Validation and Screening Capacity of the European Portuguese Version of the SUNFRAIL Tool for Community-Dwelling Older Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Instruments and Procedures
2.3. Ethical-Legal Considerations
2.4. Statistical Analyses
3. Results
3.1. Sociodemographic and Clinical Characteristics
3.2. Internal Consistency of the SUNFRAIL Tool
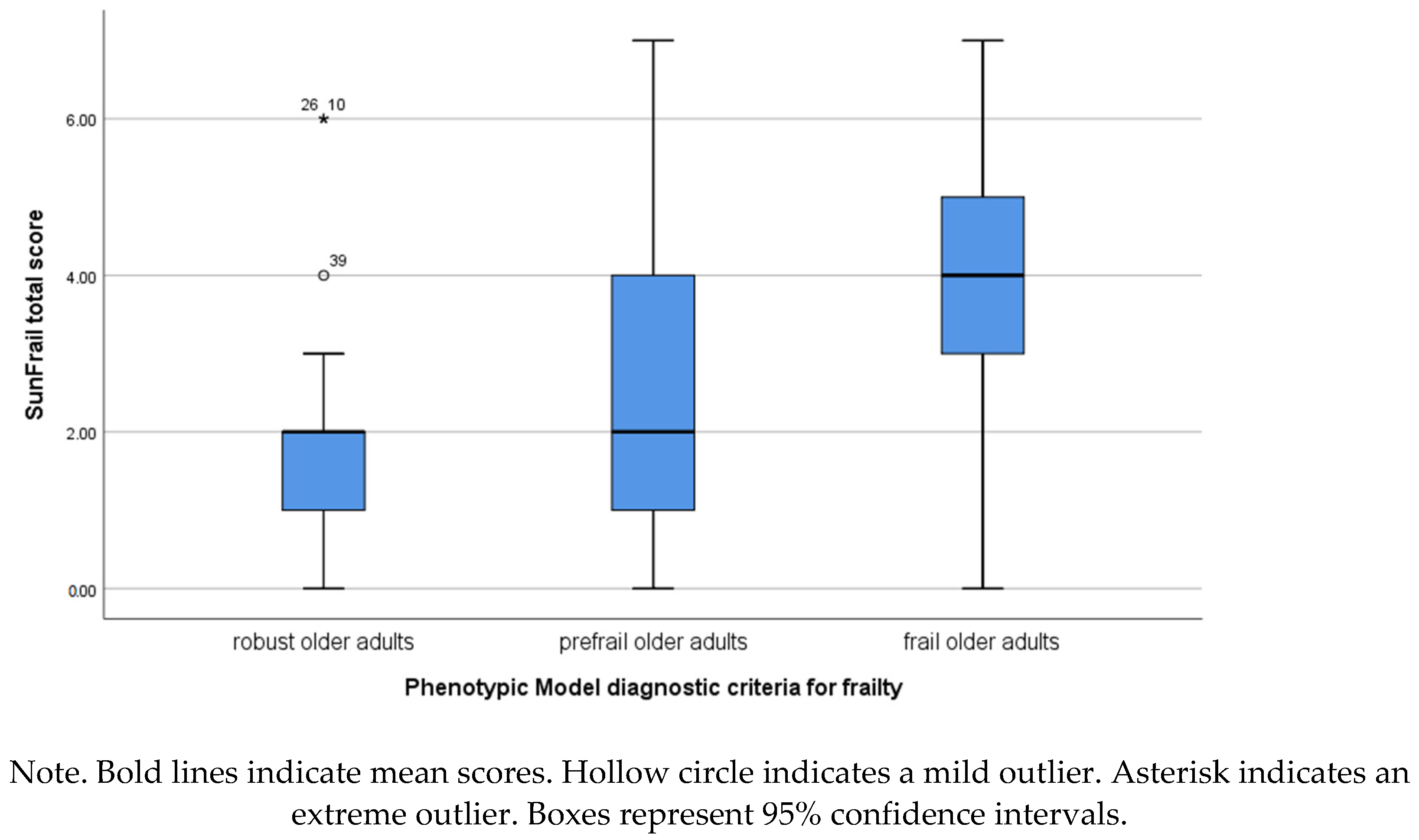
3.3. SUNFRAIL Score in Robust and Non-Robust Older Adults
3.4. SUNFRAIL Tool and 6-CIT
3.5. Concurrent Validity of the SUNFRAIL Tool
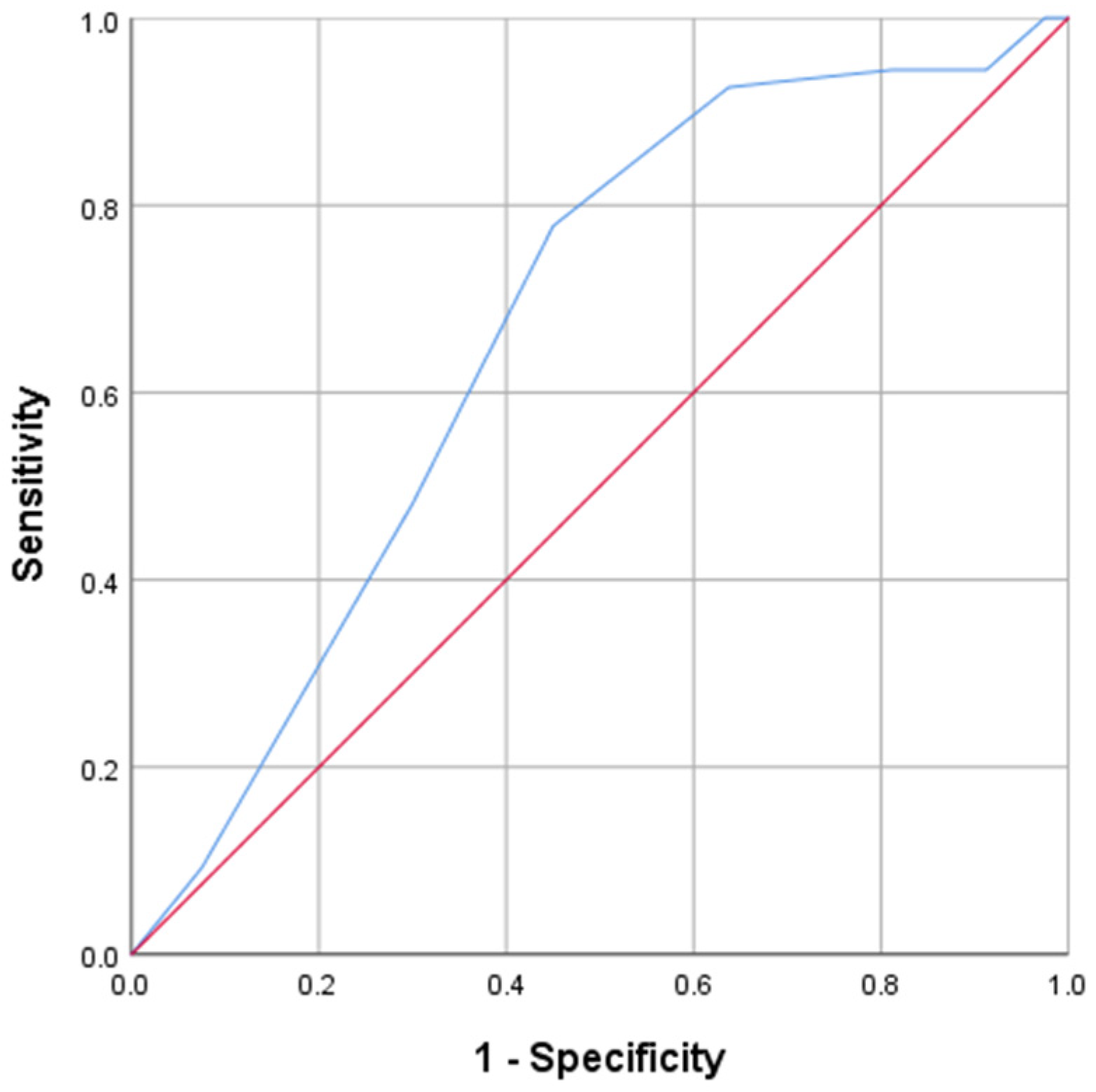
3.6. Sensitivity and Specificity of the SUNFRAIL Tool
4. Discussion
Strengths and Limitation of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boreskie, K.F.; Hay, J.L.; Duhamel, T.A. Preventing Frailty Progression during the COVID-19 Pandemic. J. Frailty Aging 2020, 9, 130–131. [Google Scholar] [CrossRef] [PubMed]
- Lesser, I.A.; Nienhuis, C.P. The Impact of COVID-19 on Physical Activity Behavior and Well-Being of Canadians. Int. J. Environ. Res. Public Health 2020, 17, 3899. [Google Scholar] [CrossRef] [PubMed]
- Giustino, V.; Parroco, A.M.; Gennaro, A.; Musumeci, G.; Palma, A.; Battaglia, G. Physical activity levels and related energy expenditure during COVID-19 quarantine among the sicilian active population: A cross-sectional online survey study. Sustainability 2020, 12, 4356. [Google Scholar] [CrossRef]
- Suzuki, Y.; Maeda, N.; Hirado, D.; Shirakawa, T.; Urabe, Y. Physical activity changes and its risk factors among community-dwelling japanese older adults during the COVID-19 epidemic: Associations with subjective well-being and health-related quality of life. Int. J. Environ. Res. Public Health 2020, 17, 6591. [Google Scholar] [CrossRef] [PubMed]
- Jürschik, P.; Nunin, C.; Botigué, T.; Escobar, M.A.; Lavedán, A.; Viladrosa, M. Prevalence of frailty and factors associated with frailty in the elderly population of Lleida, Spain: The FRALLE survey. Arch. Gerontol. Geriatr. 2012, 55, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Bonaga, B.; Sánchez-Jurado, P.M.; Martínez-Reig, M.; Ariza, G.; Rodríguez-Mañas, L.; Gnjidic, D.; Salvador, T.; Abizanda, P. Frailty, Polypharmacy, and Health Outcomes in Older Adults: The Frailty and Dependence in Albacete Study. J. Am. Med. Dir. Assoc. 2018, 19, 46–52. [Google Scholar] [CrossRef]
- Ding, Y.Y.; Kuha, J.; Murphy, M. Multidimensional predictors of physical frailty in older people: Identifying how and for whom they exert their effects. Biogerontology 2017, 18, 237–252. [Google Scholar] [CrossRef]
- Gobbens, R.J.; van Assen, M.A.; Luijkx, K.G.; Schols, J.M. Testing an integral conceptual model of frailty. J. Adv. Nurs. 2012, 68, 2047–2060. [Google Scholar] [CrossRef]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in Older People. Lancet 2014, 381, 752–762. [Google Scholar] [CrossRef]
- Gwyther, H.; Bobrowicz-Campos, E.; Luis Alves Apóstolo, J.; Marcucci, M.; Cano, A.; Holland, C. A realist review to understand the efficacy and outcomes of interventions designed to minimise, reverse or prevent the progression of frailty. Health Psychol. Rev. 2018, 12, 382–404. [Google Scholar] [CrossRef]
- Apóstolo, J.; Cooke, R.; Bobrowicz-Campos, E.; Santana, S.; MArcucci, M.; Cano, A.; Vollenbroek-Hutten, M.; Germini, F.; Holland, C. Predicting risk and outcomes for frail older adults: An umbrella review of frailty screening tools. JBI Database Syst. Rev. Implement. Rep. 2017, 15, 1154–1208. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Maggio, M.; Palummeri, E.; Poli, S.; Barbolini, M.; Moda, G. Sunfrail Tools for the Identification of Frailty and Multimorbidity; European Union: Emilia-Romagna, Italy, 2018; Volume 10, p. 46, Call identifier: H2020-HP-PJ-2014. [Google Scholar]
- Gobbens, R.; Maggio, M.; Longobucco, Y.; Barbolini, M. The validity of the sunfrail tool: A cross-sectional study among dutch community-dweling older people. J. Frailty Aging 2020, 9, 219–225. [Google Scholar] [CrossRef] [PubMed]
- Beaton, D.; Bombardier, C.; Guillemin, F.; Ferraz, M.B. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine 2000, 15, 3186–3191. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, A.F.; Bobrowicz-Campos, E.; Couto, F.; Cardoso, D.; Barata, A.; Apóstolo, J. Feasibility, appropriateness and meaningfulness analysis of the Sunfrail Tool to the European Portuguese population during cross-cultural adaptation process. Int. J. Evid. Based. Healthc. 2019, 17, S26–S28. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in Older Adults: Evidence for a Phenotype. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef]
- Furtado, G.; Loureiro, M.; Ferreira, J.; Teixeira, A.; Patrício, M. Predicting frail syndrome using adverse geriatric health outcomes: Comparison of different statistical classifiers. In Proceedings of the IEEE 5th Portuguese Meeting on Bioengineering (ENBENG), Coimbra, Portugal, 16–18 February 2017. [Google Scholar] [CrossRef]
- Ainsworth, B.E.; Macera, C.A.; Jones, D.A.; Reis, J.P.; Addy, C.L.; Bowles, H.R.; Kohl, H.W. Comparison of the 2001 BRFSS and the IPAQ physical activity questionnaires. Med. Sci. Sports Exerc. 2006, 38, 1584–1592. [Google Scholar] [CrossRef]
- Gonçalves, B.; Fagulha, T. The Portuguese version of the center of epidemologic studies depression scale (CES-D). Eur. J. Psychol. Assess. 2004, 20, 339–348. [Google Scholar] [CrossRef]
- Brooke, P.; Bullock, R. Validation of a 6 Item Cognitive Impairment Test with a view to primary care usage. Int. J. Geriatr. Psychiatry 1999, 14, 936–940. [Google Scholar] [CrossRef]
- Apóstolo, J.; Paiva, D.; Silva, R.; Santos, E.; Schultz, T. Adaptation and validation into Portuguese language of the six-item cognitive impairment test (6CIT). Aging Ment. Health 2018, 22, 1184–1189. [Google Scholar] [CrossRef]
- Marôco, J. Análise Estatística com o SPSS Statistics, 5th ed.; ReportNumber: Pero Pinheiro, Portugal, 2011; 990p. [Google Scholar]
- Crow, R.S.; Lohman, M.C.; Titus, A.J.; Cook, S.B.; Bruce, M.L.; Mackenzie, T.A.; Bartels, S.J.; Batsis, J.A. Association of Obesity and Frailty in Older Adults: NHANES 1999–2004. J. Nutr. Health Aging 2019, 23, 138–144. [Google Scholar] [CrossRef]
- Afonso, C.; Sousa-Santos, A.R.; Santos, A.; Borges, N.; Padrão, P.; Moreira, P.; Amaral, T.F. Frailty status is related to general and abdominal obesity in older adults. Nutr. Res. 2021, 85, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Le Cossec, C.; Perrine, A.L.; Beltzer, N.; Fuhrman, C.; Carcaillon-Bentata, L. Pre-frailty, frailty, and multimorbidity: Prevalences and associated characteristics from two French national surveys. J. Nutr. Health Aging 2016, 20, 860–869. [Google Scholar] [CrossRef] [PubMed]
| Robust Older Adults (n = 54) | Pre-Frail Older Adults (n = 56) | Frail Older Adults (n = 24) | |||
|---|---|---|---|---|---|
| % | % | % | χ2 | Cramer’s V | |
| Gender: Female/Male | 72/28 | 80/20 | 75/25 | 1.021 | 0.600 |
| Marital Status: single/married/widowed/divorced | 4/69/13.5 /13.5 | 4/54.5/36 /5.5 | 4/46/37.5 /12.5 | 9.628 | 0.141 |
| 6-CIT: without cognitive decline/with mild cognitive decline | 81.5/18.5 | 79/21 | 42/58 | 14.853 | 0.001 |
| Mean (SD) | Mean (SD) | Mean (SD) | Kruskal-Wallis (p) | Pairwise comparisons | |
| Age | 70.83 (4.50) | 72.15 (5.36) | 72.39 (3.86) | 0.321 | -------- |
| Education level | 8.76 (3.99) | 7.84 (4.14) | 7.25 (4.20) | 0.299 | -------- |
| Medication intake | 1.5 (1.28) | 1.91 (1.37) | 2.63 (1.31) | 0.003 | R < F * |
| Comorbidities | 3.81 (1.84) | 3.84 (2.09) | 4.88 (1.87) | 0.078 | -------- |
| Non-Robust Older Adults | |||||
|---|---|---|---|---|---|
| Robust Older Adults (n = 54) | Total (n = 80) | Pre-Frail Older Adults (n = 56) | Frail Older Adults (n = 24) | ||
| SUNFRAIL Total score | Mean ± SD (range) | 1.83 ± 1.37 (0–6) | 2.84 ± 1.81 (0–7) | 2.36 ± 1.63 (0–7) | 3.96 ± 1.73 (0–7) |
| SUNFRAIL -Biological | Mean ± SD (range) | 1.07 ± 0.91 (0–4) | 1.56 ± 1.21 (0–4) | 1.18 ± 1.05 (0–4) | 2.46 ± 1.10 (0–4) |
| SUNFRAIL -Psychological | Mean ± SD (range) | 0.57 ± 0.69 (0–2) | 0.94 ± 0.68 (0–2) | 0.88 ± 0.69 (0–2) | 1.08 ± 0.65 (0–2) |
| SUNFRAIL -Social | Mean ± SD (range) | 0.19 ± 0.48 (0–2) | 0.34 ± 0.50 (0–2) | 0.30 ± 0.50 (0–2) | 0.42 ± 0.50 (0–1) |
| Fried’s Phenotype Model criteria | Mean ± SD (range) | 0.00 ± 0.00 (0.00–0.00) | 1.89 ± 1.06 (1–4) | 1.27 ± 0.45 (1–2) | 3.33 ± 0.48 (3–4) |
| Weight loss | % of persons with symptom | ------- | 1.32% | 0.00% | 4.35% |
| Fatigue | % of persons with symptom | ------- | 27.63% | 7.55% | 73.91% |
| Reduced activity | % of persons with symptom | ------- | 51.32% | 39.62% | 78.26% |
| Reduced speed | % of persons with symptom | ------- | 40.79% | 20.75% | 86.96% |
| Reduced hangrip strength | % of persons with symptom | ------- | 75.00% | 66.04% | 95.65% |
| Cutoff Point | Sensitivity | Specificity | Youden Index |
|---|---|---|---|
| >0 | 9.26% | 92.50% | 0.02 |
| >1 | 48.15% | 70.00% | 0.18 |
| >2 | 77.78% | 55.00% | 0.33 |
| >3 | 92.59% | 36.25% | 0.29 |
| >4 | 94.44% | 18.75% | 0.13 |
| >5 | 94.44% | 8.75% | 0.03 |
| >6 | 100.00% | 2.50% | 0.02 |
| >8 | 100.00% | 0.00% | 0.00 |
| Sensitivity | Specificity | AUC | PPV | NPV | LR+ | LR- |
|---|---|---|---|---|---|---|
| 0.78 (0.64–0.88) | 0.55 (0.43–0.66) | 0.666 * (0.57–0.76) | 0.79 (0.68–0.86) | 0.54 (0.47–0.61) | 2.48 (1.45–4.23) | 0.58 (0.44–0.77) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardoso, A.F.; Bobrowicz-Campos, E.; Teixeira-Santos, L.; Cardoso, D.; Couto, F.; Apóstolo, J. Validation and Screening Capacity of the European Portuguese Version of the SUNFRAIL Tool for Community-Dwelling Older Adults. Int. J. Environ. Res. Public Health 2021, 18, 1394. https://doi.org/10.3390/ijerph18041394
Cardoso AF, Bobrowicz-Campos E, Teixeira-Santos L, Cardoso D, Couto F, Apóstolo J. Validation and Screening Capacity of the European Portuguese Version of the SUNFRAIL Tool for Community-Dwelling Older Adults. International Journal of Environmental Research and Public Health. 2021; 18(4):1394. https://doi.org/10.3390/ijerph18041394
Chicago/Turabian StyleCardoso, Ana Filipa, Elzbieta Bobrowicz-Campos, Luísa Teixeira-Santos, Daniela Cardoso, Filipa Couto, and João Apóstolo. 2021. "Validation and Screening Capacity of the European Portuguese Version of the SUNFRAIL Tool for Community-Dwelling Older Adults" International Journal of Environmental Research and Public Health 18, no. 4: 1394. https://doi.org/10.3390/ijerph18041394
APA StyleCardoso, A. F., Bobrowicz-Campos, E., Teixeira-Santos, L., Cardoso, D., Couto, F., & Apóstolo, J. (2021). Validation and Screening Capacity of the European Portuguese Version of the SUNFRAIL Tool for Community-Dwelling Older Adults. International Journal of Environmental Research and Public Health, 18(4), 1394. https://doi.org/10.3390/ijerph18041394







