Does Delaying Time in Cancer Treatment Affect Mortality? A Retrospective Cohort Study of Korean Lung and Gastric Cancer Patients
Abstract
:1. Introduction
2. Methods
2.1. Study Population
2.2. Variables
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Barta, J.A.; Powell, C.A.; Wisnivesky, J.P. Global Epidemiology of Lung Cancer. Ann. Glob. Health 2019, 85, 8. [Google Scholar] [CrossRef] [Green Version]
- Hong, S.; Won, Y.J.; Lee, J.J.; Park, Y.R.; Jung, K.W.; Kong, H.J.; Lee, E.S.; Im, J.-S.; Seo, H.G. Cancer Statistics in Korea: Incidence, Mortality, Survival, and Prevalence in 2017. Cancer Res. Treat. 2020, 52, 335–350. [Google Scholar] [CrossRef] [PubMed]
- Global Burden of Disease Cancer Collaboration; Fitzmaurice, C.; Allen, C.; Barber, R.M.; Barregard, L.; Bhutta, Z.A.; Brenner, H.; Dicker, D.J.; Chimed-Orchir, O.; Dandona, R.; et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived with Disability, and Disa-bility-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Dis-ease Study. JAMA Oncol. 2017, 3, 524–548. [Google Scholar] [CrossRef]
- Brenkman, H.; Visser, E.; van Rossum, P.; Siesling, S.; van Hillegersberg, R.; Ruurda, J. Association between wait-ing time from diagnosis to treatment and survival in patients with curable gastric cancer: A population-based study in the Netherlands. Ann. Surg. Oncol. 2017, 24, 1761–1769. [Google Scholar] [CrossRef] [Green Version]
- Miles, A.; McClements, P.L.; Steele, R.J.; Redeker, C.; Sevdalis, N.; Wardle, J. Perceived diagnostic delay and cancer-related distress: A cross-sectional study of patients with colorectal cancer. Psychooncology 2017, 26, 29–36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neal, R.; Tharmanathan, P.; France, B.; Din, N.; Cotton, S.; Fallon-Ferguson, J.; Hamilton, W.; Hendry, A.; Hendry, M.; Lewis, R.; et al. Is increased time to diagno-sis and treatment in symptomatic cancer associated with poorer outcomes? Systematic review. Br. J. Cancer 2015, 112, S92–S107. [Google Scholar] [CrossRef] [Green Version]
- Samson, P.; Patel, A.; Garrett, T.; Crabtree, T.; Kreisel, D.; Krupnick, A.S.; Patterson, G.A.; Broderick, S.; Meyers, B.F.; Puri, V. Effects of Delayed Surgical Resec-tion on Short-Term and Long-Term Outcomes in Clinical Stage I Non-Small Cell Lung Cancer. Ann. Thorac Surg. 2015, 99, 1906–1912, discussion 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khorana, A.A.; Tullio, K.; Elson, P.; Pennell, N.A.; Grobmyer, S.R.; Kalady, M.F.; Raymond, D.; Abraham, J.; Klein, E.A.; Walsh, R.M.; et al. Time to initial cancer treatment in the United States and association with survival over time: An observational study. PLoS ONE 2019, 14, e0213209. [Google Scholar]
- Malalasekera, A.; Nahm, S.; Blinman, P.L.; Kao, S.C.; Dhillon, H.M.; Vardy, J.L. How long is too long? A scoping review of health system delays in lung cancer. Eur. Respir. Rev. 2018, 27, 180045. [Google Scholar] [CrossRef] [PubMed]
- Yun, Y.H.; Kim, Y.A.; Min, Y.H.; Park, S.; Won, Y.J.; Kim, D.Y.; Choi, I.J.; Kim, J.H.; Lee, D.H.; Yoon, S.J.; et al. The influence of hospital volume and surgical treatment delay on long-term survival after cancer surgery. Ann. Oncol. 2012, 23, 2731–2737. [Google Scholar] [CrossRef]
- Shin, D.W.; Cho, J.; Kim, S.Y.; Guallar, E.; Hwang, S.S.; Cho, B.; Oh, J.H.; Jung, K.W.; Seo, H.G.; Park, J.H. Delay to curative surgery greater than 12 weeks is associated with increased mortality in patients with colorectal and breast cancer but not lung or thy-roid cancer. Ann. Surg. Oncol. 2013, 20, 2468–2476. [Google Scholar] [CrossRef]
- Buchmueller, T.C.; Jacobson, M.; Wold, C. How far to the hospital?: The effect of hospital closures on access to care. J. Health Econ. 2006, 25, 740–761. [Google Scholar] [CrossRef] [PubMed]
- Hung, P.; Casey, M.M.; Kozhimannil, K.B.; Karaca-Mandic, P.; Moscovice, I.S. Rural-urban differences in access to hospital obstetric and neonatal care: How far is the closest one? J. Perinatol. 2018, 38, 645–652. [Google Scholar] [CrossRef]
- Gomez, D.; Liao, K.-P.; Swisher, S.; Blumenschein, G.; Erasmus, J.R.J.; Buchholz, T.A.; Giordano, S.H.; Smith, B.D. Time to treatment as a quality metric in lung cancer: Staging studies, time to treatment, and patient survival. Radiother. Oncol. 2015, 115, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.H.; Kung, P.T.; Kuo, W.Y.; Tsai, W.C. Effect of time interval from diagnosis to treatment for non-small cell lung cancer on survival: A national cohort study in Taiwan. BMJ Open 2020, 10, e034351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radzikowska, E.; Roszkowski-Sliz, K.; Glaz, P. The impact of timeliness of care on survival in non-small cell lung cancer patients. Pneumonol. Alergol. Pol. 2012, 80, 422–429. [Google Scholar] [PubMed]
- Myrdal, G.; Lambe, M.; Hillerdal, G.; Lamberg, K.; Agustsson, T.; Stahle, E. Effect of delays on prognosis in pa-tients with non-small cell lung cancer. Thorax 2004, 59, 45–49. [Google Scholar]
- Maiga, A.W.; Deppen, S.A.; Pinkerman, R.; Callaway-Lane, C.; Massion, P.P.; Dittus, R.S.; Lambright, E.S.; Nesbitt, J.C.; Baker, D.; Grogan, E.L. Timeliness of Care and Lung Cancer Tumor-Stage Progression: How Long Can We Wait? Ann. Thorac. Surg. 2017, 104, 1791–1797. [Google Scholar] [CrossRef] [Green Version]
- Ahn, G.T.; Baek, S.K.; Han, J.J.; Kim, H.J.; Jeong, S.J.; Maeng, C.H. Optimal time interval from surgery to adjuvant chemotherapy in gastric cancer. Oncol. Lett. 2020, 20, 32. [Google Scholar] [CrossRef]
- Park, J.M.; Kim, Y.H. Current approaches to gastric cancer in Korea. Gastrointest Cancer Res. 2008, 2, 137–144. [Google Scholar]
- Lee, J.W.; Ali, B.; Yoo, H.M.; Park, C.H.; Song, K.Y. Conditional survival analysis in Korean patients with gastric cancer undergoing curative gastrectomy. BMC Cancer 2015, 15, 1005. [Google Scholar] [CrossRef] [Green Version]
- Yang, P. Epidemiology of lung cancer prognosis: Quantity and quality of life. Methods Mol. Biol. 2009, 471, 469–486. [Google Scholar] [PubMed] [Green Version]
- Leduc, C.; Antoni, D.; Charloux, A.; Falcoz, P.E.; Quoix, E. Comorbidities in the management of patients with lung cancer. Eur. Respir. J. 2017, 49, 1601721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rim, C.H.; Lee, J.; Kim, W.C.; Yang, D.; Yoon, W.S.; Koom, W.S.; Kim, C.Y. A Survey of Radiation Therapy Utilization in Korea from 2010 to 2016: Focusing on Use of Intensity-Modulated Radiation Therapy. J. Korean Med. Sci. 2018, 33, e67. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Park, I.K.; Kim, E.R.; Hwang, Y.; Lee, H.J.; Kang, C.H.; Kim, Y.T. Current Trends of Lung Cancer Surgery and Demographic and Social Factors Related to Changes in the Trends of Lung Cancer Surgery: An Analysis of the National Database from 2010 to 2014. Cancer Res. Treat. 2017, 49, 330–337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.; Ahn, S.; Lee, B.; Lee, K.; Yoo, S.; Lee, K.; Suh, D.H.; No, J.H.; Kim, Y.B. Factors associated with patients’ choice of physician in the Korean population: Database analyses of a tertiary hospital. PLoS ONE 2018, 13, e0190472. [Google Scholar] [CrossRef] [Green Version]
- Han, K.T.; Kim, S.J. Instability in daily life and depression: The impact of sleep variance between weekday and weekend in South Korean workers. Health Soc. Care Community 2020, 28, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Yang, S.-Y.; Cho, H.-S.; Kim, W.; Park, E.-C.; Han, K.-T. Mortality differences by surgical volume among patients with stomach cancer: A threshold for a favorable volume-outcome relationship. World J. Surg. Oncol. 2017, 15, 1–9. [Google Scholar] [CrossRef]
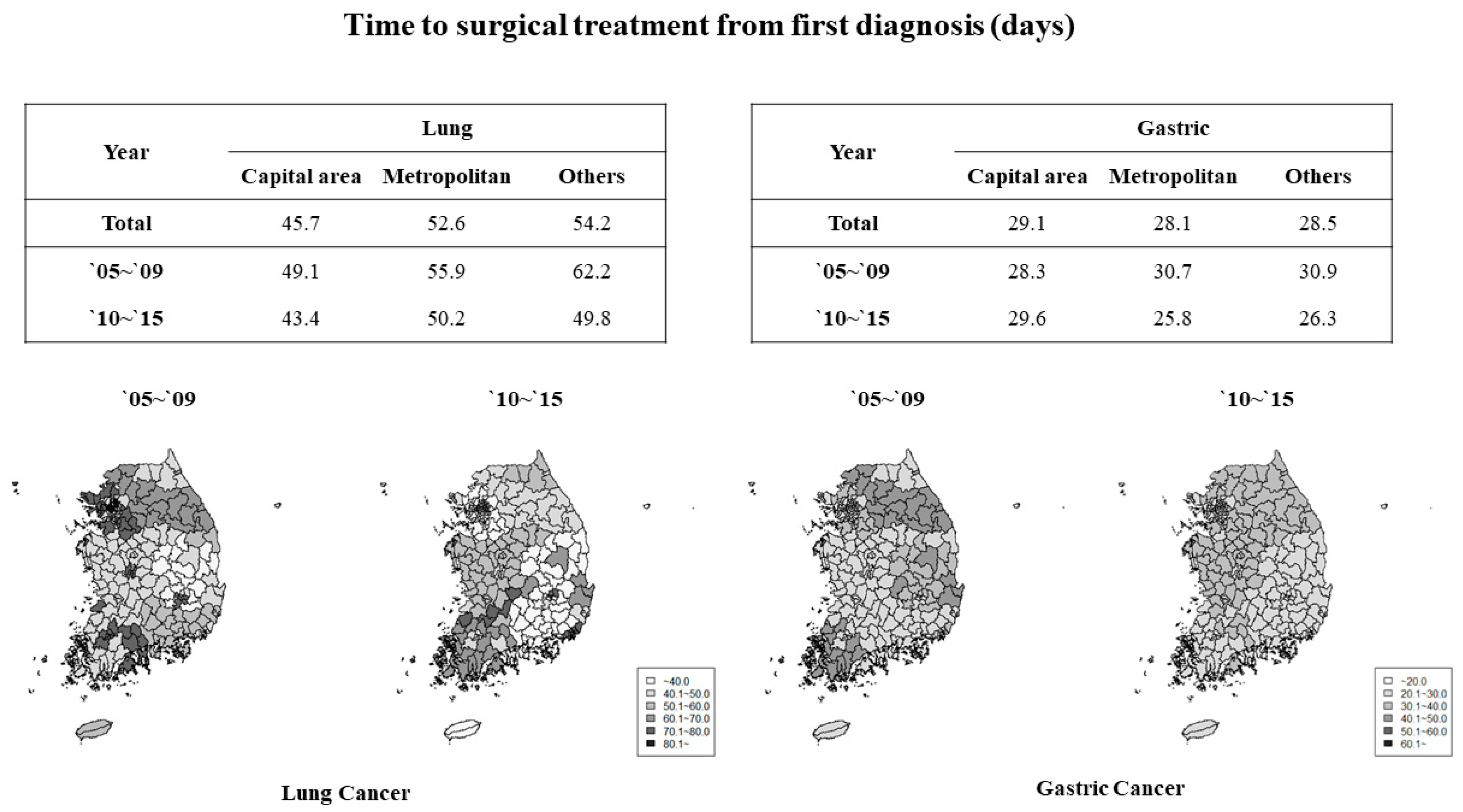

| Variables | Lung Cancer | Gastric Cancer | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Within 50 Days | After 51 Days | p-Value | Within 30 Days | After 31 Days | p-Value | |||||
| N/Mean | %/SD | N/Mean | %/SD | N/Mean | %/SD | N/Mean | %/SD | |||
| Sex | ||||||||||
| Male | 409 | 73.7 | 146 | 26.3 | 0.278 | 1252 | 69.5 | 549 | 30.5 | 0.006 |
| Female | 197 | 77.3 | 58 | 22.7 | 641 | 74.7 | 217 | 25.3 | ||
| Age (Years) | ||||||||||
| ~49 | 40 | 80.0 | 10 | 20.0 | 0.500 | 221 | 72.5 | 84 | 27.5 | 0.145 |
| 50~54 | 29 | 67.4 | 14 | 32.6 | 201 | 74.4 | 69 | 25.6 | ||
| 54~59 | 50 | 70.4 | 21 | 29.6 | 251 | 71.3 | 101 | 28.7 | ||
| 60~64 | 101 | 73.7 | 36 | 26.3 | 278 | 75.3 | 91 | 24.7 | ||
| 65~69 | 109 | 80.7 | 26 | 19.3 | 257 | 67.6 | 123 | 32.4 | ||
| 70~74 | 112 | 76.7 | 34 | 23.3 | 274 | 67.7 | 131 | 32.3 | ||
| 75+ | 165 | 72.4 | 63 | 27.6 | 411 | 71.1 | 167 | 28.9 | ||
| Income Level | ||||||||||
| ~20 Percentile | 72 | 66.7 | 36 | 33.3 | 0.066 | 286 | 72.8 | 107 | 27.2 | 0.196 |
| 21~40 Percentile | 68 | 70.8 | 28 | 29.2 | 274 | 74.1 | 96 | 25.9 | ||
| 41~60 Percentile | 105 | 77.8 | 30 | 22.2 | 338 | 69.5 | 148 | 30.5 | ||
| 61~80 Percentile | 138 | 72.6 | 52 | 27.4 | 424 | 74.0 | 149 | 26.0 | ||
| 81 Percentile~ | 223 | 79.4 | 58 | 20.6 | 571 | 68.2 | 266 | 31.8 | ||
| Types of Insurance Coverage | ||||||||||
| NHI, Self-employed | 192 | 71.6 | 76 | 28.4 | 0.144 | 671 | 71.0 | 274 | 29.0 | 0.875 |
| NHI, Employee | 414 | 76.4 | 128 | 23.6 | 1222 | 71.3 | 492 | 28.7 | ||
| Residence Area | ||||||||||
| Capital area | 250 | 78.4 | 69 | 21.6 | 0.041 | 686 | 70.9 | 282 | 29.1 | 0.740 |
| Metropolitan | 132 | 68.4 | 61 | 31.6 | 512 | 72.3 | 196 | 27.7 | ||
| Others | 224 | 75.2 | 74 | 24.8 | 695 | 70.7 | 288 | 29.3 | ||
| Charlson Comorbidity Index | ||||||||||
| ~3 | 136 | 81.0 | 32 | 19.0 | 0.058 | 703 | 74.9 | 236 | 25.1 | 0.005 |
| 4~6 | 202 | 75.7 | 65 | 24.3 | 660 | 68.1 | 309 | 31.9 | ||
| 7~ | 268 | 71.5 | 107 | 28.5 | 530 | 70.6 | 221 | 29.4 | ||
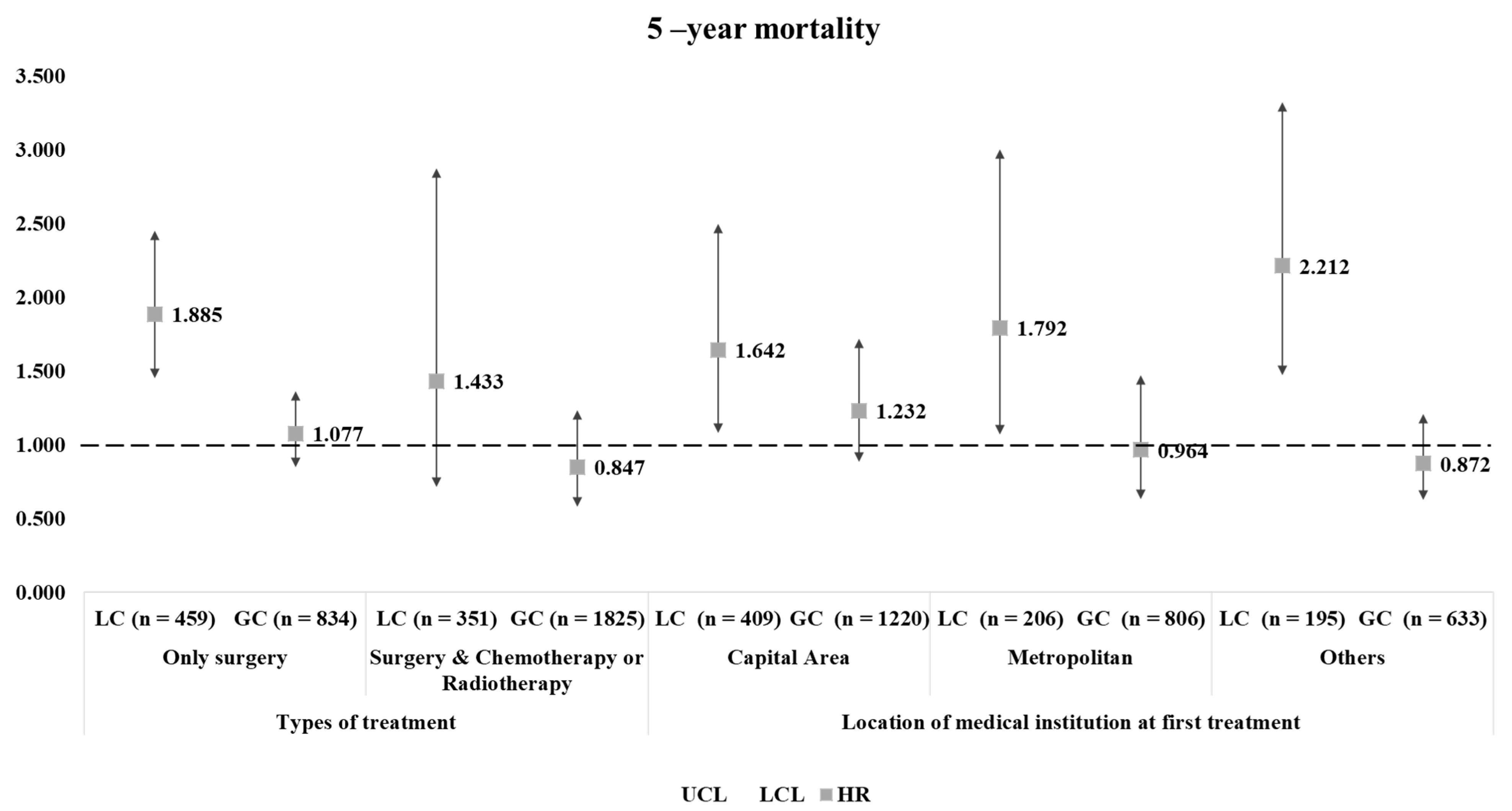
| Types of Treatment | ||||||||||
| Only surgery | 328 | 71.5 | 131 | 28.5 | 0.012 | 626 | 75.1 | 208 | 24.9 | 0.003 |
| Surgery & Chemotherapy or Radiotherapy | 278 | 79.2 | 73 | 20.8 | 1267 | 69.4 | 558 | 30.6 | ||
| Types of Medical Institution at First Treatment | ||||||||||
| General Hospital | 217 | 81.3 | 50 | 18.7 | 0.003 | 546 | 69.1 | 244 | 30.9 | 0.124 |
| Others | 389 | 71.6 | 154 | 28.4 | 1347 | 72.1 | 522 | 27.9 | ||
| Location of Medical Institution at First Treatment | ||||||||||
| Capital area | 329 | 80.4 | 80 | 19.6 | 0.001 | 859 | 70.4 | 361 | 29.6 | 0.711 |
| Metropolitan | 143 | 69.4 | 63 | 30.6 | 580 | 72.0 | 226 | 28.0 | ||
| Others | 134 | 68.7 | 61 | 31.3 | 454 | 71.7 | 179 | 28.3 | ||
| Total | 606 | 74.8 | 204 | 25.2 | 1893 | 71.2 | 766 | 28.8 | ||
| Variables | Lung Cancer | Gastric Cancer | ||||||
|---|---|---|---|---|---|---|---|---|
| HR | 95% CI | p-Value | HR | 95% CI | p-Value | |||
| Time to Surgical Treatment | ||||||||
| Below average time | 1.000 | - | - | - | 1.000 | - | - | - |
| Above average time | 1.826 | 1.437 | 2.321 | <0.001 | 1.003 | 0.822 | 1.225 | 0.974 |
| Sex | ||||||||
| Male | 1.280 | 0.982 | 1.669 | 0.068 | 1.142 | 0.935 | 1.396 | 0.193 |
| Female | 1.000 | - | - | - | 1.000 | - | - | - |
| Age (Years) | ||||||||
| ~49 | 1.000 | - | - | - | 1.000 | - | - | - |
| 50~54 | 0.975 | 0.416 | 2.287 | 0.954 | 0.621 | 0.390 | 0.991 | 0.046 |
| 54~59 | 1.197 | 0.559 | 2.563 | 0.643 | 0.671 | 0.441 | 1.021 | 0.062 |
| 60~64 | 1.416 | 0.709 | 2.831 | 0.325 | 0.788 | 0.515 | 1.206 | 0.272 |
| 65~69 | 1.356 | 0.681 | 2.700 | 0.386 | 0.849 | 0.572 | 1.261 | 0.418 |
| 70~74 | 1.271 | 0.639 | 2.530 | 0.495 | 1.230 | 0.852 | 1.774 | 0.269 |
| 75+ | 2.932 | 1.513 | 5.681 | 0.001 | 2.216 | 1.571 | 3.125 | <0.001 |
| Income Level | ||||||||
| ~20 Percentile | 0.813 | 0.526 | 1.257 | 0.351 | 0.818 | 0.591 | 1.132 | 0.226 |
| 21~40 Percentile | 0.793 | 0.525 | 1.198 | 0.270 | 0.822 | 0.611 | 1.107 | 0.197 |
| 41~60 Percentile | 0.750 | 0.512 | 1.098 | 0.139 | 0.721 | 0.540 | 0.962 | 0.026 |
| 61~80 Percentile | 0.773 | 0.536 | 1.113 | 0.166 | 0.768 | 0.584 | 1.010 | 0.059 |
| 81 Percentile~ | 1.000 | - | - | - | 1.000 | - | - | - |
| Types of Insurance Coverage | ||||||||
| NHI, Self-employed | 1.212 | 0.956 | 1.536 | 0.112 | 1.082 | 0.897 | 1.306 | 0.410 |
| NHI, Employee | 1.000 | - | - | - | 1.000 | - | - | - |
| Residence Area | ||||||||
| Capital area | 1.336 | 0.848 | 2.104 | 0.212 | 1.234 | 0.844 | 1.803 | 0.278 |
| Metropolitan | 1.371 | 0.917 | 2.049 | 0.124 | 0.879 | 0.634 | 1.218 | 0.439 |
| Others | 1.000 | - | - | - | 1.000 | - | - | - |
| Charlson Comorbidity Index | ||||||||
| ~3 | 1.000 | - | - | - | 1.000 | - | - | - |
| 4~6 | 1.201 | 0.789 | 1.829 | 0.393 | 1.428 | 1.071 | 1.903 | 0.015 |
| 7~ | 2.114 | 1.436 | 3.112 | 0.0001 | 2.950 | 2.267 | 3.838 | <0.001 |
| Types of Treatment | ||||||||
| Only surgery | 1.000 | - | - | - | 1.000 | - | - | - |
| Surgery & Chemotherapy or Radiotherapy | 3.884 | 2.836 | 5.320 | <0.001 | 5.800 | 4.710 | 7.142 | <0.001 |
| Types of Medical Institution at First Treatment | ||||||||
| General Hospital | 1.059 | 0.745 | 1.505 | 0.750 | 0.813 | 0.611 | 1.082 | 0.156 |
| Others | 1.000 | - | - | - | 1.000 | - | - | - |
| Location of Medical Institution at First Treatment | ||||||||
| Capital area | 1.000 | - | - | - | 1.000 | - | - | - |
| Metropolitan | 0.962 | 0.600 | 1.540 | 0.870 | 0.769 | 0.513 | 1.152 | 0.203 |
| Others | 0.934 | 0.582 | 1.497 | 0.776 | 1.117 | 0.757 | 1.648 | 0.577 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, K.-T.; Kim, W.; Kim, S. Does Delaying Time in Cancer Treatment Affect Mortality? A Retrospective Cohort Study of Korean Lung and Gastric Cancer Patients. Int. J. Environ. Res. Public Health 2021, 18, 3462. https://doi.org/10.3390/ijerph18073462
Han K-T, Kim W, Kim S. Does Delaying Time in Cancer Treatment Affect Mortality? A Retrospective Cohort Study of Korean Lung and Gastric Cancer Patients. International Journal of Environmental Research and Public Health. 2021; 18(7):3462. https://doi.org/10.3390/ijerph18073462
Chicago/Turabian StyleHan, Kyu-Tae, Woorim Kim, and Seungju Kim. 2021. "Does Delaying Time in Cancer Treatment Affect Mortality? A Retrospective Cohort Study of Korean Lung and Gastric Cancer Patients" International Journal of Environmental Research and Public Health 18, no. 7: 3462. https://doi.org/10.3390/ijerph18073462






