In Vitro Anti-Leptospiral Activity of Phyllanthus amarus Extracts and Their Combinations with Antibiotics
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling and Plant Extraction
2.2. Ellinghausen McCullough Johnson Harris Broth Preparation
2.3. Antibiotic Solution Preparation
2.4. Leptospira Inoculum Preparation
2.5. Antimicrobial Susceptibility Testing
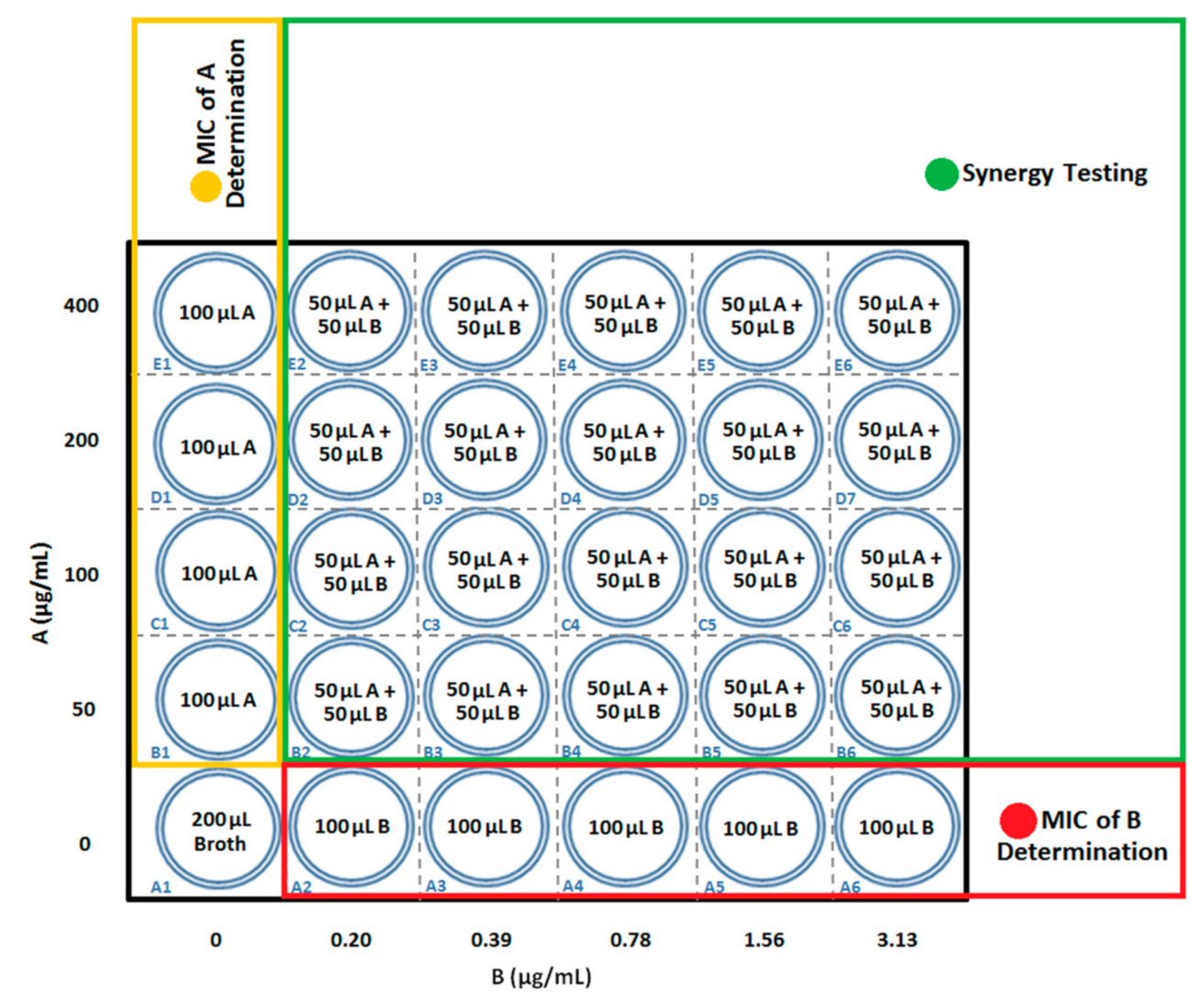
2.6. Synergy Testing by Checkerboard Assay
2.7. Scanning Electron Microscope Analysis
2.8. Statistical Analysis
3. Results
3.1. Antimicrobial Susceptibility Testing
3.2. Synergy Testing
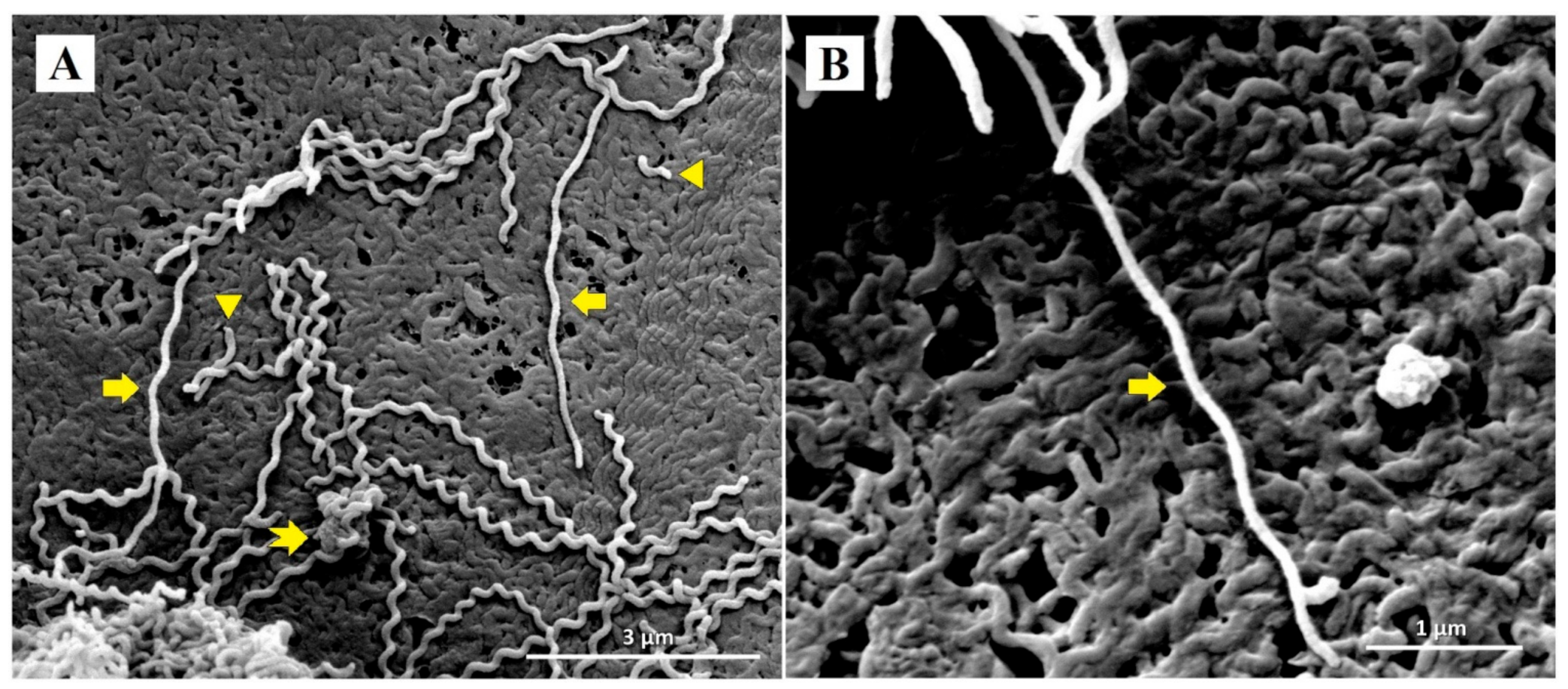
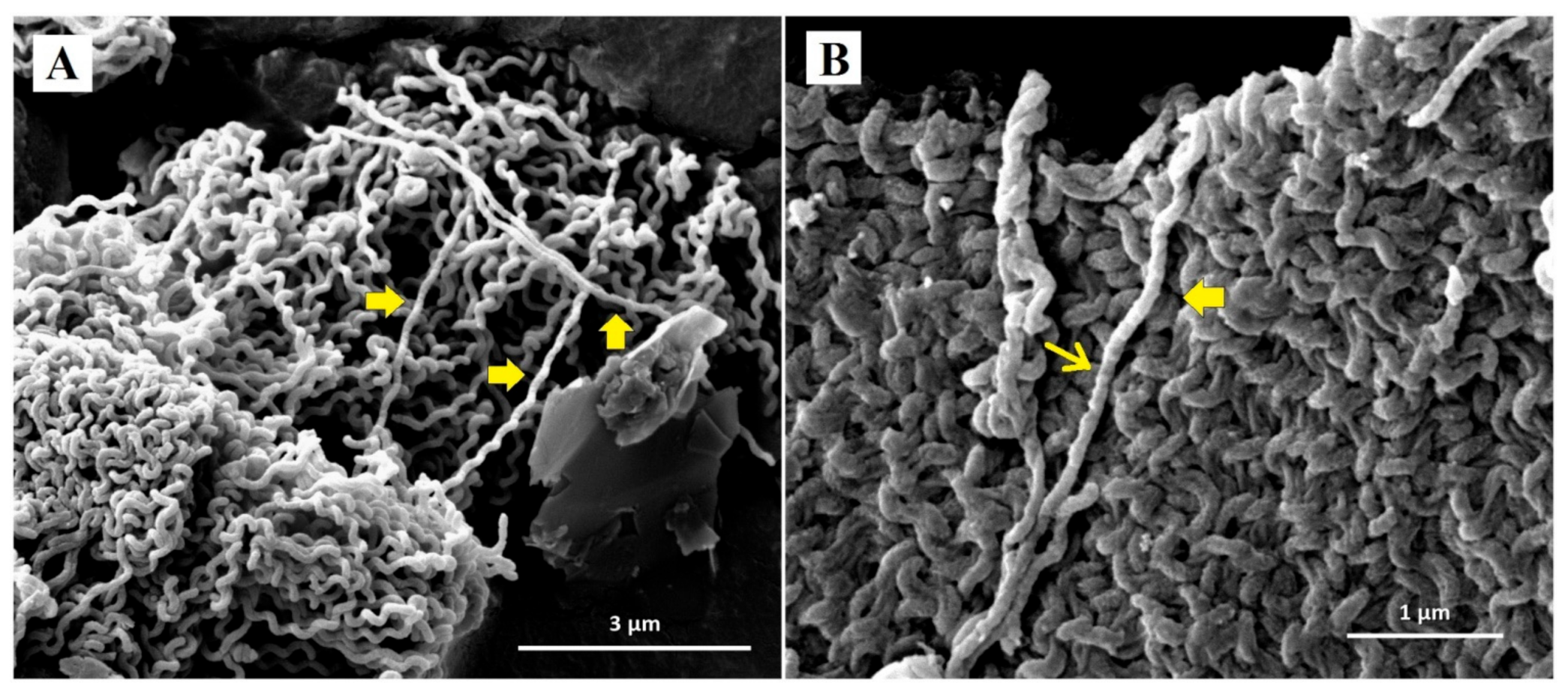
3.3. SEM Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lim, V.K.E. Leptospirosis: A re-emerging infection. Malays. J. Pathol. 2011, 33, 1–5. [Google Scholar]
- Prabhakaran, S.G.; Shanmughapriya, S.; Dhanapaul, S.; James, A.; Natarajaseenivasan, K. Risk factors associated with rural and urban epidemics of leptospirosis in Tiruchirappalli district of Tamilnadu, India. J. Public Health 2014, 22, 323–333. [Google Scholar] [CrossRef]
- Haake, D.A.; Levett, P.N. Leptospirosis in humans. Curr. Top. Microbiol. Immunol. 2015, 387, 65–97. [Google Scholar] [CrossRef]
- World Health Organization. Leptospirosis: Fact Sheet; Regional Office for South-East Asia: New Delhi, India, 2009; pp. 1–12. [Google Scholar]
- Levett, P.N. Leptospirosis. Clin. Microbiol. Rev. 2001, 14, 296–326. [Google Scholar] [CrossRef] [PubMed]
- Costa, F.; Hagan, J.E.; Calcagno, J.; Kane, M.; Torgerson, P.; Martinez-Silveira, M.S.; Stein, C.; Abela-Ridder, B.; Ko, A.I. Global morbidity and mortality of leptospirosis: A Systematic Review. PLOS Negl. Trop. Dis. 2015, 9, e0003898. [Google Scholar] [CrossRef]
- Marchiori, E.; Lourenço, S.; Setúbal, S.; Zanetti, G.; Gasparetto, T.D.; Hochhegger, B. Clinical and imaging manifestations of hemorrhagic pulmonary leptospirosis: A state-of-the-art review. Lung 2011, 189, 1–9. [Google Scholar] [CrossRef]
- Fraga, T.R.; Carvalho, E.; Isaac, L.; Barbosa, A.S. Leptospira and leptospirosis. In Molecular Medical Microbiology; Yi-Wei, T., Liu, D., Schwartzman, J., Sussman, M., Poxton, I., Eds.; Academic Press: Amsterdam, The Netherlands, 2015; pp. 1973–1990. [Google Scholar]
- Gyawali, R.; Ibrahim, S.A. Natural products as antimicrobial agents. Food Control 2014, 46, 412–429. [Google Scholar] [CrossRef]
- Joseph, B.; Raj, S.J. Overview pharmacognostic properties of Phyllanthus Amarus Linn. Int. J. Pharmacol. 2011, 7, 40–45. [Google Scholar] [CrossRef]
- Sarin, B.; Verma, N.; Martín, J.P.; Mohanty, A. An overview of important ethnomedicinal herbs of Phyllanthus species: Present status and future prospects. Sci. World J. 2014, 2014, 839172. [Google Scholar] [CrossRef]
- Oladosu, S.A.; Coker, A.O.; Nwaokorie, F. Antibacterial effects of Phyllantus amarus on urinary tract pathogens. Int. Clin. Pathol. J. 2019, 7, 1–10. [Google Scholar] [CrossRef]
- Bharathi, T.; Kolanjinathan, K.; Saranraj, P. Antimicrobial activity of solvent extracts of Ocimum sanctum, Azadirachta indica and Phyllanthus amarus against clinical pathogens. Glob. J. Pharmacol. 2014, 8, 294–305. [Google Scholar] [CrossRef]
- Mazumder, A.; Mahato, A.; Mazumder, R. Antimicrobial potentiality of Phyllanthus amarus against drug resistant pathogens. Nat. Prod. Res. 2006, 20, 323–326. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.S.; Hegde, K.S.; Chandrashekhar, S.; Rao, S.N.; Manikkoth, S. Preclinical screening of Phyllanthus amarus ethanolic extract for its analgesic and antimicrobial activity. Pharmacogn. Res. 2015, 7, 378–384. [Google Scholar] [CrossRef]
- Sekar, D.; Kolanjinathan, K.; Saranraj, P.; Gajendiran, K. Screening of Phyllanthus amarus, Acalypha indica and Datura metel for its antimicrobial activity against selected pathogens. Int. J. Pharm. Biol. Arch. 2012, 3, 1231–1235. [Google Scholar]
- Oluwafemi, F.; Debiri, F. Antimicrobial effect of Phyllanthus amarus and Parquetina nigrescens on Salmonella typhi. Afr. J. Biomed. Res. 2008, 11, 215–219. [Google Scholar] [CrossRef]
- Chandan, S.; Umesha, S.; Balamurugan, V. Antileptospiral, antioxidant and dna damaging properties of Eclipta alba and Phyllanthus amarus. Open Access Sci. Rep. 2012, 1, 231. [Google Scholar] [CrossRef]
- Suwari, H.Z.K.; Buang, Y. Optimization of soxhlet extraction and physicochemical analysis of crop oil from seed kernel of Feun kase (Thevetia peruviana). AIP Conf. Proc. 2017, 1911, 1–7. [Google Scholar] [CrossRef]
- Azwanida, N.N. A review on the extraction methods use in medicinal plants, principle, strength and limitation. Med. Aromat. Plants 2015, 4, 196. [Google Scholar] [CrossRef]
- Murray, C.K.; Ellis, M.W.; Hospenthal, D.R. Susceptibility of Leptospira serovars to antimalarial agents. Am. J. Trop. Med. Hyg. 2004, 71, 685–686. [Google Scholar] [CrossRef]
- Drancourt, M.; Raoult, D. Methodology of antibiotics testing for intracellular pathogens. In Antimicrobial Agents and Intracellular Pathogens; Raoult, D., Ed.; CRC Press, Inc.: Boca Raton, FL, USA, 1993; pp. 73–85. [Google Scholar]
- European Committee for Antimicrobial Susceptibility Testing of the European Society of Clinical Microbiology Infectious Diseases. Determination of minimum inhibitory concentrations (mics) of antibacterial agents by broth dilution. Clin. Microbiol. Infect. 2003, 9, ix–xv. [Google Scholar] [CrossRef]
- Van Vuuren, S.; Viljoen, A. Plant-based antimicrobial studies–methods and approaches to study the interaction between natural products. Planta Med. 2011, 77, 1168–1182. [Google Scholar] [CrossRef] [PubMed]
- Den Hollander, J.G.; Mouton, J.W. The predictive value of laboratory tests for efficacy of antibiotic combination therapy. In Antimicrobial Pharmacodynamics in Theory and Clinical Practice; Nightingale, C.H., Ambrose, P.G., Drusano, G.L., Murakawa, T., Eds.; Informa Healthcare USA, Inc.: New York, NY, USA, 2007; pp. 103–127. [Google Scholar]
- Alli, A.I.; Ehinmidu, J.O.; Ibrahim, Y.K.E. Preliminary phytochemical screening and antimicrobial activities of some medicinal plants used in Ebiraland. Bayero J. Pure Appl. Sci. 2011, 4, 10–16. [Google Scholar] [CrossRef]
- Sen, A.; Batra, A. Determination of antimicrobial potentialities of different solvent extracts of the medicinal plant: Phyllanthus amarus Schum. and Thonn. Int. J. Gree Pharm. 2012, 6, 50–56. [Google Scholar] [CrossRef]
- Tiwari, U.; Cummins, E. Factors influencing levels of phytochemicals in selected fruit and vegetables during pre-and post-harvest food processing operations. Food Res. Int. 2013, 50, 497–506. [Google Scholar] [CrossRef]
- Ogunjobi, A.A.; Abiala, M.A. Antimicrobial activity of Senna alata and Phyllanthus amarus. Glob. J. Pharmacol. 2013, 7, 198–202. [Google Scholar] [CrossRef]
- Dhandapani, R.; Lakshmi, D.; Balakrishnan, V.; Jayakumar, S.; Kumar, A. Preliminary phytochemical investigation and antibacterial activity of Phyllanthus amarus Schum & Thorn. Anc. Sci. Life 2007, 27, 1–5. [Google Scholar]
- Zubair, M.; Atolani, O.; Ibrahim, S.; Adebisi, O.; Hamid, A.; Sowunmi, R. Chemical constituents and antimicrobial properties of Phyllanthus amarus (Schum & Thonn). Bayero J. Pure Appl. Sci. 2017, 10, 238–246. [Google Scholar] [CrossRef]
- Shirahatti, P.S.; Syed, A.; Elgorban, A.M. Potential antileptospiral constituents from Phyllanthus amarus. Pharmacogn. Mag. 2020, 16, S371–S378. [Google Scholar] [CrossRef]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Lahiri, D.; Dash, S.; Dutta, R.; Nag, M. Elucidating the effect of anti-biofilm activity of bioactive compounds extracted from plants. J. Biosci. 2019, 44, 1–19. [Google Scholar] [CrossRef]
- Mahmood, Z.; Yameen, M.; Jahangeer, M.; Riaz, M.; Ghaffar, A.; Javid, I. Lignin as Natural Antioxidant Capacity. In Lignin—Trends and Application; Poletto, M., Ed.; IntechOpen: Rijeka, Croatia, 2018; pp. 181–205. [Google Scholar]
- Dong, S.; Yang, X.; Zhao, L.; Zhang, F.; Hou, Z.; Xue, P. Antibacterial activity and mechanism of action saponins from Chenopodium quinoa Willd. husks against foodborne pathogenic bacteria. Ind. Crops Prod. 2020, 149, 112350. [Google Scholar] [CrossRef]
- Yu, Z.-H.; Ding, X.-Z.; Xia, L.-Q.; Xiao, X.-Q.; Cao, Z.-P.; Xu, S.; Liu, S.; Liu, X.-M. Antimicrobial activity and mechanism of total saponins from Allium chinense. Food Sci. 2013, 34, 75–80. [Google Scholar] [CrossRef]
- Wei, Y.; Liu, Q.; Yu, J.; Feng, Q.; Zhao, L.; Song, H.; Wang, W. Antibacterial mode of action of 1, 8-dihydroxy-anthraquinone from Porphyra haitanensis against Staphylococcus aureus. Nat. Prod. Res. 2015, 29, 976–979. [Google Scholar] [CrossRef] [PubMed]
- Doğan, A.; Otlu, S.; Çelebi, Ö.; Aksu, P.; Sağlam, A.G.; Doğan, A.N.C.; Mutlu, N. An investigation of antibacterial effects of steroids. Turkish J. Vet. Anim. Sci. 2017, 41, 302–305. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, J.-G. Health functions and structure–activity relationships of natural anthraquinones from plants. Food Funct. 2018, 9, 6063–6080. [Google Scholar] [CrossRef]
- Liegeon, G.; Delory, T.; Picardeau, M.A. Antibiotic susceptibilities of livestock isolates of Leptospira. Int. J. Antimicrob. Agents 2018, 51, 693–699. [Google Scholar] [CrossRef]
- Chitra, M.A. Susceptibility of Leptospira serotypes against various antimicrobials by broth microdilution method. Indian J. Anim. Sci. 2013, 83, 775–778. [Google Scholar]
- Ressner, R.A.; Griffith, M.E.; Beckius, M.L.; Pimentel, G.; Miller, R.S.; Mende, K.; Fraser, S.L.; Galloway, R.L.; Hospenthal, D.R.; Murray, C.K. Antimicrobial susceptibilities of geographically diverse clinical human isolates of Leptospira. Antimicrob. Agents Chemother. 2008, 52, 2750–2754. [Google Scholar] [CrossRef] [PubMed]
- Benacer, D.; Mohd Zain, S.N.; Ooi, P.T.; Thong, K.L. Antimicrobial susceptibility of Leptospira spp. isolated from environmental, human and animal sources in Malaysia. Indian J. Med. Microbiol. 2017, 35, 124–128. [Google Scholar] [CrossRef]
- Murray, C.K.; Hospenthal, D.R. Determination of susceptibilities of 26 Leptospira sp. serovars to 24 antimicrobial agents by a broth microdilution technique. Antimicrob. Agents Chemother. 2004, 48, 4002–4005. [Google Scholar] [CrossRef]
- Seesom, W.; Jaratrungtawee, A.; Suksamrarn, S.; Mekseepralard, C.; Ratananukul, P.; Sukhumsirichart, W. Antileptospiral activity of xanthones from Garcinia mangostana and synergy of gamma-mangostin with penicillin G. BMC Complement. Altern. Med. 2013, 13, 182. [Google Scholar] [CrossRef] [PubMed]
- Kersten, A.; Poitschek, C.; Rauch, S.; Aberer, E. Effects of Penicillin, Ceftriaxone, and Doxycycline on Morphology of Borrelia burgdorferi. Antimicrob. Agents Chemother. 1995, 39, 1127–1133. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meriläinen, L.; Herranen, A.; Schwarzbach, A.; Gilbert, L. Morphological and biochemical features of Borrelia burgdorferi pleomorphic forms. Microbiology 2015, 161, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Toyofuku, M.; Nomura, N.; Eberl, L. Types and origins of bacterial membrane vesicles. Nat. Rev. Microbiol. 2019, 17, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Girschick, H.J. Lyme borreliosis: From infection to autoimmunity. Clin. Microbiol. Infect. 2004, 10, 598–614. [Google Scholar] [CrossRef]


| Leptospira Serovars | AE (µg/mL) | ME (µg/mL) | ||
|---|---|---|---|---|
| MIC | MBC | MIC | MBC | |
| L. interrogans serovar Australis | 400 ± 0.0 | 400 ± 0.0 | 800 ± 0.0 | 800 ± 0.0 |
| L. interrogans serovar Bataviae | 100 ± 0.0 | 100 ± 0.0 | 400 ± 0.0 | 400 ± 0.0 |
| L. interrogans serovar Caniola | 100 ± 0.0 | 100 ± 0.0 | 400 ± 0.0 | 400 ± 0.0 |
| L. interrogans serovar Javanica | 200 ± 0.0 | 200 ± 0.0 | 800 ± 0.0 | 800 ± 0.0 |
| Leptospira Serovars | Doxycycline (µg/mL) | Penicillin G (µg/mL) | Ceftriaxone (µg/mL) | |||
|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | |
| L. interrogans serovar Australis | 0.78 ± 0.00 | 12.50 ± 0.00 | <0.01 ± 0.00 | <0.01 ± 0.00 | 0.20 ± 0.00 | 0.39 ± 0.00 |
| L. interrogans serovar Bataviae | 0.39 ± 0.00 | 12.50 ± 0.00 | <0.01 ± 0.00 | 0.05 ± 0.00 | 0.05 ± 0.00 | 0.05 ± 0.00 |
| L. interrogans serovar Canicola | 1.56 ± 0.00 | 12.50 ± 0.00 | 0.10 ± 0.00 | 0.20 ± 0.00 | 0.20 ± 0.00 | 0.39 ± 0.00 |
| L. interrogans serovar Javanica | 3.13 ± 0.00 | 25.00 ± 0.00 | 0.78 ± 0.00 | 3.13 ± 0.00 | 0.78 ± 0.00 | 0.78 ± 0.00 |
| Leptospira Serovars | Combination of AE and Doxycycline | Combination of ME and Doxycycline | ||||||
|---|---|---|---|---|---|---|---|---|
| MIC AE (µg/mL) | MIC DOX (µg/mL) | FICI | Activity | MIC ME (µg/mL) | MIC DOX (µg/mL) | FICI | Activity | |
| L. interrogans serovar Australis | 800 ± 0.00 | 0.78 ± 0.00 | 1.59 ± 0.00 | Indifferent | 800 ± 0.00 | 0.39 ± 0.00 | 2.82 ± 0.00 | Indifferent |
| L. interrogans serovar Bataviae | 50 ± 0.00 | 0.78 ± 0.00 | 3.09 ± 0.00 | Indifferent | 200 ± 0.00 | 0.39 ± 0.00 | 4.22 ± 0.00 | Antagonistic |
| L. interrogans serovar Canicola | 100 ± 0.00 | 1.56 ± 0.00 | 2.09 ± 0.00 | Indifferent | 400 ± 0.00 | 1.56 ± 0.00 | 2.65 ± 0.00 | Indifferent |
| L. interrogans serovar Javanica | 100 ± 0.00 | 1.56 ± 0.00 | 2.46 ± 0.00 | Indifferent | 400 ± 0.00 | 0.78 ± 0.00 | 3.58 ± 0.00 | Indifferent |
| Leptospira Serovars | Combination of AE and Penicillin G | Combination of ME and Penicillin G | ||||||
|---|---|---|---|---|---|---|---|---|
| MIC AE (µg/mL) | MIC PEN (µg/mL) | FICI | Activity | MIC ME (µg/mL) | MIC PEN (µg/mL) | FICI | Activity | |
| L. interrogans serovar Australis | 200 ± 0.00 | 0.02 ± 0.00 | 2.92 ± 0.00 | Indifferent | 800 ± 0.00 | 0.01 ± 0.00 | 3.32 ± 0.00 | Indifferent |
| L. interrogans serovar Bataviae | 50 ± 0.00 | 0.02 ± 0.00 | 3.41 ± 0.00 | Indifferent | 400 ± 0.00 | 0.01 ± 0.00 | 2.95 ± 0.00 | Indifferent |
| L. interrogans serovar Canicola | 100 ± 0.00 | 0.05 ± 0.00 | 2.42 ± 0.00 | Indifferent | 400 ± 0.00 | 0.05 ± 0.00 | 4.87 ± 0.00 | Antagonistic |
| L. interrogans serovar Javanica | 100 ± 0.00 | 0.78 ± 0.00 | 2.61 ± 0.00 | Indifferent | 400 ± 0.00 | 0.78 ± 0.00 | 3.38 ± 0.00 | Indifferent |
| Leptospira Serovars | Combination of AE and Ceftriaxone | Combination of ME and Ceftriaxone | ||||||
|---|---|---|---|---|---|---|---|---|
| MIC AE (µg/mL) | MIC CEF (µg/mL) | FICI | Activity | MIC ME (µg/mL) | MIC CEF (µg/mL) | FICI | Activity | |
| L. interrogans serovar Australis | 400 ± 0.00 | 0.10 ± 0.00 | 2.39 ± 0.00 | Indifferent | 400 ± 0.00 | 0.20 ± 0.00 | 3.19 ± 0.00 | Indifferent |
| L. interrogans serovar Bataviae | 100 ± 0.00 | 0.05 ± 0.00 | 2.24 ± 0.00 | Indifferent | 400 ± 0.00 | 0.02 ± 0.00 | 2.51 ± 0.00 | Indifferent |
| L. interrogans serovar Canicola | 100 ± 0.00 | 0.20 ± 0.00 | 2.36 ± 0.00 | Indifferent | 100 ± 0.00 | 0.10 ± 0.00 | 3.22 ± 0.00 | Indifferent |
| L. interrogans serovar Javanica | 200 ± 0.00 | 1.56 ± 0.00 | 2.40 ± 0.00 | Indifferent | 800 ± 0.00 | 1.56 ± 0.00 | 2.75 ± 0.00 | Indifferent |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ismail, C.A.M.; Deris, Z.Z.; Bakar, R.A.; Ismail, N. In Vitro Anti-Leptospiral Activity of Phyllanthus amarus Extracts and Their Combinations with Antibiotics. Int. J. Environ. Res. Public Health 2021, 18, 2834. https://doi.org/10.3390/ijerph18062834
Ismail CAM, Deris ZZ, Bakar RA, Ismail N. In Vitro Anti-Leptospiral Activity of Phyllanthus amarus Extracts and Their Combinations with Antibiotics. International Journal of Environmental Research and Public Health. 2021; 18(6):2834. https://doi.org/10.3390/ijerph18062834
Chicago/Turabian StyleIsmail, Che Ain Munirah, Zakuan Zainy Deris, Ruzilawati Abu Bakar, and Nabilah Ismail. 2021. "In Vitro Anti-Leptospiral Activity of Phyllanthus amarus Extracts and Their Combinations with Antibiotics" International Journal of Environmental Research and Public Health 18, no. 6: 2834. https://doi.org/10.3390/ijerph18062834
APA StyleIsmail, C. A. M., Deris, Z. Z., Bakar, R. A., & Ismail, N. (2021). In Vitro Anti-Leptospiral Activity of Phyllanthus amarus Extracts and Their Combinations with Antibiotics. International Journal of Environmental Research and Public Health, 18(6), 2834. https://doi.org/10.3390/ijerph18062834






