Predicting the Severity of Parkinson’s Disease Dementia by Assessing the Neuropsychiatric Symptoms with an SVM Regression Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
2.2. Methods
3. Results
3.1. The General Characteristics of the Subjects
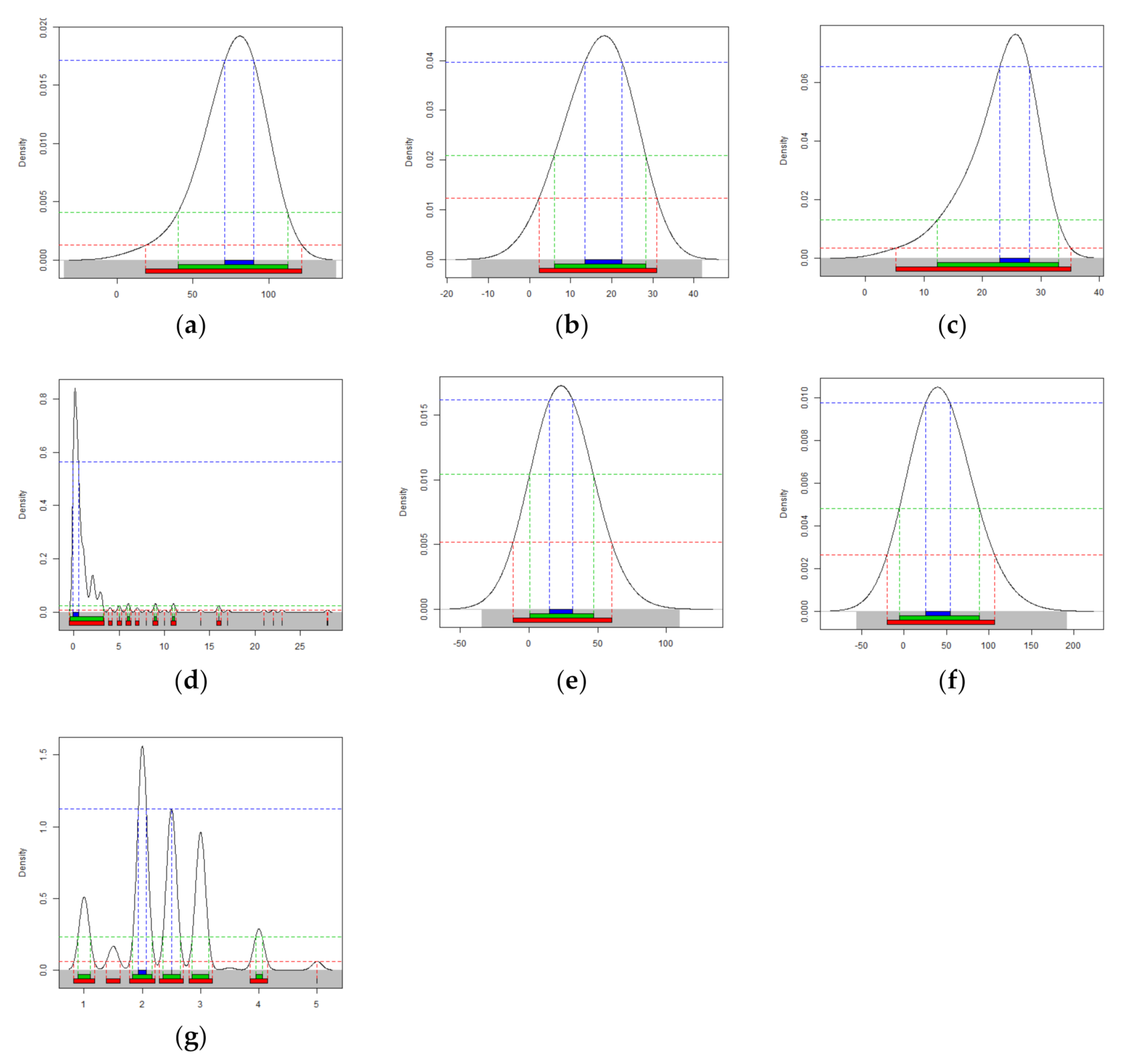
3.2. Comparing the Convergence Rate of Dementia Severity Prediction Model with the SVR Classification Algorithm
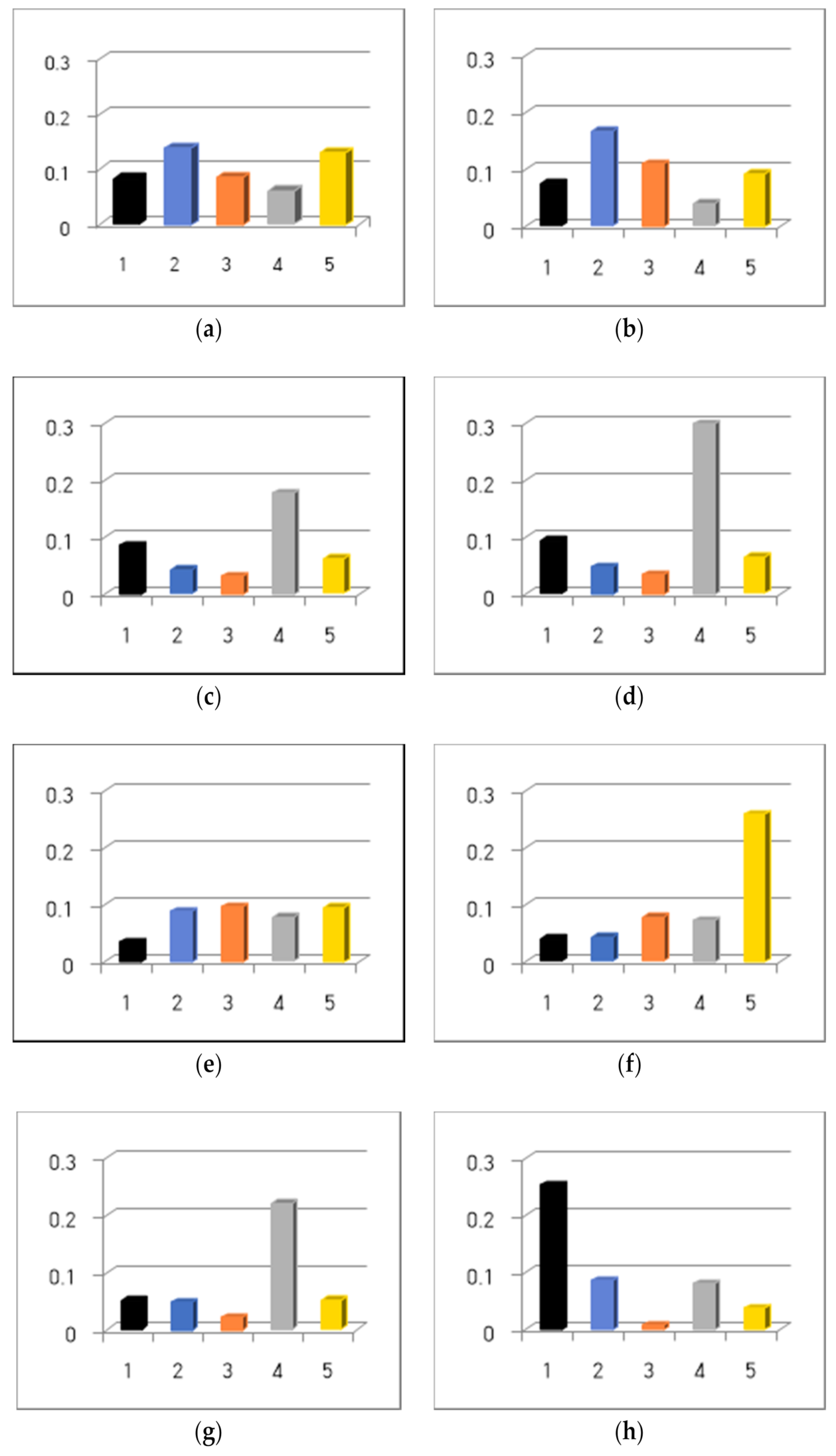
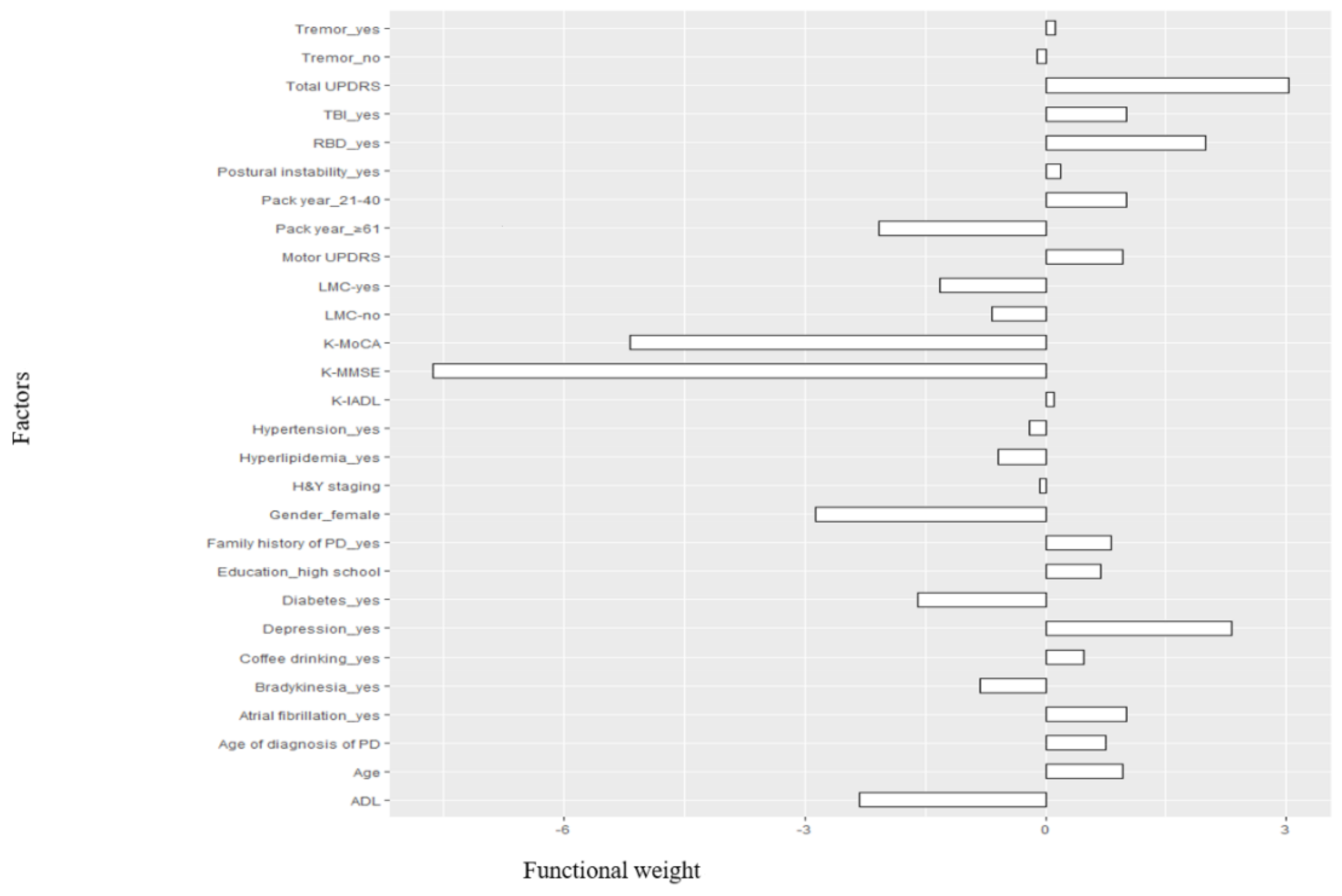
3.3. Factors Related to the Severity of PDD Using the SVR Models
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Papagno, C.; Trojano, L. Cognitive and behavioral disorders in Parkinson’s disease: An update. I: Cognitive impairments. J. Neurol Sci. 2018, 39, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Schwarzschild, M.A. The epidemiology of Parkinson’s disease: Risk factors and prevention. Lancet Neurol. 2016, 15, 1257–1272. [Google Scholar] [CrossRef]
- Byeon, H. Development of a depression in Parkinson’s disease prediction model using machine learning. World J. Psychiatr. 2020, 10, 234–244. [Google Scholar] [CrossRef]
- Wilczyński, J.; Habik, N. The effect of L-dopa on postural stability in Parkinson’s disease patients. Appl. Sci. 2019, 9, 409–419. [Google Scholar] [CrossRef]
- Korczyn, A.D. Dementia in Parkinson’s disease. J. Neurol. 2001, 248, 1–4. [Google Scholar]
- Borrione, P.; Tranchita, E.; Sansone, P.; Parisi, A. Effects of physical activity in Parkinson’s disease: A new tool for rehabilitation. World J. Methodol. 2014, 4, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Jellinger, K.A. Dementia with Lewy bodies and Parkinson’s disease-dementia: Current concepts and controversies. J. Neural Transm. 2018, 125, 615–650. [Google Scholar] [CrossRef]
- Madhusoodanan, S.; Ting, M.B. Pharmacological management of behavioral symptoms associated with dementia. World J. Psychiatr. 2014, 4, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Woo, B.K. Association between recognizing dementia as a mental illness and dementia knowledge among elderly Chinese Americans. World J. Psychiatr. 2016, 6, 233–238. [Google Scholar] [CrossRef]
- Choi, S.H.; Na, D.L.; Lee, B.H.; Hahm, D.S.; Jeong, J.H.; Yoon, S.J.; Yoo, K.H.; Ha, C.K.; Han, I.W. Estimating the validity of the Korean version of expanded Clinical Dementia Rating (CDR) scale. J. Korean Neurol. Assoc. 2001, 19, 585–591. [Google Scholar]
- Huang, H.C.; Tseng, Y.M.; Chen, Y.C.; Chen, P.Y.; Chiu, H.Y. Diagnostic accuracy of the Clinical Dementia Rating Scale for detecting mild cognitive impairment and dementia: A bivariate meta-analysis. Int. J. Geriatr. Psychiatry 2020, 36, 239–251. [Google Scholar] [CrossRef]
- Seppi, K.; Ray Chaudhuri, K.; Coelho, M.; Fox, S.H.; Katzenschlager, R.; Perez Lloret, S.; Weintraub, D.; Sampaio, C. The collaborators of the Parkinson’s disease update on non-motor symptoms study group on behalf of the movement disorders society evidence-based medicine committee. Update on treatments for nonmotor symptoms of Parkinson’s disease—An evidence-based medicine review. Mov. Disord. 2019, 34, 180–198. [Google Scholar] [PubMed]
- Lam, K.; Chan, W.S.; Luk, J.K.; Leung, A.Y. Assessment and diagnosis of dementia: A review for primary healthcare professionals. Hong Kong Med. J. 2019, 25, 473–482. [Google Scholar] [CrossRef]
- Liu, G.Y.; Liu, K.H.; Zhang, Y.; Wang, Y.Z.; Wu, X.H.; Lu, Y.Z.; Pan, C.; Yin, P.; Liao, H.F.; Su, J.Q.; et al. Alterations of tumor-related genes do not exactly match the histopathological grade in gastric adenocarcinomas. World J. Gastroenterol. 2010, 16, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Shankar, K.; Lakshmanaprabu, S.K.; Gupta, D.; Maseleno, A.; De Albuquerque, V.H.C. Optimal feature-based multi-kernel SVM approach for thyroid disease classification. J. Supercomput. 2020, 76, 1128–1143. [Google Scholar] [CrossRef]
- Li, D.; Ju, Y.; Zou, Q. Protein folds prediction with hierarchical structured SVM. Curr. Proteom. 2016, 13, 79–85. [Google Scholar] [CrossRef]
- Dolatabadi, A.D.; Khadem, S.E.Z.; Asl, B.M. Automated diagnosis of coronary artery disease (CAD) patients using optimized SVM. Comput. Methods Programs Biomed. 2017, 138, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Byeon, H. Best early-onset Parkinson dementia predictor using ensemble learning among Parkinson’s symptoms, rapid eye movement sleep disorder, and neuropsychological profile. World J. Psychiatr. 2020, 10, 245–259. [Google Scholar] [CrossRef]
- Hughes, A.J.; Daniel, S.E.; Kilford, L.; Lees, A.J. Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: A clinico-pathological study of 100 cases. J. Neurol. Neurosurg. Psychiatry 1992, 55, 181–184. [Google Scholar] [CrossRef]
- Dubois, B.; Burn, D.; Goetz, C.; Aarsland, D.; Brown, R.G.; Broe, G.A.; Dickson, D.; Duyckaerts, C.; Cummings, J.; Gauthier, S.; et al. Diagnostic procedures for Parkinson’s disease dementia: Recommendations from the movement disorder society task force. Mov. Disord. 2007, 22, 2314–2324. [Google Scholar] [CrossRef]
- Schwab, R.S.; England, A.C.J. Projection Technique for Evaluating Surgery in Parkinson’s Disease. In Third Symposium on Parkinson’s Disease; Gillingham, F.J., Donaldson, I.M.L., Eds.; E&S Livingstone: Edinburgh, UK, 1969. [Google Scholar]
- Kang, Y.; Park, J.; Yu, K.H.; Lee, B.C. The validity of the Korean-Montreal Cognitive Assessment (K-MoCA) as a screening test for both MCI and VCI. In Proceedings of the 20th Annual Rotman Research Institute Conference, The Frontal Lobes, Toronto, ON, Canada, 22–26 March 2010. [Google Scholar]
- Kang, Y.; Na, D.L.; Hahn, S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J. Korean Neurol. Assoc. 1997, 15, 300–308. [Google Scholar]
- Kang, S.J.; Choi, S.H.; Lee, B.H.; Kwon, J.C.; Na, D.L.; Han, S.H. Korean Dementia Research Group. The reliability and validity of the Korean Instrumental Activities of Daily Living (K-IADL). J. Korean Neurol. Assoc. 2002, 20, 8–14. [Google Scholar]
- Richards, M.; Marder, K.; Cote, L.; Mayeux, R. Interrater reliability of the Unified Parkinson’s Disease Rating Scale motor examination. Mov. Disord. 1994, 9, 89–91. [Google Scholar] [CrossRef]
- Movement Disorder Society Task Force on Rating Scales for Parkinson’s Disease. The unified Parkinson’s disease rating scale (UPDRS): Status and recommendations. Mov. Disord. 2003, 18, 738–750. [Google Scholar] [CrossRef]
- Hoehn, M.M.; Yahr, M.D. Parkinsonism: Onset, progression and mortality. Neurology 1976, 17, 427–442. [Google Scholar] [CrossRef] [PubMed]
- Smola, A.J.; Schölkopf, B. A tutorial on support vector regression. Stat. Comput. 2004, 14, 199–222. [Google Scholar] [CrossRef]
- Xu, Y.; Yang, J.; Shang, H. Meta-analysis of risk factors for Parkinson’s disease dementia. Transl. Neurodegener. 2016, 5, 11. [Google Scholar] [CrossRef]
- Hoogland, J.; Boel, J.A.; de Bie, R.M.A.; Schmand, B.A.; Geskus, R.B.; Dalrymple-Alford, J.C.; Marras, C.; Adler, C.H.; Weintraub, D.; Junque, C.; et al. Validation of Mild Cognitive Impairment in Parkinson Disease. Risk of Parkinson’s disease dementia related to level I MDS PD-MCI. Mov. Disord. 2019, 34, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Marinus, J.; Zhu, K.; Marras, C.; Aarsland, D.; van Hilten, J.J. Risk factors for non-motor symptoms in Parkinson’s disease. Lancet Neurol. 2018, 17, 559–568. [Google Scholar] [CrossRef]
- Goetz, C.G.; Emre, M.; Dubois, B. Parkinson’s disease dementia: Definitions, guidelines, and research perspectives in diagnosis. Ann. Neurol. 2008, 64, S81–S92. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, M. Comparison of early stage clinical symptoms, cognitive, affective and ADL functions between PDD and DLB. J. Neurol. Sci. 2017, 381, 1021. [Google Scholar] [CrossRef]
- Leroi, I.; Pantula, H.; McDonald, K.; Harbishettar, V. Neuropsychiatric symptoms in Parkinson’s disease with mild cognitive impairment and dementia. Parkinsons Dis. 2012, 2012, 308097. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.A.; Kim, J.H.; Lee, M.S. Status of Korean patients with Parkinson’s disease before diagnosis. J. Korean Neurol. Assoc. 2000, 18, 687–693. [Google Scholar]
- Park, J.H.; Kim, D.H.; Kwon, D.Y.; Choi, M.; Kim, S.; Jung, J.H.; Han, K.; Park, Y.G. Trends in the incidence and prevalence of Parkinson’s disease in Korea: A nationwide, population-based study. BMC Geriatr. 2019, 19, 320. [Google Scholar] [CrossRef] [PubMed]
- Llebaria, G.; Pagonabarraga, J.; Kulisevsky, J.; García-Sánchez, C.; Pascual-Sedano, B.; Gironell, A.; Martínez-Corral, M. Cut-off score of the Mattis Dementia Rating Scale for screening dementia in Parkinson’s disease. Mov. Disord. 2008, 23, 1546–1550. [Google Scholar] [CrossRef]
- Lamorski, K.; Sławiński, C.; Moreno, F.; Barna, G.; Skierucha, W.; Arrue, J.L. Modelling soil water retention using support vector machines with genetic algorithm optimisation. Sci. World J. 2014, 2014, 740521. [Google Scholar] [CrossRef]
- Pal, A.; Pegwal, N.; Kaur, S.; Mehta, N.; Behari, M.; Sharma, R. Deficit in specific cognitive domains associated with dementia in Parkinson’s disease. J. Clin. Neurosci. 2018, 57, 116–120. [Google Scholar] [CrossRef] [PubMed]
| SVR | Kernel Function | |||
|---|---|---|---|---|
| Linear | Polynomial | RBF | Sigmoid | |
| Eps | 0.101 | 0.095 | 0.079 | 0.110 |
| Nu | 0.079 | 0.102 | 0.078 | 0.091 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byeon, H. Predicting the Severity of Parkinson’s Disease Dementia by Assessing the Neuropsychiatric Symptoms with an SVM Regression Model. Int. J. Environ. Res. Public Health 2021, 18, 2551. https://doi.org/10.3390/ijerph18052551
Byeon H. Predicting the Severity of Parkinson’s Disease Dementia by Assessing the Neuropsychiatric Symptoms with an SVM Regression Model. International Journal of Environmental Research and Public Health. 2021; 18(5):2551. https://doi.org/10.3390/ijerph18052551
Chicago/Turabian StyleByeon, Haewon. 2021. "Predicting the Severity of Parkinson’s Disease Dementia by Assessing the Neuropsychiatric Symptoms with an SVM Regression Model" International Journal of Environmental Research and Public Health 18, no. 5: 2551. https://doi.org/10.3390/ijerph18052551
APA StyleByeon, H. (2021). Predicting the Severity of Parkinson’s Disease Dementia by Assessing the Neuropsychiatric Symptoms with an SVM Regression Model. International Journal of Environmental Research and Public Health, 18(5), 2551. https://doi.org/10.3390/ijerph18052551






