Exercise, Immune System, Nutrition, Respiratory and Cardiovascular Diseases during COVID-19: A Complex Combination
Abstract
1. Introduction
2. The Immune System
2.1. The Innate Immunity
2.2. The Acquired Immunity
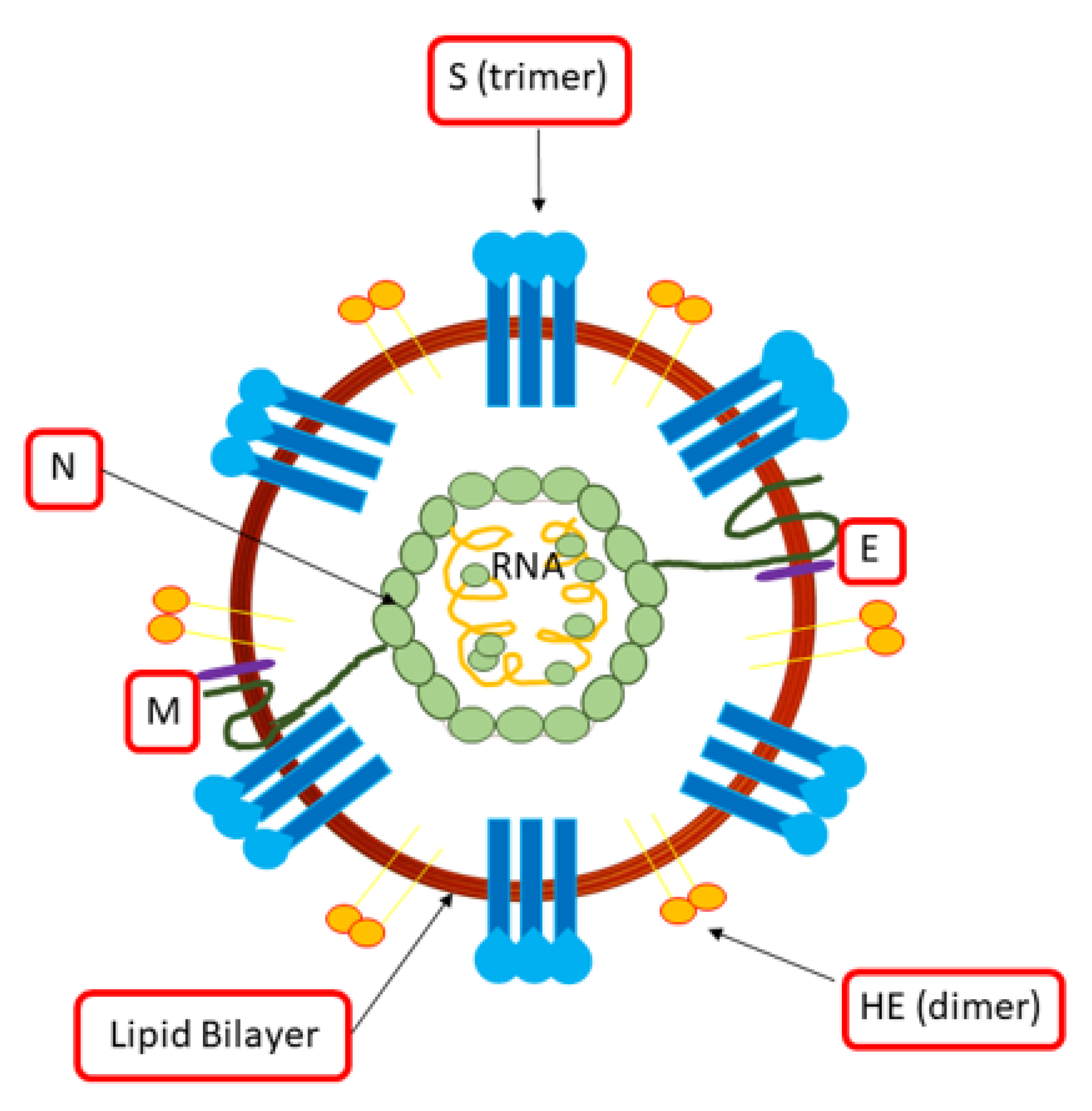
2.3. Mechanism of Immune Systems in the Human Body against COVID-19
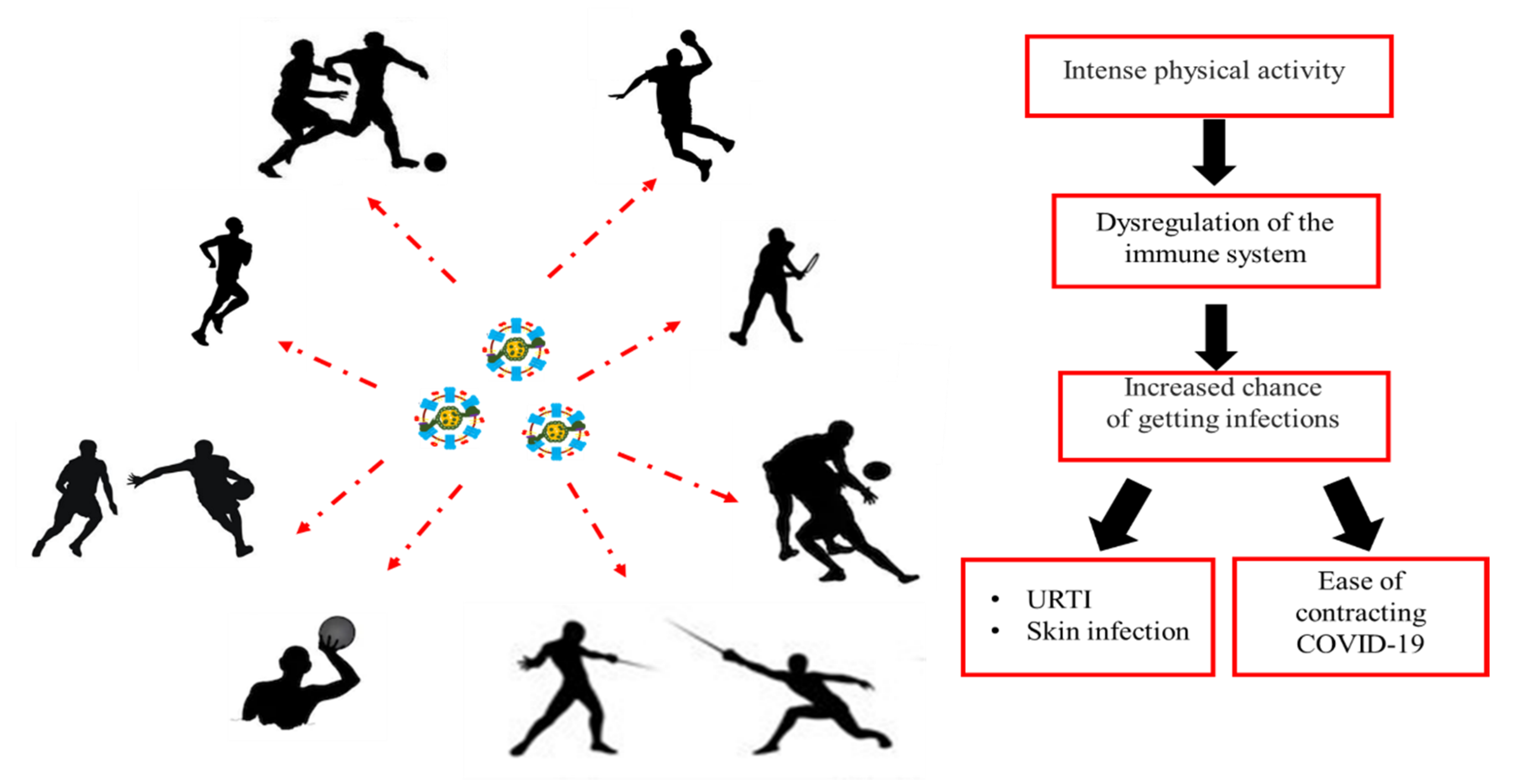
3. Effect of Exercise on Immunity: Open Window Theory
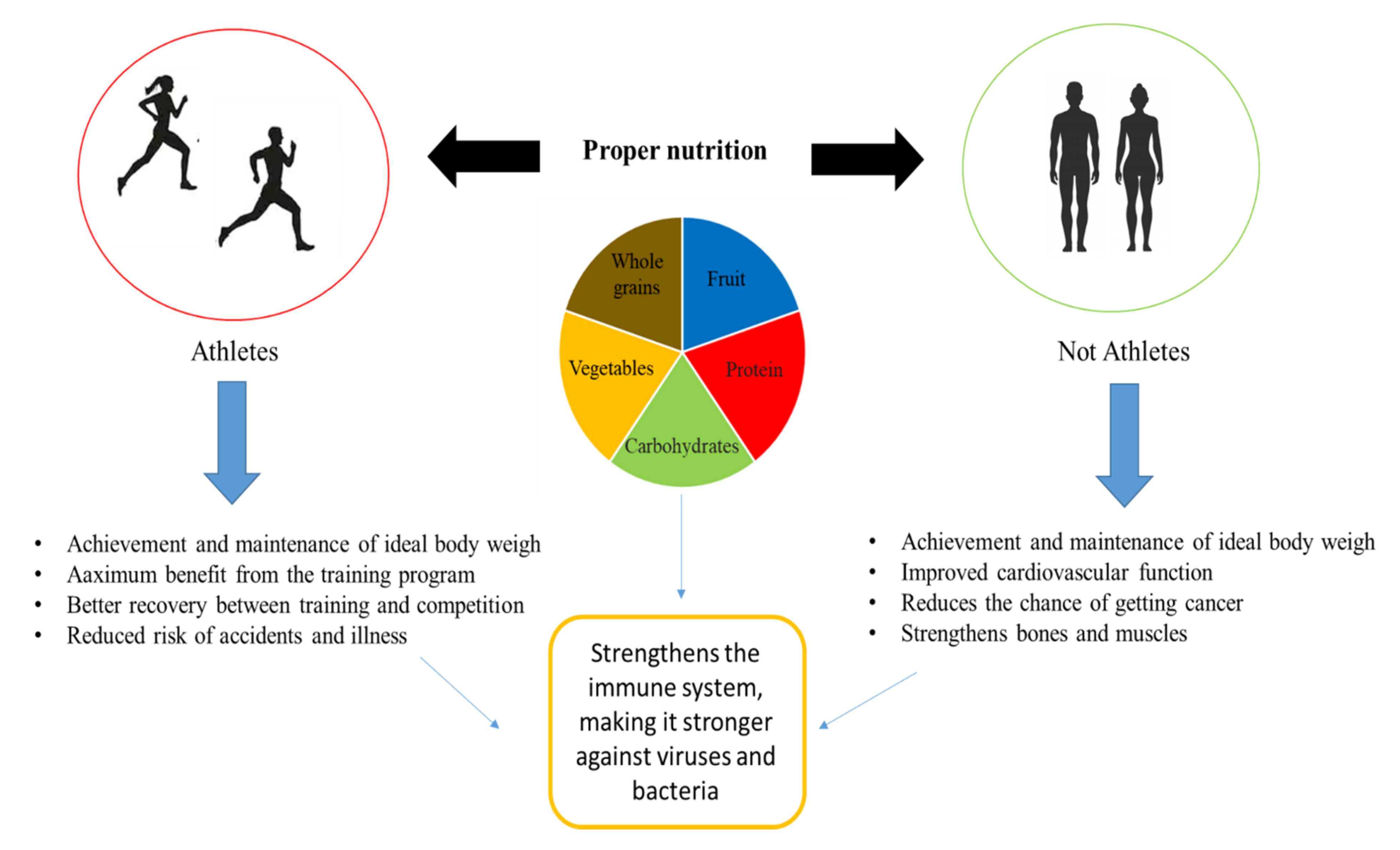
4. Nutrition and Prevention
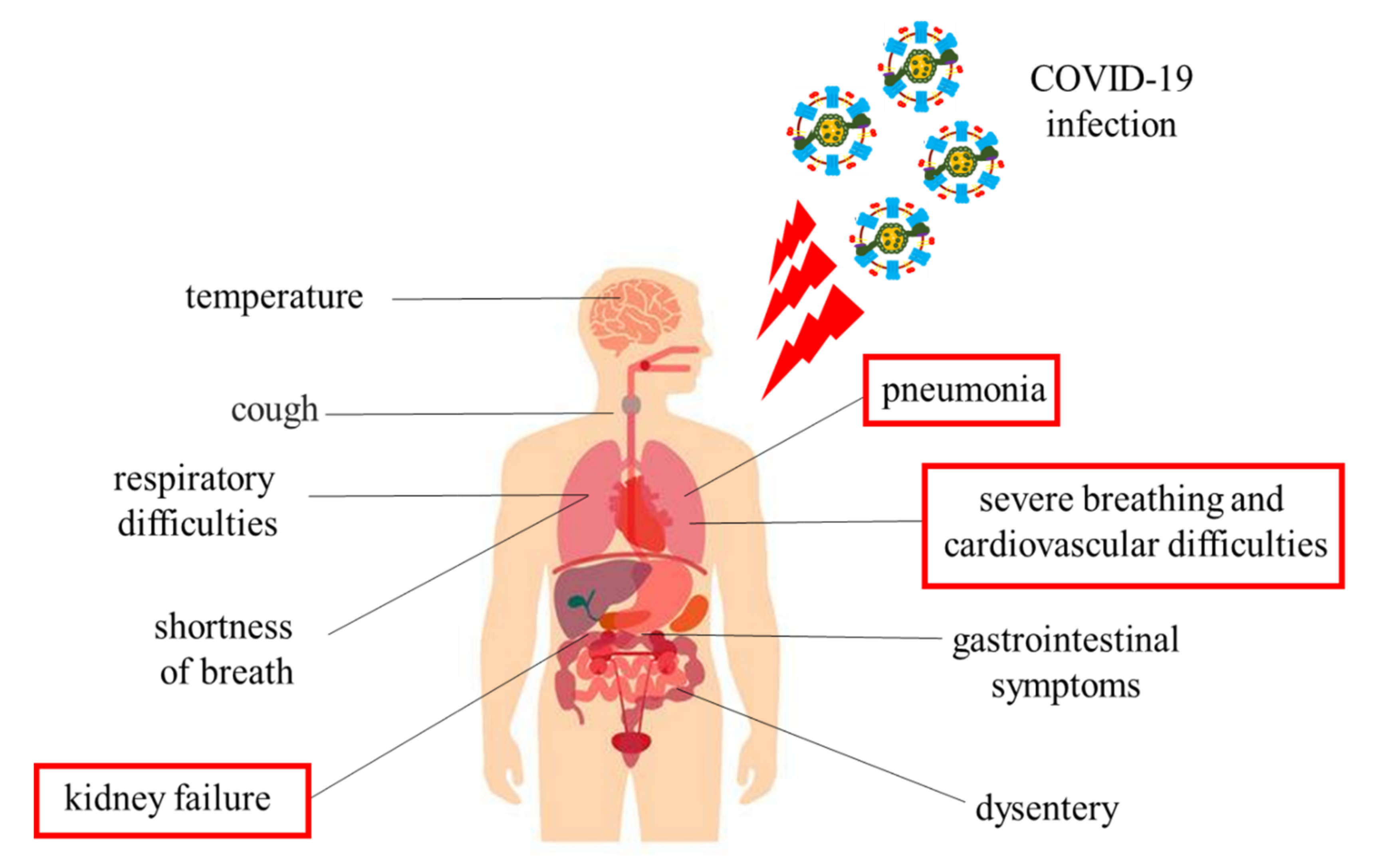
5. Respiratory Infection and Exercise: Risk and Complications of COVID-19
6. Cardiovascular Involvement in COVID-19: Direct Injury and Impact of Cardiac Comorbidities
7. The Association between Sport and Venous Thromboembolism
8. Genetic Predisposition to COVID-19 Infection and to Athlete Performance
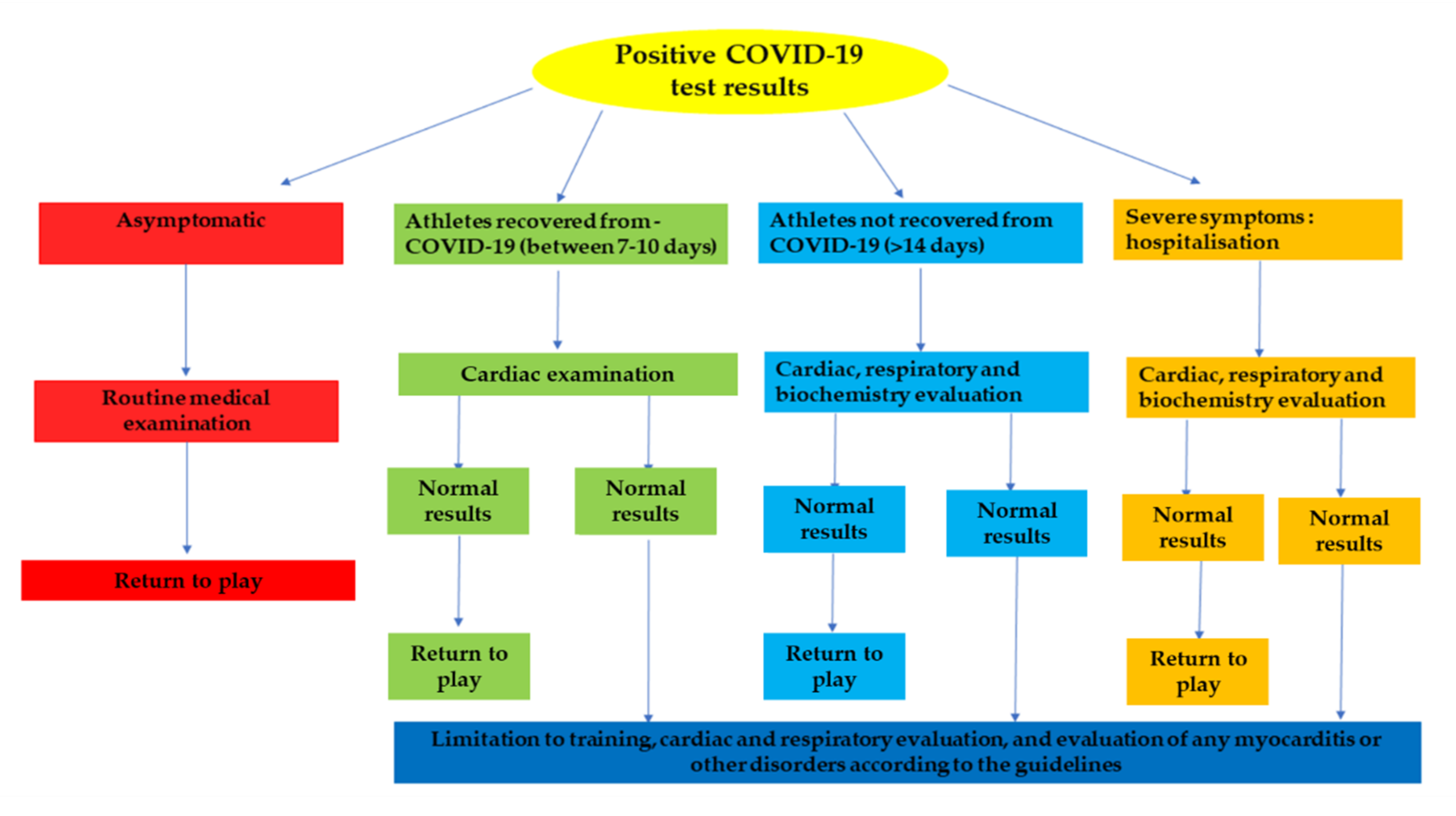
9. The State of Art about Sport and COVID-19
10. Physical and Mental Rehabilitation through Moderate Exercise
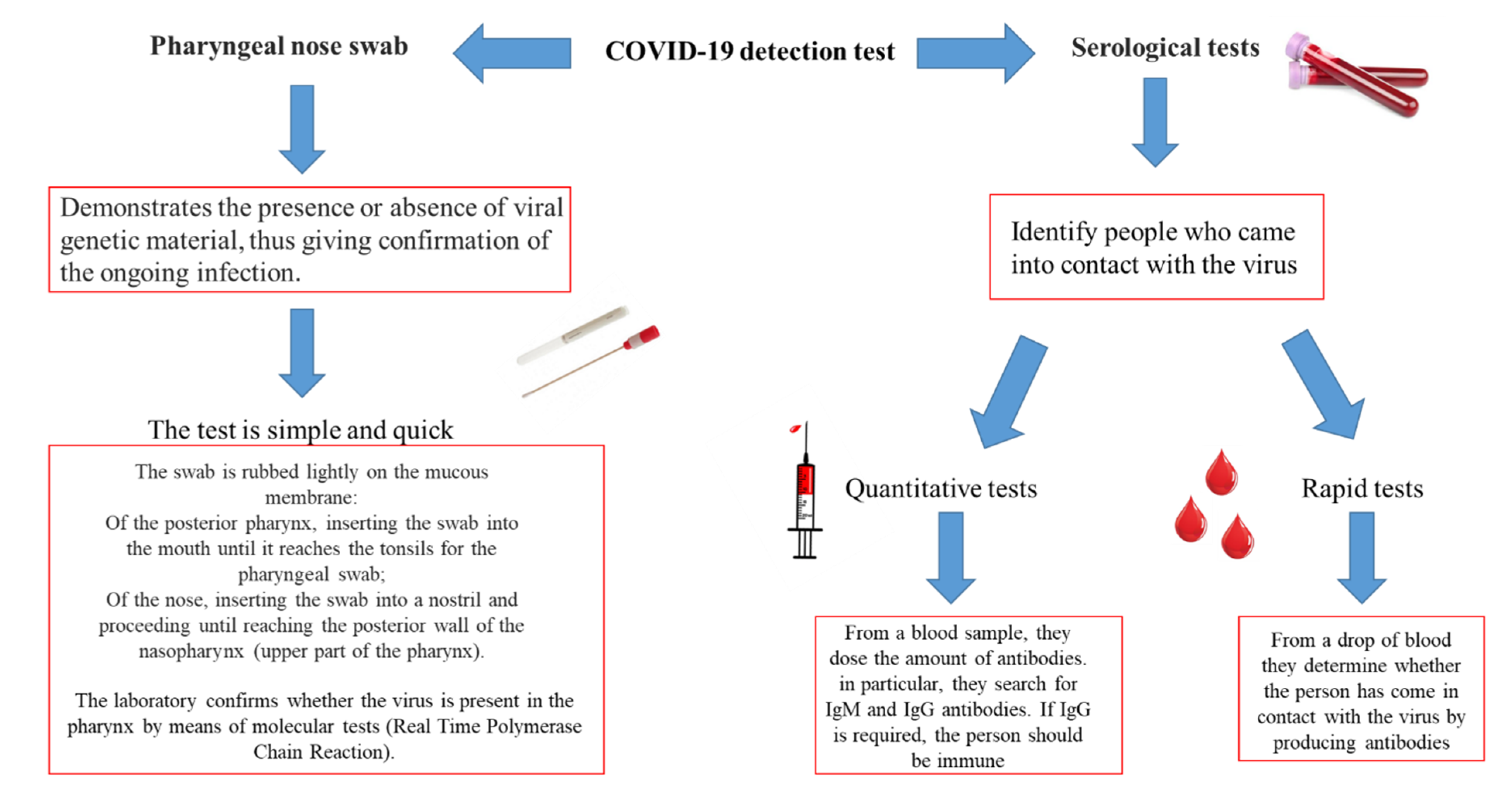
11. Diagnostic Methods for Detecting COVID-19
12. Current Therapies to Fight COVID-19 Disease
13. Future Directions
14. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CoVs | Coronaviruses |
| SARS-CoV | Severe acute respiratory syndrome—coronavirus |
| MERS-CoV | Middle East respiratory syndrome—coronavirus |
| COVID-19 | Coronavirus disease 2019 |
| URTI | Inflammation of the upper respiratory tract |
| AMP | Antimicrobial peptides |
| HNP1 | Human alpha-defensin 1 |
| HBD2 | Human beta-defensin 2 |
| HBD3 | Human beta-defensin 3 |
| HSV-1 | Herpes virus simplex type I |
| HSV-2 | Herpes virus simplex type II |
| CMV | Cytomegalovirus |
| AHSG | Human α-2-Heremans-Schmid Glycoprotein |
| IL-17 | Interleukin-17 |
| NF-kB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| IL-1 | Interleukin-1 |
| IL-6 | Interleukin-6 |
| IL-8 | Interleukin-8 |
| IL–21 | Interleukin-21 |
| IL–10 | Interleukin-10 |
| TNF–α | Tumor necrosis factor-alpha |
| TNF–β | Tumor necrosis factor-beta |
| ARDS | Acute respiratory distress syndrome |
| ACE2 | Angiotensin-converting enzyme 2 |
| TMPRSS2 | Transmembrane Serine Protease 2 |
| ACE1 | Angiotensin-converting enzyme 1 |
| TLR7/8 | Toll-like receptors 7 and 8 |
References
- Zaki, A.M.; van Boheemen, S.; Bestebroer, M.T.; Osterhaus, A.D.M.E.; Fouchier, R.A.M. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012, 367, 1814–1820. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Laboratory Testing of Human Suspected Cases of Novel Coronavirus (nCoV) Infection. Available online: https://www.who.int/publications/i/item/10665-330374 (accessed on 10 January 2020).
- World Health Organization. Novel Coronavirus (2019-nCoV) Situation Report-2. Available online: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200122-sitrep-2-2019-ncov.pdf?sfvrsn=4d5bcbca_2 (accessed on 22 January 2020).
- World Health Organization. Middle East Respiratory Syndrome Coronavirus (MERS-CoV). Available online: https://www.who.int/health-topics/middle-east-respiratory-syndrome-coronavirus-mers#tab=tab_1 (accessed on 19 January 2020).
- World Health Organization. WHO MERS Global Summary and Assessment of Risk. Available online: https://apps.who.int/iris/bitstream/handle/10665/326126/WHO-MERS-RA-19.1-eng.pdf?ua=1 (accessed on 19 July 2019).
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Hui, D.S.; Azhar, E.I.; Madani, T.A.; Ntoumi, F.; Kock, R.; Dar, O.; Ippolito, G.; Mchugh, T.D.; Memish, Z.A.; Drosten, C.; et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health—The latest 2019 novel coronavirus outbreak in Wuhan, China. Int. J. Infect. Dis. 2020, 91, 264–266. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Morales, A.J.; Bonilla-Aldana, D.K.; Balbin-Ramon, G.J.; Rabaan, A.A.; Sah, R.; Paniz-Mondolfi, A.; Pagliano, P.; Esposito, S. History is repeating itself: Probable zoonotic spillover as the cause of the 2019 novel Coronavirus Epidemic. Infez. Med. 2020, 28, 3–5. [Google Scholar]
- Rothan, H.A.; Byrareddy, S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J. Autoimmun. 2020, 109, 102433. [Google Scholar] [CrossRef]
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus infections and immune responses. J. Med. Virol. 2020, 92, 424–432. [Google Scholar] [CrossRef]
- Scudiero, O.; Brancaccio, M.; Mennitti, C.; Laneri, S.; Lombardo, B.; De Biasi, M.G.; De Gregorio, E.; Pagliuca, C.; Colicchio, R.; Salvatore, P.; et al. Human Defensins: A Novel Approach in the Fight against Skin Colonizing Staphylococcus aureus. Antibiotics 2020, 9, 198. [Google Scholar] [CrossRef]
- Nicholson, L.B. The immune system. Essays Biochem. 2016, 60, 275–301. [Google Scholar] [CrossRef]
- Kumar, H.; Kawai, T.; Akira, S. Pathogen Recognition by the Innate Immune System. Int. Rev. Immunol. 2011, 30, 16–34. [Google Scholar] [CrossRef]
- Uthaisangsook, S.; Day, N.K.; Bahna, S.L.; Good, R.A.; Haraguchi, S. Innate immunity and its role against infections. Ann. Allergy Asthma Immunol. 2002, 88, 253–264. [Google Scholar] [CrossRef]
- Pero, R.; Angrisano, T.; Brancaccio, M.; Falanga, A.; Lombardi, L.; Natale, F.; Laneri, S.; Lombardo, B.; Galdiero, S.; Scudiero, O. Beta-defensins and analogs in Helicobacter pylori infections: mRNA expression levels, DNA methylation, and antibacterial activity. PLoS ONE 2019, 14, e0222295. [Google Scholar] [CrossRef] [PubMed]
- Pero, R.; Brancaccio, M.; Laneri, S.; De Biasi, M.G.; Lombardo, B.; Scudiero, O. A Novel View of Human Helicobacter pylori Infections: Interplay between Microbiota and Beta-Defensins. Biomolecules 2019, 9, 237. [Google Scholar] [CrossRef] [PubMed]
- Pero, R.; Coretti, L.; Nigro, E.; Lembo, F.; Laneri, S.; Lombardo, B.; Daniele, A.; Scudiero, O. β-Defensins in the Fight against Helicobacter pylori. Molecules 2017, 22, 424. [Google Scholar] [CrossRef] [PubMed]
- Colavita, I.; Nigro, E.; Sarnataro, D.; Scudiero, O.; Granata, V.; Daniele, A.; Zagari, A.; Pessi, A.; Salvatore, F. Membrane protein 4F2/CD98 is a cell surface receptor involved in the internalization and trafficking of human β-Defensin 3 in epithelial cells. Chem. Biol. 2015, 22, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Falanga, A.; Nigro, E.; De Biasi, M.G.; Daniele, A.; Morelli, G.; Galdiero, S.; Scudiero, O. Cyclic Peptides as Novel Therapeutic Microbicides: Engineering of Human Defensin Mimetics. Molecules 2017, 20, 1217. [Google Scholar] [CrossRef]
- Scudiero, O.; Nigro, E.; Cantisani, M.; Colavita, I.; Leone, M.; Mercurio, F.A.; Galdiero, M.; Pessi, A.; Daniele, A.; Salvatore, F.; et al. Design and activity of a cyclic mini-β-defensin analog: A novel antimicrobial tool. Int. J. Nanomed. 2015, 10, 6523–6539. [Google Scholar]
- Falanga, A.; Valiante, S.; Galdiero, E.; Franci, G.; Scudiero, O.; Morelli, G.; Galdiero, S. Dimerization in tailoring uptake efficacy of the HSV-1 derived membranotropic peptide gH625. Sci. Rep. 2017, 7, 9434. [Google Scholar] [CrossRef]
- Daher, K.A.; Selsted, M.E.; Lehrer, R.I. Direct inactivation of viruses by human granulocyte defensins. J. Virol. 1986, 60, 1068–1074. [Google Scholar] [CrossRef]
- Wilson, S.S.; Wiens, M.E.; Smith, J.G. Antiviral Mechanisms of Human Defensins. J. Mol. Biol. 2013, 425, 4965–4980. [Google Scholar] [CrossRef]
- Chan, J.F.; Lau, S.K.; To, K.K.; Cheng, V.C.; Woo, P.C.; Yuen, K.Y. Middle East respiratory syndrome coronavirus: Another zoonotic betacoronavirus causing SARS-like disease. Clin. Microbiol. Rev. 2015, 28, 465–522. [Google Scholar] [CrossRef]
- Manni, M.L.; Robinson, K.M.; Alcorn, J.F. A tale of two cytokines: IL-17 and IL-22 in asthma and infection. Expert Rev. Respir. Med. 2014, 8, 25–42. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.; Wong, P.; Klein, J.; Castro, T.B.R.; Silva, J.; Sundaram, M.; Ellingson, M.K.; Mao, T.; Oh, J.E. Longitudinal analyses reveal immunological misfiring in severe COVID-19. Nature 2020, 584, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Pero, R.; Brancaccio, M.; Mennitti, C.; Gentile, L.; Franco, A.; Laneri, S.; De Biasi, M.G.; Pagliuca, C.; Colicchio, R.; Salvatore, P.; et al. HNP-1 and HBD-1 as Biomarkers for the Immune Systems of Elite Basketball Athletes. Antibiotics 2020, 9, 306. [Google Scholar] [CrossRef] [PubMed]
- Brancaccio, M.; Mennitti, C.; Laneri, S.; Franco, A.; De Biasi, M.G.; Cesaro, A.; Fimiani, F.; Moscarella, E.; Gragnano, F.; Mazzaccara, C.; et al. Methicillin-Resistant Staphylococcus aureus: Risk for General Infection and Endocarditis Among Athletes. Antibiotics 2020, 9, 332. [Google Scholar] [CrossRef] [PubMed]
- Mennitti, C.; Brancaccio, M.; Gentile, L.; Ranieri, A.; Terracciano, D.; Cennamo, M.; La Civita, E.; Liotti, A.; D’Alicandro, G.; Mazzaccara, C.; et al. Athlete’s Passport: Prevention of Infections, Inflammations, Injuries and Cardiovascular Diseases. J. Clin. Med. 2020, 9, 2540. [Google Scholar] [CrossRef]
- Pero, R.; Brancaccio, M.; Mennitti, C.; Gentile, L.; Arpino, S.; De Falco, R.; Leggiero, E.; Ranieri, A.; Pagliuca, C.; Colicchio, R.; et al. Urinary Biomarkers: Diagnostic Tools for Monitoring Athletes’ Health Status. Int. J. Environ. Res. Public Health 2020, 17, 6065. [Google Scholar] [CrossRef]
- Simpson, R.J.; Kunz, H.; Agha, N.; Graff, R. Chapter Fifteen—Exercise and the Regulation of Immune Functions. Prog. Mol. Biol. Transl. Sci. 2015, 135, 355–380. [Google Scholar]
- Nieman, D.C.; Pedersen, B.K. Exercise and immune function. Recent Dev. 1999, 27, 73–80. [Google Scholar] [CrossRef]
- Kakanis, M.W.; Peake, J.; Brenu, E.W.; Simmonds, M.; Gray, B.; Hooper, S.L.; Marshall-Gradisnik, S.M. The open window of susceptibility to infection after acute exercise in healthy young male elite athletes. Exerc. Immunol. Rev. 2010, 16, 119–137. [Google Scholar] [CrossRef]
- Dhabhar, F.S. Stress-induced augmentation of immune function-The role of stress hormones, leukocyte trafficking, and cytokines. Brain Behav. Immun. 2002, 16, 785–798. [Google Scholar] [CrossRef]
- Ostrowski, K.; Rohde, T.; Asp, S.; Schjerling, P.; Pedersen, B.K. Pro- and anti-inflammatory cytokine balance in strenuous exercise in humans. J. Physiol. 1999, 515, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Schouten, W.; Verschuur, R.; Kemper, H.C.G. Habitual Physical Activity, Strenuous Exercise, and Salivary Immunoglobulin A Levels in Young Adults: The Amsterdam Growth and Health Study. Int. J. Sports Med. 1988, 4, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Trochimiak, T.; Hübner-Woźniak, E. Effect of Exercise on the Level of Immunoglobulin A in Saliva. Biol. Sport 2012, 4, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.M.; Qiao, X.L.; Ai, L.; Zhai, J.J.; Wang, X.X. Isolation of antimicrobial resistant bacteria in upper respiratory tract infections of patients. 3 Biotech 2016, 6, 166. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nieman, D.C. Exercise, infection and immunity. Int. J. Sports Med. 1994, 15, S131–S141. [Google Scholar] [CrossRef]
- Schwellnus, M.; Soligard, T.; Alonso, J.M.; Bahr, R.; Clarsen, B.; Dijkstra, H.P.; Gabbett, T.J.; Gleeson, M.; Hägglund, M.; Hutchinson, M.R.; et al. How much is too much? (Part 2) International Olympic Committee consensus statement on load in sport and risk of illness. Br. J. Sports Med. 2016, 50, 1043–1052. [Google Scholar] [CrossRef]
- Mårtensson, S.; Nordebo, K.; Malm, C. High training volumes are associated with a low number of self-reported sick days in elite endurance athletes. J. Sports Sci. Med. 2014, 13, 929–933. [Google Scholar]
- Brancaccio, M.; D’Argenio, G.; Lembo, V.; Palumbo, A.; Castellano, I. Antifibrotic Effect of Marine Ovothiol in an In Vivo Model of Liver Fibrosis. Oxidative Med. Cell. Longev. 2018, 2018, 5045734. [Google Scholar] [CrossRef]
- Brancaccio, M.; Russo, M.; Masullo, M.; Palumbo, A.; Russo, G.L.; Castellano, I. Sulfur-containing histidine compounds inhibit gamma-glutamyl transpeptidase activity in human cancer cells. J. Biol. Chem. 2019, 294, 14603–14614. [Google Scholar] [CrossRef]
- Brancaccio, M.; Mennitti, C.; Cesaro, A.; Fimiani, F.; Moscarella, E.; Caiazza, M.; Gragnano, F.; Ranieri, A.; D’Alicandro, G.; Tinto, N.; et al. Dietary Thiols: A Potential Supporting Strategy against Oxidative Stress in Heart Failure and Muscular Damage during Sports Activity. Int. J. Environ. Res. Public Health 2020, 17, 9424. [Google Scholar] [CrossRef]
- Guloyan, V.; Oganesian, B.; Baghdasaryan, N.; Yeh, C.; Singh, M.; Guilford, F.; Ting, S.T.; Venketaraman, V. Glutathione Supplementation as an Adjunctive Therapy in COVID-19. Antioxidants 2020, 10, 914. [Google Scholar] [CrossRef] [PubMed]
- Fortmann, S.P.; Burda, B.U.; Senger, C.A.; Lin, J.S.; Whitlock, E.P. Vitamin and mineral supplements in the primary prevention of cardiovascular disease and cancer: An updated systematic evidence review for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2013, 159, 824–834. [Google Scholar] [CrossRef] [PubMed]
- Lakhan, S.E.; Vieira, K.F. Nutritional therapies for mental disorders. Nutr. J. 2008, 7, 2. [Google Scholar] [CrossRef] [PubMed]
- Murdaca, G.; Pioggia, G.; Negrini, S. Vitamin D and Covid-19: An update on evidence and potential therapeutic implications. Clin. Mol. Allergy 2020, 18, 23. [Google Scholar] [CrossRef]
- Shakoor, H.; Feehan, H.S.J.; Al Dhaheri, A.S.; Ali, H.I.; Platat, C.; Ismail, C.L.; Apostolopoulos, V.; Stojanovska, L. Immune-boosting role of vitamins D, C, E, zinc, selenium and omega-3 fatty acids: Could they help against COVID-19? Maturitas 2021, 143, 1–9. [Google Scholar] [CrossRef]
- Hunt, C.; Chakravorty, N.; Annan, G.; Habibzadeh, N.; Schorah, C. The clinical effects of vitamin C supplementation in elderly hospitalised patients with acute respiratory infections. Int. J. Vitam. Nutr. Res. 1994, 64, 212–219. [Google Scholar]
- Hiedra, R.; Lo, K.B.; Elbashabsheh, M.; Gul, F.; Wright, R.M.; Albano, J.; Azmaiprashvili, Z.; Patarroyo Aponte, G. The use of IV vitamin C for patients with COVID-19: A case series. Expert Rev. Anti-Infect. Ther. 2020, 12, 1259–1261. [Google Scholar] [CrossRef]
- Coretti, L.; Natale, A.; Cuomo, M.; Florio, E.; Keller, S.; Lembo, F.; Chiariotti, L.; Pero, R. The Interplay between Defensins and Microbiota in Crohn’s Disease. Mediat. Inflamm. 2017, 2017, 8392523. [Google Scholar] [CrossRef]
- Angrisano, T.; Pero, R.; Brancaccio, M.; Coretti, L.; Florio, E.; Pezone, A.; Calabrò, V.; Falco, G.; Keller, S.; Lembo, F.; et al. Cyclical DNA Methylation and Histone Changes Are Induced by LPS to Activate COX-2 in Human Intestinal Epithelial Cells. PLoS ONE 2016, 11, e0156671. [Google Scholar] [CrossRef]
- Chiariotti, L.; Coretti, L.; Pero, R.; Lembo, F. Epigenetic Alterations Induced by Bacterial Lipopolysaccharides. Adv. Exp. Med. Biol. 2016, 879, 91–105. [Google Scholar]
- Nieman, D.C. Exercise immunology: Future directions for research related to athletes, nutrition, and the elderly. Int. J. Sports Med. 2000, 1, S61–S68. [Google Scholar] [CrossRef] [PubMed]
- Dini, I.; Laneri, S. Nutricosmetics: A brief overview. Phytother. Res. 2019, 33, 3054–3063. [Google Scholar] [CrossRef] [PubMed]
- Spence, L.; Brown, W.J.; Pyne, D.B.; Nissen, M.D.; Sloots, T.P.; McCormack, J.G.; Locke, A.S.; Fricker, P.A. Incidence, etiology, and symptomatology of upper respiratory illness in elite athletes. Med. Sci. Sports Exerc. 2007, 39, 577–586. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Tian, S.; Hu, W.; Niu, L.; Liu, H.; Xu, H.; Xiao, S.Y. Pulmonary pathology of early phase 2019novel coronavirus (COVID-19) pneumonia in two patients with lung cancer. J. Thorac. Oncol. 2020, 15, 700–704. [Google Scholar] [CrossRef]
- Liu, K.; Fang, Y.Y.; Deng, Y.; Liu, W.; Wang, M.F.; Ma, J.P.; Xiao, W.; Wang, Y.N.; Zhong, M.H.; Li, C.H.; et al. Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province. Chin. Med. J. 2020, 133, 1025–1031. [Google Scholar] [CrossRef]
- Walsh, N.P.; Gleeson, M.; Shephard, R.J.; Gleeson, M.; Woods, J.A.; Bishop, N.C.; Fleshner, M.; Green, C.; Pedersen, B.K.; Hoffman-Goetz, L.; et al. Position statement. Part one: Immune function and exercise. Exerc. Immunol. Rev. 2011, 17, 6–63. [Google Scholar]
- Shaw, D.M.; Merien, F.; Braakhuis, A.; Dulson, D. T-cells and their cytokine production: The anti-inflammatory and immunosuppressive effects of strenuous exercise. Cytokine 2017, 104, 136–142. [Google Scholar] [CrossRef]
- Cox, A.J.; Pyne, D.B.; Saunders, P.U.; Callister, R.; Gleeson, M. Cytokine responses to treadmill running in healthy and illness prone athletes. Med. Sci. Sports Exerc. 2007, 39, 1918–1926. [Google Scholar] [CrossRef]
- Cox, A.J.; Gleeson, M.; Pyne, D.B.; Callister, R.; Fricker, P.A.; Scott, R.J. Cytokine gene polymorphisms and risk for upper respiratory symptoms in highly-trained athletes. Exerc. Immunol. Rev. 2010, 16, 8–21. [Google Scholar]
- Zehsaz, F.; Farhangi, N.; Monfaredan, A.; Tabatabaei Seyed, M. IL-10 G-1082A gene polymorphism and susceptibility to upper respiratory tract infection among endurance athletes. J. Sports Med. Phys. Fitness 2015, 55, 128–134. [Google Scholar]
- Vollmer-Conna, U.; Piraino, B.F.; Cameron, B.; Davenport, T.; Hickie, I.; Wakefield, D.; Lloyd, A.R. Cytokine polymorphisms have a synergistic effect on severity of the acute sickness response to infection. Clin. Infect. Dis. 2008, 47, 1418–1425. [Google Scholar] [CrossRef]
- Hull, J.H.; Loosemore, M.; Schwellnus, M. Respiratory health in athletes: Facing the COVID-19 challenge. Lancet Respir. Med. 2020, 8, 557–558. [Google Scholar] [CrossRef]
- Driggin, E.; Madhavan, M.V.; Bikdeli, B.; Chuich, T.; Laracy, J.; Bondi-Zoccai, G.; Brown, T.S.; Der Nigoghossian, C.; Zidar, D.A.; Haythe, J.; et al. Cardiovascular Considerations for Patients, Health Care Workers, and Health Systems During the Coronavirus Disease 2019 (COVID-19) Pandemic. J. Am. Coll. Cardiol. 2020, 75, 2352–2371. [Google Scholar] [CrossRef]
- Nishiga, M.; Wang, D.W.; Han, Y.; Lewis, D.B.; Wu, J.C. COVID-19 and cardiovascular disease: From basic mechanisms to clinical perspectives. Nat. Rev. Cardiol. 2020, 17, 543–558. [Google Scholar] [CrossRef]
- Davalgi, S.; Undi, M.; Annadani, R.; Nawaz, A.S. Comparison of Measures adopted to combat COVID 19 Pandemic by different countries in WHO regions. Indian J. Community Health 2020, 32, 288–299. [Google Scholar]
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef]
- Long, B.; Brady, W.J.; Koyfman, A.; Gottlieb, M. Cardiovascular complications in COVID-19. Am. J. Emerg. Med. 2020, 38, 1504–1507. [Google Scholar] [CrossRef]
- Libby, P. The Heart in COVID-19: Primary Target or Secondary Bystander? JACC Basic Transl. Sci. 2020, ì5, 537–542. [Google Scholar] [CrossRef]
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of Cardiac Injury with Mortality in Hospitalized Patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020, 5, 802–810. [Google Scholar] [CrossRef]
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients with Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 811–818. [Google Scholar] [CrossRef]
- Wang, D.; Hu, B.; Hu, C.; Zhu, F.; Liu, X.; Zhang, J.; Wang, B.; Xiang, H.; Cheng, Z.; Xiong, Y.; et al. Clinical Characteristics of 138 Hospitalized Patients with 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA 2020, 323, 1061–1069. [Google Scholar] [CrossRef]
- Li, B.; Yang, J.; Zhao, F.; Zhi, L.; Wang, X.; Liu, L.; Bi, Z.; Zhao, Y. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin. Res. Cardiol. 2020. [Google Scholar] [CrossRef]
- Scudiero, O.; Gentile, L.; Ranieri, A.; Coppola, E.; Di Micco, P.; Mazzaccara, C.; D’alicandro, G.; Leggiero, E.; Frisso, G.; Pastore, L.; et al. Physical Activity and Thrombophilic Risk in a Short Series. J. Blood Med. 2020, 11, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Toresdahl, B.G.; Asif, I.M. Coronavirus Disease 2019 (COVID-19): Considerations for the Competitive Athlete. Sport Health A Multidiscip. Approach 2020, 12, 221–224. [Google Scholar] [CrossRef] [PubMed]
- Parnell, D.; Widdop, P.; Bond, A.; Wilson, R. COVID-19, networks and sport. Manag. Sport Leis. 2020, 2020, 1–7. [Google Scholar] [CrossRef]
- Grabowski, G.; Whiteside, W.K.; Kanwisher, M. Venous thrombosis in athletes. J. Am. Acad. Orthop. Surg. 2013, 21, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Hoch, A.Z.; Lal, S.; Jurva, J.W.; Gutterman, D.D. The female athlete triad and cardiovascular dysfunction. Phys. Med. Rehabil. Clin. N. Am. 2007, 18, 385. [Google Scholar] [CrossRef]
- Zadow, E.K.; Adams, M.J.; Kitic, C.M.; Wu, S.S.X.; Fell, J.W. Acquired and Genetic Thrombotic Risk Factors in the Athlete. Semin. Thromb. Hemost. 2018, 44, 723–733. [Google Scholar]
- Řádek, M.; Babuňková, E.; Špaček, M.; Kvasnička, T.; Kvasnička, J. Determination of Circulating Endothelial Cells and Endothelial Progenitor Cells Using Multicolor Flow Cytometry in Patients with Thrombophilia. Acta Haematol. 2019, 142, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Hilberg, T.; Jeschke, D.; Gabriel, H.H. Hereditary thrombophilia in elite athletes. Med. Sci. Sports Exerc. 2002, 34, 218–221. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.; Sale, C.; Cooper, S.B.; Elliott-Sale, K.J. Period Prevalence and Perceived Side Effects of Hormonal Contraceptive Use and the Menstrual Cycle in Elite Athletes. Int. J. Sports Physiol. Perform. 2018, 13, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Mao, L.; Nassis, G.P.; Harmer, P.; Ainsworth, B.E.; Li, F. Coronavirus disease (COVID-19): The need to maintain regular physical activity while taking precautions. J. Sport Health Sci. 2020, 9, 103–104. [Google Scholar] [CrossRef] [PubMed]
- Handelsman, D.J.; Hirschberg, A.L.; Bermon, S. Circulating Testosterone as the Hormonal Basis of Sex Differences in Athletic Performance. Endocr. Rev. 2018, 39, 803–829. [Google Scholar] [CrossRef]
- Freedman, J.; Glueck, C.J.; Prince, M.; Riaz, R.; Wang, P. Testosterone, thrombophilia, thrombosis. Transl. Res. 2015, 165, 537–548. [Google Scholar] [CrossRef]
- Ranieri, A.; Benetti, E.; Tita, R.; Spiga, O.; Ciolfi, A.; Birolo, G.; Bruselles, A.; Doddato, G.; Giliberti, A.; Marcomni, C.; et al. ACE2 variants underlie inter-individual variability and susceptibility to COVID-19 in Italian population. medRxiv 2020. [Google Scholar] [CrossRef]
- Russo, R.; Andolfo, I.; Lasorsa, V.A.; Iolascon, A.; Capasso, M. Genetic analysis of the novel SARS-CoV-2 host receptor TMPRSS2 in different populations. bioRxiv 2020. [Google Scholar] [CrossRef]
- Laneri, S.; Di Ronza, C.; Bernardi, A.; Ostacolo, C.; Sacchi, A.; Cervone, C.; D’Amico, M.; Di Filippo, C.; Trincavelli, M.L.; Panighini, A.; et al. Synthesis and Antihypertensive Action of New Imidazo [1,2-a] pyridine Derivatives, non Peptidic Angiotensin II Receptor Antagonists. Cardiovasc. Haematol. Disord. -Drug Targets 2011, 11, 87–96. [Google Scholar] [CrossRef]
- Zheng, H.; Cao, J.J. Angiotensin-Converting Enzyme Gene Polymorphism and Severe Lung Injury in Patients with Coronavirus Disease 2019. Am. J. Pathol. 2020, 190, 2013–2017. [Google Scholar] [CrossRef]
- Delanghe, J.R.; Speeckaert, M.M.; De Buyzere, M.L. The host’s angiotensin-converting enzyme polymorphism may explain epidemiological findings in COVID-19 infections. Clin. Chim. Acta 2020, 505, 192–193. [Google Scholar] [CrossRef] [PubMed]
- Delanghe, J.R.; Speeckaert, M.M.; Marc, L. COVID-19 infections are also affected by human ACE1 D/I polymorphism. Clin. Chem. Lab Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.; Davide, J.K.; Maden, S.K.; Wood, M.A.; Weeder, B.J.; Nellore, A.; Thompson, R.T. Human leukocyte antigen susceptibility map for SARS-CoV-2. J. Virol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Eutimio, M.A.; Lopez-Macias, C.; Pastelin-Palacios, R. Bioinformatic analysis and identification of single-stranded RNA sequences recognized by TLR7/8 in the SARS-CoV-2, SARS-CoV, and MERS-CoV genomes. Microbes Infect. 2020, 22, 226–229. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, Y.; Huang, H.; Li, D.; Gu, D.; Lu, X.; Zhang, Z.; Liu, L.; Liu, T.; Liu, Y.; et al. Relationship between the ABO Blood Group and the COVID-19 Susceptibility. medRxiv 2020. [Google Scholar] [CrossRef]
- Eynon, N.; Ruiz, J.R.; Oliveira, J.; Duarte, J.A.; Birk, R.; Lucia, A. Genes and elite athletes: A roadmap for future research. J. Physiol. 2011, 589 Pt 13, 3063–3307. [Google Scholar] [CrossRef]
- Ahmetov, I.I.; Fedotovskaya, O.N. Current Progress in Sports Genomics. Adv. Clin. Chem. 2015, 70, 247–314. [Google Scholar]
- Zheng, Q.; Cui, G.; Chen, J.; Gao, H.; Wei, Y.; Uede, T.; Chen, Z.; Diao, H. Regular Exercise Enhances the Immune Response Against Microbial Antigens Through Up-Regulation of Toll-like Receptor Signaling Pathways. Cell Physiol. Biochem. 2015, 37, 735–746. [Google Scholar] [CrossRef]
- Gleeson, M.; Mc Farlin, B.; Flynn, M. Exercise and Toll-like receptors. Exerc. Immunol. Rev. 2006, 12, 34–53. [Google Scholar]
- Lippi, G.; Gandini, G.; Salvagno, G.L.; Skafidas, S.; Festa, L.; Danese, E.; Montagnana, M.; Sanchis-Gomar, F.; Tarperi, C.; Schena, F. Influence of ABO blood group on sports performance. Ann. Transl. Med. 2017, 5, 255. [Google Scholar] [CrossRef]
- Wong, A.Y.-Y.; Lin, S.K.-K.; Louie, L.H.-T.; Law, G.Y.-K.; So, R.C.-H.; Lee, D.C.-W.; Yau, F.C.-F.; Yung, S.-H. Impact of the COVID-19 pandemic on sports and exercise. Asia Pac. J. Sports Med. Arthrosc. Rehabil. Technol. 2020, 22, 39–44. [Google Scholar] [CrossRef] [PubMed]
- BBC Sport. Coronavirus: How the Virus has Impacted Sporting Events around the World. 2020. Available online: https://www.bbc.com/sport/51605235 (accessed on 12 June 2020).
- Samuel, D.R.; Tenenbaum, G.; Galily, Y. The 2020 Coronavirus Pandemic as a Change-Evet in Sport Performers’ Careers: Conceptual and Applied Practice Considerations. Front. Psychol. 2020, 11, 56796. [Google Scholar] [CrossRef] [PubMed]
- Pillay, L.; van Rensburg, D.C.C.J.; van Rensburg, A.J.; Ramagole, D.A.; Holtzhausen, L.; Dijkstra, H.P.; Cronje, T. Nowhere to hide: The significant impact of coronavirus disease 2019 (COVID-19) measures on elite and semi-elite South African athletes. J. Sci. Med. Sport 2020, 23, 670–679. [Google Scholar] [CrossRef]
- Rajpal, S.; Tong, M.S.; Borchers, J.; Zareba, K.M.; Obarski, T.P.; Simonetti, O.P.; Daniels, C.J. Cardiovascular Magnetic Resonance Findings in Competitive Athletes Recovering From COVID-19 Infection. JAMA Cardiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Verwoert, G.C.; de Vries, S.T.; Bijsterveld, N.; Willems, A.R.; vd Borgh, R.; Jongman, J.K.; Kemps, H.M.; Snoek, J.A.; Rienks, R.; Jorstad, H.T. Return to sports after COVID-19: A position paper from the Dutch Sports Cardiology Section of the Netherlands Society of Cardiology. Neth. Heart J. 2020, 28, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Shaukat, N.; Ali, D.M.; Razzak, J. Physical and mental health impacts of COVID-19 on healthcare workers: A scoping review. Int. J. Emerg. Med. 2020, 13, 40. [Google Scholar] [CrossRef]
- Troosters, T. Managing the Respiratory Care of Patients with COVID-19: Italian Recommendations; European Respiratory Society: Lausanne, Switzerland, 2020. [Google Scholar]
- Balbi, B.; Berney, S.; Brooks, D. Report of an Ad-Hoc International Task Force to Develop an Expert-Based Opinion on Early and Short-Term Rehabilitative Interventions; European Respiratory Society: Lausanne, Switzerland, 2020. [Google Scholar]
- Boldrini, P.; Bernetti, A.; Fiore, P. SIMFER Executive Committee and SIMFER Committee for international affairs. Impact of COVID-19 outbreak on rehabilitation services and Physical and Rehabilitation Medicine (PRM) physicians’ activities in Italy. Eur. J. Phys. Rehabil. Med. 2020, 10, S1973-9087. [Google Scholar]
- Ragozzino, E.; Brancaccio, M.; Di Costanzo, A.; Scalabri, F.; Andolfi, G.; Wanderlingh, L.G.; Patriarca, E.J.; Minchiotti, G.; Altamura, S.; Varrone, F.; et al. 6-Bromoindirubin- 3’-oxime intercepts GSK3 signaling to promote and enhance skeletal muscle differentiation affecting miR-206 expression in mice. Sci. Rep. 2019, 9, 18091. [Google Scholar] [CrossRef]
- Gaspersz, R.; Lamers, F.; Wittenberg, G.; Beekman, A.T.; van Hemert, A.M.; Schoevers, R.A.; Penninx, B.W. The role of anxious distress in immune dysregulation in patients with major depressive disorder. Transl. Psychiatry 2017, 7, 1268. [Google Scholar] [CrossRef]
- Hogan, C.L.; Mata, J.; Carstensen, L.L. Exercise Holds Immediate Benefits for Affect and Cognition in Younger and Older Adults. Psychol. Aging 2013, 28, 587–594. [Google Scholar] [CrossRef]
- Asmundson, G.J.G.; Fetzner, M.G.; DeBoer, L.B.; Powers, M.B.; Otto, M.W.; Smits, J.A.J. Let’s Get Physical: A Contemporary Review of the Anxiolytic Effects of Exercise for Anxiety and Its Disorders. Depress. Anxiety 2013, 30, 362–373. [Google Scholar] [CrossRef]
- AminJafari, A.; Ghasemi, S. The Possible of Immunotherapy for COVID-19: A Systematic Review. Int. Immunopharmacol. 2020, 83, 106455. [Google Scholar] [CrossRef] [PubMed]
- Schijns, V.; Lavelle, E.D. Prevention and Treatment of COVID-19 Disease by Controlled Modulation of Innate Immunity. Eur. J. Immunol. 2020, 50, 932–938. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, Y. Potential Interventions for Novel Coronavirus in China: A Systematic Review. J. Med. Virol. 2020, 92, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Catanzaro, M.; Fagiani, F.; Racchi, M.; Corsini, E.; Govoni, S.; Lanni, C. Immune Response in COVID-19: Addressing a Pharmacological Challenge by Targeting Pathways Triggered by SARS-CoV-2. Signal Transduct. Target. Ther. 2020, 5, 84. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Lewis, E.D.; Pae, M.; Meydani, S.N. Nutritional Modulation of Immune Function: Analysis of Evidence, Mechanisms, and Clinical Relevance. Front. Immunol. 2018, 9, 3160. [Google Scholar] [CrossRef] [PubMed]
| Company | Type | Doses | How Effective | Storage | Cost for Doses |
|---|---|---|---|---|---|
| Oxford Uni-AstraZeneca | Viral vector 8 genetically modified virus) | X2 | 62–90% | 4 °C | $4 |
| Moderna | RNA (part of virus genetic code) | X2 | 95% | −20 °C up to 6 months | $33 |
| Pfizer-BioNTech | RNA | X2 | 95% | −70 °C | $20 |
| Gamaleya (Sputnik V) | Viral vector | X2 | 92% | 4 °C | $10 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scudiero, O.; Lombardo, B.; Brancaccio, M.; Mennitti, C.; Cesaro, A.; Fimiani, F.; Gentile, L.; Moscarella, E.; Amodio, F.; Ranieri, A.; et al. Exercise, Immune System, Nutrition, Respiratory and Cardiovascular Diseases during COVID-19: A Complex Combination. Int. J. Environ. Res. Public Health 2021, 18, 904. https://doi.org/10.3390/ijerph18030904
Scudiero O, Lombardo B, Brancaccio M, Mennitti C, Cesaro A, Fimiani F, Gentile L, Moscarella E, Amodio F, Ranieri A, et al. Exercise, Immune System, Nutrition, Respiratory and Cardiovascular Diseases during COVID-19: A Complex Combination. International Journal of Environmental Research and Public Health. 2021; 18(3):904. https://doi.org/10.3390/ijerph18030904
Chicago/Turabian StyleScudiero, Olga, Barbara Lombardo, Mariarita Brancaccio, Cristina Mennitti, Arturo Cesaro, Fabio Fimiani, Luca Gentile, Elisabetta Moscarella, Federica Amodio, Annaluisa Ranieri, and et al. 2021. "Exercise, Immune System, Nutrition, Respiratory and Cardiovascular Diseases during COVID-19: A Complex Combination" International Journal of Environmental Research and Public Health 18, no. 3: 904. https://doi.org/10.3390/ijerph18030904
APA StyleScudiero, O., Lombardo, B., Brancaccio, M., Mennitti, C., Cesaro, A., Fimiani, F., Gentile, L., Moscarella, E., Amodio, F., Ranieri, A., Gragnano, F., Laneri, S., Mazzaccara, C., Di Micco, P., Caiazza, M., D’Alicandro, G., Limongelli, G., Calabrò, P., Pero, R., & Frisso, G. (2021). Exercise, Immune System, Nutrition, Respiratory and Cardiovascular Diseases during COVID-19: A Complex Combination. International Journal of Environmental Research and Public Health, 18(3), 904. https://doi.org/10.3390/ijerph18030904

















