Metabolic Syndrome and Endocrine Disrupting Chemicals: An Overview of Exposure and Health Effects
Abstract
:1. Introduction
2. Methods
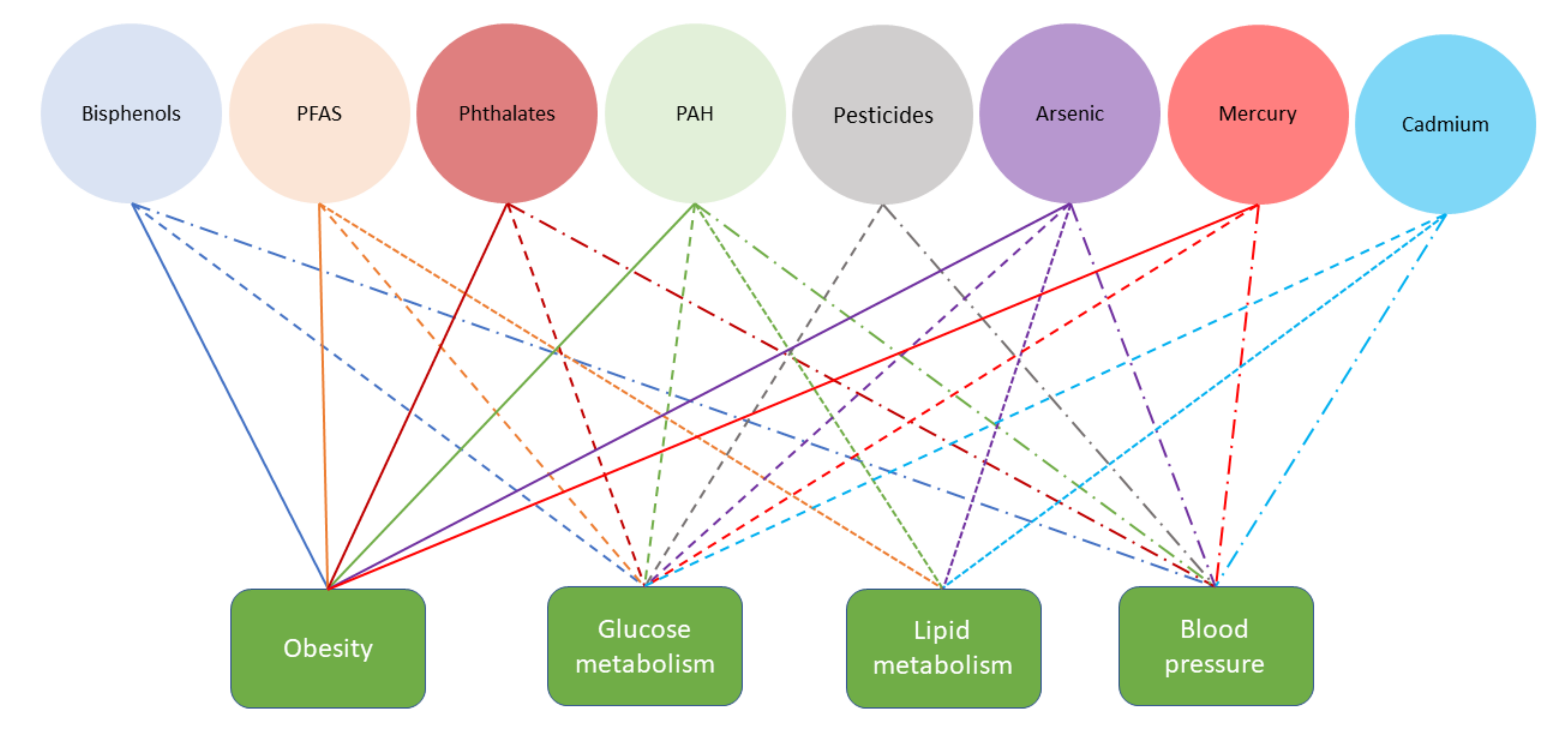
3. Results
3.1. Bisphenols
3.2. Per- and Polyfluoroalkyl Substances
3.3. Phthalates
3.4. Polycyclic Aromatic Hydrocarbons (PAH)
3.5. Pesticides
3.6. Heavy Metals
3.6.1. Arsenic (As)
3.6.2. Mercury (Hg)
3.6.3. Cadmium (Cd)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Noncommunicable Diseases; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/health-topics/noncommunicable-diseases#tab=tab_1 (accessed on 1 December 2021).
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.; James, W.P.T.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [PubMed] [Green Version]
- Qiao, Q.; D.S. Group. Comparison of different definitions of the metabolic syndrome in relation to cardiovascular mortality in European men and women. Diabetologia 2006, 49, 2837–2846. [Google Scholar] [PubMed]
- Kassi, E.; Pervanidou, P.; Kaltsas, G.; Chrousos, G. Metabolic syndrome: Definitions and controversies. BMC Med. 2011, 9, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Neill, S.; O’Driscoll, L. Metabolic syndrome: A closer look at the growing epidemic and its associated pathologies. Obes. Rev. 2015, 16, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Binetti, R.; Costamagna, F.M.; Marcello, I. Exponential growth of new chemicals and evolution of information relevant to risk control. Annali dell’Istituto Supeiore di Sanità 2008, 44, 13–15. [Google Scholar]
- Tudurí, E.; Marroqui, L.; Dos Santos, R.S.; Quesada, I.; Fuentes, E.; Alonso-Magdalena, P. Timing of Exposure and Bisphenol-A: Implications for Diabetes Development. Front. Endocrinol. 2018, 9, 648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ndaw, S.; Remy, A.; Jargot, D.; Robert, A. Occupational exposure of cashiers to Bisphenol A via thermal paper: Urinary biomonitoring study. Int. Arch. Occup. Environ. Health 2016, 89, 935–946. [Google Scholar] [CrossRef]
- Serrazina, D.C.; De Andrade, V.L.; Cota, M.; Mateus, M.L.; Aschner, M.; dos Santos, A.P.M. Biomarkers of exposure and effect in a working population exposed to lead, manganese and arsenic. J. Toxicol. Environ. Health Part. A 2018, 81, 983–997. [Google Scholar] [CrossRef] [PubMed]
- Tavakkoli, L.; Khanjani, N. Environmental and occupational exposure to cadmium in Iran: A systematic review. Rev. Environ. Health 2016, 31, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Goldenman, G.; Liezmann J Meura, L.; Camboni, M.; Reihlen, A.; Bakker, J. Study for the Strategy for a Non-Toxic Environment of the 7th Environment Action Programme; Final Report; European Union: Luxembourg, 2017; Available online: https://op.europa.eu/fi/publication-detail/-/publication/89fbbb74-969c-11e7-b92d-01aa75ed71a1 (accessed on 1 December 2021).
- Zoeller, R.T.; Brown, T.R.; Doan, L.L.; Gore, A.C.; Skakkebaek, N.E.; Soto, A.M.; Woodruff, T.J.; Vom Saal, F.S. Endocrine-disrupting chemicals and public health protection: A statement of principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef] [PubMed]
- Bergman, A.; Heindel, J.J.; Jobling, S.; Kidd, K.A.; Zoeller, R.T. State of the Science of Endocrine Disrupting Chemicals 2012: Summary for Decision-Makers. United Nations Environment Programme and the World Health Organization: 2013. Available online: https://apps.who.int/iris/bitstream/handle/10665/78102/WHO_HSE_PHE_IHE_2013.1_eng.pdf?sequence=1&isAllowed=y (accessed on 1 December 2021).
- Waterhouse, D.F.; McLaughlin, A.M.; Sheehan, F.; O’Shea, D. An examination of the prevalence of IDF- and ATPIII-defined metabolic syndrome in an Irish screening population. Ir. J. Med. Sci. 2009, 178, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, P.M.; Engström, G.; Hedblad, B. The metabolic syndrome and incidence of cardiovascular disease in non-diabetic subjects-a population-based study comparing three different definitions. Diabet. Med. 2007, 24, 464–472. [Google Scholar] [CrossRef]
- Jacobson, M.H.; Woodward, M.; Bao, W.; Liu, B.; Trasande, L. Urinary Bisphenols and Obesity Prevalence Among U.S. Children and Adolescents. J. Endocr. Soc. 2019, 3, 1715–1726. [Google Scholar] [CrossRef] [PubMed]
- Trasande, L.; Zoeller, R.T.; Hass, U.; Kortenkamp, A.; Grandjean, P.; Myers, J.P.; DiGangi, J.; Bellanger, M.; Hauser, R.; Legler, J.; et al. Estimating Burden and Disease Costs of Exposure to Endocrine-Disrupting Chemicals in the European Union. J. Clin. Endocrinol. Metab. 2015, 100, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Legler, J.; Fletcher, T.; Govarts, E.; Porta, M.; Blumberg, B.; Heindel, J.J.; Trasande, L. Obesity, diabetes, and associated costs of exposure to endocrine-disrupting chemicals in the European Union. J. Clin. Endocrinol. Metab. 2015, 100, 1278–1288. [Google Scholar] [CrossRef]
- Ganzleben, C.; Antignac, J.-P.; Sepai, O.; Tolonen, H.; Kolossa-Gehring, M.; Barouki, R.; Castaño, A.; Fiddicke, U.; Klanova, J.; Lebret, E.; et al. Human biomonitoring as a tool to support chemicals regulation in the European Union. Int. J. Hyg. Environ. Health 2017, 220, 94–97. [Google Scholar] [CrossRef] [PubMed]
- HBM4EU. 2021. Available online: https://www.hbm4eu.eu/ (accessed on 30 September 2021).
- Schoeters, G.; Lange, R.; Kolossa, M.; Barouki, R.; Tarroja, E.; Uhl, M.; Klanova, J.; Melymuk, L.; Horvat, M.; Bocca, B.; et al. Scoping Document for 2021 for the First and Second Round HBM4EU Priority Substances: HBM4EU. 2020. Available online: https://www.hbm4eu.eu/wp-content/uploads/2021/03/HBM4EU_D4.9_Scoping_Documents_HBM4EU_priority_substances_v1.0.pdf (accessed on 1 December 2021).
- Sucharew, H.; Macaluso, M. Progress Notes: Methods for Research Evidence Synthesis: The Scoping Review Approach. J. Hosp. Med. 2019, 14, 416–418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Braver-Sewradj, S.P.D.; Van Spronsen, R.; Hessel, E.V.S. Substitution of bisphenol A: A review of the carcinogenicity, reproductive toxicity, and endocrine disruption potential of alternative substances. Crit. Rev. Toxicol. 2020, 50, 128–147. [Google Scholar] [CrossRef]
- Rochester, J.R.; Bolden, A.L. Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes. Environ. Health Perspect. 2015, 123, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Wan, Y.; Kannan, K. Occurrence of bisphenols, bisphenol A diglycidyl ethers (BADGEs), and novolac glycidyl ethers (NOGEs) in indoor air from Albany, New York, USA, and its implications for inhalation exposure. Chemosphere 2016, 151, 1–8. [Google Scholar] [CrossRef]
- Valentino, R.; D’Esposito, V.; Ariemma, F.; Cimmino, I.; Beguinot, F.; Formisano, P. Bisphenol A environmental exposure and the detrimental effects on human metabolic health: Is it necessary to revise the risk assessment in vulnerable population? J. Endocrinol. Investig. 2016, 39, 259–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, B.; Lehmler, H.-J.; Sun, Y.; Xu, G.; Sun, Q.; Snetselaar, L.G.; Wallace, R.B.; Bao, W. Association of Bisphenol A and Its Substitutes, Bisphenol F and Bisphenol S, with Obesity in United States Children and Adolescents. Diabetes Metab. J. 2019, 43, 59–75. [Google Scholar] [CrossRef] [PubMed]
- Stojanoska, M.M.; Milosevic, N.; Milic, N.; Abenavoli, L. The influence of phthalates and bisphenol A on the obesity development and glucose metabolism disorders. Endocrine 2016, 55, 666–681. [Google Scholar] [CrossRef]
- European Commission. Commission Directive 2011/8/EU of 28 January 2011 Amending Directive 2002/72/EC as Regards the Restriction of Use of Bisphenol A in Plastic Infrant Feeding Bottles; European Commission: Brussels, Belgium, 2011; Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2011:026:0011:0014:EN:PDF (accessed on 1 December 2021).
- European Commission. Commission regulation (EU) 2018/213 of 12 February 2018 on the Use of Bisphenol A in Varnishes and Coating Intended to come into Contact with Food and Amending Regulation (EU) No 10/2011 as Regards the Use of that Substance in Plastic Food Contact Materials; European Commission: Brussels, Belgium, 2018; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32018R0213&from=FI. (accessed on 1 December 2021).
- European Commission. Commission Directive (EU) 2017/898 of 24 May 2017 Amending, for the Purpose of Adopting Specific Limit Values for Chemicals used in Toys, Appendix C to Annex II of Directive 2009/48/EC of the European Parliament and the Council on the Safety of Toys, as Regards Bisphenol A; European Commission: Brussels, Belgium, 2017; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32017L0898&from=ES (accessed on 1 December 2021).
- European Commission. Commission Regulation (EU) 2016/2235 of 12 December 2016 Ameding Annex XVII to Regulation (EC) No 1907/2006 on the European Parliament and of the Council Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) as Regards Bisphenol A; European Commission: Brussels, Belgium, 2016; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32016R2235&from=EN (accessed on 1 December 2021).
- Song, Y.; Chou, E.L.; Baecker, A.; You, N.Y.; Song, Y.; Sun, Q.; Liu, S. Endocrine-disrupting chemicals, risk of type 2 diabetes, and diabetes-related metabolic traits: A systematic review and meta-analysis. J. Diabetes 2016, 8, 516–532. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R. Bisphenol A and human health: A review of the literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef] [PubMed]
- Rancière, F.; Lyons, J.G.; Loh, V.H.; Botton, J.; Galloway, T.; Wang, T.; Shaw, J.E.; Magliano, D. Bisphenol A and the risk of cardiometabolic disorders: A systematic review with meta-analysis of the epidemiological evidence. Environ. Health 2015, 14, 1–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mustieles, V.; Arrebola, J.P. How polluted is your fat? What the study of adipose tissue can contribute to environmental epidemiology. J. Epidemiol. Commun. Health 2020, 74, 401–407. [Google Scholar] [CrossRef]
- Aekplakorn, W.; Chailurkit, L.-O.; Ongphiphadhanakul, B. Association of Serum Bisphenol A with Hypertension in Thai Population. Int. J. Hypertens. 2015, 2015, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Liu, H.; Zhou, S.; Zhang, X.; Peng, C.; Zhou, H.; Tong, Y.; Lu, Q. Association of bisphenol A and its alternatives bisphenol S and F exposure with hypertension and blood pressure: A cross-sectional study in China. Environ. Pollut. 2020, 257, 113639. [Google Scholar] [CrossRef] [PubMed]
- Shankar, A.; Teppala, S. Urinary Bisphenol A and Hypertension in a Multiethnic Sample of US Adults. J. Environ. Public Health 2012, 2012, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Teppala, S.; Madhavan, S.; Shankar, A. Bisphenol A and Metabolic Syndrome: Results from NHANES. Int. J. Endocrinol. 2012, 2012, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.Y.; Lee, E.; Kim, Y. The Association between Bisphenol a Exposure and Obesity in Children—A Systematic Review with Meta-Analysis. Int. J. Environ. Res. Public Health 2019, 16, 2521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ribeiro, C.M.; Beserra, B.T.S.; Silva, N.G.; Lima, C.L.; Rocha, P.R.S.; Coelho, M.S.; Neves, F.D.A.R.; Amato, A.A. Exposure to endocrine-disrupting chemicals and anthropometric measures of obesity: A systematic review and meta-analysis. BMJ Open 2020, 10, e033509. [Google Scholar] [CrossRef] [PubMed]
- Mustieles, V.; D’Cruz, S.C.; Couderq, S.; Rodríguez-Carrillo, A.; Fini, J.-B.; Hofer, T.; Steffensen, I.-L.; Dirven, H.; Barouki, R.; Olea, N.; et al. Bisphenol A and its analogues: A comprehensive review to identify and prioritize effect biomarkers for human biomonitoring. Environ. Int. 2020, 144, 105811. [Google Scholar] [CrossRef] [PubMed]
- Provvisiero, D.P.; Pivonello, C.; Muscogiuri, G.; Negri, M.; De Angelis, C.; Simeoli, C.; Pivonello, R.; Colao, A. Influence of Bisphenol A on Type 2 Diabetes Mellitus. Int. J. Environ. Res. Public Health 2016, 13, 989. [Google Scholar] [CrossRef] [Green Version]
- Warembourg, C.; Maitre, L.; Uria, I.T.; Fossati, S.; Roumeliotaki, T.; Aasvang, G.M.; Andrusaityte, S.; Casas, M.; Cequier, E.; Chatzi, L.; et al. Early-Life Environmental Exposures and Blood Pressure in Children. J. Am. Coll. Cardiol. 2019, 74, 1317–1328. [Google Scholar] [CrossRef] [PubMed]
- Chiu, Y.-H.; Mínguez-Alarcón, L.; Ford, J.B.; Keller, M.; Seely, E.W.; Messerlian, C.; Petrozza, J.; Williams, P.L.; Ye, X.; Calafat, A.M.; et al. Trimester-Specific Urinary Bisphenol A Concentrations and Blood Glucose Levels Among Pregnant Women From a Fertility Clinic. J. Clin. Endocrinol. Metab. 2017, 102, 1350–1357. [Google Scholar] [CrossRef] [Green Version]
- Shapiro, G.; Dodds, L.; Arbuckle, T.; Ashley-Martin, J.; Fraser, W.; Fisher, M.; Taback, S.; Keely, E.; Bouchard, M.; Monnier, P.; et al. Exposure to phthalates, bisphenol A and metals in pregnancy and the association with impaired glucose tolerance and gestational diabetes mellitus: The MIREC study. Environ. Int. 2015, 83, 63–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sol, C.M.; Santos, S.; Duijts, L.; Asimakopoulos, A.G.; Martinez-Moral, M.-P.; Kannan, K.; Jaddoe, V.W.; Trasande, L. Fetal phthalates and bisphenols and childhood lipid and glucose metabolism. A population-based prospective cohort study. Environ. Int. 2020, 144, 106063. [Google Scholar] [CrossRef] [PubMed]
- Silver, M.K.; O’Neill, M.S.; Sowers, M.R.; Park, S.K. Urinary Bisphenol A and Type-2 Diabetes in U.S. Adults: Data from NHANES 2003–2008. PLoS ONE 2011, 6, e26868. [Google Scholar] [CrossRef] [Green Version]
- Tai, X.; Chen, Y. Urinary bisphenol A concentrations positively associated with glycated hemoglobin and other indicators of diabetes in Canadian men. Environ. Res. 2016, 147, 172–178. [Google Scholar] [CrossRef] [PubMed]
- Beydoun, H.A.; Khanal, S.; Zonderman, A.B.; Beydoun, M.A. Sex differences in the association of urinary bisphenol-A concentration with selected indices of glucose homeostasis among U.S. adults. Ann. Epidemiol. 2014, 24, 90–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sabanayagam, C.; Teppala, S.; Shankar, A. Relationship between urinary bisphenol A levels and prediabetes among subjects free of diabetes. Acta Diabetol. 2013, 50, 625–631. [Google Scholar] [CrossRef]
- Shankar, A.; Teppala, S. Relationship between Urinary Bisphenol A Levels and Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2011, 96, 3822–3826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bi, Y.; Wang, W.; Xu, M.; Wang, T.; Lu, J.; Xu, Y.; Dai, M.; Chen, Y.; Zhang, D.; Sun, W.; et al. Diabetes Genetic Risk Score Modifies Effect of Bisphenol A Exposure on Deterioration in Glucose Metabolism. J. Clin. Endocrinol. Metab. 2016, 101, 143–150. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain (EFSA CONTAM Panel). Risk to human health related to the presence of perfluoroalkyl substances in food. ESFA J. 2020, 18, e06223. [CrossRef]
- Barry, V.; Winquist, A.; Steenland, K. Perfluorooctanoic Acid (PFOA) Exposures and Incident Cancers among Adults Living Near a Chemical Plant. Environ. Health Perspect. 2013, 121, 1313–1318. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, M.J.; Moon, S.; Oh, B.-C.; Jung, D.; Ji, K.; Choi, K.; Park, Y.J. Association between perfluoroalkyl substances exposure and thyroid function in adults: A meta-analysis. PLoS ONE 2018, 13, e0197244. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Zhang, B.; Furtado, J.D.; Sun, Q.; Hu, Y.; Rood, J.; Liang, L.; Qi, L.; Bray, G.A.; DeJonge, L.; et al. Associations of Perfluoroalkyl substances with blood lipids and Apolipoproteins in lipoprotein subspecies: The POUNDS-lost study. Environ. Health 2020, 19, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qi, W.; Clark, J.M.; Timme-Laragy, A.R.; Park, Y. Per- and polyfluoroalkyl substances and obesity, type 2 diabetes and non-alcoholic fatty liver disease: A review of epidemiologic findings. Toxicol. Environ. Chem. 2020, 102, 1–36. [Google Scholar] [CrossRef]
- Rappazzo, K.M.; Coffman, E.; Hines, E.P. Exposure to Perfluorinated Alkyl Substances and Health Outcomes in Children: A Systematic Review of the Epidemiologic Literature. Int. J. Environ. Res. Public Health 2017, 14, 691. [Google Scholar] [CrossRef]
- Christensen, K.Y.; Raymond, M.; Meiman, J. Perfluoroalkyl substances and metabolic syndrome. Int. J. Hyg. Environ. Health 2019, 222, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Mondal, D.; Lopez-Espinosa, M.-J.; Armstrong, B.; Stein, C.R.; Fletcher, T. Relationships of Perfluorooctanoate and Perfluorooctane Sulfonate Serum Concentrations between Mother–Child Pairs in a Population with Perfluorooctanoate Exposure from Drinking Water. Environ. Health Perspect. 2012, 120, 752–757. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M.; Chen, A.; Romano, M.; Calafat, A.M.; Webster, G.M.; Yolton, K.; Lanphear, B.P. Prenatal perfluoroalkyl substance exposure and child adiposity at 8 years of age: The HOME study. Obesity 2016, 24, 231–237. [Google Scholar] [CrossRef] [Green Version]
- Halldorsson, T.I.; Rytter, D.; Haug, L.S.; Bech, B.H.; Danielsen, I.; Becher, G.; Henriksen, T.B.; Olsen, S. Prenatal Exposure to Perfluorooctanoate and Risk of Overweight at 20 Years of Age: A Prospective Cohort Study. Environ. Health Perspect. 2012, 120, 668–673. [Google Scholar] [CrossRef] [Green Version]
- Averina, M.; Brox, J.; Huber, S.; Furberg, A.-S. Exposure to perfluoroalkyl substances (PFAS) and dyslipidemia, hypertension and obesity in adolescents. The Fit Futures study. Environ. Res. 2021, 195, 110740. [Google Scholar] [CrossRef] [PubMed]
- Geiger, S.D.; Xiao, J.; Shankar, A. Positive Association Between Perfluoroalkyl Chemicals and Hyperuricemia in Children. Am. J. Epidemiol. 2013, 177, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Santander, N.M.; Valvi, D.; Lopez-Espinosa, M.-J.; Manzano-Salgado, C.B.; Ballester, F.; Ibarluzea, J.; Marina, L.S.; Schettgen, T.; Guxens, M.; Sunyer, J.; et al. Exposure to Perfluoroalkyl Substances and Metabolic Outcomes in Pregnant Women: Evidence from the Spanish INMA Birth Cohorts. Environ. Health Perspect. 2017, 125, 117004. [Google Scholar] [CrossRef] [PubMed]
- Preston, E.V.; Rifas-Shiman, S.L.; Hivert, M.-F.; Zota, A.R.; Sagiv, S.K.; Calafat, A.M.; Oken, E.; James-Todd, T. Associations of Per- and Polyfluoroalkyl Substances (PFAS) With Glucose Tolerance During Pregnancy in Project Viva. J. Clin. Endocrinol. Metab. 2020, 105. [Google Scholar] [CrossRef]
- Xu, H.; Zhou, Q.; Zhang, J.; Chen, X.; Zhao, H.; Lu, H.; Ma, B.; Wang, Z.; Wu, C.; Ying, C.; et al. Exposure to elevated per- and polyfluoroalkyl substances in early pregnancy is related to increased risk of gestational diabetes mellitus: A nested case-control study in Shanghai, China. Environ. Int. 2020, 143, 105952. [Google Scholar] [CrossRef]
- Huang, R.; Chen, Q.; Zhang, L.; Luo, K.; Chen, L.; Zhao, S.; Feng, L.; Zhang, J. Prenatal exposure to perfluoroalkyl and polyfluoroalkyl substances and the risk of hypertensive disorders of pregnancy. Environ. Health 2019, 18, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Bangma, J.; Eaves, L.A.; Oldenburg, K.; Reiner, J.L.; Manuck, T.; Fry, R.C. Identifying Risk Factors for Levels of Per- and Polyfluoroalkyl Substances (PFAS) in the Placenta in a High-Risk Pregnancy Cohort in North Carolina. Environ. Sci. Technol. 2020, 54, 8158–8166. [Google Scholar] [CrossRef]
- Wen, Y.; Mirji, N.; Irudayaraj, J. Epigenetic toxicity of PFOA and GenX in HepG2 cells and their role in lipid metabolism. Toxicol. Vitr. 2020, 65, 104797. [Google Scholar] [CrossRef] [PubMed]
- Mi, X.; Yang, Y.-Q.; Zeeshan, M.; Wang, Z.-B.; Zeng, X.-Y.; Zhou, Y.; Yang, B.-Y.; Hu, L.-W.; Yu, H.-Y.; Liu, R.-Q.; et al. Serum levels of per- and polyfluoroalkyl substances alternatives and blood pressure by sex status: Isomers of C8 health project in China. Chemosphere 2020, 261, 127691. [Google Scholar] [CrossRef] [PubMed]
- Fragki, S.; Dirven, H.; Fletcher, T.; Grasl-Kraupp, B.; Bjerve Gützkow, K.; Hoogenboom, R.; Kersten, S.; Lindeman, B.; Louisse, J.; Peijnenburg, A.; et al. Systemic PFOS and PFOA exposure and disturbed lipid homeostasis in humans: What do we know and what not? Crit. Rev. Toxicol. 2021, 51, 141–164. [Google Scholar] [CrossRef]
- Benjamin, S.; Masai, E.; Kamimura, N.; Takahashi, K.; Anderson, R.C.; Faisal, P.A. Phthalates impact human health: Epidemiological evidences and plausible mechanism of action. J. Hazard. Mater. 2017, 340, 360–383. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Regulaton (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), Establishing a European Chemical Agency, Amending Directive 1999/45/EC and Repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as Well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC; European Commission: Brussels, Belgium, 2006; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006R1907&from=EN (accessed on 1 December 2021).
- European Commission. Commission Delegated Directive (EU) 2015/863 of 31 March 2015 Amending Annex II to Directive 2011/65/EU of the Parliament and of the Council as Regards the List of Restricted Substances; European Commission: Brussels, Belgium, 2015; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32015L0863&from=EN (accessed on 1 December 2021).
- European Commission. Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products (Recast); European Commission: Brussels, Belgium, 2009; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32009R1223&from=FI (accessed on 1 December 2021).
- Lange, R.; Apel, P.; Rousselle, C.; Charles, S.; Sissoko, F.; Kolossa-Gehring, M.; Ougier, E. The European Human Biomonitoring Initiative (HBM4EU): Human biomonitoring guidance values for selected phthalates and a substitute plasticizer. Int. J. Hyg. Environ. Health 2021, 234, 113722. [Google Scholar] [CrossRef]
- Kolossa-Gehring, M.; Fiddicke, U.; Leng, G.; Angerer, J.; Wolz, B. New human biomonitoring methods for chemicals of concern—the German approach to enhance relevance. Int. J. Hyg. Environ. Health 2017, 220, 103–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Golestanzadeh, M.; Riahi, R.; Kelishadi, R. Association of exposure to phthalates with cardiometabolic risk factors in children and adolescents: A systematic review and meta-analysis. Environ. Sci. Pollut. Res. 2019, 26, 35670–35686. [Google Scholar] [CrossRef] [PubMed]
- Radke, E.; Galizia, A.; Thayer, K.A.; Cooper, G.S. Phthalate exposure and metabolic effects: A systematic review of the human epidemiological evidence. Environ. Int. 2019, 132, 104768. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-C.; Moon, K.A.; Wang, S.-L.; Silbergeld, E.; Navas-Acien, A. The Association of Arsenic Metabolism with Cancer, Cardiovascular Disease, and Diabetes: A Systematic Review of the Epidemiological Evidence. Environ. Health Perspect. 2017, 125, 087001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, X.; Xu, X.; Lin, Y.; Zhang, Y.; Huo, X. Phthalate exposure as a risk factor for hypertension. Environ. Sci. Pollut. Res. 2018, 25, 20550–20561. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Zhang, C.; Tao, F.-B. Association between prenatal phthalate exposure and gestational metabolic syndrome parameters: A systematic review of epidemiological studies. Environ. Sci. Pollut. Res. 2021, 28, 20921–20938. [Google Scholar] [CrossRef]
- Gaston, S.A.; Tulve, N.S. Urinary phthalate metabolites and metabolic syndrome in U.S. adolescents: Cross-sectional results from the National Health and Nutrition Examination Survey (2003–2014) data. Int. J. Hyg. Environ. Health 2019, 222, 195–204. [Google Scholar] [CrossRef]
- Hatch, E.E.; Nelson, J.W.; Qureshi, M.M.; Weinberg, J.; Moore, L.L.; Singer, M.; Webster, T.F. Association of urinary phthalate metabolite concentrations with body mass index and waist circumference: A cross-sectional study of NHANES data, 1999–2002. Environ. Health 2008, 7, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- James-Todd, T.M.; Huang, T.; Seely, E.W.; Saxena, A.R. The association between phthalates and metabolic syndrome: The National Health and Nutrition Examination Survey 2001–2010. Environ. Health 2016, 15, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Perng, W.; Watkins, D.J.; Cantoral, A.; Mercado-García, A.; Meeker, J.D.; Téllez-Rojo, M.M.; Peterson, K.E. Exposure to phthalates is associated with lipid profile in peripubertal Mexican youth. Environ. Res. 2017, 154, 311–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stahlhut, R.W.; Van Wijngaarden, E.; Dye, T.D.; Cook, S.; Swan, S.H. Concentrations of Urinary Phthalate Metabolites Are Associated with Increased Waist Circumference and Insulin Resistance in Adult U.S. Males. Environ. Health Perspect. 2007, 115, 876–882. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trasande, L.; Attina, T.M. Association of Exposure to Di-2-Ethylhexylphthalate Replacements With Increased Blood Pressure in Children and Adolescents. Hypertension 2015, 66, 301–308. [Google Scholar] [CrossRef]
- Trasande, L.; Sathyanarayana, S.; Spanier, A.J.; Trachtman, H.; Attina, T.M.; Urbina, E.M. Urinary Phthalates Are Associated with Higher Blood Pressure in Childhood. J. Pediatr. 2013, 163, 747–753.e1. [Google Scholar] [CrossRef] [Green Version]
- Sarigiannis, D.A.; Karakitsios, S.P.; Gotti, A.; Liakos, I.L.; Katsoyiannis, A. Exposure to major volatile organic compounds and carbonyls in European indoor environments and associated health risk. Environ. Int. 2011, 37, 743–765. [Google Scholar] [CrossRef] [PubMed]
- De León-Martínez, L.D.; Flores-Ramírez, R.; Rodriguez-Aguilar, M.; Berumen-Rodríguez, A.; Pérez-Vázquez, F.J.; Díaz-Barriga, F. Analysis of urinary metabolites of polycyclic aromatic hydrocarbons in precarious workers of highly exposed occupational scenarios in Mexico. Environ. Sci. Pollut. Res. Int. 2021, 28, 23087–23098. [Google Scholar] [CrossRef] [PubMed]
- Gearhart-Serna, L.M.; Jayasundara, N.; Tacam, M.; Di Giulio, R.; Devi, G.R. Assessing Cancer Risk Associated with Aquatic Polycyclic Aromatic Hydrocarbon Pollution Reveals Dietary Routes of Exposure and Vulnerable Populations. J. Environ. Public Health 2018, 2018, 1–10. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Directive 2001/81/EC of the European Parliament and of the Council of 23 October 2001 on National Emission Ceilings for Certain Atmospheric Pollutants; European Commission: Brussels, Belgium, 2001; Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2001:309:0022:0030:EN:PDF (accessed on 1 December 2021).
- Khosravipour, M.; Khosravipour, H. The association between urinary metabolites of polycyclic aromatic hydrocarbons and diabetes: A systematic review and meta-analysis study. Chemosphere 2020, 247, 125680. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.-M.; Poursafa, P.; Moosazadeh, M.; Abedini, E.; Hajizadeh, Y.; Mansourian, M.; Pourzamani, H. A systematic review on the effects of polycyclic aromatic hydrocarbons on cardiometabolic impairment. Int. J. Prev. Med. 2017, 8, 19. [Google Scholar] [CrossRef] [PubMed]
- Bangia, K.S.; Symanski, E.; Strom, S.S.; Bondy, M. A cross-sectional analysis of polycyclic aromatic hydrocarbons and diesel particulate matter exposures and hypertension among individuals of Mexican origin. Environ. Health 2015, 14, 51. [Google Scholar] [CrossRef] [Green Version]
- Lee, T.-W.; Kim, D.H.; Ryu, J.Y. Association between urinary polycyclic aromatic hydrocarbons and hypertension in the Korean population: Data from the Second Korean National Environmental Health Survey (2012–2014). Sci. Rep. 2020, 10, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ranjbar, M.; Rotondi, M.A.; Ardern, C.I.; Kuk, J.L. Urinary Biomarkers of Polycyclic Aromatic Hydrocarbons Are Associated with Cardiometabolic Health Risk. PLoS ONE 2015, 10, e0137536. [Google Scholar] [CrossRef]
- Shiue, I. Are urinary polyaromatic hydrocarbons associated with adult hypertension, heart attack, and cancer? USA NHANES, 2011–2012. Environ. Sci. Pollut. Res. 2015, 22, 16962–16968. [Google Scholar] [CrossRef]
- Wang, B.; Li, Z.; Ma, Y.; Qiu, X.; Ren, A. Association of polycyclic aromatic hydrocarbons in housewives’ hair with hypertension. Chemosphere 2016, 153, 315–321. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Kan, H.; Kearney, G.; Xu, X. Associations between exposure to polycyclic aromatic hydrocarbons and glucose homeostasis as well as metabolic syndrome in nondiabetic adults. Sci. Total Environ. 2015, 505, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Pan, B.; Zhao, X.; Fu, Y.; Li, X.; Yang, A.; Li, Q.; Dong, J.; Nie, J.; Yang, J. The interaction effects of smoking and polycyclic aromatic hydrocarbons exposure on the prevalence of metabolic syndrome in coke oven workers. Chemosphere 2020, 247, 125880. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Yan, K.; Zeng, D.; Lai, X.; Chen, X.; Fang, Q.; Guo, H.; Wu, T.; Zhang, X. Association of polycyclic aromatic hydrocarbons metabolites and risk of diabetes in coke oven workers. Environ. Pollut. 2017, 223, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Sancini, A.; Caciari, T.; Sinibaldi, F.; Sacco, C.; Boscolo, P.; Giubilati, R.; Scala, B.; Tomei, G.; Tomei, F.; Rosati, M.V. Blood pressure changes and polycyclic aromatic hydrocarbons in outdoor workers. Clin. Ter. 2014, 165, 295–303. [Google Scholar]
- Poursafa, P.; Dadvand, P.; Amin, M.; Hajizadeh, Y.; Ebrahimpour, K.; Mansourian, M.; Pourzamani, H.; Sunyer, J.; Kelishadi, R. Association of polycyclic aromatic hydrocarbons with cardiometabolic risk factors and obesity in children. Environ. Int. 2018, 118, 203–210. [Google Scholar] [CrossRef]
- Scinicariello, F.; Buser, M. Urinary Polycyclic Aromatic Hydrocarbons and Childhood Obesity: NHANES (2001–2006). Environ. Health Perspect. 2014, 122, 299–303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trasande, L.; Urbina, E.M.; Khoder, M.; Alghamdi, M.; Shabaj, I.; Alam, M.S.; Harrison, R.M.; Shamy, M. Polycyclic aromatic hydrocarbons, brachial artery distensibility and blood pressure among children residing near an oil refinery. Environ. Res. 2015, 136, 133–140. [Google Scholar] [CrossRef] [Green Version]
- Kaur, R.; Kaur Mavi, G.; Raghav, S. Pesticipdes Classification and its Impact on Environmet. J. Curr. Microbiol. Appl. Sci. 2019, 8, 1889–1897. [Google Scholar] [CrossRef]
- European Commission. Commission Directive 2006/141/EC of 22 December 2006 on Infant Formulae and Follow-On Formulae and Amending Directive 1999/21/EC; European Commission: Brussels, Belgium, 2006; Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32006L0141&from=EN (accessed on 1 December 2021).
- Evangelou, E.; Ntritsos, G.; Chondrogiorgi, M.; Kavvoura, F.K.; Hernández, A.F.; Ntzani, E.E.; Tzoulaki, I. Exposure to pesticides and diabetes: A systematic review and meta-analysis. Environ. Int. 2016, 91, 60–68. [Google Scholar] [CrossRef]
- Jaacks, L.; Staimez, L.R. Association of persistent organic pollutants and non-persistent pesticides with diabetes and diabetes-related health outcomes in Asia: A systematic review. Environ. Int. 2015, 76, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Clark, J.M.; Park, Y. Potential contribution of insecticide exposure and development of obesity and type 2 diabetes. Food Chem. Toxicol. 2017, 105, 456–474. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.-K.; Park, S.; Chang, S.-J.; Kim, S.-K.; Song, J.-S.; Kim, H.-R.; Oh, S.-S.; Koh, S.-B. Pesticides as a risk factor for metabolic syndrome: Population-based longitudinal study in Korea. Mol. Cell. Toxicol. 2019, 15, 431–441. [Google Scholar] [CrossRef]
- Park, S.-K.; Son, H.-K.; Lee, S.-K.; Kang, J.-H.; Chang, Y.-S.; Jacobs, D.R.; Lee, D.-H. Relationship Between Serum Concentrations of Organochlorine Pesticides and Metabolic Syndrome Among Non-Diabetic Adults. J. Prev. Med. Public Health 2010, 43, 1–8. [Google Scholar] [CrossRef]
- Rosenbaum, P.F.; Weinstock, R.S.; Silverstone, A.E.; Sjödin, A.; Pavuk, M. Metabolic syndrome is associated with exposure to organochlorine pesticides in Anniston, AL, United States. Environ. Int. 2017, 108, 11–21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mustieles, V.; Fernandez, M.F.; Martin-Olmedo, P.; Alzaga, B.G.; Fontalba-Navas, A.; Hauser, R.; Olea, N.; Arrebola, J.P. Human adipose tissue levels of persistent organic pollutants and metabolic syndrome components: Combining a cross-sectional with a 10-year longitudinal study using a multi-pollutant approach. Environ. Int. 2017, 104, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-H.; Steffes, M.W.; Sjödin, A.; Jones, R.S.; Needham, L.L. Low Dose Organochlorine Pesticides and Polychlorinated Biphenyls Predict Obesity, Dyslipidemia, and Insulin Resistance among People Free of Diabetes. PLoS ONE 2011, 6, e15977. [Google Scholar] [CrossRef] [Green Version]
- Suarez-Lopez, J.R.; Gross, M.D.; Lee, D.-H. Summary data of serum concentrations of 32 persistent organic pollutants in young adults in relation to summary scores of persistent organic pollutants. Data Brief. 2019, 23, 103720. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-C.; Moon, K.; Thayer, K.A.; Navas-Acien, A. Environmental Chemicals and Type 2 Diabetes: An Updated Systematic Review of the Epidemiologic Evidence. Curr. Diabetes Rep. 2013, 13, 831–849. [Google Scholar] [CrossRef]
- Kim, Y.A.; Park, J.B.; Woo, M.S.; Lee, S.Y.; Kim, H.Y.; Yoo, Y. Persistent Organic Pollutant-Mediated Insulin Resistance. Int. J. Environ. Res. Public Health 2019, 16, 448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- González, M.C. Prenatal exposure to persistent organic pollutants as a risk factor of offspring metabolic syndrome development during childhood. Rev. Environ. Health 2021. [Google Scholar] [CrossRef] [PubMed]
- Kahn, L.; Trasande, L. Environmental Toxicant Exposure and Hypertensive Disorders of Pregnancy: Recent Findings. Curr. Hypertens. Rep. 2018, 20, 87. [Google Scholar] [CrossRef]
- Saldana, T.M.; Basso, O.; Baird, D.; Hoppin, J.; Weinberg, C.; Blair, A.; Alavanja, M.C.; Sandler, D.P. Pesticide Exposure and Hypertensive Disorders During Pregnancy. Environ. Health Perspect. 2009, 117, 1393–1396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Long, N.; Holloway, A.C. Early-life chemical exposures and risk of metabolic syndrome. Diabetes Metab. Syndr. Obes. Targets Ther. 2017, 10, 101–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- La Merrill, M.; Cirillo, P.M.; Terry, M.B.; Krigbaum, N.; Flom, J.D.; Cohn, B.A. Prenatal Exposure to the Pesticide DDT and Hypertension Diagnosed in Women before Age 50: A Longitudinal Birth Cohort Study. Environ. Health Perspect. 2013, 121, 594–599. [Google Scholar] [CrossRef] [Green Version]
- IARC. Arsenic, Metals, Fibres, and Dusts. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans Volume 100C; IARC Monograph; IARC: Lyon, France, 2012; Available online: https://publications.iarc.fr/120 (accessed on 1 December 2021).
- WHO. Preventing Disease through Healthy Environments. Exposure to Arsenic: A Major Public Health Concern; WHO: Geneva, Switzerland, 2019; Available online: https://www.who.int/publications/i/item/WHO-CED-PHE-EPE-19.4.1 (accessed on 1 December 2021).
- Baker, B.A.; Cassano, V.A.; Murray, C. Arsenic Exposure, Assessment, Toxicity, Diagnosis, and Management: Guidance for Occupational and Environmental Physicians. J. Occup. Environ. Med. 2018, 60, e634–e639. [Google Scholar] [CrossRef]
- Freire, B.M.; Lange, C.N.; Pedron, T.; Monteiro, L.R.; Sanches, L.R.; Pedreira-Filho, W.D.R.; Batista, B.L. Occupational exposure of foundry workers assessed by the urinary concentrations of 18 elements and arsenic species. J. Trace Elem. Med. Biol. 2020, 62, 126593. [Google Scholar] [CrossRef] [PubMed]
- Leso, V.; Capitanelli, I.; Lops, E.A.; Ricciardi, W.; Iavicoli, I. Occupational chemical exposure and diabetes mellitus risk. Toxicol. Ind. Health 2017, 33, 222–249. [Google Scholar] [CrossRef]
- Vimercati, L.; Gatti, M.F.; Gagliardi, T.; Cuccaro, F.; De Maria, L.; Caputi, A.; Quarato, M.; Baldassarre, A. Environmental exposure to arsenic and chromium in an industrial area. Environ. Sci. Pollut. Res. 2017, 24, 11528–11535. [Google Scholar] [CrossRef] [Green Version]
- Eick, S.M.; Steinmaus, C. Arsenic and Obesity: A Review of Causation and Interaction. Curr. Environ. Health Rep. 2020, 7, 343–351. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Samarghandian, S.; Azimi-Nezhad, M. The role of arsenic in obesity and diabetes. J. Cell. Physiol. 2019, 234, 12516–12529. [Google Scholar] [CrossRef] [PubMed]
- Navas-Acien, A.; Silbergeld, E.K.; Streeter, R.A.; Clark, J.M.; Burke, T.A.; Guallar, E. Arsenic Exposure and Type 2 Diabetes: A Systematic Review of the Experimental and Epidemiologic Evidence. Environ. Health Perspect. 2006, 114, 641–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abhyankar, L.N.; Jones, M.R.; Guallar, E.; Navas-Acien, A. Arsenic Exposure and Hypertension: A Systematic Review. Environ. Health Perspect. 2012, 120, 494–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Li, M.; Tian, X.; Xie, J.; Liu, P.; Ying, X.; Wang, M.; Yuan, J.; Gao, Y.; Tian, F.; et al. Effects of arsenic exposure on lipid metabolism: A systematic review and meta-analysis. Toxicol. Mech. Methods 2021, 31, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Farzan, S.F.; Gossai, A.; Chen, Y.; Chasan-Taber, L.; Baker, E.; Karagas, M. Maternal arsenic exposure and gestational diabetes and glucose intolerance in the New Hampshire birth cohort study. Environ. Health 2016, 15, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Salmeri, N.; Villanacci, R.; Candiani, M.; Ottolina, J.; Bartiromo, L.; Cavoretto, P.; Dolci, C.; Lembo, R.; Schimberni, M.; Valsecchi, L.; et al. Maternal Arsenic Exposure and Gestational Diabetes: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 3094. [Google Scholar] [CrossRef] [PubMed]
- Young, J.; Cai, L.; States, J.C. Impact of prenatal arsenic exposure on chronic adult diseases. Syst. Biol. Reprod. Med. 2018, 64, 469–483. [Google Scholar] [CrossRef]
- Lin, H.-C.; Huang, Y.-K.; Shiue, H.-S.; Chen, L.-S.; Choy, C.-S.; Huang, S.-R.; Han, B.-C.; Hsueh, Y.-M. Arsenic methylation capacity and obesity are associated with insulin resistance in obese children and adolescents. Food Chem. Toxicol. 2014, 74, 60–67. [Google Scholar] [CrossRef]
- United National Environmental Programme. Minamata Convention on Mercury: Text. and Annexes; United National Environmental Programme: Paris, France, 2019; Available online: https://www.mercuryconvention.org/sites/default/files/2021-06/Minamata-Convention-booklet-Sep2019-EN.pdf (accessed on 1 December 2021).
- Višnjevec, A.M.; Kocman, D.; Horvat, M. Human mercury exposure and effects in Europe. Environ. Toxicol. Chem. 2014, 33, 1259–1270. [Google Scholar] [CrossRef]
- Planchart, A.; Green, A.; Hoyo, C.; Mattingly, C.J. Heavy Metal Exposure and Metabolic Syndrome: Evidence from Human and Model System Studies. Curr. Environ. Health Rep. 2018, 5, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.; Tremblay, P.-Y.; Ayotte, P. Is mercury exposure causing diabetes, metabolic syndrome and insulin resistance? A systematic review of the literature. Environ. Res. 2017, 156, 747–760. [Google Scholar] [CrossRef] [PubMed]
- Tinkov, A.A.; Ajsuvakova, O.; Skalnaya, M.; Popova, E.; Sinitskii, A.; Nemereshina, O.; Gatiatulina, E.; Nikonorov, A.; Skalny, A.V. Mercury and metabolic syndrome: A review of experimental and clinical observations. BioMetals 2015, 28, 231–254. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.F.; Singh, K.; Chan, H.M. Mercury Exposure, Blood Pressure, and Hypertension: A Systematic Review and Dose-response Meta-analysis. Environ. Health Perspect. 2018, 126, 076002. [Google Scholar] [CrossRef]
- Gallego-Viñas, G.; Ballester, F.; Llop, S. Chronic mercury exposure and blood pressure in children and adolescents: A systematic review. Environ. Sci. Pollut. Res. 2019, 26, 2238–2252. [Google Scholar] [CrossRef]
- He, K.; Xun, P.; Liu, K.; Morris, S.; Reis, J.; Guallar, E. Mercury Exposure in Young Adulthood and Incidence of Diabetes Later in Life: The CARDIA Trace Element Study. Diabetes Care 2013, 36, 1584–1589. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Huang, Y.; Xing, Y.; Hu, C.; Zhang, W.; Tang, Y.; Su, W.; Huo, X.; Zhou, A.; Xia, W.; et al. Association of urinary cadmium, circulating fatty acids, and risk of gestational diabetes mellitus: A nested case-control study in China. Environ. Int. 2020, 137, 105527. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, B.; Xia, W.; Xu, S.; Li, Y.; Huang, Z.; Pan, X.; Chen, X.; Hu, C.; Liu, H.; et al. Cadmium Body Burden and Gestational Diabetes Mellitus: A Prospective Study. Environ. Health Perspect. 2018, 126, 027006. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Xia, W.; Zhang, B.; Zhou, A.; Huang, Z.; Zhang, H.; Liu, H.; Jiang, Y.; Hu, C.; Chen, X.; et al. Relation between cadmium exposure and gestational diabetes mellitus. Environ. Int. 2018, 113, 300–305. [Google Scholar] [CrossRef]
- Jeong, H.-S. The Relationship between Workplace Environment and Metabolic Syndrome. Int. J. Occup. Environ. Med. 2018, 9, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Tinkov, A.A.; Filippini, T.; Ajsuvakova, O.P.; Aaseth, J.; Gluhcheva, Y.G.; Ivanova, J.; Bjørklund, G.; Skalnaya, M.G.; Gatiatulina, E.R.; Popova, E.V.; et al. The role of cadmium in obesity and diabetes. Sci. Total. Environ. 2017, 601, 741–755. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Vesey, D.; Gobe, G.C. Kidney Cadmium Toxicity, Diabetes and High Blood Pressure: The Perfect Storm. Tohoku J. Exp. Med. 2017, 241, 65–87. [Google Scholar] [CrossRef] [Green Version]
- Caciari, T.; Sancini, A.; Fioravanti, M.; Capozzella, A.; Casale, T.; Montuori, L.; Fiaschetti, M.; Schifano, M.P.; Andreozzi, G.; Nardone, N.; et al. Cadmium and hypertension in exposed workers: A meta-analysis. Int. J. Occup. Med. Environ. Health 2013, 26, 440–456. [Google Scholar] [CrossRef]
- Martins, A.C.; Lopes, A.C.B.A.; Urbano, M.R.; Carvalho, M.D.F.H.; Silva, A.M.R.; Tinkov, A.A.; Aschner, M.; Mesas, A.E.; Silbergeld, E.K.; Paoliello, M.M. An updated systematic review on the association between Cd exposure, blood pressure and hypertension. Ecotoxicol. Environ. Saf. 2021, 208, 111636. [Google Scholar] [CrossRef] [PubMed]
- Obeng-Gyasi, E. Chronic cadmium exposure and cardiovascular disease in adults. J. Environ. Sci. Health Part A 2020, 55, 726–729. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Lu, Y.-H.; Pi, H.-F.; Gao, P.; Li, M.; Zhang, L.; Pei, L.-P.; Mei, X.; Liu, L.; Zhao, Q.; et al. Cadmium Exposure is Associated with the Prevalence of Dyslipidemia. Cell. Physiol. Biochem. 2016, 40, 633–643. [Google Scholar] [CrossRef] [PubMed]
- Sargis, R.M.; Simmons, R. Environmental neglect: Endocrine disruptors as underappreciated but potentially modifiable diabetes risk factors. Diabetologia 2019, 62, 1811–1822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin, O.; Scholze, M.; Ermler, S.; McPhie, J.; Bopp, S.K.; Kienzler, A.; Parissis, N.; Kortenkamp, A. Ten years of research on synergisms and antagonisms in chemical mixtures: A systematic review and quantitative reappraisal of mixture studies. Environ. Int. 2021, 146, 106206. [Google Scholar] [CrossRef] [PubMed]
- Kortenkamp, A.; Faust, M. Regulate to reduce chemical mixture risk. Science 2018, 361, 224–226. [Google Scholar] [CrossRef] [Green Version]
- Kolossa-Gehring, M.; Becker, K.; Conrad, A.; Lüdecke, A.; Riedel, S.; Seiwert, M.; Schulz, C.; Szewzyk, R. German Environmental Survey for Children (GerES IV)—First results. Int. J. Hyg. Environ. Health 2007, 210, 535–540. [Google Scholar] [CrossRef]
- Balicco, A.; Oleko, A.; Boschat, L.; Deschamps, V.; Saoudi, A.; Zeghnoun, A.; Fillol, C. Esteban design: A cross-sectional health survey about environment biomonitoring, physical activity and nutrition (2014–2016). Toxicol. Anal. Clin. 2017, 29, 517–537. [Google Scholar]
- López, M.E.; Göen, T.; Vaccher, V.; Elbers, I.; Thomsen, C.; Vorkamp, K.; Díaz, S.P.; Kolossa-Gehring, M.; Castaño, A.; Mol, H.; et al. The European human biomonitoring platform—Design and implementation of a laboratory quality assurance/quality control (QA/QC) programme for selected priority chemicals. Int. J. Hyg. Environ. Health 2021, 234, 113740. [Google Scholar] [CrossRef]
- Louro, H.; Heinälä, M.; Alvito, P.; Martins, C.; Assunção, R.; Silva, M.J.; Pronk, A.; Schaddelee-Scholten, B.; Gonzalez, M.D.C.; de Alba, M.; et al. Human biomonitoring in health risk assessment in Europe: Current practices and recommendations for the future. Int. J. Hyg. Environ. Health 2019, 222, 727–737. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haverinen, E.; Fernandez, M.F.; Mustieles, V.; Tolonen, H. Metabolic Syndrome and Endocrine Disrupting Chemicals: An Overview of Exposure and Health Effects. Int. J. Environ. Res. Public Health 2021, 18, 13047. https://doi.org/10.3390/ijerph182413047
Haverinen E, Fernandez MF, Mustieles V, Tolonen H. Metabolic Syndrome and Endocrine Disrupting Chemicals: An Overview of Exposure and Health Effects. International Journal of Environmental Research and Public Health. 2021; 18(24):13047. https://doi.org/10.3390/ijerph182413047
Chicago/Turabian StyleHaverinen, Elsi, Mariana F. Fernandez, Vicente Mustieles, and Hanna Tolonen. 2021. "Metabolic Syndrome and Endocrine Disrupting Chemicals: An Overview of Exposure and Health Effects" International Journal of Environmental Research and Public Health 18, no. 24: 13047. https://doi.org/10.3390/ijerph182413047
APA StyleHaverinen, E., Fernandez, M. F., Mustieles, V., & Tolonen, H. (2021). Metabolic Syndrome and Endocrine Disrupting Chemicals: An Overview of Exposure and Health Effects. International Journal of Environmental Research and Public Health, 18(24), 13047. https://doi.org/10.3390/ijerph182413047






