Hazardous Heavy Metals Accumulation and Health Risk Assessment of Different Vegetable Species in Contaminated Soils from a Typical Mining City, Central China
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Areas
2.2. Vegetable Samples and Rhizosphere Soils Collection
2.3. Sample Preparation and Analysis
2.4. Data Analysis
2.4.1. Nemerow’s Synthetical Pollution Index (Pn)
2.4.2. Potential Ecological Risk Index (RI)
2.4.3. Geo-Accumulation Index (Igeo)
2.4.4. Health Risk Assessment
2.4.5. Statistical Analysis
3. Results
3.1. Heavy Metal Concentrations in Soils
3.2. Contaminated Evaluation of HMs in Soils
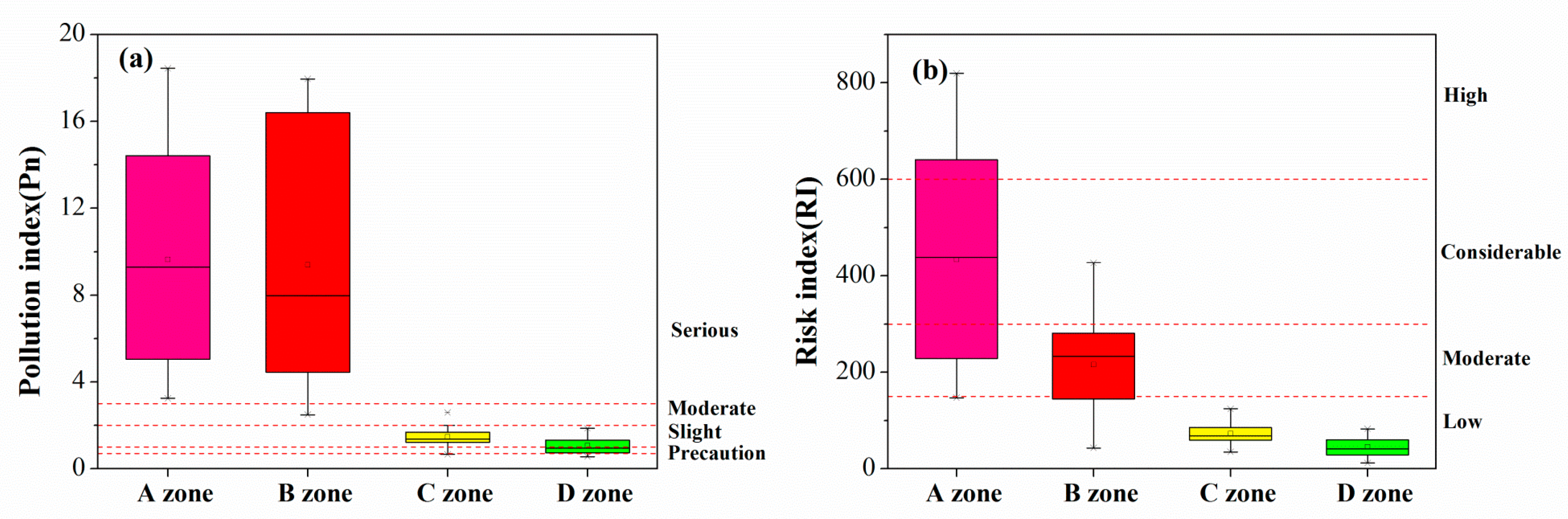
3.2.1. Nemerow’s Synthetical Pollution Index (Pn) and Potential Ecological Risk (RI)
3.2.2. Geo-Accumulation Index (Igeo)
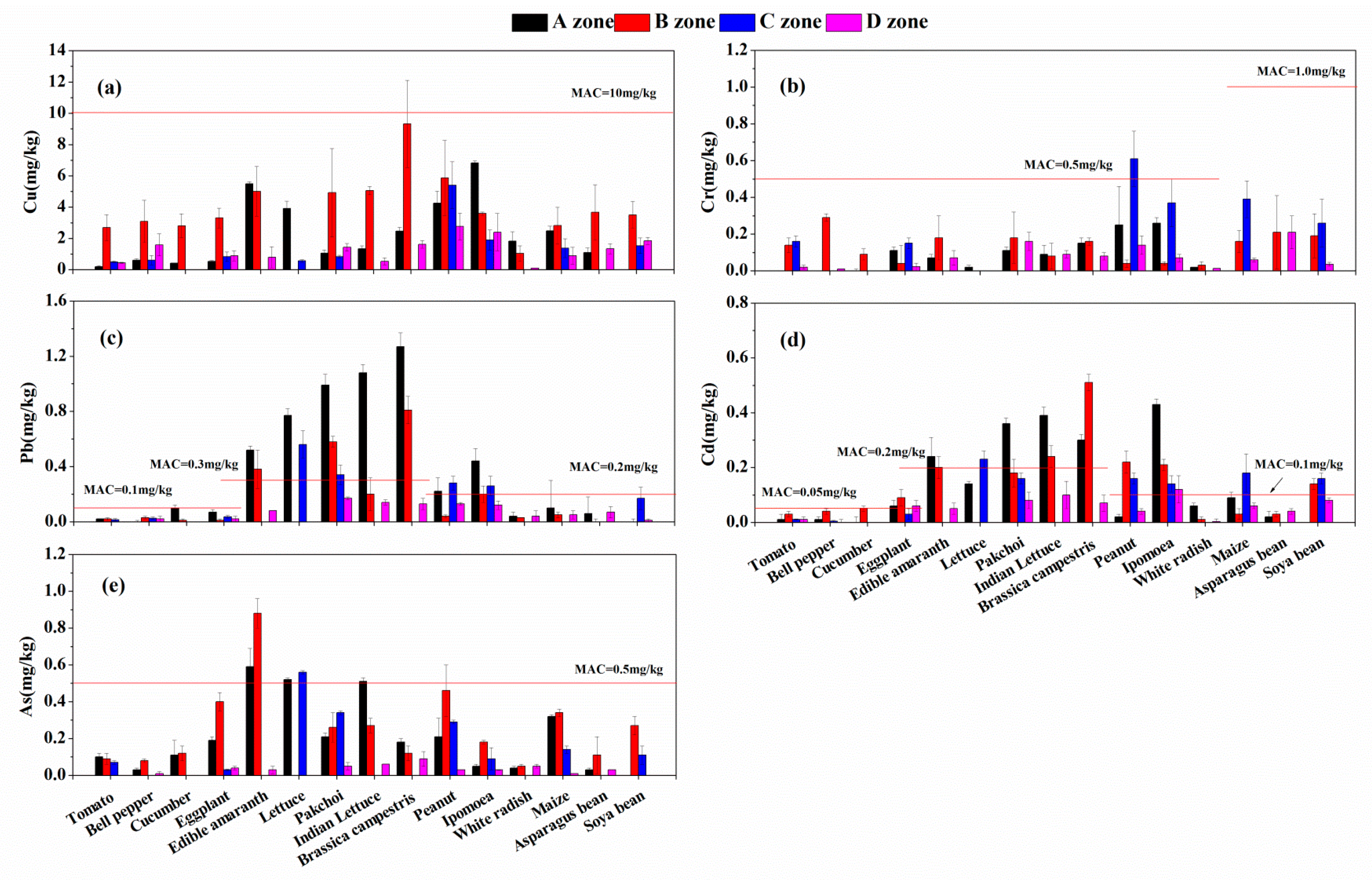
3.3. Heavy Metals in Vegetables and Crops
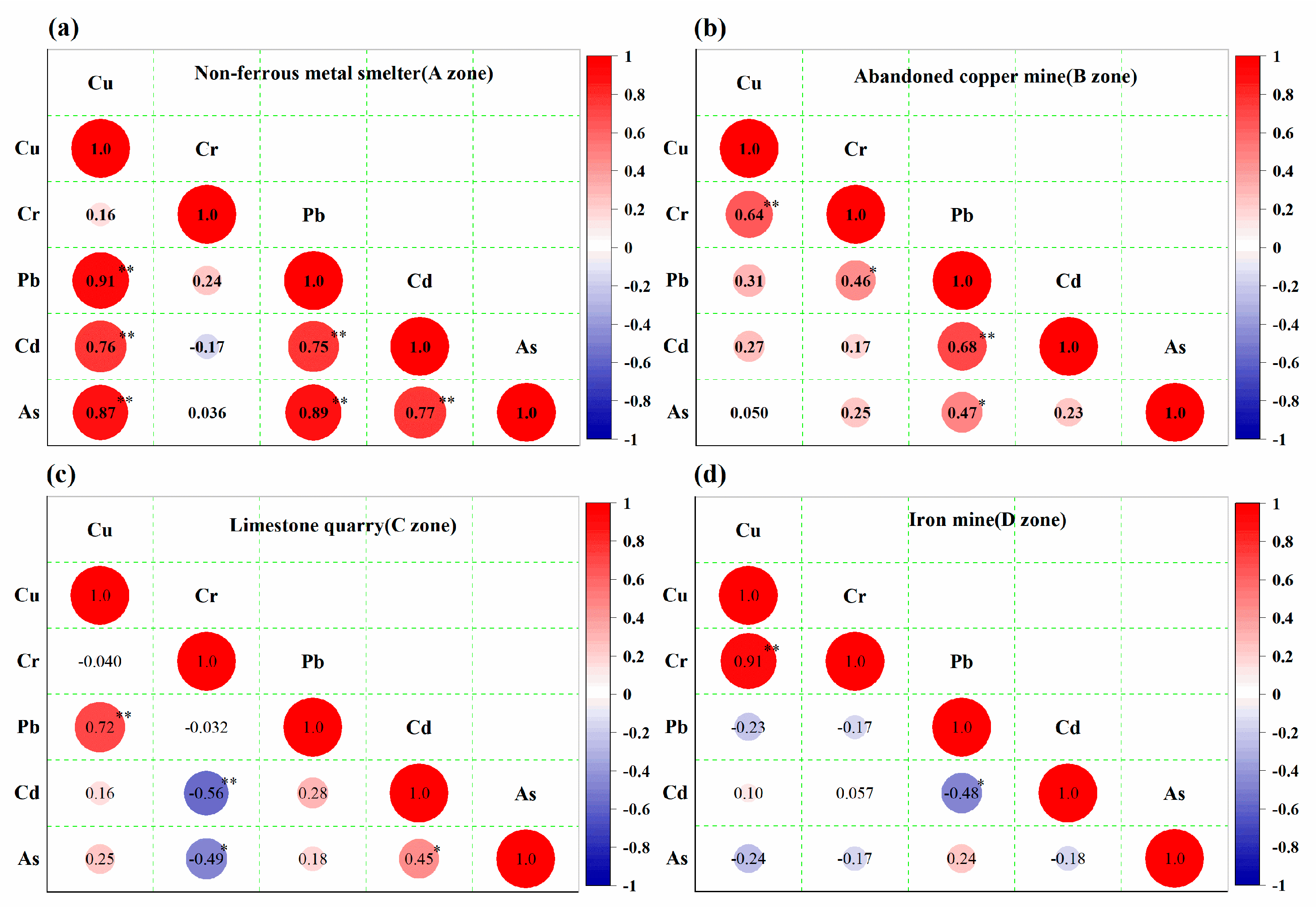
3.4. Source Analysis of HMs in Different Contaminated Soils
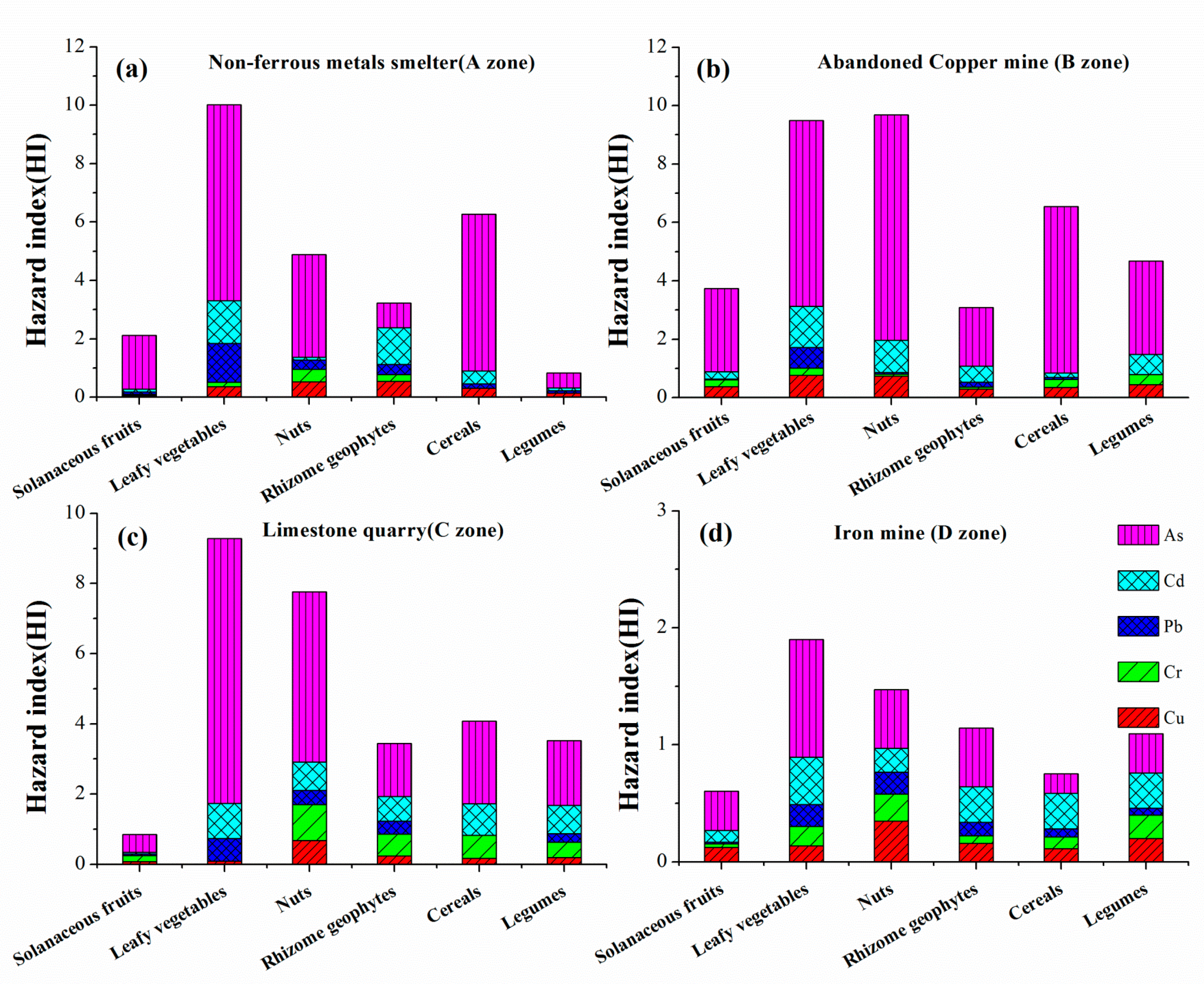
3.5. Health Risk Assessment of Dietary Intake
4. Discussion
4.1. Pollution and Source Identification in Soils
4.2. Comparisons of HMs Accumulation in Different Vegetable and Crop Species
4.3. Health Risk Assessment for Local Children
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhuang, P.; McBride, M.B.; Xia, H. Health Risk from Heavy Metals Via Consumption of Food Crops in the Vicinity of Dabaoshan Mine, South China. Sci. Total Environ. 2009, 407, 1551–1561. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Chen, Q.Q.; Deng, M.H.; Japenga, J.; Li, T.Q.; Yang, X.E.; He, Z. Heavy Metal Pollution and Health Risk Assessment of Agricultural Soils in a Typical Peri-Urban Area in Southeast China. J. Environ. Manag. 2018, 207, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.M.; Xu, Z.C.; Qi, J.Y.; Feng, Z.Z.; Xiang, T.S. Assessment of Exposure to Heavy Metals and Health Risks among Residents near Tonglushan Mine in Hubei, China. Chemosphere 2015, 127, 127–135. [Google Scholar] [CrossRef]
- Ahmed, M.; Matsumoto, M.; Kurosawa, K. Heavy Metal Contamination of Irrigation Water, Soil, and Vegetables in a Multi-Industry District of Bangladesh. Int. J. Environ. Res. 2018, 12, 531–542. [Google Scholar] [CrossRef]
- Vatanpour, N.; Malvandi, A.M.; Talouki, H.H.; Gattinoni, P.; Scesi, L. Impact of Rapid Urbanization on the Surface Water’s Quality: A Long-term Environmental and Physicochemical Investigation of Tajan River, Iran (2007–2017). Environ. Sci. Pollut. Res. Int. 2020, 27, 8439–8450. [Google Scholar] [CrossRef] [PubMed]
- MEP of China. National Soil Pollution Survey Bulletin. 2014. Available online: http://www.zhb.gov.cn/gkml/hbb/qt/201404/t20140417-270670.htm (accessed on 10 February 2020).
- Zhao, F.J.; Ma, Y.; Zhu, Y.G. Soil Contamination in China: Current Status and Mitigation Strategies. Environ. Sci. Technol. 2015, 49, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.Y.; Ma, Z.W.; van der Kuijp, T.J.; Yuan, Z.W.; Huang, L. A Review of Soil Heavy Metal Pollution from Mines in China: Pollution and Health Risk Assessment. Sci. Total Environ. 2014, 468, 843–853. [Google Scholar] [CrossRef]
- Anju, M.; Banerjee, D.K. Multivariate Statistical Analysis of Heavy Metals in Soils of a Pbezn Mining Area, India. Environ. Monit. Assess. 2012, 184, 4191–4206. [Google Scholar] [CrossRef]
- Gu, Y.G.; Li, Q.S.; Fang, J.H.; He, B.Y.; Fu, H.B.; Tong, Z.J. Identification of Heavy Metal Sources in the Reclaimed Farmland Soils of the Pearl River Estuary in China Using a Multivariate Geostatistical Approach. Ecotox. Environ. Saf. 2014, 105, 7–12. [Google Scholar] [CrossRef]
- Facchinelli, A.; Sacchi, E.; Mallen, L. Multivariate Statistical and Gis-Based Approach to Identify Heavy Metal Sources in Soils. Environ. Pollut. 2001, 114, 313–324. [Google Scholar] [CrossRef]
- Chen, L.; Zhou, S.L.; Shi, Y.X.; Wang, C.H.; Li, B.J.; Li, Y.; Wu, S.H. Heavy metals in Food Crops, Soil, Andwater in the Lihe River Watershed of the Taihu Region and Their Potential Health Risks When Ingested. Sci. Total Environ. 2018, 615, 141–149. [Google Scholar] [CrossRef]
- Xiao, W.; Ye, X.Z.; Zhang, Q.; Chen, D.; Hu, J.; Gao, N. Evaluation of Cadmium Transfer from Soil to Leafy Vegetables: Influencing Factors, Transfer Models, and Indication of Soil Threshold Contents. Ecotox. Environ. Saf. 2018, 164, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.R.; Li, H.Z.; Zhou, Y.Z.; Dou, L.; Cai, L.M.; Mo, L.P.; You, J. Bioavailability and Soil-to-Crop Transfer of Heavy Metals in Farmland Soils: A Case Study in the Pearl River Delta, South China. Environ. Pollut. 2018, 235, 710–719. [Google Scholar] [CrossRef]
- Kumar, S.; Prasad, S.; Yadav, K.K.; Shrivastava, M.; Gupta, N.; Nagar, S.; Bach, Q.V.; Kamyab, H.; Khan, S.A.; Yadav, S.; et al. Hazardous Heavy Metals Contamination of Vegetables and Food Chain: Role of Sustainable Remediation Approaches—A Review. Environ. Res. 2019, 179, 108792. [Google Scholar] [CrossRef] [PubMed]
- Bi, C.J.; Zhou, Y.; Chen, Z.L.; Jia, J.P.; Bao, X.Y. Heavy Metals and Lead Isotopes in Soils, Road Dust and Leafy Vegetables and Health Risks Via Vegetable Consumption in the Industrial Areas of Shanghai, China. Sci. Total Environ. 2018, 619, 1349–1357. [Google Scholar] [CrossRef]
- Rahmdel, S.; Rezaei, M.; Ekhlasi, J.; Zarei, S.H.; Akhlaghi, M.; Abdollahzadeh, S.M.; Sefidkar, R.; Mazloomi, S.M. Heavy Metals (Pb, Cd, Cu, Zn, Ni, Co) in Leafy Vegetables Collected from Production Sites: Their Potential Health Risk to the General Population in Shiraz, Iran. Environ. Monit. Assess. 2018, 190, 650. [Google Scholar] [CrossRef]
- Huang, L.K.; Wang, Q.; Zhou, Q.Y.; Ma, L.Y.; Wu, Y.J.; Liu, Q.Z.; Wang, S.; Feng, Y. Cadmium Uptake from Soil and Transport by Leafy Vegetables: A Meta-Analysis. Environ. Pollut. 2020, 264, 114677. [Google Scholar] [CrossRef] [PubMed]
- Jia, Z.M.; Li, S.Y.; Wang, L. Assessment of Soil Heavy Metals for Eco-environment and Human Health in a Rapidly Urbanization Area of the Upper Yangtze Basin. Sci. Rep. 2018, 8, 3256. [Google Scholar] [CrossRef]
- Mandal, R.; Kaur, S. Impact of Environmental Pollution on Trace Elements in Vegetables and Associated Potential Risk to Human Health in Industrial Town Mandi-Gobindgarh (India). Chemosphere 2019, 219, 574–587. [Google Scholar] [CrossRef]
- Chen, M.; Li, F.G.; Hu, L.W.; Yang, T.; Yang, Q.; Tao, M.X.; Deng, Y.W. Heavy Metal Pollution in Topsoil and Vegetables in the Typical Mining Area near Gannan, Jiangxi Province, China. Environ. Eng. Sci. 2019, 10, 1307–1314. [Google Scholar] [CrossRef]
- Li, X.Y.; Li, Z.G.; Lin, C.J.; Bi, X.Y.; Liu, J.L.; Feng, X.; Zhang, H.; Chen, J.; Wu, T.T. Health Risks of Heavy Metal Exposure through Vegetable Consumption near a Large-Scale Pb/Zn Smelter in Central China. Ecotox. Environ. Saf. 2018, 161, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Guo, G.H.; Zhang, D.G.; Wang, Y.T. Probabilistic Human Health Risk Assessment of Heavy Metal Intake Via Vegetable Consumption around Pb/Zn Smelters in Southwest China. Int. J. Environ. Res. Public Health 2019, 16, 3267. [Google Scholar] [CrossRef]
- Antoniadis, V.; Shaheen, S.M.; Boersch, J.; Frohne, T.; Laing, G.D.; Rinklebe, J. Bioavailability and Risk Assessment of Potentially Toxic Elements in Garden Edible Vegetables and Soils around a Highly Contaminated Former Mining Area in Germany. J. Environ. Manag. 2017, 186, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Douay, F.; Pelfrene, A.; Planque, J.; Fourrier, H.; Richard, A.; Roussel, H.; Girondelot, B. Assessment of Potential Health Risk for Inhabitants Living near a Former Lead Smelter. Part 1: Metal Concentrations in Soils, Agricultural Crops, and Homegrown Vegetables. Environ. Monit. Assess. 2013, 185, 3665–3680. [Google Scholar] [CrossRef]
- Boussen, S.; Soubrand, M.; Bril, H. Transfer of Lead, Zinc and Cadmium from Mine Tailings to Wheat (Triticum Aestivum) in Carbonated Mediterranean (Northern Tunisia) Soils. Geoderma 2013, 192, 227–236. [Google Scholar] [CrossRef]
- Ji, K.; Kim, J.; Lee, M.; Park, S.; Kwon, H.J.; Cheong, H.K.; Jang, J.Y.; Kim, D.S.; Yu, S.; Kim, Y.W.; et al. Assessment of Exposure to Heavy Metals and Health Risks among Residents near Abandoned Metal Mines in Goseong, Korea. Environ. Pollut. 2013, 178, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Vatanpour, N.; Feizy, J.; Talouki, H.H.; Es’haghi, Z.; Scesi, L.; Malvandi, A.M. The High Levels of Heavy Metal Accumulation in Cultivated Rice from the Tajan River Basin: Health and Ecological Risk Assessment. Chemosphere 2020, 245, 125639. [Google Scholar] [CrossRef]
- Chen, H.Y.; Yuan, X.Y.; Li, T.Y.; Hu, S.; Ji, J.F.; Wang, C. Characteristics of Heavy Metal Transfer and Their Influencing Factors in Different Soil-Crop Systems of the Industrialization Region, China. Ecotox. Environ. Saf. 2016, 126, 193–201. [Google Scholar] [CrossRef]
- Bi, X.Y.; Feng, X.B.; Yang, Y.G.; Li, X.D.; Shin, G.P.Y.; Li, F.L.; Qiu, G.L.; Li, G.H.; Liu, T.Z.; Fu, Z.Y. Allocation and Source Attribution of Lead and Cadmium in Maize (Zea Mays L.) Impacted by Smelting Emissions. Environ. Pollut. 2009, 157, 834–839. [Google Scholar] [CrossRef]
- Liu, W.T.; Zhou, Q.X.; Sun, Y.B.; Liu, R. Identification of Chinese Cabbage Genotypes with Low Cadmium Accumulation for Food Safety. Environ. Pollut. 2009, 157, 1961–1967. [Google Scholar] [CrossRef]
- Liu, W.T.; Zhou, Q.X.; Zhang, Y.L.; Wei, S.H. Lead Accumulation in Different Chinese Cabbage Cultivars and Screening for Pollution-Safe Cultivars. J. Environ. Manag. 2010, 91, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.M.; Tan, X.Q.; Mei, X.Q.; Li, Q.S.; Zhou, C.; Wang, L.L.; Ye, H.J.; Yang, P. Low-Cd Tomato Cultivars (Solanum Lycopersicum L.) Screened in Non-Saline Soils Also Accumulated Low Cd, Zn, and Cu in Heavy Metal-Polluted Saline Soils. Environ. Sci. Pollut. Res. Int. 2018, 25, 27439–27450. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Lv, F.H.; Zhou, J.C.; Song, Y.W.; Li, F. Health Risk Assessment of Vegetables Grown on the Contaminated Soils in Daye City of Hubei Province, China. Sustainability 2017, 9, 2141. [Google Scholar] [CrossRef]
- Du, P.; Xie, Y.F.; Wang, S.J.; Zhao, H.H.; Zhang, Z.; Wu, B.; Li, F.S. Potential Sources of and Ecological Risks from Heavy Metals in Agricultural Soils, Daye City, China. Environ. Sci. Pollut. Res. Int. 2015, 22, 3498–3507. [Google Scholar] [CrossRef]
- Wang, M.L.; Liu, R.H.; Lu, X.Y.; Zhu, Z.Y.; Wang, H.L.; Jiang, L.; Wu, Z.H. Heavy Metal Contamination and Ecological Risk Assessment of Swine Manure Irrigated Vegetable Soils in Jiangxi Province, China. Bull. Environ. Contam. Toxicol. 2018, 100, 634–640. [Google Scholar] [CrossRef]
- Sawut, R.; Kasim, N.; Maihemuti, B.; Hu, L.; Abliz, A.; Abdujappar, A.; Kurban, M. Pollution Characteristics and Health Risk Assessment of Heavy Metals in the Vegetable Bases of Northwest China. Sci. Total Environ. 2018, 642, 864–878. [Google Scholar] [CrossRef]
- Hakanson, L. An Ecological Risk Index for Aquatic Pollution Control. A Sedimentological Approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Müller, G. Heavy Metals in Sediment of the Rhine-Changes since 1971. Umsch. Wiss. Tech. 1979, 79, 778–783. [Google Scholar]
- Cai, L.M.; Wang, Q.S.; Luo, J.; Chen, L.G.; Zhu, R.L.; Wang, S.; Tang, C.H. Heavy Metal Contamination and Health Risk Assessment for Children near a Large Cu-Smelter in Central China. Sci. Total Environ. 2019, 650, 725–733. [Google Scholar] [CrossRef]
- USEPA. Human Health Risk Assessment. In Regional Screening Level-Summary Table; United States Environmental Protection Agency: Washington, DC, USA, 2018. [Google Scholar]
- Tian, K.; Biao, H.; Xing, Z.; Hu, W.Y. Geochemical Baseline Establishment and Ecological Risk Evaluation of Heavy Metals in Greenhouse Soils from Dongtai, China. Ecol. Indic. 2017, 72, 510–520. [Google Scholar] [CrossRef]
- Yang, Q.Q.; Li, Z.Y.; Lu, X.N.; Duan, Q.N.; Huang, L.; Bi, J. A Review of Soil Heavy Metal Pollution from Industrial and Agricultural Regions in China: Pollution and Risk Assessment. Sci. Total Environ. 2018, 642, 690–700. [Google Scholar] [CrossRef]
- Luo, C.; Liu, C.; Wang, Y.; Liu, X.; Li, F. Heavy Metal Contamination in Soils and Vegetables near an E-Waste Processing Site, South China. J. Hazard. Mater. 2011, 186, 481–490. [Google Scholar] [CrossRef]
- Zhang, T.; Ruan, J.J.; Zhang, B.; Lu, S.Y.; Gao, C.Z.; Huang, L.F.; Bai, X.Y.; Xie, L.; Gui, M.W.; Qiu, R.L. Heavy Metals in Human Urine, Foods and Drinking Water from an E-Waste Dismantling Area: Identification of Exposure Sources and Metal-Induced Health Risk. Ecotox. Environ. Saf. 2019, 169, 707–713. [Google Scholar] [CrossRef]
- Li, N.; Kang, Y.; Pan, W.J.; Zeng, L.X.; Zhang, Q.Y.; Luo, J.W. Concentration and Transportation of Heavy Metals in Vegetables and Risk Assessment of Human Exposure to Bioaccessible Heavy Metals in Soil near a Waste-incinerator Site, South China. Sci. Total Environ. 2015, 521–522, 144–151. [Google Scholar] [CrossRef]
- Sun, Z.H.; Hu, Y.N.; Cheng, H.F. Public Health Risk of Toxic Metal (Loid) Pollution to the Population Living near an Abandoned Small-Scale Polymetallic Mine. Sci. Total Environ. 2020, 718, 137434. [Google Scholar] [CrossRef]
- Ma, C.Y.; Cai, D.J.; Yan, H. Soil Cd Pollution and Research Progress of Treatment Techniques. Henan Chem. Ind. 2013, 30, 17–22, (In Chinese with English Abstract). [Google Scholar]
- Bhattacharya, P.; Welch, A.H.; Stollenwerk, K.G.; McLaughlin, M.J.; Bundschuh, J.; Panaullah, G. Arsenic in the Environment: Biology and Chemistry. Sci. Total Environ. 2007, 379, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.S.; Liao, Q.L.; Hua, M.; Wu, X.M.; Bi, K.S.; Yan, C.Y.; Chen, B.; Zhang, X.Y. Survey of Heavy Metal Pollution and Assessment of Agricultural Soil in Yangzhong District, Jiangsu Province, China. Chemosphere 2007, 67, 2148–2155. [Google Scholar] [CrossRef] [PubMed]
- Imperato, M. Spatial Distribution of Heavy Metals in Urban Soils of Naples City (Italy). Environ. Pollut. 2003, 124, 247–256. [Google Scholar] [CrossRef]
- Moryani, H.T.; Kong, S.Q.; Du, J.K.; Bao, J.G. Health Risk Assessment of Heavy Metals Accumulated on PM2.5 Fractioned Road Dust from Two Cities of Pakistan. Int. J. Environ. Res. Public Health 2020, 17, 7124. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Y.; Wang, H.B.; Wang, H.J.; Yin, F.; Yang, X.Y.; Hu, Y.J. Heavy Metal Pollution in Vegetables Grown in the Vicinity of a Multi-Metal Mining Area in Gejiu, China: Total Concentrations, Speciation Analysis, and Health Risk. Environ. Sci. Pollut. Res. Int. 2014, 21, 12569–12582. [Google Scholar] [CrossRef]
- Hu, B.F.; Xue, J.; Zhou, Y.; Shao, S.; Fu, Z.Y.; Li, Y.; Chen, S.C.; Qi, L.; Shi, Z. Modelling Bioaccumulation of Heavy Metals in Soil-Crop Ecosystems and Identifying Its Controlling Factors Using Machine Learning. Environ. Pollut. 2020, 262, 114308. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Yang, W.T.; Zhou, X.; Liu, L.; Gu, J.F.; Wang, W.L.; Zou, J.L.; Tian, T.; Peng, P.Q.; Liao, B.H. Accumulation of Heavy Metals in Vegetable Species Planted in Contaminated Soils and the Health Risk Assessment. Int. J. Environ. Res. Public Health 2016, 13, 289. [Google Scholar] [CrossRef]
- Hu, W.Y.; Huang, B.; Tian, K.; Holm, P.E.; Zhang, Y.X. Heavy Metals in Intensive Greenhouse Vegetable Production Systems Along Yellow Sea of China: Levels, Transfer and Health Risk. Chemosphere 2017, 167, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Wang, Y.H.; Jiang, Y.; Li, G.C.; Cui, J.H.; Wang, Y.; Zhang, H.; Wang, S.C.; Xu, S.; Wang, R.Z. The Accumulation and Health Risk of Heavy Metals in Vegetables around a Zinc Smelter in Northeastern China. Environ. Sci. Pollut. Res. Int. 2016, 23, 25114–25126. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Huang, Q.; Liu, L.; Cai, P.; Liang, W.; Li, M. Poultry Manure Compost Alleviates the Phytotoxicity of Soil Cadmium: Influence on Growth of Pakchoi (Brassica Chinensis L.). Pedosphere 2010, 20, 63–70. [Google Scholar] [CrossRef]
- Vandecasteele, B.; Willekens, K.; Laing, G.D.; Waes, J.V.; Tack, F.M.G. Effect of Feedstock, Organic Matter Content and pH on Cd, Zn and Mn Availability in Farm Compost Based on Bark and Wood Chips. Acta Hortic. 2014, 1018, 661–668. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, W.; Wang, M.; Peng, C. Regional Accumulation Characteristics of Cadmium in Vegetables: Influencing Factors, Transfer Model and Indication of Soil Threshold Content. Environ. Pollut. 2016, 219, 1036–1043. [Google Scholar] [CrossRef]
- Clemens, S. Toxic Metal Accumulation, Responses to Exposure and Mechanisms to Tolerance in Plants. Biochimie 2006, 88, 1707–1719. [Google Scholar] [CrossRef]
- Harrison, R.M.; Chirgawi, M.B. The Assessment of Air and Soil as Contributors of Some Trace Metals to Vegetable Plants. I. Use of a Filtered Air Growth Cabinet. Sci. Total Environ. 1989, 83, 13–34. [Google Scholar] [CrossRef]
- Fan, Y.; Li, Y.L.; Li, H.; Cheng, F.Q. Evaluating Heavy Metal Accumulation and Potential Risks in Soil-Plant Systems Applied with Magnesium Slag-Based Fertilizer. Chemosphere 2018, 197, 382–388. [Google Scholar] [CrossRef]
- Ji, Y.; Wu, P.; Zhang, J.; Zhang, J.; Zhou, Y.; Peng, Y.; Zhang, S.; Cai, G.; Gao, G. Heavy Metal Accumulation, Risk Assessment and Integrated Biomarker Responses of Local Vegetables: A Case Study Along the Le’an River. Chemosphere 2018, 199, 361–371. [Google Scholar] [CrossRef]
- Zhou, Q.; Yang, Y.C.; Yang, Z.Y. Molecular Dissection of Cadmium-Responsive Transcriptome Profile in a Low Cadmium-Accumulating Cultivar of Brassica Parachinensis. Ecotox. Environ. Saf. 2019, 176, 85–94. [Google Scholar] [CrossRef]
- USEPA. Risk Based Concentration Table; United States Environmental Protection Agency: Washington, DC, USA, 2000.
- USEPA. Risk Assessment Guidance for Superfund. Human Health Evaluation Manual, (Part a); Office of Emergency and Remedial Response [Epa/540/1-89/002]; United States Environmental Protection Agency: Washington, DC, USA, 1989; Volume I.
- USEPA. Exposure Factors Handbook; Office of Research and Development, National Center for Environmental Assessment [Epa/600/P-95/002f a-C]; United States Environmental Protection Agency: Washington, DC, USA, 1997.
- Tao, X.Q.; Shen, D.S.; Shentu, J.L. Bioaccessibility and Health Risk of Heavy Metals in Ash Fromthe Incineration of Different E-Waste Residues. Environ. Sci. Pollut. Res. Int. 2015, 22, 3558–3569. [Google Scholar] [CrossRef]
- White, P.D.; Marcus, A.H. The Conceptual Structure of the Integrated Exposure Uptake Biokinetic Model for Lead in Children. Environ. Health Persp. 1998, 106, 1513. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.Z.; Duan, X.L.; Zhao, X.G.; Wang, B.B.; Ma, J.; Fan, D.L.; Sun, C.Y.; He, B.; Wei, F.S.; Jiang, G.B. Health Risk Assessment of Various Metal(Loid)S Via Multiple Exposure Pathways on Children Living near a Typical Lead-Acid Battery Plant, China. Environ. Pollut. 2015, 200, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Dhankher, O.P.; Pilon-Smits, E.A.; Meagher, R.B.; Doty, S. Biotechnological Approaches for Phytoremediation. Plant Biotechnol. Agric. 2012, 2012, 309–328. [Google Scholar]
- Vahter, M. Species Differences in the Metabolism of Arsenic Compounds. Appl. Organomet. Chem. 1994, 8, 175–182. [Google Scholar] [CrossRef]
- Zhou, X.; Yoshida, K.; Kuroda, K.; Endo, Y.; Endo, G. Effects of Cysteine on the Cytotoxicity of Arsenic Compounds. Arch. Environ. Con. Tox. 2003, 45, 324–330. [Google Scholar] [CrossRef]
- Bakkali, K.; Martos, N.R.; Souhail, B.; Ballesteros, E. Determination of Heavy Metal Content in Vegetables and Oils from Spain and Morocco by Inductively Coupled Plasmass Spectrometry. Anal. Lett. 2012, 45, 907–919. [Google Scholar] [CrossRef]
- Pajevic, S.; Arsenov, D.; Nikolic, N.; Borisev, M.; Orcic, D.; Zupunski, M.; Mimica-Dukic, N. Heavy Metal Accumulation in Vegetable Species and Health Risk Assessment in Serbia. Environ. Monit. Assess. 2018, 190, 459. [Google Scholar] [CrossRef] [PubMed]
- Hussain, R.; Luo, K.L.; Liang, H.D.; Hong, X.P. Impact of the Coal Mining-Contaminated Soil on the Food Safety in Shaanxi, China. Environ. Geochem. Health 2019, 41, 1521–1544. [Google Scholar] [CrossRef] [PubMed]
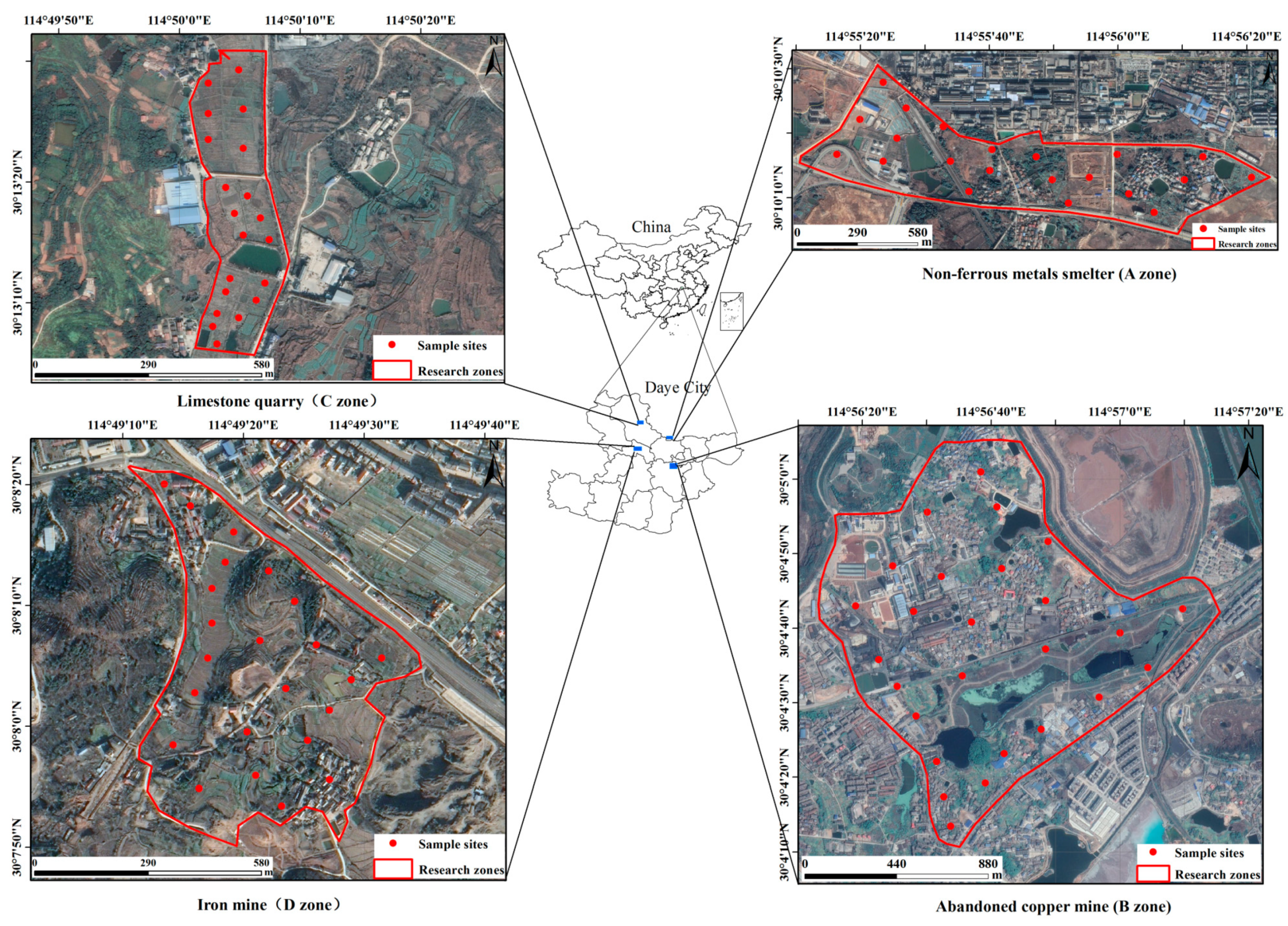

| NO. | Types | Vegetable Species | Harvest Season |
|---|---|---|---|
| 1 | Solanaceous fruits | Tomato (Lycopersicum esculentum Mill.) | Spring and summer |
| 2 | Bell pepper (Capsicum annuum L.) | Spring and summer | |
| 3 | Cucumber (Cucumis sativus L.) | Spring and summer | |
| 4 | Eggplant (Solanum melongena L.) | Spring and summer | |
| 5 | Leafy vegetables | Edible amaranth (Amaranthus tricolor L.) | Spring and summer |
| 6 | Lettuce (Lactuca sativa L.) | Spring and summer | |
| 7 | Pakchoi (Brassica Chinensis L.) | Spring and summer | |
| 8 | Indian Lettuce (Lactuca sativa L. var longifoliaf. Lam) | Spring and summer | |
| 9 | Brassica campestris (Brassica campestris L. var. purpurea Baileysh) | Spring and summer | |
| 10 | Nuts | Peanut (Arachis hypogaea Linn.) | Autumn and winter |
| 11 | Rhizome geophytes | Ipomoea (Ipomoea batatas L.) | Autumn and winter |
| 12 | White radish (Raphanus sativus L.) | Autumn and winter | |
| 13 | Cereals | Maize (Zea mays L.) | Autumn and winter |
| 14 | Legumes | Asparagus bean (Vigna unguiculata Linn.) | Spring and summer |
| 15 | Soya bean (Glycine max Linn.) | Autumn and winter |
| Sampling Areas | pH | Cu (mg/kg) | Cr (mg/kg) | Pb (mg/kg) | Cd (mg/kg) | As (mg/kg) | |
|---|---|---|---|---|---|---|---|
| Non-ferrous metals smelter (n = 21) | Range | 6.51–6.87 | 116.17–858.97 | 61.33–94.82 | 27.04–219.51 | 0.44–7.51 | 4.49–75.54 |
| Mean ± SD | 6.71 ± 0.13 | 475.39 ± 226.10 | 72.27 ± 7.55 | 136.89 ± 59.99 | 3.80 ± 2.10 | 42.48 ± 21.26 | |
| CV/% | 1.94 | 47.56 | 10.45 | 43.82 | 55.26 | 50.05 | |
| K-Sp | — | 0.200 * | 0.200 * | 0.006 | 0.190 * | 0.144 * | |
| a Excessive rate % | — | 100.00 | 0.00 | 61.90 | 100.00 | 71.4 | |
| Abandoned copper mine (n = 26) | Range | 6.58–7.35 | 213.36–1446.10 | 66.21–131.70 | 33.25–223.48 | 0.15–3.28 | 4.09–86.04 |
| Mean ± SD | 7.03 ± 0.30 | 729.39 ± 383.83 | 95.69 ± 19.37 | 107.76 ± 56.64 | 1.34 ± 0.78 | 41.96 ± 21.68 | |
| CV/% | 4.25 | 52.62 | 20.24 | 52.56 | 58.21 | 51.67 | |
| K-Sp | — | 0.062 * | 0.200 * | 0.000 | 0.200 * | 0.200 * | |
| a Excessive rate % | — | 100.00 | 0.00 | 42.31 | 88.46 | 53.85 | |
| Limestone quarry (n = 20) | Range | 7.56–7.83 | 74.53–123.75 | 5.10–99.78 | 28.01–103.25 | 0.40–2.06 | 2.93–45.98 |
| Mean ± SD | 7.72 ± 0.11 | 94.45 ± 14.93 | 55.00 ± 31.22 | 60.57 ± 19.77 | 1.10 ± 0.45 | 26.45 ± 11.20 | |
| CV/% | 1.41 | 15.81 | 56.76 | 32.64 | 40.91 | 42.34 | |
| K-Sp | — | 0.134 * | 0.200 * | 0.200 * | 0.200 * | 0.200 * | |
| a Excessive rate % | — | 30.00 | 0.00 | 0.00 | 85.00 | 60.00 | |
| Iron mine (n = 23) | Range | 4.76–5.42 | 20.17–64.48 | 49.12–80.77 | 36.09–76.25 | 0.05–1.75 | 0.79–39.24 |
| Mean ± SD | 5.05 ± 0.22 | 36.31 ± 11.02 | 58.23 ± 7.72 | 52.34 ± 13.53 | 0.45 ± 0.41 | 8.33 ± 9.49 | |
| CV/% | 4.29 | 30.35 | 13.26 | 25.85 | 91.11 | 113.93 | |
| K-Sp | — | 0.125 * | 0.005 | 0.011 | 0.021 | 0.000 | |
| a Excessive rate % | — | 8.70 | 0.00 | 21.74 | 47.83 | 0.00 | |
| b Background values | 30.7 | 86 | 26.7 | 0.17 | 12.3 | ||
| c Soil pollutant standards | 50, 100 | 150, 200, 250 | 70, 90, 120, 170 | 0.3, 0.6 | 40, 30, 25 | ||
| Total excessive rate % | 61.11 | 0.00 | 32.22 | 80.00 | 45.56 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; Bao, J.; Wang, T.; Moryani, H.T.; Kang, W.; Zheng, J.; Zhan, C.; Xiao, W. Hazardous Heavy Metals Accumulation and Health Risk Assessment of Different Vegetable Species in Contaminated Soils from a Typical Mining City, Central China. Int. J. Environ. Res. Public Health 2021, 18, 2617. https://doi.org/10.3390/ijerph18052617
Wang Z, Bao J, Wang T, Moryani HT, Kang W, Zheng J, Zhan C, Xiao W. Hazardous Heavy Metals Accumulation and Health Risk Assessment of Different Vegetable Species in Contaminated Soils from a Typical Mining City, Central China. International Journal of Environmental Research and Public Health. 2021; 18(5):2617. https://doi.org/10.3390/ijerph18052617
Chicago/Turabian StyleWang, Zhen, Jianguo Bao, Tong Wang, Haseeb Tufail Moryani, Wei Kang, Jin Zheng, Changlin Zhan, and Wensheng Xiao. 2021. "Hazardous Heavy Metals Accumulation and Health Risk Assessment of Different Vegetable Species in Contaminated Soils from a Typical Mining City, Central China" International Journal of Environmental Research and Public Health 18, no. 5: 2617. https://doi.org/10.3390/ijerph18052617
APA StyleWang, Z., Bao, J., Wang, T., Moryani, H. T., Kang, W., Zheng, J., Zhan, C., & Xiao, W. (2021). Hazardous Heavy Metals Accumulation and Health Risk Assessment of Different Vegetable Species in Contaminated Soils from a Typical Mining City, Central China. International Journal of Environmental Research and Public Health, 18(5), 2617. https://doi.org/10.3390/ijerph18052617







