The Impact of the COVID-19 Pandemic on Immunization Campaigns and Programs: A Systematic Review
Abstract
:1. Introduction
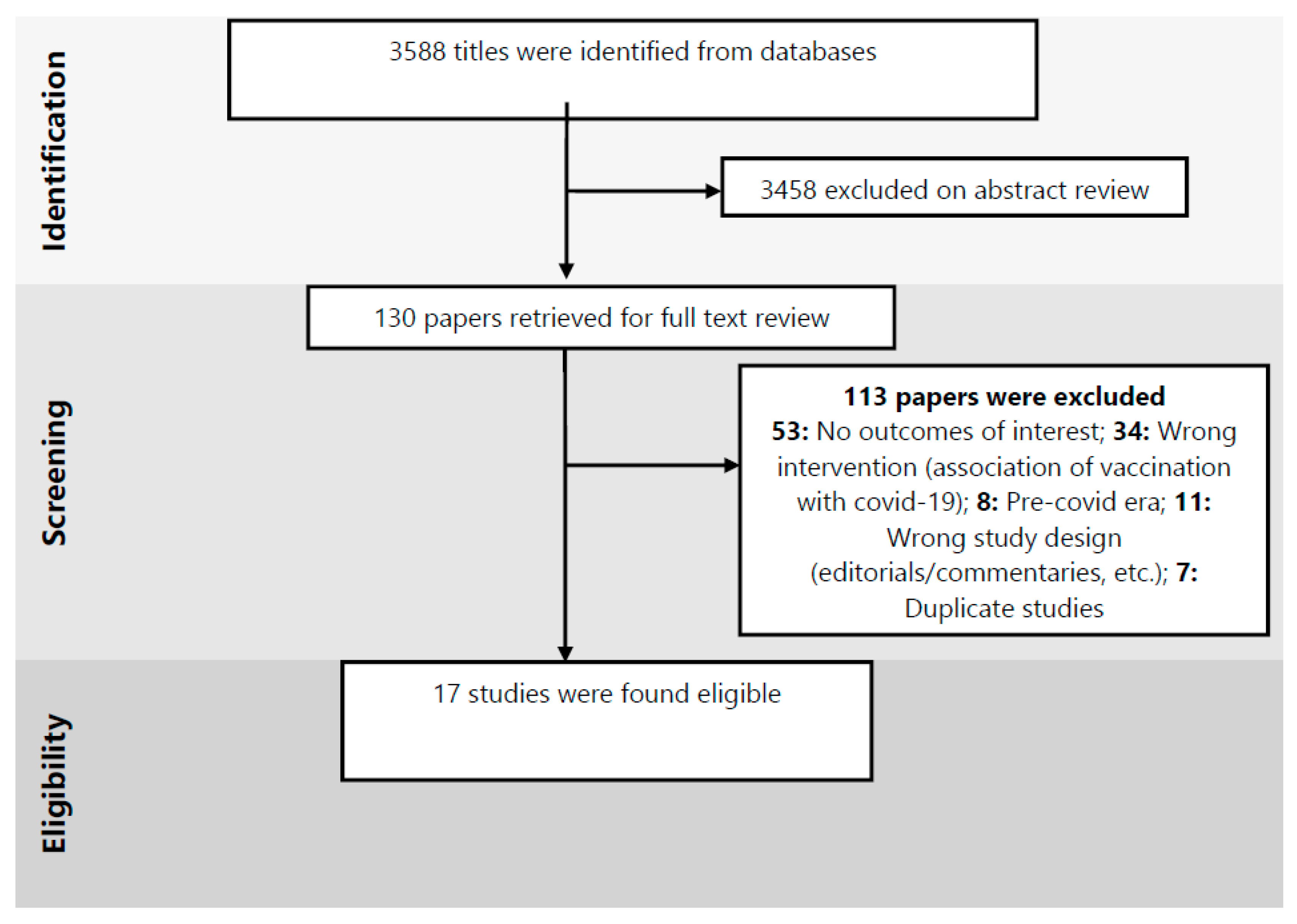
2. Methods
- Impact on routine immunization (including immunization coverage; and pediatric clinic visits);
- Impact on mass immunizations or supplementary immunization activities; and
- Factors affecting immunization coverage
3. Results
3.1. Characteristics of the Included Studies
3.2. Impact on Routine Immunization
Immunization Coverage
3.3. Impact on Mass Immunization or Supplementary Immunization Activities (SIAs)
3.4. Factors Affecting Immunization Coverage
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- WHO Director-General’s Opening Remarks at the Media Briefing on COVID-19—11 March 2020. Available online: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-media-briefing-on-covid-19---11-march-2020 (accessed on 31 July 2020).
- Atique, S.; Zarour, A.; Siddiqui, T.; El-Menyar, A.; Maull, K.; Al Thani, H.; Latifi, R. Trauma caused by falling objects at construction sites. J. Trauma Acute Care Surg. 2012, 73, 704–708. [Google Scholar] [CrossRef] [PubMed]
- Oyo-Ita, A.; Wiysonge, C.S.; Oringanje, C.; Nwachukwu, C.E.; Oduwole, O.; Meremikwu, M.M. Interventions for improving coverage of childhood immunisation in low- and middle-income countries. Cochrane Database Syst. Rev. 2016, 7, Cd008145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- PAHO. The Immunization Program in the Context of the COVID-19 Pandemic, 26 March 2020; PAHO: Washington, DC, USA, 2020. [Google Scholar]
- WHO. Guiding Principles for Immunization Activities during the COVID-19 Pandemic. Interim Guidance. 26 March 2020. Available online: https://apps.who.int/iris/bitstream/handle/10665/331590/WHO-2019-nCoV-immunization_services-2020.1-eng.pdf (accessed on 31 July 2020).
- WHO. Progress and Challenges with Achieving Universal Immunization Coverage. 2017 WHO/UNICEF Estimates of National Immunization Coverage [internet]; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- WHO. At Least 80 Million Children under One at Risk of Diseases Such as Diphtheria, Measles and Polio as COVID-19 Disrupts Routine Vaccination Efforts, Warn GAVI, WHO and UNICEF. Available online: https://www.who.int/news-room/detail/22-05-2020-at-least-80-million-children-under-one-at-risk-of-diseases-such-as-diphtheria-measles-and-polio-as-covid-19-disrupts-routine-vaccination-efforts-warn-gavi-who-and-unicef (accessed on 5 August 2020).
- Nelson, R. COVID-19 disrupts vaccine delivery. Lancet Infect. Dis. 2020, 20, 546. [Google Scholar] [CrossRef]
- Zar, H.J.; Dawa, J.; Fischer, G.B.; Castro-Rodriguez, J.A. Challenges of COVID-19 in children in low- and middle-income countries. Paediatr. Respir. Rev. 2020, 35, 70–74. [Google Scholar] [CrossRef]
- GAVI. COVID-19: Massive Impact on Lower-Income Countries Threatens More Disease Outbreaks. Available online: https://www.gavi.org/news/media-room/covid-19-massive-impact-lower-income-countries-threatens-more-disease-outbreaks (accessed on 9 August 2020).
- Hoffman, J.M.R. Slowing the Coronavirus is Speeding the Spread of Other Diseases. New York Times. Available online: https://www.nytimes.com/2020/06/14/health/coronavirus-vaccines-measles.html (accessed on 5 August 2020).
- This Week. Polio This Week as of 13 January 2020. Available online: http://polioeradication.org/polio-today/polio-now/this-week/ (accessed on 5 August 2020).
- Bagcchi, S. COVID-19 and measles: Double trouble for Burundi. Lancet Microbe 2020, 1, e65. [Google Scholar] [CrossRef]
- Roberts, L. Why measles deaths are surging- and coronavirus could make it worse. Nature 2020, 580, 446–447. [Google Scholar] [CrossRef] [PubMed]
- Initiative, M.a.R. More than 117 Million Children at Risk of Missing Out on Measles Vaccine, as COVID-19 Surges. Available online: https://measlesrubellainitiative.org/measles-news/more-than-117-million-children-at-risk-of-missing-out-on-measles-vaccines-as-covid-19-surges/ (accessed on 31 July 2020).
- Navarro, J.; Arrivillaga-Henríquez, J.; Salazar-Loor, J.; Rodriguez-Morales, A.J. COVID-19 and dengue, co-epidemics in Ecuador and other countries in Latin America: Pushing strained health care systems over the edge. Travel Med. Infect. Dis. 2020, 37, 101656. [Google Scholar] [CrossRef] [PubMed]
- Nacher, M.; Douine, M.; Gaillet, M.; Flamand, C.; Rousset, D.; Rousseau, C.; Mahdaoui, C.; Carroll, S.; Valdes, A.; Passard, N.; et al. Simultaneous dengue and COVID-19 epidemics: Difficult days ahead? PLoS Negl. Trop. Dis. 2020, 14, e0008426. [Google Scholar] [CrossRef] [PubMed]
- Adepoju, P. Lessons from Ebola as DRC grapples with conflict, measles, and covid-19. BMJ 2020, 370, m2879. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Samba, T.T.; Yao, J.; Yin, W.; Xiao, L.; Liu, F.; Liu, X.; Zhou, J.; Kou, Z.; Fan, H.; et al. Impact of the Ebola outbreak on routine immunization in western area, Sierra Leone—A field survey from an Ebola epidemic area. BMC Public Health 2017, 17, 363. [Google Scholar] [CrossRef]
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cuomo-Dannenburg, G.; Thompson, H.; Walker, P.G.T.; Fu, H.; et al. Estimates of the severity of coronavirus disease 2019: A model-based analysis. Lancet Infect Dis 2020, 20, 669–677. [Google Scholar] [CrossRef]
- WHO. The Potential Impact of Health Service Disruptions on the Burden of Malaria: A Modelling Analysis for Countries in Sub-Saharan Africa; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Vitek, C.R.; Wharton, M. Diphtheria in the former Soviet Union: Reemergence of a pandemic disease. Emerg. Infect. Dis. 1998, 4, 539–550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Galvagno, S.M., Jr.; Massey, M.; Bouzat, P.; Vesselinov, R.; Levy, M.J.; Millin, M.G.; Stein, D.M.; Scalea, T.M.; Hirshon, J.M. Correlation between the revised trauma score and injury severity score: Implications for prehospital trauma triage. Prehospital Emerg. Care 2019, 23, 263–270. [Google Scholar] [CrossRef]
- Grabenstein, J.D.; Nevin, R.L. Mass immunization programs: Principles and standards. Curr. Top Microbiol. Immunol. 2006, 304, 31–51. [Google Scholar] [CrossRef]
- Covidence. Covidence Systematic Review Software; Covidence: Melbourne, VIC, Australia, 2017. [Google Scholar]
- NHLBI. Study Quality Assessment Tools; NHLBI: Bethesda, MD, USA, 2019. [Google Scholar]
- Haqqi, A.; Zahoor, S.; Aftab, M.N.; Tipu, I.; Rehman, Y.; Ahmed, H.; Afzal, M.S. COVID-19 in Pakistan: Impact on global polio eradication initiative. J. Med. Virol. 2021, 93, 141–143. [Google Scholar] [CrossRef]
- Bechini, A.; Garamella, G.; Giammarco, B.; Zanella, B.; Flori, V.; Bonanni, P.; Boccalini, S. Paediatric activities and adherence to vaccinations during the COVID-19 epidemic period in Tuscany, Italy: A survey of paediatricians. J. Prev. Med. Hyg. 2020, 61, E125–E129. [Google Scholar] [CrossRef]
- Bramer, C.A.; Kimmins, L.M.; Swanson, R.; Kuo, J.; Vranesich, P.; Jacques-Carroll, L.A.; Shen, A.K. Decline in Child Vaccination Coverage During the COVID-19 Pandemic—Michigan Care Improvement Registry, May 2016–May 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 630–631. [Google Scholar] [CrossRef]
- Buonsenso, D.; Cinicola, B.; Kallon, M.N.; Iodice, F. Child Healthcare and Immunizations in Sub-Saharan Africa During the COVID-19 Pandemic. Front. Pediatr. 2020, 8, 517. [Google Scholar] [CrossRef]
- Chanchlani, N.; Buchanan, F.; Gill, P.J. Addressing the indirect effects of COVID-19 on the health of children and young people. CMAJ 2020, 192, E921–E927. [Google Scholar] [CrossRef]
- Chandir, S.; Siddiqi, D.A.; Setayesh, H.; Khan, A.J. Impact of COVID-19 lockdown on routine immunisation in Karachi, Pakistan. Lancet Glob. Health 2020, 8, e1118–e1120. [Google Scholar] [CrossRef]
- Chard, A.N.; Datta, S.D.; Tallis, G.; Burns, C.C.; Wassilak, S.G.F.; Vertefeuille, J.F.; Zaffran, M. Progress Toward Polio Eradication-Worldwide, January 2018–March 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 784–789. [Google Scholar] [CrossRef] [PubMed]
- Kirmani, S.; Saleem, A. Impact of COVID-19 pandemic on paediatric services at a referral centre in Pakistan: Lessons from a low-income and middle-income country setting. Arch. Dis. Child. 2020. [Google Scholar] [CrossRef] [PubMed]
- Langdon-Embry, M.; Papadouka, V.; Cheng, I.; Almashhadani, M.; Ternier, A.; Zucker, J.R. Notes from the Field: Rebound in Routine Childhood Vaccine Administration Following Decline During the COVID-19 Pandemic—New York City, March 1–June 27, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 999–1001. [Google Scholar] [CrossRef]
- McDonald, H.I.; Tessier, E.; White, J.M.; Woodruff, M.; Knowles, C.; Bates, C.; Parry, J.; Walker, J.L.; Scott, J.A.; Smeeth, L.; et al. Early impact of the coronavirus disease (COVID-19) pandemic and physical distancing measures on routine childhood vaccinations in England, January to April 2020. Euro Surveill. 2020, 25. [Google Scholar] [CrossRef] [PubMed]
- Moraga-Llop, F.A.; Fernández-Prada, M.; Grande-Tejada, A.M.; Martínez-Alcorta, L.I.; Moreno-Pérez, D.; Pérez-Martín, J.J. Recovering lost vaccine coverage due to COVID-19 pandemic. Vacunas 2020, 21, 129–135. [Google Scholar] [CrossRef]
- Santoli, J.M.; Lindley, M.C.; DeSilva, M.B.; Kharbanda, E.O.; Daley, M.F.; Galloway, L.; Gee, J.; Glover, M.; Herring, B.; Kang, Y.; et al. Effects of the COVID-19 Pandemic on Routine Pediatric Vaccine Ordering and Administration—United States, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 591–593. [Google Scholar] [CrossRef] [PubMed]
- Saxena, S.; Skirrow, H.; Bedford, H. Routine vaccination during covid-19 pandemic response. BMJ 2020, 369, m2392. [Google Scholar] [CrossRef]
- Siedner, M.J.; Kraemer, J.D.; Meyer, M.J.; Harling, G.; Mngomezulu, T.; Gabela, P.; Dlamini, S.; Gareta, D.; Majozi, N.; Ngwenya, N.; et al. Access to primary healthcare during lockdown measures for COVID-19 in rural South Africa: A longitudinal cohort study. medRxiv 2020. [Google Scholar] [CrossRef]
- Vogt, T.M.; Zhang, F.; Banks, M.; Black, C.; Arthur, B.; Kang, Y.; Lucas, P.; Lamont, B. Provision of Pediatric Immunization Services During the COVID-19 Pandemic: An Assessment of Capacity Among Pediatric Immunization Providers Participating in the Vaccines for Children Program—United States, May 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 859–863. [Google Scholar] [CrossRef]
- GAVI. COVID-19: Situation Report #14. In COVID-19 Situation Reports; GAVI: Geneva, Switzerland, 2020. [Google Scholar]
- Weill, A.; Drouin, J.; Desplas, D.; Cuenot, F.; Dray Spira, R.; Zureik, M. Usage des Médicaments de Ville en France Durant L’épidémie de la Covid-19–Point de Situation Après les 8 Semaines de Confinement et une Semaine de Post-Confinement (jusqu’au 17 mai 2020). Étude Pharmaco-Épidémiologique à Partir des Données de Remboursement du SNDS; GIS ANSM–Cnam: Saint-Denis, France, 2020; 312p. [Google Scholar]
- The Lancet Child. Pandemic school closures: Risks and opportunities. Lancet Child. Adolesc. Health 2020, 4, 341. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Lee, P.I.; Hu, Y.L.; Chen, P.Y.; Huang, Y.C.; Hsueh, P.R. Are children less susceptible to COVID-19? J. Microbiol. Immunol. Infect. 2020, 53, 371–372. [Google Scholar] [CrossRef] [PubMed]
- Roberton, T.; Carter, E.D.; Chou, V.B.; Stegmuller, A.R.; Jackson, B.D.; Tam, Y.; Sawadogo-Lewis, T.; Walker, N. Early estimates of the indirect effects of the COVID-19 pandemic on maternal and child mortality in low-income and middle-income countries: A modelling study. Lancet Glob. Health 2020, 8, e901–e908. [Google Scholar] [CrossRef]
- Macpherson, L.; Ogero, M.; Akech, S.; Aluvaala, J.; Gathara, D.; Irimu, G.; English, M.; Agweyu, A. Risk factors for death among children aged 5-14 years hospitalised with pneumonia: A retrospective cohort study in Kenya. BMJ Glob. Health 2019, 4, e001715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clark, H.; Coll-Seck, A.M.; Banerjee, A.; Peterson, S.; Dalglish, S.L.; Ameratunga, S.; Balabanova, D.; Bhan, M.K.; Bhutta, Z.A.; Borrazzo, J.; et al. A future for the world’s children? A WHO-UNICEF-Lancet Commission. Lancet 2020, 395, 605–658. [Google Scholar] [CrossRef] [Green Version]
- Donohue, J.M.; Miller, E. COVID-19 and School Closures. JAMA 2020, 324, 845–847. [Google Scholar] [CrossRef]
- Van Lancker, W.; Parolin, Z. COVID-19, school closures, and child poverty: A social crisis in the making. Lancet Public Health 2020, 5, e243–e244. [Google Scholar] [CrossRef]
- Mills, A. Health Care Systems in Low- and Middle-Income Countries. N. Engl. J. Med. 2014, 370, 552–557. [Google Scholar] [CrossRef] [Green Version]
- Abbas, K.; Procter, S.R.; van Zandvoort, K.; Clark, A.; Funk, S.; Mengistu, T.; Hogan, D.; Dansereau, E.; Jit, M.; Flasche, S. Routine childhood immunisation during the COVID-19 pandemic in Africa: A benefit-risk analysis of health benefits versus excess risk of SARS-CoV-2 infection. Lancet Glob. Health 2020, 8, e1267–e1272. [Google Scholar] [CrossRef]
- Vigo, D.; Thornicroft, G.; Gureje, O. The Differential Outcomes of Coronavirus Disease 2019 in Low- and Middle-Income Countries vs High-Income Countries. JAMA Psychiatry 2020, 77, 1207–1208. [Google Scholar] [CrossRef] [PubMed]
- Nic Lochlainn, L.M.; de Gier, B.; van der Maas, N.; Strebel, P.M.; Goodman, T.; van Binnendijk, R.S.; de Melker, H.E.; Hahné, S.J.M. Immunogenicity, effectiveness, and safety of measles vaccination in infants younger than 9 months: A systematic review and meta-analysis. Lancet Infect. Dis. 2019, 19, 1235–1245. [Google Scholar] [CrossRef] [Green Version]
- Hosseinpoor, A.R.; Bergen, N.; Schlotheuber, A.; Gacic-Dobo, M.; Hansen, P.M.; Senouci, K.; Boerma, T.; Barros, A.J. State of inequality in diphtheria-tetanus-pertussis immunisation coverage in low-income and middle-income countries: A multicountry study of household health surveys. Lancet Glob. Health 2016, 4, e617–e626. [Google Scholar] [CrossRef] [Green Version]
- Restrepo-Méndez, M.C.; Barros, A.J.; Wong, K.L.; Johnson, H.L.; Pariyo, G.; França, G.V.; Wehrmeister, F.C.; Victora, C.G. Inequalities in full immunization coverage: Trends in low- and middle-income countries. Bull. World Health Organ. 2016, 94, 794. [Google Scholar] [CrossRef]
- G.P.E.I. Endemic Countries. Available online: http://polioeradication.org/where-we-work/polio-endemic-countries/ (accessed on 6 September 2020).
- Niger Reports New Polio Outbreak. Available online: https://www.afro.who.int/news/niger-reports-new-polio-outbreak (accessed on 6 September 2020).
- Singh, V.K.; Mishra, A.; Singh, S.; Kumar, P.; Singh, M.; Jagannath, C.; Khan, A. Emerging prevention and treatment strategies to control COVID-19. Pathogens 2020, 9, 501. [Google Scholar] [CrossRef] [PubMed]
- Słomka, A.; Kowalewski, M.; Żekanowska, E. Coronavirus Disease 2019 (COVID–19): A Short Review on Hematological Manifestations. Pathogens 2020, 9, 493. [Google Scholar] [CrossRef]
| Author | Objective | Country/Region | Method of Retrieval | Source of Data Retrieval |
|---|---|---|---|---|
| High income countries | ||||
| Bechini 2020 [29] | Evaluate the impact of the COVID-19 epidemic on pediatric vaccinations administered | Tuscany, Italy, Europe and Central Asia | Members of Italian Federation of Pediatricians participated in a semi-structured online survey | Project-level data |
| Bramer 2020 [30] | i. Evaluate whether vaccination coverage has changed during the pandemic. ii. Changes in vaccine doses administered to children and the effects of those changes | Michigan, United States, North America | Michigan Care Improvement Registry | State-level: Data derived from a state-level immunization information system, i.e., Michigan Care Improvement Registry. |
| Chanchlani 2020 [32] | Discussed how limited access to health care, parental fear of seeking health care, school closures & financial instability as a result of COVID-19 may lead to increased morbidity and mortality. | United States, North America | Reports by US Vaccines for Children (VFC ) Program | National level—VFC Program is a federally funded program through the CDC” |
| Langdon-Embry 2020 [36] | Weekly number of routine vaccine administered to persons aged <24 months & 2–18 years in 2020 vs. 2019 | New York City, United States, North America | Citywide immunization registry—2.7 million patients records | State-level: Data derived from a state-level immunization information system |
| McDonald 2020 [37] | Assess the early impact of COVID-19 on routine childhood vaccination in England to 26 April 2020. | England & Scotland, United Kingdom; Europe and Central Asia | EHR (electronic health records) records—electronic patient records for over 2600 primary care practices in the UK and over 35 child health providers | State/national level Data derived from a SystmOne software system. |
| Moraga-Llop 2020 [38] | Discusses aspects of recovery of vaccination coverage | Spain, Europe and Central Asia | Not defined | - |
| Santoli 2020 [39] | Assess the impact of the pandemic on pediatric vaccination in the United States. | United States, North America | VFC Program provider order data from CDC’s Vaccine tracking System. | National level—“The VFC Program and Vaccine Tracking System |
| Saxena 2020 [40] | Assess the impact of COVID-19 on routine immunizations | England & Scotland, United Kingdom; Europe and Central Asia | Not defined | Not defined |
| Vogt 2020 [42] | Assess the capacity of pediatric health care practices to provide immunization services to children during the covid-19 pandemic. | United States, North America | Survey of practices participating in the Vaccines for Children (VFC) program | Project data |
| Alain Weill 2020 [44] | Report quantifies the evolution of drug use prescribed in France, since the start of the epidemic | France; Europe and Central Asia | Number of users measured & compared to the ‘expected’ number of consumers-estimated from consumption data during the same weeks in 2018 and 2019 | Project data; data from National Hospital Discharge Survey (NHDS)-EPI-PHARE reported dispensing of reimbursed drugs in pharmacies since the start of the epidemic |
| Low- and middle-income countries | ||||
| Haqqi 2020 [28] | Assess the impact on global polio eradication initiative | Pakistan, South Asia | Not defined | Not defined |
| Buonsenso 2020 [31] | Understand the potential impact of COVID-19 on child vaccinations in a typical poor peripheral area of Sierra Leone | Kent, Rural Western Area, Sierra Leone; Sub Saharan Africa | Cases retrospectively collected by the center’s health workers registry book | Project- level data |
| Chandir/GAVI 2020 [33] | Understand the impact of COVID-19 restrictions on routine immunization coverage in Karachi | Karachi, Pakistan, South Asia; | Used data from the provincial Electronic Immunization Registry. | Project- level data |
| Chard 2020 [34] | Summarizes progress toward polio eradication during 1 January 2018 and 31 March 2020 | Afghanistan & Pakistan; South Asia | Global Polio Eradication Initiative (GPEI) | Project level—derived from GPEI |
| Kirmani 2020 [35] | Study the disruption in clinical activity at the Children’s Hospital | Karachi, Pakistan, South Asia; | Systematically studied the disruption in clinical activity | Project-level data |
| Siedner 2020 [41] | Assess the impact of the lockdown order in response to the COVID-19 epidemic in South Africa | Kwa-Zulu Natal, South Africa, Sub-Saharan Africa | Data collected by the Africa Health Research Institute (AHRI) | National-level data—data on clinic visitation at 11 public health clinics |
| GAVI 2020 [43] | Situation report #14 | Pakistan, South Asia | Expanded program on immunization | Project- level data |
| Author | Time Period | Vaccine | Outcome | |
|---|---|---|---|---|
| Mass immunization or supplementary immunization activities (SIAs) | ||||
| New polio cases | ||||
| Haqqi 2020 [28] | COVID-19 until June 2020 | Polio | New polio cases | Polio cases as of June 2020: 51 cases |
| Chard 2020 [34] | 1 January 2018–31 March 2020 | Polio | New polio cases | Jan–March 2020: 54 cases- 12 in Afghanistan and 42 in Pakistan Jan–March 2019: 12 cases- 6 in Afghanistan and 6 in Pakistan |
| Routine Immunization | ||||
| Immunization coverage | ||||
| Bramer 2020 [30] | a point in time in May 2020 compared to points in time in May 2016–May 2019 | HepB; rotavirus (Rota); Diphtheria, Tetanus, Pertussis (DTaP); Haemophilus influenzae type b; pneumococcal conjugate; inactivated poliovirus; MMR; varicella; hepatitis A | Doses of vaccines administered Percentage of children vaccinated | Number of doses administered Jan–April 2020 vs. 2018 & 2019: children aged ≤18 years decreased 21.5% children aged ≤24 months decreased 15.5% Percentage of infants and children vaccinated: 1 month: 2016–19: 82; 83; 82; 84, 2020: 85 3 months: 2016–19: 74; 75; 74; 65, 2020: 61 5 months: 2016–19: 66.6; 67.4; 67.3; 67.9, 2020: 49.7 7 months: 2016–19: 52; 54; 55; 54, 2020: 44 16 months: 2016–19: 52; 54; 54; 55; 2020: 40 19 months: 2016–19: 60; 62; 60; 2020: 5524 months: 2016–19: 45; 47; 49. 50, 2020: 43 |
| Buosenso 2020 [31] | 1 March 2019 to 26 April 2019 v1 March 2020 to 26 April 2020 | Bacillus Calmette–Guérin (BCG), Oral poliovirus vaccines (OPV), Pneumococcal vaccine (PCV),Tetanus txoid (TT), pentavalent vaccine (PENTA): diphtheria, pertussis, tetanus, hep b, hemophilus, TT, Intermittent preventive treatment in infant | Vaccines administered at a health centre | BCG: Pre-covid 36; COVID: 17; Change −52.7; p-value < 0.0005 OPV0 Pre-covid 36; COVID: 17; Change −52.7; p-value < 0.0005 OPV1 Pre-covid 58; COVID: 17; Change −70.7; p-value < 0.0005 PENTA1 Pre-covid: 58; COVID: 17; Change −70.7; p-value: < 0.0005 PCV1 Pre-covid 58; COVID: 17; Change −70.7; p-value: < 0.0005 ROTA1 Pre-covid: 58; COVID: 17; Change −70.7; p-value: < 0.0005 OPV2 Pre-covid: 71; COVID: 15; Change −78.9; p-value: < 0.0005 PENTA2 Pre-covid: 71; COVID: 15; Change −78.9; p-value: < 0.0005 PCV2 Pre-covid: 71; COVID: 15; Change −78.9; p-value: < 0.0005 ROTA2 Pre-covid: 71; COVID: 15; Change −78.9; p-value: < 0.0005 IPTI1 Pre-covid: 49; COVID: 15; Change −69.4; p-value: < 0.0005 OPV3 Pre-covid: 67; COVID: 15; Change −77.6; p-value: < 0.0005 PENTA3 Pre-covid: 67; COVID: 15; Change −77.6; p-value: < 0.0005 PCV3 Pre-covid: 67; COVID: 15; Change −77.6; p-value: < 0.0005 IPTI2 Pre-covid: 44; COVID: 15; Change −65.9; p-value: < 0.0005 IPV Pre-covid: 67; COVID: 15; Change −77.6; p-value: < 0.0005 IPTI3 Pre-covid: 45; COVID: 22; Change −51.1; p-value: < 0.0005 Measles Pre-covid: 64; COVID: 22; Change −65.6; p-value: < 0.0005 Yellow fever Pre-covid 64; COVID 22; Change −65.6; p-value < 0.0005 Measles 2nd Pre-covid 49; COVID 8; Change −83.7; p-value < 0.0005 |
| Chandir/GAVI 2020 [33] | 6 months before the lockdown (23 Sept 2019–22 March 2020) vs. first 6 weeks of the lockdown (March 23–May 9, 2020) | General | Immunization coverage Visit to immunization clinic | Daily average number of immunization 6 months before COVID-19 lockdown: Total: 16, 649 Enrollments: 32% Follow-ups: 68% During COVID-19 lockdown: Total: 5836 Enrollments: 31% Follow-ups: 69% Immunization of children (aged 0–23 months): Baseline: 608,832 children Lockdown: 92,492 children decrease in mean number of daily immunization visits vs baseline: Lockdown (March–May 9): −52.8% (5184 to 2450) Post-lockdown (May 10–June 2020): −27.2% (5184 to 3772). 2734 children per day missed routine immunization during the lockdown Immunization centre: 16% of 321 immunization centres had no flow reduction in immunization doses: 88.6% for outreach; 38.7% for fixed Vaccinators attending work: Baseline: 91.6% Lockdown: 78.7% |
| Langdon-Embry 2020 [36] | 2020 vs. 2019 | General | Doses administerednumber of facilities that administered at least one vaccine in 2020 | Mar 1: First confirmed COVID-19 case in NYC <24 months: 2019: 32,000; 2020: 32,0002–18 years: 2019: 19,000; 2020: 20,000 Mar 13: National emergency declared <24 months: 2019: 35,000; 2020: 31,0002–18 years: 2019: 20,000; 2020: 16,000 Mar 22: NYC on PAUSE—required New Yorkers to stay at home <24 months: 2019: 33,000; 2020: 15,0002–18 years: 2019: 21,000; 2020: 3000 April 5–11: <24 months: 62% decrease: 2019: 33,000; 2020: 13,0002–18 years: 96% decrease: 2019: 24,000; 2020: 1000 facilities administering at least one vaccine, <24 months: 46% decrease, from 900 in 2019 to 488 in 2020 facilities administering at least one vaccine, 2–18 years: decreased 78%, from 1238 in 2019 to 275 in 2020 April 19–25: number of new COVID-19 cases declined <24 months: 2019: 27,000; 2020: 20,0002–18 years: 2019: 25,000; 2020: 3000 June 21–27: facilities administering at least one vaccine, <24 months:increased 69% from the lowest point to 825 Vaccines administered to persons 2–18 years:35% fewer vaccines (17,947 doses versus 27,405). |
| McDonald 2020 [37] | first 17 weeks of 2019 and 2020: | i. Hexavalent: DTaP, polio, Hib and Hep B ii. MMR | Immunization coverage | 1. Hexavalent: i. 2019: 69,568; ii. 2020: 67,116, Percentage change: −3.5 (−3.7 to −3.4) 2. MMR: i. 2019: 65, 341; ii. 2020: 61, 832. Percentage change: −3.7 (−3.8 to −3.6) Week 1–9: prior to social distancing Hexavalent: −5.8% (95% CI—6.0 to −5.5%) MMR: −1.0% (95% CI −1.1 to −0.9%) Week 10–12: instruction of self isolation by UK government Hexavalent: 4.4% (95% CI −4.8 to −4.0%) MMR: −7.2% (95% CI −7.7 to −6.7%) Week 13: Strict social distancing introduced Hexavalent: −5.1% (−5.9 to −4.4) MMR: −24.2 (−25.9 to −22.5) Week 13–15: Hexavalent: −6.7% (95% CI −7.1 to −6.2%) MMR: −19.8% (95% CI −20.7 to −18.9%) |
| Moraga-Llop 2020 [38] | COVID-19 era vs. Pre-COVID-19 | Hexavalent, pneumococcus, meningococcus, MMR, Varicella, Men B, Rota, Tdap | Immunization coverage | Coverage decreased in all autonomous communities between 5% and 60%Reduction in vaccines administered (March 2020 vs. 2019) 2 & 4 mths- hexavalent, pneumococcus & meningococcus C: 8–13% 11-month booster doses -hexavalent and pneumococcus: 15% 12-months -MMR & meningococcus ACWY: 12% 15 months- varicella: 20% Rota virus: 19% Men B: 40% pertussis vaccination in pregnant women was not affected (−0.6%) decrease more evident for vaccines not funded: Valencian: 1st dose of Men B: −68.4% Andalusia: total doses of Men B: −39% Rotavirus: −18% decrease accentuated with age; Valencian Community: tetanus & diphtheria (>64 years of age): −67.5%. private pediatrics did not register the same effects |
| GAVI 2020 [43] | Mar–April 2019 vs. 2020 | Pentavalent 3, Oral polio vaccine type 3 (OPV3), Measles, Bacillus Calmette–Guérin (BCG) | Immunization coverage | % target children covered: Pentavalent 2019: 89, 2020: 40 Oral polio vaccine (OPV) 2019: 88, 2020: 62 Measles 2019: 84, 2020: 61 BCG 2019: 88, 2020: 64 |
| Alain Weil 2020 [44] | Lockdown: March 16–May 10, 2020Post-lockdown: May 11–17, 2020 | Anti- human papilloma virus (HPV) Combined Penta/Hexavalent vaccines for infants: Hæmophilus influenzæ B, pertussis, poliomyelitis, tetanus, hep B MMR Anti-tetanus (excluding infants) | Drugs prescribed since the start of the epidemic | March 2–8: HPV: −3.6; Comb: −0.4; MMR: −13.6; Tetanus: +0.3 March 9–15: HPV: −6.8; Comb: −2.5; MMR: −12.3; Tetanus: −6.5March 16–22: HPV: −21.9; Comb: −3.6; MMR: −28.5; Tetanus: −26.2 March 23–29: HPV: −67.4; Comb: −23; MMR: −49; Tetanus: −64.9 March 30-April 5: HPV: −78.1; Comb: −28.9; MMR: −50.8; Tetanus: −77.3April 6–12: HPV: −74.4; Comb: −21.3; MMR: −50.8; Tetanus: −76.5 April 13–19: HPV: −69.6; Comb: −18.7; MMR: −46.9; Tetanus: −72.9 April 20–26: HPV: −45.6; Comb: + 6.1; MMR: −21.7; Tetanus: −55.2April 30–May 3: HPV: −45.6; Comb: + 6.1; MMR: −21.7; Tetanus: −55.2 May 4–10: HPV: −43.2; Comb: −5.6; MMR: −15.7; Tetanus: −48.4 Difference: HPV: −89,508; Comb: −44,171; MMR: −123,966; Tetanus: −446,580 Post-lockdown May 11–17: HPV: −34.4; Comb: −6.4; MMR: −4.7; Tetanus:−37,9 |
| Vaccine orders | ||||
| Chanchlani 2020 [32] | 6 Jan.–19 Apr. 2020, v. 2019 | General, MMR, diptheria | decrease in orders | Decrease in vaccination: Apr. 5, 2020 vs Feb 16 (pre-coronavirus): 1. 50% for MMR 2. 42% for diphtheria and whooping cough |
| Santoli 2020 [39] | Period 1: 7 January–21 April 2019Period 2: 6 January–19 April 2020 | DTaP; Hib; Hep A; Hep B; HPV: influenza; MMR; Meningococcal; Pneumococcal 13-& 23 valent; IPV; Rota; Tdap; Var | decrease in vaccine orders in period 2, compared to period 1 | measles-containing vaccine administered: 21.5% decline (Mar, 2020)Children ≤24 months: March 9th (before US national emergency): 2000 March 16th (After US national emergency): 1100 Children >24 months through 18 years: March 9th (before US national emergency): 2250 March 16th (After US national emergency): 500 difference in doses of vaccines ordered by health care providers:non-influenza vaccines Jan 20th (first U.S. COVID-19 case reported): −500,000Apr 13th: −3,000,000 measles-containing vaccinesJan 20th: −100,000Apr 13th: −400,000 |
| Health care providers providing immunization services during the pandemic | ||||
| Bechini 2020 [29] | Lockdown: 11 March 11–4 May 2020 | Not identified | Activities & behavior of pediatricians Visits to clinic | 1) 208 (93.3%) pediatricians continued to vaccinate 66 (31.7%): reduction in compliance to mandatory vaccines (hexavalent and MMRV) 88 (42.3%): reduction in compliance to non-mandatory vaccines 37 (17.8%) administrated only the first doses of the vaccines 82.2%) reported administering all the scheduled vaccine doses. 2) 15 (7%) had suspended vaccinations 98.2%- pediatricians reported a general decline in outpatient visits during the COVID-19 epidemic period |
| Vogt 2020 [42] | 12–20 May 2020 | Not identified | Practices providing immunization | Of 1933 practices: open: 1727 (89.8%) providing immunization services: 1397 (81.1%) |
| Visits to clinic | ||||
| Bechini 2020 [29] | Lockdown: 11 March–4 May 2020v pre-COVID era | Not identified | Visits to clinic | (98.2%) reported a general decline in outpatient pediatric visits during the COVID-19 epidemic period65.8% reported a more than 60% reduction in comparison with the situation before the COVID-19 pandemic. |
| Kirmani 2020 [35] | February 2020 to post-lockdown | Not identified | Visits to clinic | Week 7 (pre-COVID): 2600 Week 8 (26 Feb, First case of COVID reported): 2200Week 11 (21 March, Sindh Lockdown): 1800Week 13: 400Week 15: 1300Week 17: 1200Week 19: 1300 |
| Saxena 2020 [40] | - | Not identified | Visits to clinic;Parental concern;Vaccine coverage | Children Visits to ED: fell by over 90% during April 2020 Parents expressed concerns about overburdening the NHS and fear of exposure to covid-19 when attending for vaccination. 60% of 752 health visitors surveyed in May 2020 reported families who had considered cancelling or postponing their child’s vaccinations |
| Siedner 2020 [41] | Pre lockdown: 60 days prior to lockdown Lockdown: 27th March–30th April’ 20: Post lockdown: 35 days after the lockdown. | Not identified | Visits to clinic | overall clinic visits during lockdown: −0.2, (95%CI −3.4, 3.1) visits/day/child-health clinic: pre-lockdown period: 11.8 (8.4, 15.1) lockdown period: 4.5 mean change: −7.2 (95%Cl −9.2, −5.3); p-value: <0.001 mean change post-lockdown: +1.1 (0.5, 1.7); p-value: 0.001Age-stratified: Total visits Jan–Apr. 2020: 6194 <1 year: 4270 (68.9%) 1−5 years:1786 (28.8%) 6–19 years: 103 (1.7%) 20–45 years: 29 (0.5%) >45 years: 6 (0.1%)children under 1: mean decrease of −5.3 visits, 95%CI −7.1, −3.61–5 years: mean decrease of −5.5 visits, 95%CI −6.8, −4.2 |
| Study ID | 1. Study Question or Objective | 2. Eligibility/ Selection Criteria | 3. Representative Participants | 4. Eligible Participants Met Entry Criteria | 5. Sample Size | 6. Test/ Service/ Intervention Clearly Described | 7. Outcome Measures | 8. Outcomes Blinded | 9. Loss to Follow-up | 10. Statistical Methods | 11. Interrupted Time-Series Design | 12. Use of Individual-Level Data |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Haqqi 2020 [28] | Yes | No | No | NR | NR | NR | No | N/A | N/A | No | No | No |
| Bechini 2020 [29] | Yes | Yes | Yes | NR | CD | Yes | Yes | N/A | N/A | No | No | No |
| Bramer 2020 [30] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | N/A | No | No | No |
| Buonsenso 2020 [31] | Yes | Yes | Yes | NR | CD | Yes | Yes | N/A | N/A | Yes | No | No |
| Chanchlani 2020 [32] | Yes | Yes | NR | N/A | NR | NR | No | N/A | N/A | No | No | No |
| Chandir/GAVI 2020 [33] | Yes | Yes | NR | NR | NR | Yes | Yes | N/A | N/A | No | No | No |
| Chard 2020 [34] | Yes | No | No | NR | Yes | Yes | CD | N/A | N/A | No | No | No |
| Kirmani 2020 [35] | Yes | Yes | NR | NR | NR | N/A | Yes | N/A | N/A | No | No | No |
| Langdon-Embry 2020 [36] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | N/A | No | No | No |
| McDonald 2020 [37] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | N/A | CD | NR | No |
| Moraga-Llop 2020 [38] | Yes | No | CD | N/A | NR | NA | NR | N/A | N/A | No | No | No |
| Santoli 2020 [39] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | N/A | No | No | No |
| Saxena 2020 [40] | Yes | NR | NR | NR | NR | No | No | N/A | N/A | No | No | No |
| Siedner 2020 [41] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N/A | N/A | Yes | Yes | No |
| Vogt 2020 [42] | Yes | Yes | Yes | NR | Yes | Yes | Yes | N/A | N/A | No | No | No |
| GAVI 2020 [43] | Yes | No | NR | NR | CD | N/A | NR | N/A | N/A | No | No | No |
| Alain Weill 2020 [44] | Yes | Yes | Yes | N/A | NR | CD | NR | N/A | N/A | No | No | No |
Detailed Criteria:
| ||||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lassi, Z.S.; Naseem, R.; Salam, R.A.; Siddiqui, F.; Das, J.K. The Impact of the COVID-19 Pandemic on Immunization Campaigns and Programs: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 988. https://doi.org/10.3390/ijerph18030988
Lassi ZS, Naseem R, Salam RA, Siddiqui F, Das JK. The Impact of the COVID-19 Pandemic on Immunization Campaigns and Programs: A Systematic Review. International Journal of Environmental Research and Public Health. 2021; 18(3):988. https://doi.org/10.3390/ijerph18030988
Chicago/Turabian StyleLassi, Zohra S., Rabia Naseem, Rehana A. Salam, Faareha Siddiqui, and Jai K. Das. 2021. "The Impact of the COVID-19 Pandemic on Immunization Campaigns and Programs: A Systematic Review" International Journal of Environmental Research and Public Health 18, no. 3: 988. https://doi.org/10.3390/ijerph18030988





