The Effect of Lifestyle Intervention on Health-Related Quality of Life in Adults with Metabolic Syndrome: A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Overview
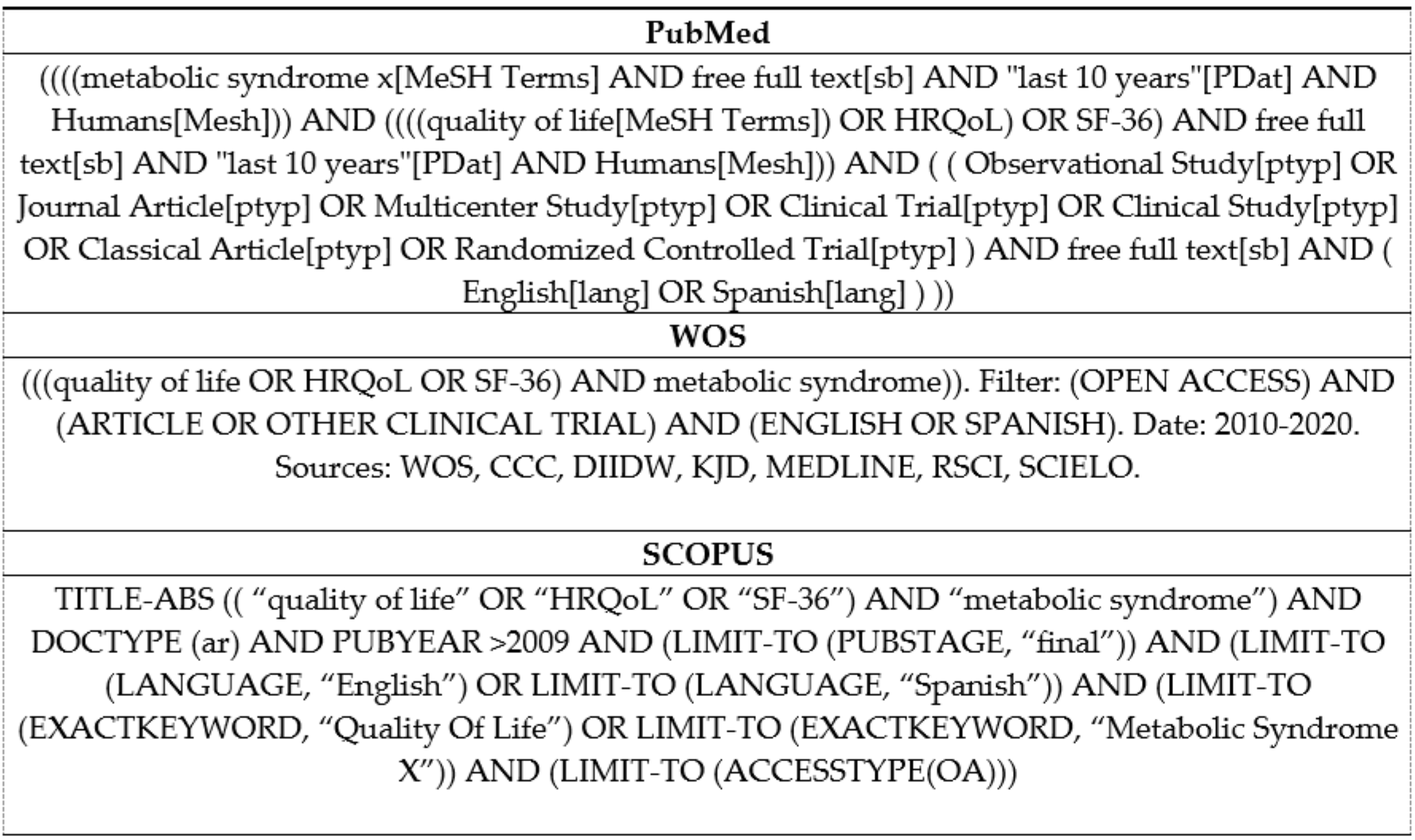
2.2. Data Sources and Searches
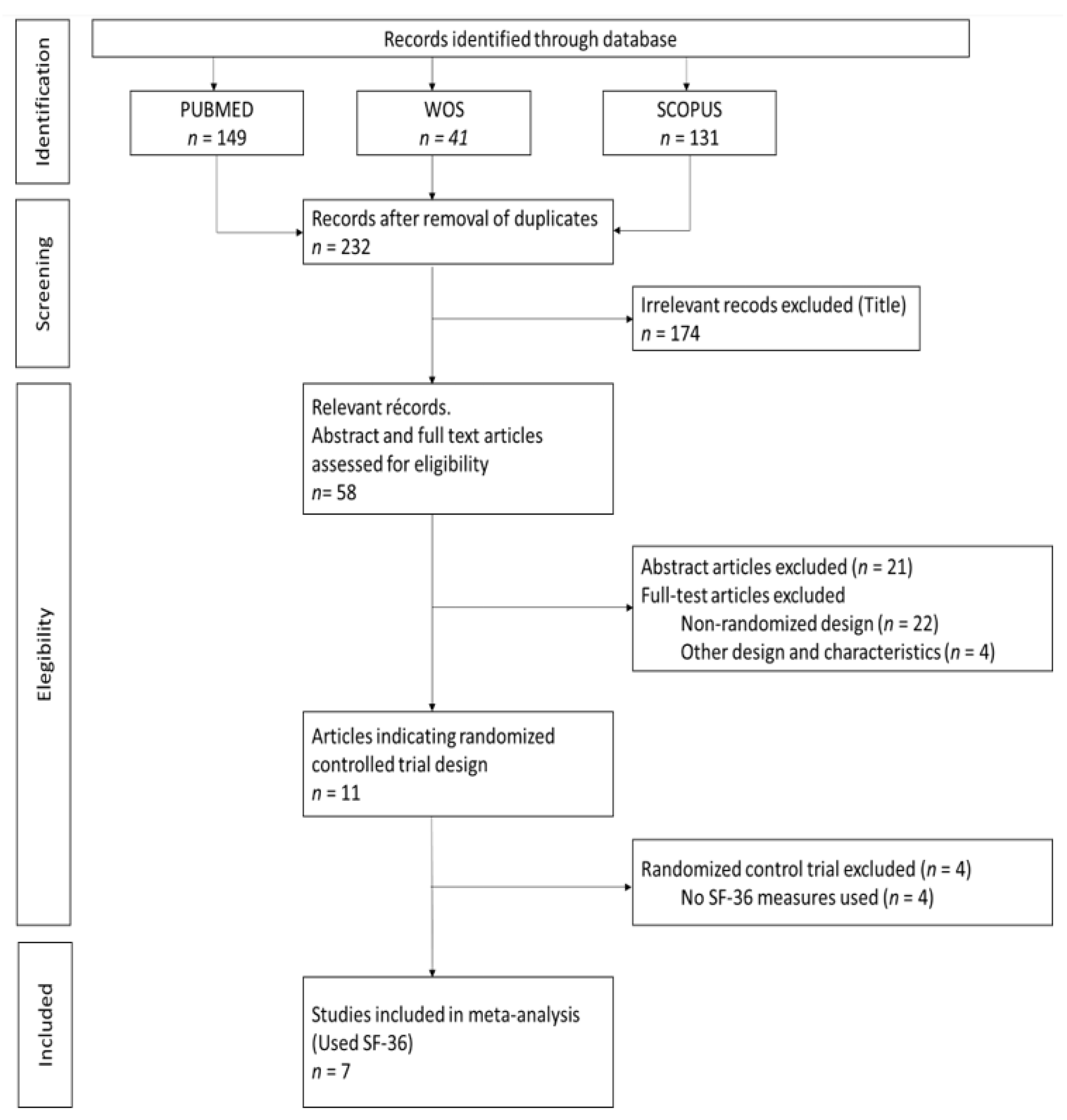
2.3. Study Selection
2.4. Data Extraction
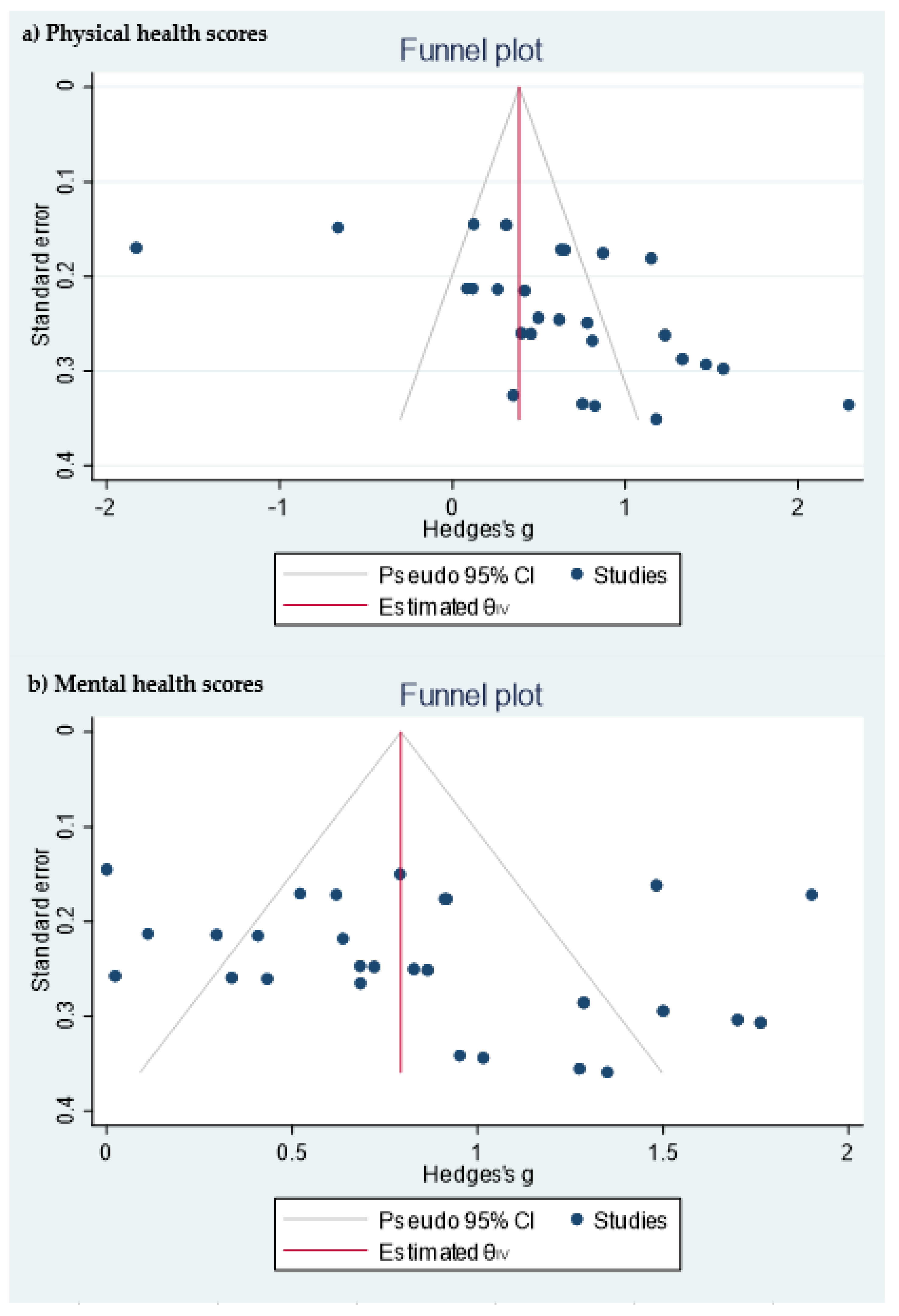
2.5. Meta-Analysis
3. Results
3.1. Description of Studies
3.2. Study Quality and Risk of Bias
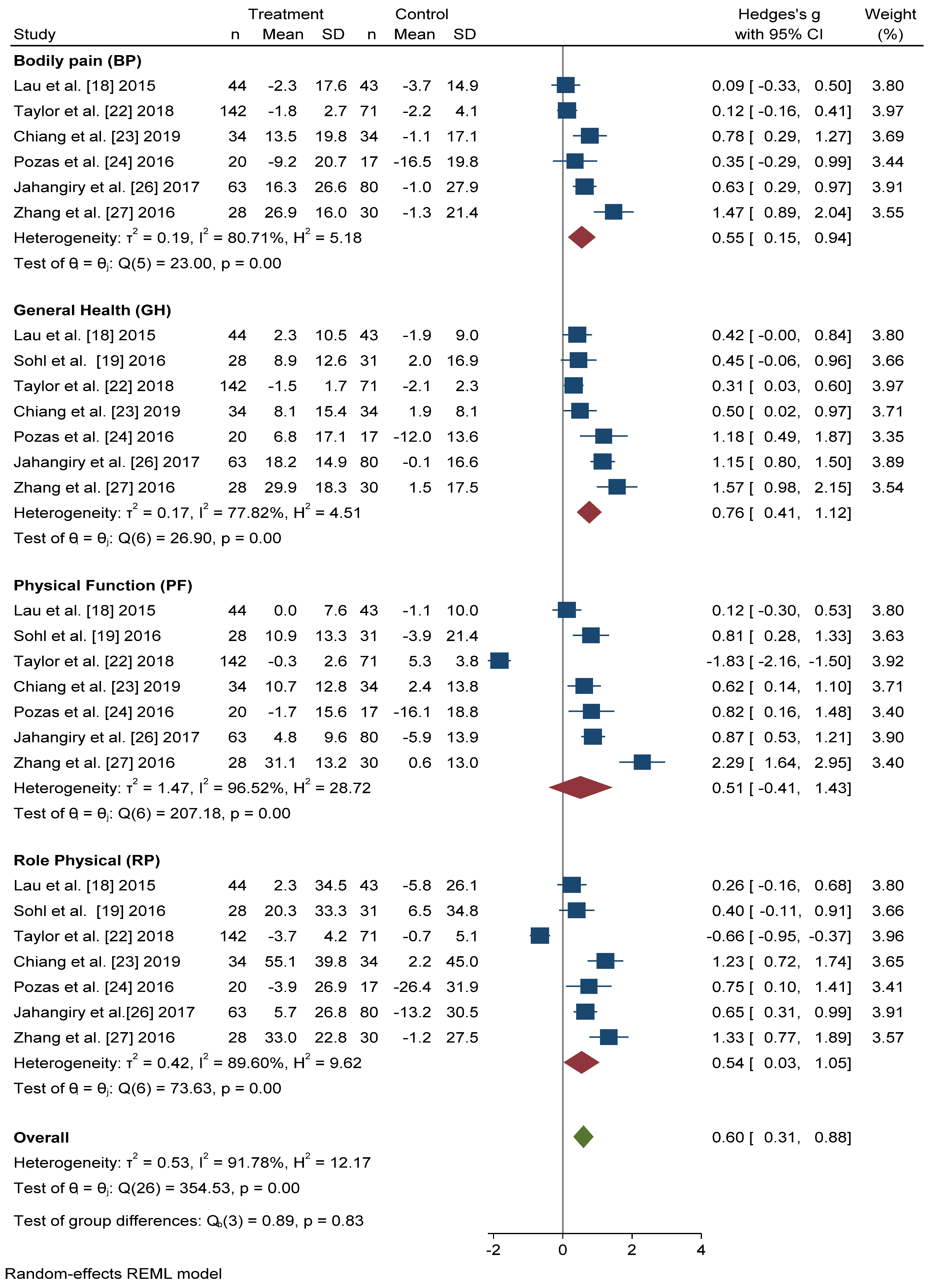
3.3. Physical Health-Related Quality of Life
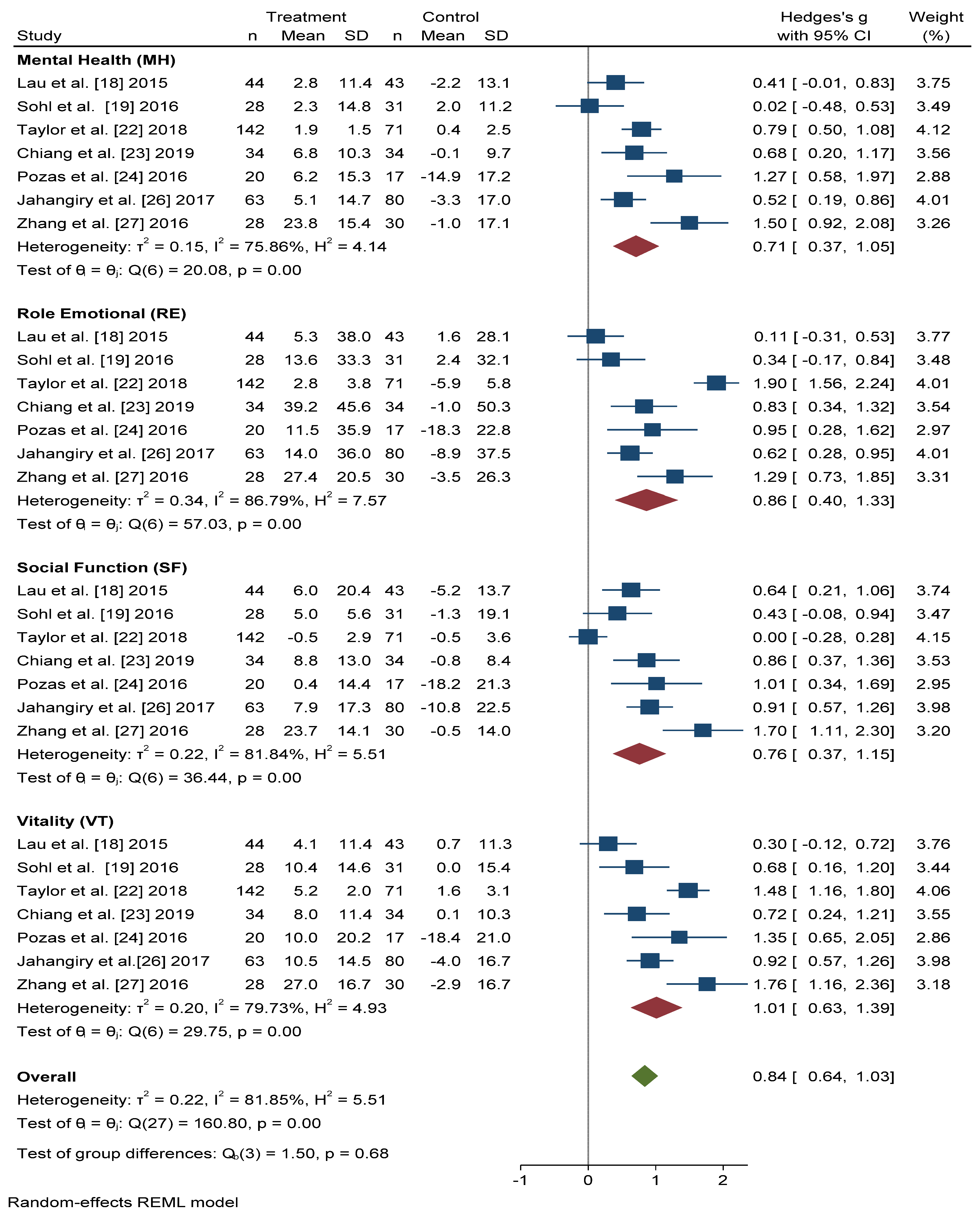
3.4. Mental Health-Related Quality of Life
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- WHO Causes of Death. Available online: https://www.who.int/data/gho/data/themes/topics/causes-of-death/GHO/causes-of-death (accessed on 15 October 2020).
- Elabbassi, W.N.; Haddad, H.A. The epidemic of the metabolic syndrome. Saudi Med. J. 2005, 26, 373–375. [Google Scholar]
- Parikh, R.; Mohan, V. Changing definitions of metabolic syndrome. Indian J. Endocrinol. Metab. 2012, 16, 7. [Google Scholar]
- Maloberti, A.; Bruno, R.M.; Facchetti, R.; Grassi, G.; Taddei, S.; Ghiadoni, L.; Giannattasio, C. THE role of metabolic syndrome in blood pressure control and pulse wave velocity progression over a 3.5 years in treated hypertensive PATIENTS. Eur. J. Intern. Med. 2020, 76, 107–109. [Google Scholar]
- Sigit, F.S.; Tahapary, D.L.; Trompet, S.; Sartono, E.; Van Dijk, K.W.; Rosendaal, F.R.; De Mutsert, R. The prevalence of metabolic syndrome and its association with body fat distribution in middle-aged individuals from Indonesia and the Netherlands: A cross-sectional analysis of two population-based studies. Diabetol. Metab. Syndr. 2020, 12, 1–11. [Google Scholar]
- Virani, S.S.; Alonso, A.; Benjamin, E.J.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Delling, F.N.; et al. Heart Disease and Stroke Statistics—2020 Update: A Report from the American Heart Association. Circulation 2020, 141, e139–e590. [Google Scholar]
- Shin, D.; Kongpakpaisarn, K.; Bohra, C. Trends in the prevalence of metabolic syndrome and its components in the United States 2007–2014. Int. J. Cardiol. 2018, 259, 216–219. [Google Scholar]
- Ceitlin, J.; Whitsel, L.; Romero, E.; Labarthe, D.; Pechacek, T.; Cardiovascular, M.; Program, H.; Richards, H.; Shanedling, S.B.; Foell, K.; et al. Best Practices for Cardiovascular Disease Prevention Programs A Guide to Effective Health Care System Interventions and Community Programs Linked to Clinical Services Suggested Citation. 2017. Available online: https://www.cdc.gov/dhdsp/pubs/docs/Best-Practices-Guide-508.pdf (accessed on 20 January 2021).
- Vetter, M.L.; Wadden, T.A.; Lavenberg, J.; Moore, R.H.; Volger, S.; Perez, J.L.; Sarwer, D.B.; Tsai, A.G. Relation of health-related quality of life to metabolic syndrome, obesity, depression and comorbid illnesses. Int. J. Obes. 2011, 35, 1087–1094. [Google Scholar]
- Barcones-Molero, M.F.; Sánchez-Villegas, A.; Martínez-González, M.A.; Bes-Rastrollo, M.; Martínez-Urbistondo, M.; Santabárbara, J.; Martínez, J.A. The influence of obesity and weight gain on quality of life according to the SF-36 for individuals of the dynamic follow-up cohort of the University of Navarra. Rev. Clínica Española 2018, 218, 408–416. [Google Scholar]
- Warkentin, L.M.; Das, D.; Majumdar, S.R.; Johnson, J.A.; Padwal, R.S. The effect of weight loss on health-related quality of life: Systematic review and meta-analysis of randomized trials. Obes. Rev. 2014, 15, 169–182. [Google Scholar]
- Kim, J.R.; Kim, H.N.; Song, S.W. Associations among inflammation, mental health, and quality of life in adults with metabolic syndrome. Diabetol. Metab. Syndr. 2018, 10, 4–11. [Google Scholar]
- Amiri, P.; Hosseinpanah, F.; Jalali-Farahani, S.; Mehrabi, Y.; Montazeri, A.; Azizi, F. Is persistence of metabolic syndrome associated with poor health-related quality of life in non-diabetic Iranian adults? Tehran lipid and glucose study. J. Diabetes Investig. 2014, 5, 687–693. [Google Scholar]
- Lee, Y.J.; Woo, S.Y.; Ahn, J.H.; Cho, S.; Kim, S.R. Health-related quality of life in adults with metabolic syndrome: The Korea National Health and Nutrition Examination Survey, 2007–2008. Ann. Nutr. Metab. 2012, 61, 275–280. [Google Scholar]
- Ware, J.E. SF-36 Health Survey update. Spine (Phila. Pa. 1976) 2000, 25, 3130–3139. [Google Scholar]
- McHorney, C.A.; Ware John, E.J.; Lu, J.F.R.; Sherbourne, C.D. The MOS 36-Item Short-Form Health Survey (SF-36): III. Tests of Data Quality, Scaling Assumptions, and Reliability across Diverse Patient Groups. Med. Care 1994, 32, 40–66. [Google Scholar]
- Ware, J.E.; Sherbourne, C.D. The MOS 36-item short-form health survey (Sf-36): I. conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar]
- Lau, C.; Yu, R.; Woo, J. Effects of a 12-Week Hatha Yoga Intervention on Metabolic Risk and Quality of Life in Hong Kong Chinese Adults with and without Metabolic Syndrome. PLoS ONE 2015, 10, e0130731. [Google Scholar]
- Sohl, S.J.; Wallston, K.A.; Watkins, K.; Birdee, G.S. Yoga for Risk Reduction of Metabolic Syndrome: Patient-Reported Outcomes from a Randomized Controlled Pilot Study. Evid. Based Complement. Altern. Med. 2016, 2016, 3094589. [Google Scholar] [CrossRef]
- Carvalho-lima, R.P.; Sá-caputo, D.C.; Moreira-marconi, E.; Paineiras-domingos, L.L.; Sousa-gonçalves, C.R.; Soares, D.; Frederico, E.H.; Neves, M.F.; Oliveira, R.; Oigman, W.; et al. Quality of life of patients with metabolic syndrome is improved after whole body vibration exercises. Afr. J. Tradit. Complement. Altern. Med. 2017, 14, 59–65. [Google Scholar]
- Sarwer, D.B.; Moore, R.H.; Diewald, L.K.; Chittams, J.; Berkowitz, R.I.; Vetter, M.; Volger, S.; Wadden, T.A. The impact of a primary care-based weight loss intervention on the quality of life. Int. J. Obes. 2013, 37, S25–S30. [Google Scholar]
- Taylor, T.R.; Dash, C.; Sheppard, V.; Makambi, K.; Ma, X.; Adams-Campbell, L.L. The effect of a randomized controlled physical activity trial on health related quality of life in metabolically unhealthy African-American women: Fierce study. Contemp. Clin. Trials 2018, 67, 121–128. [Google Scholar]
- Chiang, L.C.; Heitkemper, M.M.L.; Chiang, S.L.; Tzeng, W.C.; Lee, M.S.; Hung, Y.J.; Lin, C.H. Motivational Counseling to Reduce Sedentary Behaviors and Depressive Symptoms and Improve Health-Related Quality of Life among Women with Metabolic Syndrome. J. Cardiovasc. Nurs. 2019, 34, 327–335. [Google Scholar]
- Saboya, P.P.; Bodanese, L.C.; Zimmermann, P.R.; Da Silva Gustavo, A.; Macagnan, F.E.; Feoli, A.P.; Da Silva Oliveira, M. Lifestyle intervention on metabolic syndrome and its impact on quality of life: A randomized controlled trial. Arq. Bras. Cardiol. 2017, 108, 60–69. [Google Scholar]
- Prasanth, N.V.; Najiya Saheer, V.; Naina Paul, M.; Najla, P.K.; Nizar Mohamed, K.P. A study on impact of patient counseling and education on quality of life in patients with metabolic syndrome. Clin. Epidemiol. Glob. Heal. 2019, 7, 357–362. [Google Scholar]
- Jahangiry, L.; Montazeri, A.; Najafi, M.; Yaseri, M.; Farhangi, M.A. An interactive web-based intervention on nutritional status, physical activity and health-related quality of life in patient with metabolic syndrome: A randomized-controlled trial (The Red Ruby Study). Nutr. Diabetes 2017, 7, e240. [Google Scholar]
- Zhang, Y.; Mei, S.; Yang, R.; Chen, L.; Gao, H.; Li, L. Effects of lifestyle intervention using patient-centered cognitive behavioral therapy among patients with cardio-metabolic syndrome: A randomized, controlled trial. BMC Cardiovasc. Disord. 2016, 16, 1–9. [Google Scholar]
- Fanning, J.; Walkup, M.P.; Ambrosius, W.T.; Brawley, L.R.; Ip, E.H.; Marsh, A.P.; Rejeski, W.J. Change in health-related quality of life and social cognitive outcomes in obese, older adults in a randomized controlled weight loss trial: Does physical activity behavior matter? J. Behav. Med. 2018, 41, 299–308. [Google Scholar]
- Saboya, P.P.; Bodanese, L.C.; Zimmermann, P.R.; da Silva Gustavo, A.; Assumpção, C.M.; Londero, F. Metabolic syndrome and quality of life: A systematic review. Rev. Lat. Am. Enferm. 2016, 24, e2848. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Green, S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0; Wiley: Hoboken, NJ, USA, 2008; part 2. [Google Scholar]
- Hedges, L.V.; Olkin, I. Statistical Methods for Meta-Analysis; Academic Press: Cambridge, MA, USA, 1985. [Google Scholar]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. Identifying and Quantifying Heterogeneity. Introd. Meta Anal. 2009, 16, 107–125. [Google Scholar]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. Br. Med. J. 1997, 315, 629–634. [Google Scholar]
- Galilea-Zabalza, I.; Buil-Cosiales, P.; Salas-Salvadó, J.; Toledo, E.; Ortega-Azorín, C.; Díez-Espino, J.; Vázquez-Ruiz, Z.; Zomeño, M.D.; Vioque, J.; Martínez, J.A.; et al. Mediterranean diet and quality of life: Baseline cross-sectional analysis of the PREDIMED-PLUS trial. PLoS ONE 2018, 13, e0198974. [Google Scholar]
- Daimiel, L.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Schröder, H.; Vioque, J.; Romaguera, D.; Martínez, J.A.; Wärnberg, J.; Lopez-Miranda, J.; et al. Physical fitness and physical activity association with cognitive function and quality of life: Baseline cross-sectional analysis of the PREDIMED-Plus trial. Sci. Rep. 2020, 10, 1–12. [Google Scholar]
- Marcos-Delgado, A.; López-García, E.; Martínez-González, M.A.; Salas-Salvadó, J.; Corella, D.; Fitó, M.; Romaguera, D.; Vioque, J.; Alonso-Gómez, A.M.; Wärnberg, J.; et al. Health-related quality of life in individuals with metabolic syndrome: A cross-sectional study. SEMERGEN 2020, 46, 524–537. [Google Scholar]
- Kolotkin, R.L.; Andersen, J.R. A systematic review of reviews: Exploring the relationship between obesity, weight loss and health-related quality of life. Clin. Obes. 2017, 7, 273–289. [Google Scholar]
- Salas-Salvadó, J.; Díaz-López, A.; Ruiz-Canela, M.; Basora, J.; Fitó, M.; Corella, D.; Serra-Majem, L.; Wärnberg, J.; Romaguera, D.; Estruch, R.; et al. Effect of a Lifestyle Intervention Program With Energy-Restricted Mediterranean Diet and Exercise on Weight Loss and Cardiovascular Risk Factors: One-Year Results of the PREDIMED-Plus Trial. Diabetes Care 2018, 42, 777–788. [Google Scholar]
- Slagter, S.N.; van Vliet-Ostaptchouk, J.V.; van Beek, A.P.; Keers, J.C.; Lutgers, H.L.; van der Klauw, M.M.; Wolffenbuttel, B.H.R. Health-Related Quality of Life in Relation to Obesity Grade, Type 2 Diabetes, Metabolic Syndrome and Inflammation. PLoS ONE 2015, 10, e0140599. [Google Scholar]
- Amiri, P.; Deihim, T.; Nakhoda, K.; Hasheminia, M.; Montazeri, A.; Azizi, F. Metabolic syndrome and health-related quality of life in reproductive age and post-menopausal women: Tehran lipid and glucose study. Arch. Iran. Med. 2014, 17, 423–428. [Google Scholar]
- Roohafza, H.; Sadeghi, M.; Talaei, M.; Pourmoghaddas, Z.; Sarrafzadegan, N. Psychological status and quality of life in relation to the metabolic syndrome: Isfahan cohort study. Int. J. Endocrinol. 2012, 2012, 10–15. [Google Scholar]
| Source | Sequence Generation | Allocation Concealment | Blinding | Incomplete Outcome Data | Selective Outcome Reporting | Risk of Bias |
|---|---|---|---|---|---|---|
| Lau et al. 2015 [18] | No information | Yes | No | No | No | Low risk of bias |
| Sohl et al. 2016 [19] | Yes | Yes | Yes | No | No | Low risk of bias |
| Carvalho-Lima et al. 2017 [20] | No | No | No | Yes | Yes | High risk of bias |
| Sarwer et al. 2013 [21] | Yes | No information | No information | No | No | Low risk of bias |
| Taylor et al. 2018 [22] | Yes | No information | No | No | No | Low risk of bias |
| Chiang et al. 2019 [23] | Yes | Yes | Yes | No | No | Low risk of bias |
| Saboya et al. 2016 [24] | Yes | Yes | No information | No | No | Low risk of bias |
| Prasanth et al. 2018 [25] | Yes | No information | No information | Yes | Yes | High risk of bias |
| Jahangiry et al. 2017 [26] | Yes | No information | No information | No | No | Low risk of bias |
| Zhang et al. 2016 [27] | Yes | No | No | No | No | Low risk of bias |
| Fanning et al. 2018 [28] | Yes | Yes | Yes | No | Yes | Low risk of bias |
| Reference and Country | Intervention(s) | Control Treatment | Study Duration (Weeks) | % Female (Total) | MetS Criteria | Mean Age (Total), Years | Sample Size |
|---|---|---|---|---|---|---|---|
| Lau et al. [18] 2015 (China) | Yoga training consisting of 12 once-weekly, 60-min sessions (n = 44) | Maintain their routine activities and not begin any exercise (n = 43) | 12 | 63 | NCEP-ATP III | 52.0 (7.46) | 87 |
| Sohl et al. [19] 2016 (USA) | Yoga and education (n = 26) | Education only (n = 33) | 12 | 51 | Standard MetS Criteria | 58.0 (10) | 59 |
| Taylor et al. [22] 2018 (USA) | Supervised facility-based exercise intervention (n = 73) Home-based exercise intervention (n = 69) | Control group: Maintain their current daily activities and exercise habits (n = 71) | 24 | 100 | Standard MetS Criteria | 58.3 | 213 |
| Chiang et al. [23] 2019 (Taiwan) | Intervention group (IG) (n = 34) participants were given individually tailored, 12-week, telephone-based motivational counseling for modifying lifestyles. The other group (n = 32) received an educational brochure about lifestyle modification and coping with stress. | Control group only underwent routine outpatient clinical follow-up (n = 34) | 12 | 100 | NCEP-ATP III | IG: 63.1 (8.5) CG: 63.8 (7.3) | Total: 115 Groups included in the meta-analysis (CG and IG): 68 |
| Saboya et al. [24] 2016 (Brazil) | Individual Intervention group (IG) (n = 28) participated in weekly individual appointments with psychology and nutrition teams and exercised regularly and the other group (n = 25) worked the change in lifestyle through the discussion of pre-defined themes of health education | Control group was the non-pharmacological intervention recommended by the main guidelines for the clinical management of MetS (n = 19) | 36 | 55.5 | Standard MetS Criteria | CG: 52.1 (7.2) IG: 51.6 (5.6) | Total: 72 Groups included in the meta-analysis (CG and IG): 37 |
| Jahangiry et al. [26] 2017 (Iran) | Interactive lifestyle intervention with Healthy Heart Profile on nutrition, and physical activity (n = 63) | Sending e-mails every 3 weeks to visit the study website and read general information on nutrition and physical activity (n = 80) | 24 | 33.7 | NCEP-ATP III | 44.2 (10.0) | 143 |
| Zhang et al. [27] 2016 (China) | Patient-centered cognitive behavioral therapy (PC-CBT) lifestyle intervention | Control group received a letter explaining basic lifestyle advice and general information about MetS risk factors (n = 30) | 12 | 56.9 | IDF 2005 | 48.6 (5.8) | 58 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcos-Delgado, A.; Hernández-Segura, N.; Fernández-Villa, T.; Molina, A.J.; Martín, V. The Effect of Lifestyle Intervention on Health-Related Quality of Life in Adults with Metabolic Syndrome: A Meta-Analysis. Int. J. Environ. Res. Public Health 2021, 18, 887. https://doi.org/10.3390/ijerph18030887
Marcos-Delgado A, Hernández-Segura N, Fernández-Villa T, Molina AJ, Martín V. The Effect of Lifestyle Intervention on Health-Related Quality of Life in Adults with Metabolic Syndrome: A Meta-Analysis. International Journal of Environmental Research and Public Health. 2021; 18(3):887. https://doi.org/10.3390/ijerph18030887
Chicago/Turabian StyleMarcos-Delgado, Alba, Natalia Hernández-Segura, Tania Fernández-Villa, Antonio J. Molina, and Vicente Martín. 2021. "The Effect of Lifestyle Intervention on Health-Related Quality of Life in Adults with Metabolic Syndrome: A Meta-Analysis" International Journal of Environmental Research and Public Health 18, no. 3: 887. https://doi.org/10.3390/ijerph18030887
APA StyleMarcos-Delgado, A., Hernández-Segura, N., Fernández-Villa, T., Molina, A. J., & Martín, V. (2021). The Effect of Lifestyle Intervention on Health-Related Quality of Life in Adults with Metabolic Syndrome: A Meta-Analysis. International Journal of Environmental Research and Public Health, 18(3), 887. https://doi.org/10.3390/ijerph18030887








