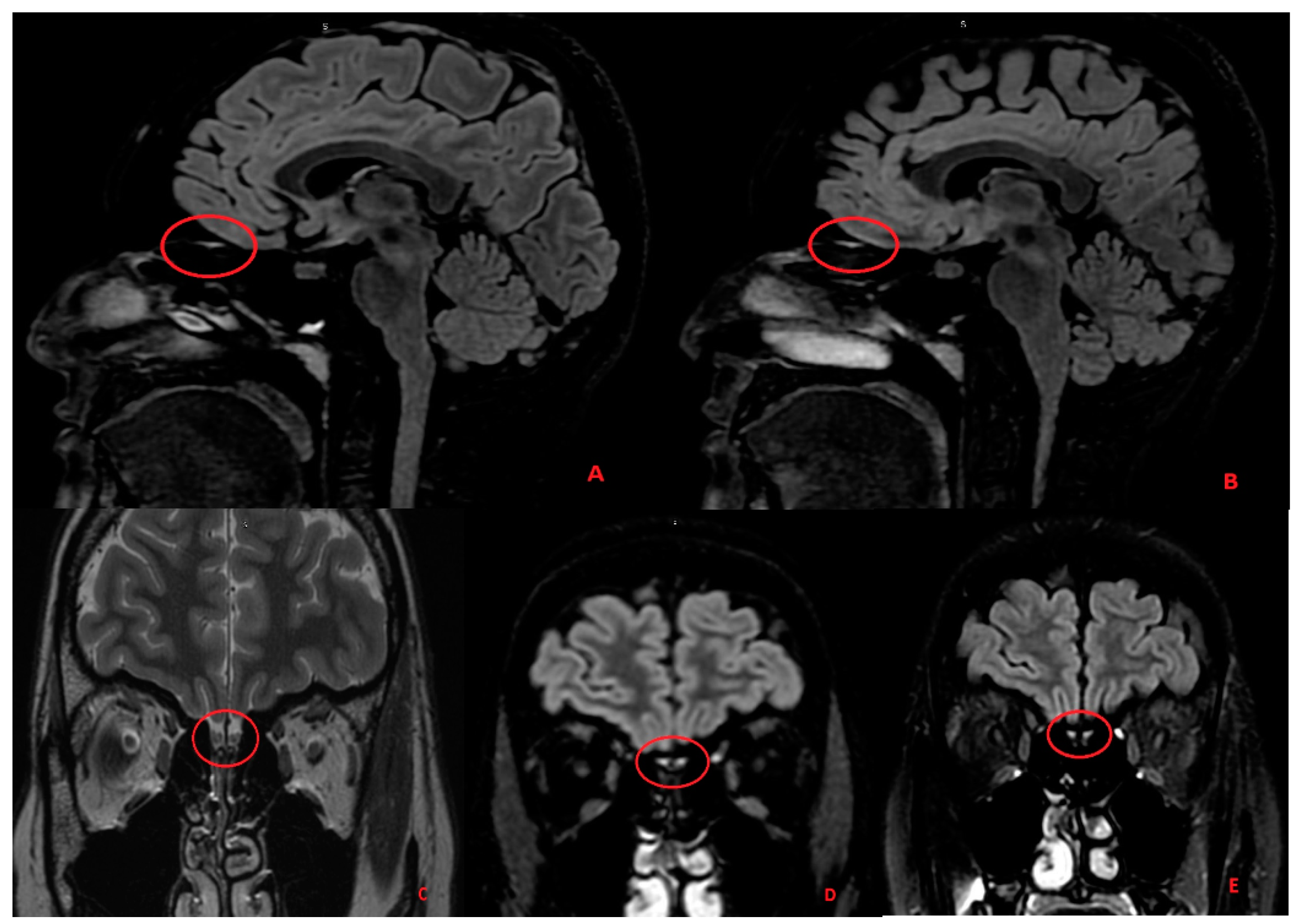
3. Case Report 1
3.1. Discussion Part I—Neuro-Invasion Mechanism: Can SARS-CoV-2 Directly Invade the CNS?
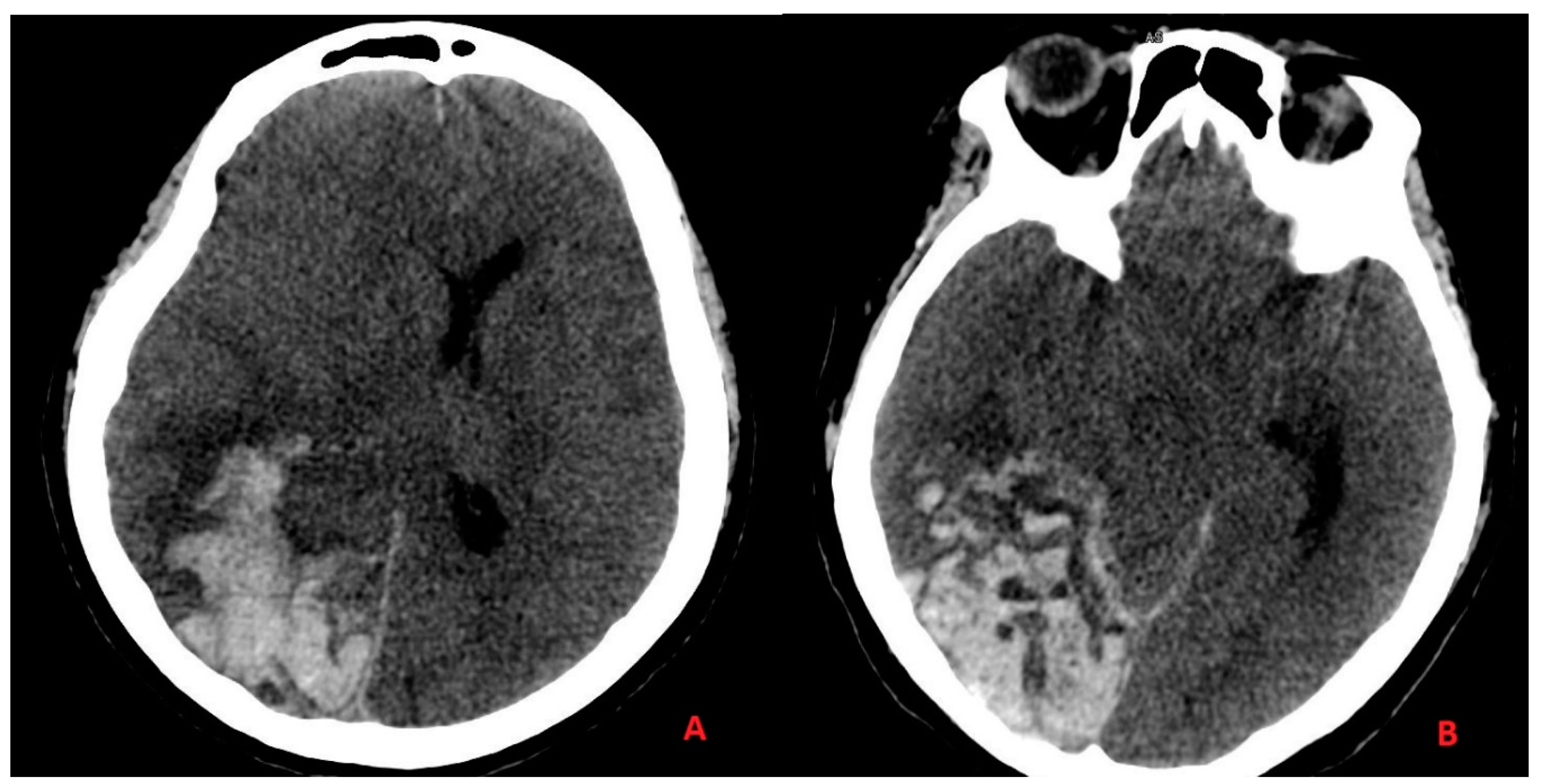
3.2. Case Report 2
3.3. Discussion Part II—Ischemic Stroke and Intracranial Hemorrhage
4. Discussion
Other Considerations and Neurological Aspects
5. Study Limitations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Spina, S.; Marrazzo, F.; Migliari, M.; Stucchi, R.; Sforza, A.; Fumagalli, R. The response of Milan’s Emergency Medical System to the COVID-19 outbreak in Italy. Lancet 2020, 395, e49–e50. [Google Scholar] [CrossRef]
- Melegari, G.; Giuliani, E.; Maini, G.; Barbieri, L.; Baffoni, P.; Bertellini, E. Novel coronavirus (2019-nCov): Do you have enough intensive care units? Med. Intensiv. 2020, 44, 583–585. [Google Scholar] [CrossRef]
- Alhazzani, W.; Møller, M.H.; Arabi, Y.M.; Loeb, M.; Gong, M.N.; Fan, E.; Oczkowski, S.; Levy, M.M.; Derde, L.; Dzierba, A.; et al. Surviving Sepsis Campaign: Guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensiv. Care Med. 2020, 46, 854–887. [Google Scholar] [CrossRef]
- Riva, G.; Nasillo, V.; Tagliafico, E.; Trenti, T.; Comoli, P.; Luppi, M. COVID-19: More than a cytokine storm. Crit. Care 2020, 24. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Tsatsakis, A.; Petrakis, D.; Nikolouzakis, T.K.; Docea, A.O.; Calina, D.; Vinceti, M.; Goumenou, M.; Kostoff, R.N.; Mamoulakis, C.; Aschner, M.; et al. COVID-19, an opportunity to reevaluate the correlation between long-term effects of anthropogenic pollutants on viral epidemic/pandemic events and prevalence. Food Chem. Toxicol. 2020, 141, 111418. [Google Scholar] [CrossRef] [PubMed]
- Olfactory Bulb MRI and Paranasal Sinus CT Findings in Persistent COVID-19 Anosmia—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/33132007/ (accessed on 14 March 2021).
- Miesbach, W.; Makris, M. COVID-19: Coagulopathy, Risk of Thrombosis, and the Rationale for Anticoagulation. Clin. Appl. Thromb. 2020, 26. [Google Scholar] [CrossRef] [PubMed]
- Bae, M.; Kim, H. Mini-Review on the Roles of Vitamin C, Vitamin D, and Selenium in the Immune System against COVID-19. Molecules 2020, 25, 5346. [Google Scholar] [CrossRef]
- Mehraeen, E.; Behnezhad, F.; Salehi, M.A.; Noori, T.; Harandi, H.; SeyedAlinaghi, S. Olfactory and gustatory dysfunctions due to the coronavirus disease (COVID-19): A review of current evidence. Eur. Arch. Oto-Rhino-Laryngol. 2021, 278, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Bai, W.Z.; Hashikawa, T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020, 92, 552–555. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Zubair, A.S.; McAlpine, L.S.; Gardin, T.; Farhadian, S.; Kuruvilla, D.E.; Spudich, S. Neuropathogenesis and neurologic manifestations of the coronaviruses in the age of coronavirus disease 2019: A review. JAMA Neurol. 2020, 77, 1018–1027. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; De Siati, D.R.; Horoi, M.; Le Bon, S.D.; Rodriguez, A.; Dequanter, D.; Blecic, S.; El Afia, F.; Distinguin, L.; et al. Olfactory and gustatory dysfunctions as a clinical presentation of mild-to-moderate forms of the coronavirus disease (COVID-19): A multicenter European study. Eur. Arch. Oto-Rhino-Laryngol. 2020, 277, 2251–2261. [Google Scholar] [CrossRef] [PubMed]
- Wee, L.E.; Chan, Y.F.Z.; Teo, N.W.Y.; Cherng, B.P.Z.; Thien, S.Y.; Wong, H.M.; Wijaya, L.; Toh, S.T.; Tan, T.T. The role of self-reported olfactory and gustatory dysfunction as a screening criterion for suspected COVID-19. Eur. Arch. Oto-Rhino-Laryngol. 2020, 277, 2389–2390. [Google Scholar] [CrossRef] [PubMed]
- Lozada-Nur, F.; Chainani-Wu, N.; Fortuna, G.; Sroussi, H. Dysgeusia in COVID-19: Possible Mechanisms and Implications. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 130, 344–346. [Google Scholar] [CrossRef]
- Vaira, L.A.; Deiana, G.; Fois, A.G.; Pirina, P.; Madeddu, G.; De Vito, A.; Babudieri, S.; Petrocelli, M.; Serra, A.; Bussu, F.; et al. Objective Evaluation of Anosmia and Ageusia in COVID-19 Patients: Single-Center Experience on 72 Cases. Head Neck 2020, 42, 1252–1258. [Google Scholar]
- Lechien, J.R.; Michel, J.; Radulesco, T.; Chiesa-Estomba, C.M.; Vaira, L.A.; De Riu, G.; Sowerby, L.; Hopkins, C.; Saussez, S. Clinical and Radiological Evaluations of COVID -19 Patients With Anosmia: Preliminary Report. Laryngoscope 2020, 130, 2526–2531. [Google Scholar] [CrossRef]
- Kanne, J.P.; Bai, H.; Bernheim, A.; Chung, M.; Haramati, L.B.; Kallmes, D.F.; Little, B.P.; Rubin, G.; Sverzellati, N. COVID-19 Imaging: What We Know Now and What Remains Unknown. Radiology 2021, 204522. [Google Scholar] [CrossRef]
- Aragão, M.; Leal, M.; Filho, O.C.; Fonseca, T.; Valença, M. Anosmia in COVID-19 Associated with Injury to the Olfactory Bulbs Evident on MRI. Am. J. Neuroradiol. 2020, 41, 1703–1706. [Google Scholar] [CrossRef]
- Han, A.Y.; Mukdad, L.; Long, J.L.; Lopez, I.A. Anosmia in COVID-19: Mechanisms and Significance. Chem. Sens. 2020, 45, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Marshall, M. The lasting misery of coronavirus long-haulers. Nature 2020, 585, 339–341. [Google Scholar] [CrossRef]
- Mendelson, M.; Nel, J.; Blumberg, L.; Madhi, S.A.; Dryden, M.; Stevens, W.; Venter, F.W.D. Long-COVID: An evolving problem with an extensive impact. S. Afr. Med. J. 2020, 111, 10–12. [Google Scholar] [CrossRef] [PubMed]
- Vagli, C.; Fisicaro, F.; Vinciguerra, L.; Puglisi, V.; Rodolico, M.S.; Giordano, A.; Ferri, R.; Lanza, G.; Bella, R. Cerebral Hemodynamic Changes to Transcranial Doppler in Asymptomatic Patients with Fabry’s Disease. Brain Sci. 2020, 10, 546. [Google Scholar] [CrossRef]
- Caricato, A.; Mignani, V.; Bocci, M.G.; Pennisi, M.A.; Sandroni, C.; Tersali, A.; Antonaci, A.; De Waure, C.; Antonelli, M. Usefulness of transcranial echography in patients with decompressive craniectomy: A comparison with computed tomography scan. Crit. Care Med. 2012, 40, 1745–1752. [Google Scholar] [CrossRef] [PubMed]
- Iba, T.; Levy, J.H.; Connors, J.M.; Warkentin, T.E.; Thachil, J.; Levi, M. The unique characteristics of COVID-19 coagulopathy. Crit. Care 2020, 24. [Google Scholar] [CrossRef] [PubMed]
- Al-Samkari, H.; Leaf, R.S.K.; Dzik, W.H.; Carlson, J.C.T.; Fogerty, A.E.; Waheed, A.; Goodarzi, K.; Bendapudi, P.K.; Bornikova, L.; Gupta, S.; et al. COVID-19 and coagulation: Bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood 2020, 136, 489–500. [Google Scholar] [CrossRef] [PubMed]
- Peyvandi, F.; Artoni, A.; Novembrino, C.; Aliberti, S.; Panigada, M.; Boscarino, M.; Gualtierotti, R.; Rossi, F.; Palla, R.; Martinelli, I.; et al. Hemostatic alterations in COVID-19. Haematologica 2020. [Google Scholar] [CrossRef]
- Connors, J.M.; Levy, J.H. COVID-19 and its implications for thrombosis and anticoagulation. Blood 2020, 135, 2033–2040. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [CrossRef]
- Fox, S.E.; Akmatbekov, A.; Harbert, J.L.; Li, G.; Brown, J.Q.; Heide, R.S.V. Pulmonary and cardiac pathology in African American patients with COVID-19: An autopsy series from New Orleans. Lancet Respir. Med. 2020, 8, 681–686. [Google Scholar] [CrossRef]
- Roncati, L.; Ligabue, G.; Nasillo, V.; Lusenti, B.; Gennari, W.; Fabbiani, L.; Malagoli, C.; Gallo, G.; Giovanella, S.; Lupi, M.; et al. A proof of evidence supporting abnormal immunothrombosis in severe COVID-19: Naked megakaryocyte nuclei increase in the bone marrow and lungs of critically ill patients. Platelets 2020, 31, 1085–1089. [Google Scholar] [CrossRef]
- Fisicaro, F.; Di Napoli, M.; Liberto, A.; Fanella, M.; Di Stasio, F.; Pennisi, M.; Bella, R.; Lanza, G.; Mansueto, G. Neurological Sequelae in Patients with COVID-19: A Histopathological Perspective. Int. J. Environ. Res. Public Health 2021, 18, 1415. [Google Scholar] [CrossRef] [PubMed]
- Atallah, B.; El Nekidy, W.; Mallah, S.I.; Cherfan, A.; AbdelWareth, L.; Mallat, J.; Hamed, F. Thrombotic events following tocilizumab therapy in critically ill COVID-19 patients: A Façade for prognostic markers. Thromb. J. 2020, 18. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; MacFadyen, J.G.; Thuren, T.; Libby, P. Residual inflammatory risk associated with interleukin-18 and interleukin-6 after successful interleukin-1β inhibition with canakinumab: Further rationale for the development of targeted anti-cytokine therapies for the treatment of atherothrombosis. Eur. Heart J. 2020, 41, 2153–2163. [Google Scholar] [CrossRef] [PubMed]
- Nannoni, S.; de Groot, R.; Bell, S.; Markus, H.S. Stroke in COVID-19: A systematic review and meta-analysis. Int. J. Stroke 2021, 16, 137–149. [Google Scholar] [CrossRef]
- Divani, A.A.; Andalib, S.; Di Napoli, M.; Lattanzi, S.; Hussain, M.S.; Biller, J.; McCullough, L.D.; Azarpazhooh, M.R.; Seletska, A.; Mayer, S.A.; et al. Coronavirus Disease 2019 and Stroke: Clinical Manifestations and Pathophysiological Insights. J. Stroke Cerebrovasc. Dis. 2020, 29, 104941. [Google Scholar] [CrossRef]
- Oxley, T.J.; Mocco, J.; Majidi, S.; Kellner, C.P.; Shoirah, H.; Singh, I.P.; De Leacy, R.A.; Shigematsu, T.; Ladner, T.R.; Yaeger, K.A.; et al. Large-Vessel Stroke as a Presenting Feature of Covid-19 in the Young. N. Engl. J. Med. 2020, 382, e60. [Google Scholar] [CrossRef]
- Nadkarni, G.N.; Lala, A.; Bagiella, E.; Chang, H.L.; Moreno, P.R.; Pujadas, E.; Arvind, V.; Bose, S.; Charney, A.W.; Chen, M.D.; et al. Anticoagulation, Bleeding, Mortality, and Pathology in Hospitalized Patients With COVID-19. J. Am. Coll. Cardiol. 2020, 76, 1815–1826. [Google Scholar] [CrossRef]
- Flumignan, R.L.; Tinôco, J.D.D.S.; Pascoal, P.I.; Areias, L.L.; Cossi, M.S.; Fernandes, M.I.; Costa, I.K.; Souza, L.; Matar, C.F.; Tendal, B.; et al. Prophylactic anticoagulants for people hospitalised with COVID-19. Cochrane Database Syst. Rev. 2020, 10, CD013739. [Google Scholar] [CrossRef]
- White, D.; Macdonald, S.; Bull, T.; Hayman, M.; De Monteverde-Robb, R.; Sapsford, D.; Lavinio, A.; Varley, J.; Johnston, A.; Besser, M.; et al. Heparin resistance in COVID-19 patients in the intensive care unit. J. Thromb. Thrombolysis 2020, 50, 287–291. [Google Scholar] [CrossRef]
- Cattaneo, M.; Bertinato, E.M.; Birocchi, S.; Brizio, C.; Malavolta, D.; Manzoni, M.; Muscarella, G.; Orlandi, M. Pulmonary embolism or pulmonary thrombosis in COVID-19? Is the recommendation to use high-dose heparin for thromboprophylaxis justified? Thromb. Haemost. 2020, 120, 1230–1232. [Google Scholar] [CrossRef]
- Mishra, S.; Choueka, M.; Wang, Q.; Hu, C.; Visone, S.; Silver, M.; Stein, E.G.; Levine, S.R.; Wang, Q.T. Intracranial Hemorrhage in COVID-19 Patients. J. Stroke Cerebrovasc. Dis. 2021, 30, 105603. [Google Scholar] [CrossRef]
- Kremer, S.; Lersy, F.; De Sèze, J.; Ferré, J.-C.; Maamar, A.; Carsin-Nicol, B.; Collange, O.; Bonneville, F.; Adam, G.; Martin-Blondel, G.; et al. Brain MRI Findings in Severe COVID-19: A Retrospective Observational Study. Radiology 2020, 297, E242–E251. [Google Scholar] [CrossRef] [PubMed]
- Chougar, L.; Shor, N.; Weiss, N.; Galanaud, D.; Leclercq, D.; Mathon, B.; Belkacem, S.; Ströer, S.; Burrel, S.; Boutolleau, D.; et al. Retrospective Observational Study of Brain MRI Findings in Patients with Acute SARS-CoV-2 Infection and Neurologic Manifestations. Radiology 2020, 297, E313–E323. [Google Scholar] [CrossRef]
- Pennisi, M.; Lanza, G.; Falzone, L.; Fisicaro, F.; Ferri, R.; Bella, R. Sars-cov-2 and the nervous system: From clinical features to molecular mechanisms. Int. J. Mol. Sci. 2020, 21, 5475. [Google Scholar] [CrossRef]
- Solomon, I.H.; Normandin, E.; Bhattacharyya, S.; Mukerji, S.S.; Keller, K.; Ali, A.S.; Adams, G.; Hornick, J.L.; Padera, R.F., Jr.; Sabeti, P. Neuropathological Features of Covid-19. N. Engl. J. Med. 2020, 383, 989–992. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.; McAuley, D.F.; Brown, M.; Sanchez, E.; Tattersall, R.S.; Manson, J.J. COVID-19: Consider cytokine storm syndromes and immunosuppression. Lancet 2020, 395, 1033–1034. [Google Scholar] [CrossRef]
- Niazkar, H.R.; Zibaee, B.; Nasimi, A.; Bahri, N. The neurological manifestations of COVID-19: A review article. Neurol. Sci. 2020, 41, 1667–1671. [Google Scholar] [CrossRef]
- Durrani, M.; Kucharski, K.; Smith, Z.; Fien, S. Acute Transverse Myelitis Secondary to Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2): A Case Report. Clin. Pract. Cases Emerg. Med. 2020, 4, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Kotfis, K.; Williams Roberson, S.; Wilson, J.E.; Dabrowski, W.; Pun, B.T.; Ely, E.W. COVID-19: ICU delirium management during SARS-CoV-2 pandemic. Crit. Care 2020, 24, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Melegari, G.; Albertini, G.; Romani, A.; Malaguti, S.; Traccitto, F.; Giuliani, E.; Cavallini, G.M.; Bertellini, E.; Barbieri, A. Why should you stay one night? Prospective observational study of enhanced recovery in elderly patients. Aging Clin. Exp. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Ozga, D.; Krupa, S.; Witt, P.; Mędrzycka-Dąbrowska, W. Nursing Interventions to Prevent Delirium in Critically Ill Patients in the Intensive Care Unit during the COVID19 Pandemic—Narrative Overview. Healthcare 2020, 8, 578. [Google Scholar] [CrossRef] [PubMed]
- Duggan, M.C.; Van, J.; Ely, E.W. Delirium Assessment in Critically Ill Older Adults: Considerations during the COVID-19 Pandemic. Crit. Care Clin. 2021, 37, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Rivi, V.; Melegari, G.; Blom, J.M. How to humanise the COVID-19 intensive care units. BMJ Evid.-Based Med. 2021. [Google Scholar] [CrossRef] [PubMed]
- Paterson, R.W.; Brown, R.L.; Benjamin, L.; Nortley, R.; Wiethoff, S.; Bharucha, T.; Jayaseelan, D.L.; Kumar, G.; Raftopoulos, R.E.; Zambreanu, L.; et al. The emerging spectrum of COVID-19 neurology: Clinical, radiological and laboratory findings. Brain 2020, 143, 3104–3120. [Google Scholar] [CrossRef]
- Henry, B.M.; de Oliveira, M.H.S.; Benoit, S.; Plebani, M.; Lippi, G. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): A meta-analysis. Clin. Chem. Lab. Med. 2020, 58, 1021–1028. [Google Scholar] [CrossRef]
- Villa, E.; Critelli, R.; Lasagni, S.; Melegari, A.; Curatolo, A.; Celsa, C.; Romagnoli, D.; Melegari, G.; Pivetti, A.; Di Marco, L.; et al. Dynamic angiopoietin-2 assessment predicts survival and chronic course in hospitalized patients with COVID-19. Blood Adv. 2021, 5, 662–673. [Google Scholar] [CrossRef]
- Monti, G.; Giovannini, G.; Marudi, A.; Bedin, R.; Melegari, A.; Simone, A.M.; Santangelo, M.; Pignatti, A.; Bertellini, E.; Trenti, T.; et al. Anti-NMDA receptor encephalitis presenting as new onset refractory status epilepticus in COVID-19. Seizure 2020, 81, 18–20. [Google Scholar] [CrossRef]
- Xu, Y.; Baylink, D.J.; Chen, C.-S.; Reeves, M.E.; Xiao, J.; Lacy, C.; Lau, E.; Cao, H. The importance of vitamin d metabolism as a potential prophylactic, immunoregulatory and neuroprotective treatment for COVID-19. J. Transl. Med. 2020, 18. [Google Scholar] [CrossRef]
- Mapelli, J.; Gandolfi, D.; Giuliani, E.; Prencipe, F.P.; Pellati, F.; Barbieri, A.; D’Angelo, E.U.; Bigiani, A. The Effect of Desflurane on Neuronal Communication at a Central Synapse. PLoS ONE 2015, 10, e0123534. [Google Scholar] [CrossRef]
- Nieuwenhuijs-Moeke, G.J.; Jainandunsing, J.S.; Struys, M.M.R.F. Sevoflurane, a sigh of relief in COVID-19? Br. J. Anaesth. 2020, 125, 118–121. [Google Scholar] [CrossRef]
- Mirro, E.A.; Halpern, C.H. Letter: Using Continuous Intracranial Electroencephalography Monitoring to Manage Epilepsy Patients during COVID-19. Neurosurgery 2020, 87, E409–E410. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melegari, G.; Rivi, V.; Zelent, G.; Nasillo, V.; De Santis, E.; Melegari, A.; Bevilacqua, C.; Zoli, M.; Meletti, S.; Barbieri, A. Mild to Severe Neurological Manifestations of COVID-19: Cases Reports. Int. J. Environ. Res. Public Health 2021, 18, 3673. https://doi.org/10.3390/ijerph18073673
Melegari G, Rivi V, Zelent G, Nasillo V, De Santis E, Melegari A, Bevilacqua C, Zoli M, Meletti S, Barbieri A. Mild to Severe Neurological Manifestations of COVID-19: Cases Reports. International Journal of Environmental Research and Public Health. 2021; 18(7):3673. https://doi.org/10.3390/ijerph18073673
Chicago/Turabian StyleMelegari, Gabriele, Veronica Rivi, Gabriele Zelent, Vincenzo Nasillo, Elena De Santis, Alessandra Melegari, Claudia Bevilacqua, Michele Zoli, Stefano Meletti, and Alberto Barbieri. 2021. "Mild to Severe Neurological Manifestations of COVID-19: Cases Reports" International Journal of Environmental Research and Public Health 18, no. 7: 3673. https://doi.org/10.3390/ijerph18073673
APA StyleMelegari, G., Rivi, V., Zelent, G., Nasillo, V., De Santis, E., Melegari, A., Bevilacqua, C., Zoli, M., Meletti, S., & Barbieri, A. (2021). Mild to Severe Neurological Manifestations of COVID-19: Cases Reports. International Journal of Environmental Research and Public Health, 18(7), 3673. https://doi.org/10.3390/ijerph18073673





