Mindfulness Meditation Improves Musical Aesthetic Emotion Processing in Young Adults
Abstract
:1. Introduction
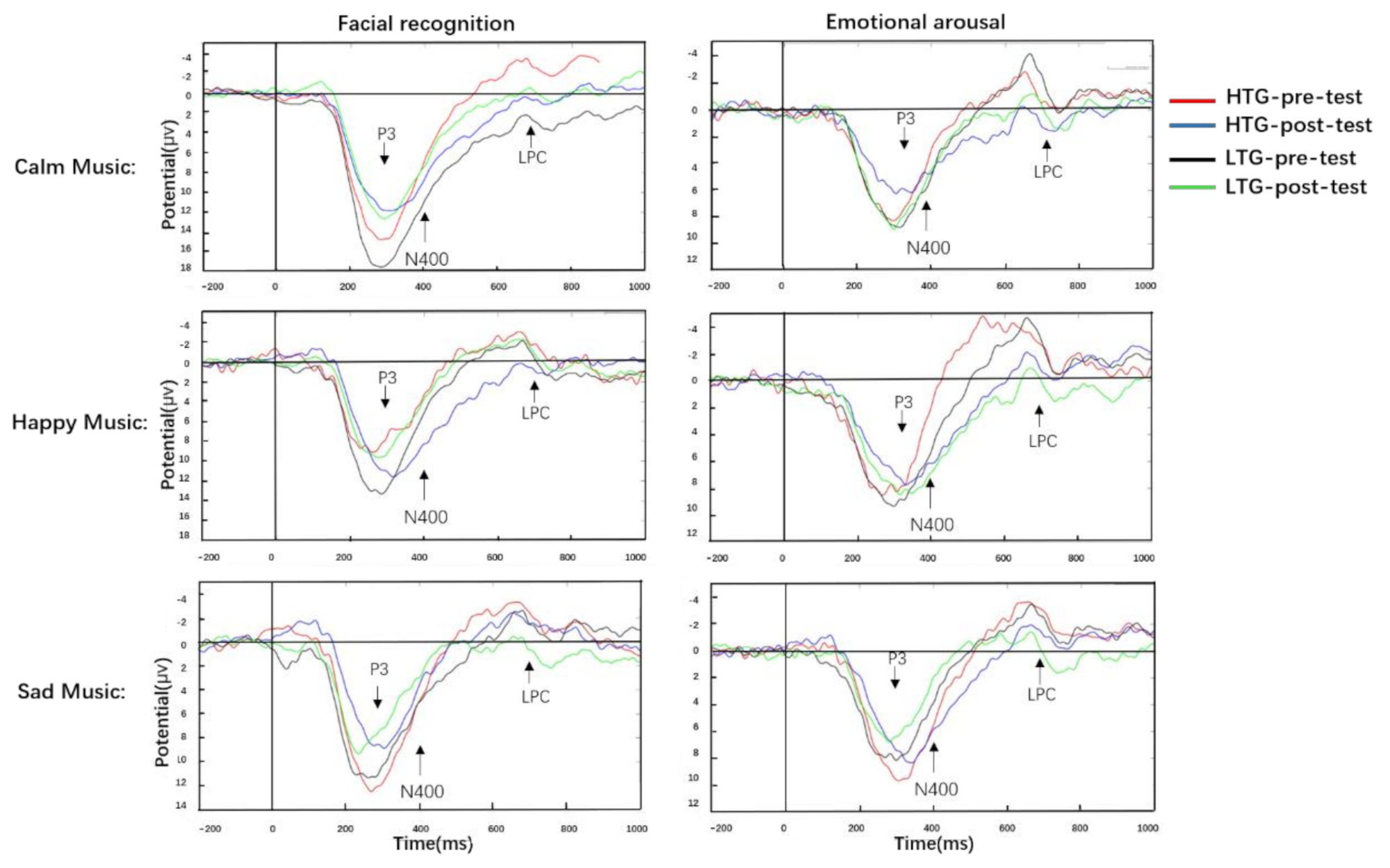
- First, compared with the pre-test, the post-test of facial recognition would have a higher accuracy (ACC) and faster response times (RTs), which would be reflected in smaller P3 and larger LPC amplitudes at the post-test.
- Second, compared with the LTG, there would be higher ACC and faster RTs in facial recognition, which would be reflected in that the smaller P3 and larger LPC amplitudes in the HTG. There would be no significant difference between the groups in the post-test of facial recognition.
- Third, compared with the pre-test, the post-test of emotional arousal would have a lower intensity and faster RTs, which would be reflected in smaller N400 and larger LPC amplitudes at the post-test.
- Fourth, compared with the LTG, there would be lower intensity and faster RTs in emotion arousal, which would be reflected in smaller N400 and larger LPC amplitudes in the HTG. There would be no significant difference between the groups in the post-test of emotional arousal.
- Fifth, in terms of music aesthetic emotions, the intensity of the post-test would be lower than that of the pre-test; the emotional intensity for the HTG would be higher than that for the LTG.
2. Methods
2.1. Participants
2.2. Stimuli
2.2.1. Musical Stimuli
2.2.2. Mindfulness Meditation Audio
2.3. Self-Reported Measures
2.3.1. The Positive and Negative Affect Schedule
2.3.2. Mindful Attention Awareness Scale
2.3.3. The Toronto Mindfulness Scale
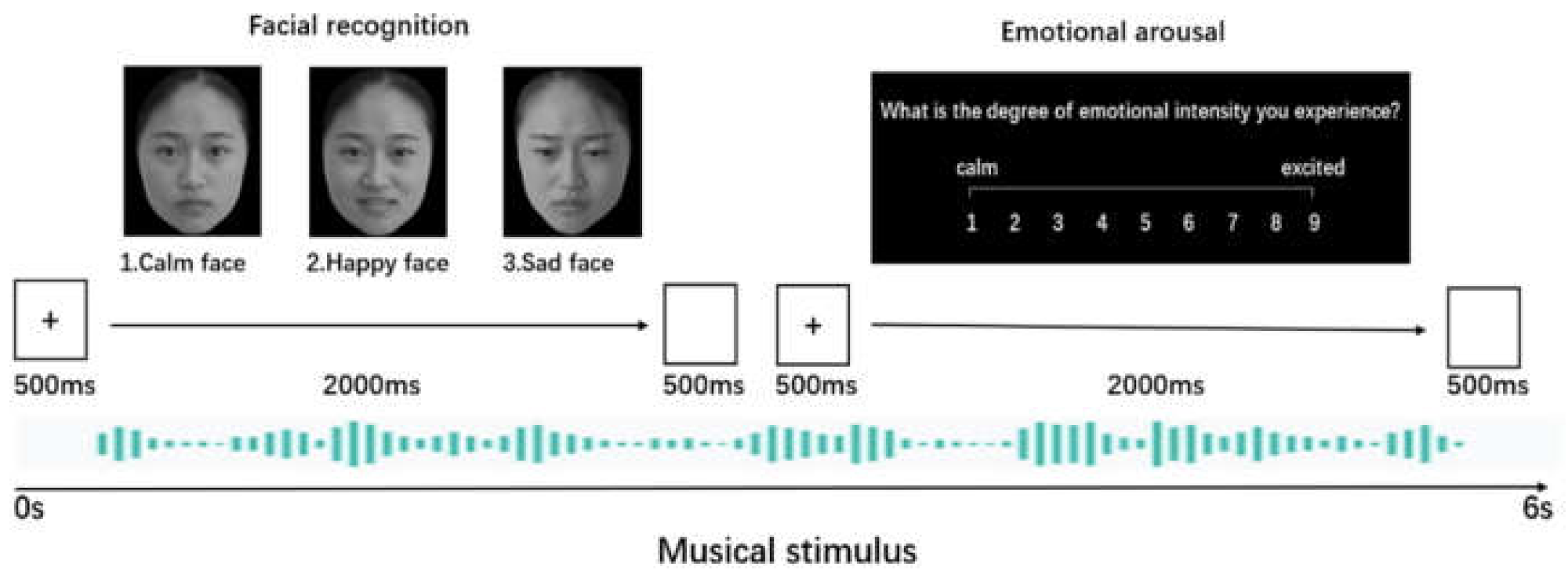
2.4. Facial Recognition and Emotional Arousal Tasks
2.5. Procedure
2.6. Behavioral Analyses
2.7. EEG Recording and Analyses
3. Results
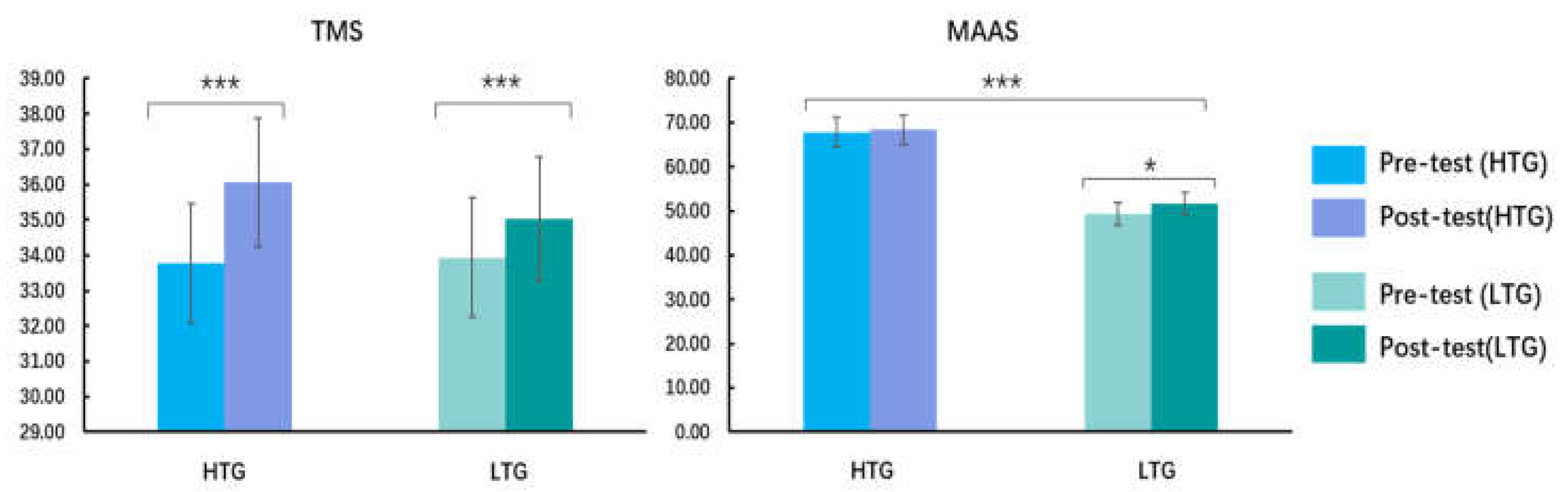
3.1. Self-Reported Results
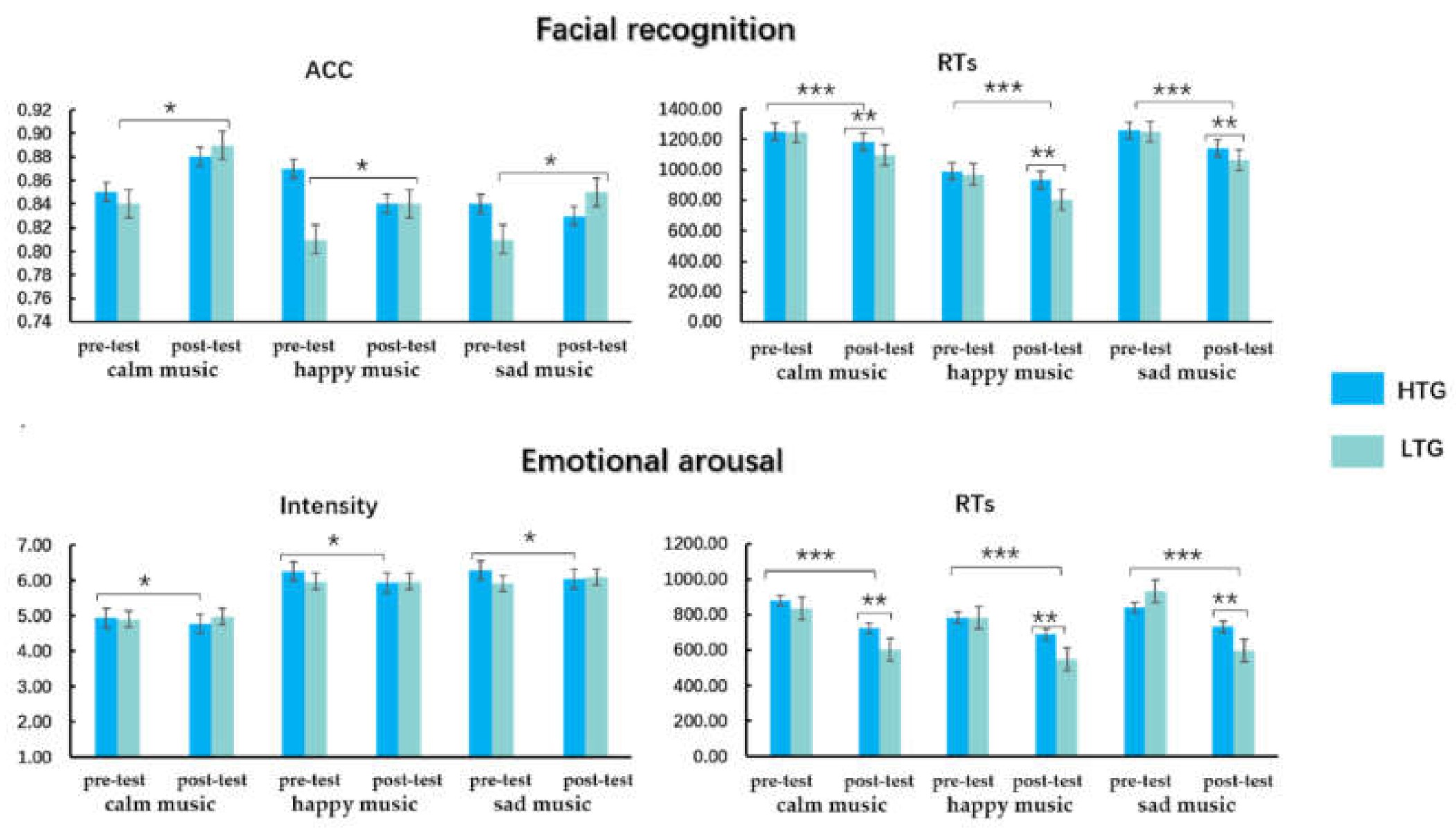
3.2. Behavioral Results
3.2.1. Facial Recognition
3.2.2. Emotional Arousal
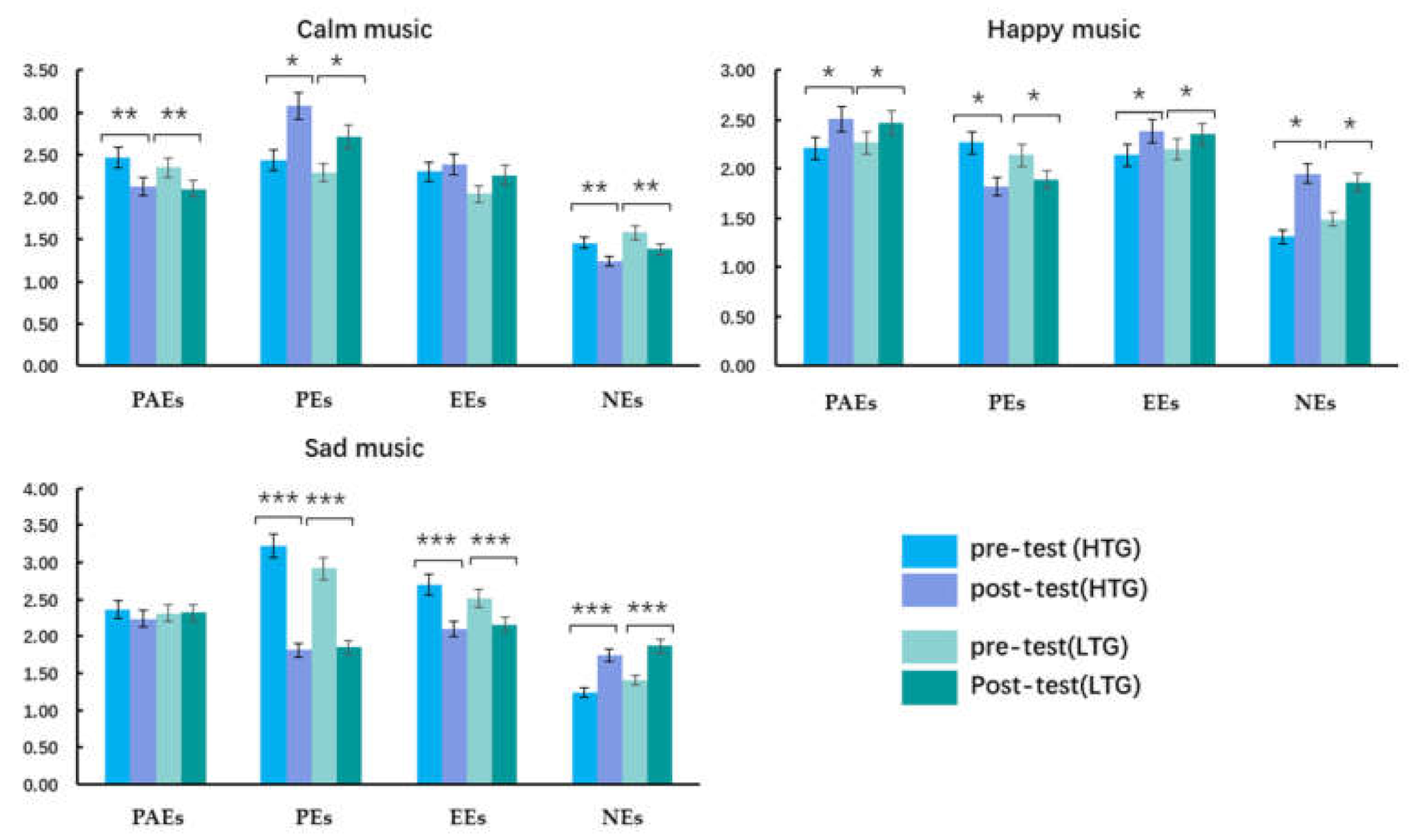
3.2.3. The AESTHEMOS Results
3.2.4. Calm Music
3.2.5. Happy Music
3.2.6. Sad Music
3.3. The ERPs Results
3.3.1. P3
3.3.2. N400
3.3.3. LPC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Wassiliwizky, E.; Menninghaus, W. Why and How Should Cognitive Science Care about Aesthetics? Trends Cogn. Sci. 2021, 25, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Eerola, T.; Vuoskoski, J.K.; Peltola, H.-R.; Putkinen, V.; Schäfer, K. An integrative review of the enjoyment of sadness associated with music. Phys. Life Rev. 2018, 25, 100–121. [Google Scholar] [CrossRef] [PubMed]
- Salimpoor, V.N.; Benovoy, M.; Larcher, K.; Dagher, A.; Zatorre, R.J. Anatomically distinct dopamine release during anticipation and experience of peak emotion to music. Nat. Neurosci. 2011, 14, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Salimpoor, V.N.; Bosch, I.v.d.; Kovacevic, N.; McIntosh, A.R.; Dagher, A.; Zatorre, R.J. Interactions between the nucleus accumbens and auditory cortices predict music reward value. Science 2013, 340, 216–219. [Google Scholar] [CrossRef]
- Liu, X.; Liu, Y.; Shi, H.; Zheng, M. Effects of mindfulness meditation on musical aesthetic emotion processing. Front. Psychol. 2021, 12, 2903. [Google Scholar] [CrossRef] [PubMed]
- Ding, W.; Chao, W.; Hong, L. The psychological mechanism of enjoying sad music. Adv. Psychol. Sci. 2018, 26, 1004–1011. [Google Scholar]
- Trost, W.; Ethofer, T.; Zentner, M.; Vuilleumier, P. Mapping aesthetic musical emotions in the brain. Cereb Cortex 2012, 22, 2769–2783. [Google Scholar] [CrossRef] [Green Version]
- Vuilleumier, P.; Trost, W. Music and emotions: From enchantment to entrainment. Ann. N. Y. Acad. Sci. 2015, 1337, 212–222. [Google Scholar] [CrossRef] [Green Version]
- Müller, M.; Höfel, L.; Brattico, E.; Jacobsen, T. Aesthetic judgments of music in experts and laypersons—An ERP study. Int. J. Psychophysiol. 2010, 76, 40–51. [Google Scholar] [CrossRef]
- Reybrouck, M.; Vuust, P.; Brattico, E. Brain Connectivity Networks and the Aesthetic Experience of Music. Brain Sci. 2018, 8, 107. [Google Scholar] [CrossRef] [Green Version]
- Juslin, P.N. From everyday emotions to aesthetic emotions: Towards a unified theory of musical emotions. Phys. Life Rev. 2013, 10, 235–266. [Google Scholar] [CrossRef] [Green Version]
- Menninghaus, W.; Wagner, V.; Wassiliwizky, E.; Schindler, I.; Hanich, J.; Jacobsen, T.; Koelsch, S. What are aesthetic emotions? Psychol. Rev. 2019, 126, 171–195. [Google Scholar] [CrossRef]
- Zatorre, R.J.; Salimpoor, V.N. From perception to pleasure: Music and its neural substrates. Proc. Natl. Acad. Sci. USA 2013, 110 (Suppl. 2), 10430–10437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eerola, T.; Vuoskoski, J.K.; Kautiainen, H. Being Moved by Unfamiliar Sad Music Is Associated with High Empathy. Front. Psychol. 2016, 7, 1176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zentner, M.; Grandjean, D.; Scherer, K.R. Emotions evoked by the sound of music: Characterization, classification, and measurement. Emotion 2008, 8, 494–521. [Google Scholar] [CrossRef] [Green Version]
- Frijda, N.H.; Sundararaja, L. Emotion Refinement: A Theory Inspired by Chinese Poetics. Perspect. Psychol. Sci. 2007, 2, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Zentner, M. Homer’s Prophecy:an Essay on Music’s Primary Emotions. Music Anal. 2010, 29, 102–125. [Google Scholar] [CrossRef]
- Ekman, P. An argument for basic emotions. Cogn. Emot. 1992, 6, 169–200. [Google Scholar] [CrossRef]
- Juslin, P.N.; Laukka, P. Expression, Perception, and Induction of Musical Emotions: A Review and a Questionnaire Study of Everyday Listening. J. New Music Res. 2004, 33, 217–238. [Google Scholar] [CrossRef]
- Russell, J.A. Core affect and the psychological construction of emotion. Psychol. Rev. 2003, 110, 145–172. [Google Scholar] [CrossRef]
- Chin, T.; Rickard, N.S. Emotion regulation strategy mediates both positive and negative relationships between music uses and well-being. Psychol. Music 2013, 42, 692–713. [Google Scholar] [CrossRef]
- Taruffi, L.; Skouras, S.; Pehrs, C.; Koelsch, S. Trait Empathy Shapes Neural Responses Toward Sad Music. Cogn. Affect. Behav. Neurosci. 2021, 21, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.; Garrido, S.; Hense, C.; McFerran, K. Music Use for Mood Regulation: Self-Awareness and Conscious Listening Choices in Young People With Tendencies to Depression. Front. Psychol. 2019, 10, 1199. [Google Scholar] [CrossRef] [PubMed]
- Diaz, F.M. Mindfulness, attention, and flow during music listening: An empirical investigation. Psychol. Music 2011, 41, 42–58. [Google Scholar] [CrossRef]
- Diaz, F.M. A Preliminary Investigation into the Effects of a Brief Mindfulness Induction on Perceptions of Attention, Aesthetic Response, and Flow during Music Listening; The Florida State University: Tallahassee, FL, USA, 2010. [Google Scholar]
- Ziegler, D.A.; Simon, A.J.; Gallen, C.L.; Skinner, S.; Janowich, J.R.; Volponi, J.J.; Rolle, C.E.; Mishra, J.; Kornfield, J.; Anguera, J.A.; et al. Closed-loop digital meditation improves sustained attention in young adults. Nat. Hum. Behav. 2019, 3, 746–757. [Google Scholar] [CrossRef]
- Liu, X.; Liu, Y.; Shi, H.; Li, L.; Zheng, M. Regulation of Mindfulness-Based Music Listening on Negative Emotions Related to COVID-19: An ERP Study. Int. J. Environ. Res. 2021, 18, 7063. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.; Panfil, K.; Bailey, C.; Kirkpatrick, K. Cognitive and behavioral training interventions to promote self-control. J. Exp. Psychol. Anim. Learn. Cogn. 2019, 45, 259–279. [Google Scholar] [CrossRef] [PubMed]
- Sayers, W.M.; Creswell, J.D.; Taren, A. The Emerging Neurobiology of Mindfulness and Emotion Processing. In Handbook of Mindfulness and Self-Regulation; Ostafin, B.D., Ed.; Springer Science+Business Media: New York, NY, USA, 2015; pp. 9–22. [Google Scholar]
- Kabat-Zinn, J. Full Catastrophe Living: Using the Wisdom of Your Body and Mind to Face Stress, Pain, and Illness.; Bantam Dell: New York, NY, USA, 1990. [Google Scholar]
- Liu, L.; Wang, H.; Chen, C. The Influence of Mindfulness Training Level on Emotional Processing. J. Psychol. Sci. 2016, 39, 1519–1524. [Google Scholar]
- Brown Warren, K.; Goodman Robert, J.; Michael, I. Dispositional mindfulness and the attenuation of neural responses to emotional stimuli. Soc. Cogn. Affect. Neurosci. 2012, 8, 93–99. [Google Scholar] [CrossRef] [Green Version]
- Carlson, L.E.; Brown, K.W. Validation of the Mindful Attention Awareness Scale in a cancer population. J. Psychosom. Res. 2005, 58, 29–33. [Google Scholar] [CrossRef]
- Deng, Y.-Q.; Li, S.; Tang, Y.-Y.; Zhu, L.-H.; Ryan, R.; Brown, K. Psychometric Properties of the Chinese Translation of the Mindful Attention Awareness Scale (MAAS). Mindfulness 2012, 3, 10–14. [Google Scholar] [CrossRef]
- Lau, M.A.; Bishop, S.R.; Segal, Z.V.; Buis, T.; Anderson, N.D.; Carlson, L.; Shapiro, S.; Carmody, J.; Abbey, S.; Devins, G. The toronto mindfulness scale: Development and validation. J. Clin. Psychol. 2006, 62, 1445–1467. [Google Scholar] [CrossRef] [PubMed]
- Bueno, V.F.; Kozasa, E.H.; da Silva, M.A.; Alves, T.M.; Louza, M.R.; Pompeia, S. Mindfulness Meditation Improves Mood, Quality of Life, and Attention in Adults with Attention Deficit Hyperactivity Disorder. Biomed. Res. Int. 2015, 2015, 962857. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.F.; Dienes, Z.; Jansari, A.; Goh, S.Y. Effect of mindfulness meditation on brain-computer interface performance. Conscious. Cogn. 2014, 23, 12–21. [Google Scholar] [CrossRef]
- Davidson, R.J.; Kabat-Zinn, J.; Schumacher, J.; Rosenkranz, M.; Muller, D.; Santorelli, S.F.; Urbanowski, F.; Harrington, A.; Bonus, K.; Sheridan, J.F. Alterations in brain and immune function produced by mindfulness meditation. Psychosom. Med. 2003, 65, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Davidson, R.J.; Kaszniak, A.W. Conceptual and methodological issues in research on mindfulness and meditation. Am. Psychol. 2015, 70, 581–592. [Google Scholar] [CrossRef]
- Tang, Y.Y.; Hölzel, B.K.; Posner, M.I. The neuroscience of mindfulness meditation. Nat. Rev. Neurosci. 2015, 16, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Hölzel, B.K.; Lazar, S.W.; Gard, T.; Schuman-Olivier, Z.; Vago, D.R.; Ott, U. How Does Mindfulness Meditation Work? Proposing Mechanisms of Action From a Conceptual and Neural Perspective. Perspect. Psychol. Sci. 2011, 6, 537–559. [Google Scholar] [CrossRef]
- Bailey, N.W.; Freedman, G.; Raj, K.; Sullivan, C.M.; Rogasch, N.C.; Chung, S.W.; Hoy, K.E.; Chambers, R.; Hassed, C.; Van Dam, N.T.; et al. Mindfulness meditators show altered distributions of early and late neural activity markers of attention in a response inhibition task. PLoS ONE 2019, 14, e0203096. [Google Scholar] [CrossRef] [Green Version]
- Diamond, E.; Zhang, Y. Cortical processing of phonetic and emotional information in speech: A cross-modal priming study. Neuropsychologia 2016, 82, 110–122. [Google Scholar] [CrossRef] [Green Version]
- Hajcak, G.; MacNamara, A.; Foti, D.; Ferri, J.; Keil, A. The dynamic allocation of attention to emotion: Simultaneous and independent evidence from the late positive potential and steady state visual evoked potentials. Biol. Psychol. 2013, 92, 447–455. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, H.; Cui, Y. The influence of overweight/obesity on adolescent executive control and intervention methods. J. Southwest Univ. (Soc. Sci. Ed.) 2020, 46, 108–118. [Google Scholar]
- Zhang, Q.; Kong, L.; Jiang, Y. The interaction of arousal and valence in affective priming: Behavioral and electrophysiological evidence. Brain Res. 2012, 1474, 60–72. [Google Scholar] [CrossRef] [Green Version]
- Steinbeis, N.; Koelsch, S. Affective priming effects of musical sounds on the processing of word meaning. J. Cogn. Neurosci. 2011, 23, 604–621. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jun, J.; Weixia, Z.; Wanqi, W. Emotional processing in vocal and instrumental music: An ERP study. Adv. Psychol. Sci. 2020, 28, 1133–1140. [Google Scholar]
- Wu, R.; Liu, L.L.; Zhu, H.; Su, W.J.; Cao, Z.Y.; Zhong, S.Y.; Liu, X.H.; Jiang, C.L. Brief Mindfulness Meditation Improves Emotion Processing. Front. Neurosci. 2019, 13, 1074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fazio, R.H.; Sanbonmatsu, D.M.; Powell, M.C.; Kardes, F.R. On the automatic activation of attitudes. J. Personal. Soc. Psychol. 1986, 50, 229. [Google Scholar] [CrossRef]
- Huang, Y.-X.; Luo, Y.-J. Temporal course of emotional negativity bias: An ERP study. Neurosci. Lett. 2006, 398, 91–96. [Google Scholar] [CrossRef]
- Ito, T.A.; Larsen, J.T.; Smith, N.K.; Cacioppo, J.T. Negative information weighs more heavily on the brain: The negativity bias in evaluative categorizations. J. Personal. Soc. Psychol. 1998, 75, 887–900. [Google Scholar] [CrossRef]
- Yuan, J.; Zhang, Q.; Chen, A.; Li, H.; Wang, Q.; Zhuang, Z.; Jia, S. Are we sensitive to valence differences in emotionally negative stimuli? Electrophysiological evidence from an ERP study. Neuropsychologia 2007, 45, 2764–2771. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, L.; Jackson, T.; Wang, J.; Yang, R.; Chen, H. Effects of negative mood state on event-related potentials of restrained eating subgroups during an inhibitory control task. Behav. Brain Res. 2020, 377, 112249. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.; Clark, L.A. Development and validation of brief measures of positive and negative affect: The PANAS scales. J. Personal. Soc. Psychol. 1988, 54, 1063–1070. [Google Scholar] [CrossRef]
- Si-yi, C.; Hong, C.; Ren-lai, Z.; Yan-yan, J. Revision of Mindful Attention Awareness Scale (MAAS). Chin. J. Clin. Psychol. 2012, 20, 148–151. [Google Scholar]
- Chung, P.-K.; Zhang, C.-Q. Psychometric Validation of the Toronto Mindfulness Scale—Trait Version in Chinese College Students. Eur. J. Psychol. 2014, 10, 726–739. [Google Scholar] [CrossRef] [Green Version]
- Ireland, M.J.; Day, J.J.; Clough, B.A. Exploring scale validity and measurement invariance of the Toronto Mindfulness Scale across levels of meditation experience and proficiency. J. Clin. Psychol. 2019, 75, 445–461. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.; Ding, F.; Zhang, W.; Li, N.; Zhang, D. Initial establishment of the same face with multi-expressions’ image database for infants and adults. Chin. Ment. Health J. 2019, 33, 918–924. [Google Scholar]
- Olichney, J.M.; Riggins, B.R.; Hillert, D.G.; Nowacki, R.; Tecoma, E.; Kutas, M.; Iragui, V.J. Reduced Sensitivity of the N400 and Late Positive Component to Semantic Congruity and Word Repetition in Left Temporal Lobe Epilepsy. Clin. EEG Neurosci. 2002, 33, 111–118. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, J.; Zhang, X.; Gao, X.; Xu, W.; Chen, H. Overweight adults are more impulsive than normal weight adults: Evidence from ERPs during a chocolate-related delayed discounting task. Neuropsychologia 2019, 133, 107181. [Google Scholar] [CrossRef] [PubMed]
- Hölzel, B.K.; Carmody, J.; Vangel, M.; Congleton, C.; Yerramsetti, S.M.; Gard, T.; Lazar, S.W. Mindfulness practice leads to increases in regional brain gray matter density. Psychiatry Res. 2011, 191, 36–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moynihan, J.A.; Chapman, B.P.; Klorman, R.; Krasner, M.S.; Talbot, N.L. Mindfulness-Based Stress Reduction for Older Adults: Effects on Executive Function, Frontal Alpha Asymmetry and Immune Function. Neuropsychobiology 2013, 68, 34–43. [Google Scholar] [CrossRef] [Green Version]
- Yuan, J.; He, Y.; Qinglin, Z.; Chen, A.; Li, H. Gender differences in behavioral inhibitory control: ERP evidence from a two-choice oddball task. Psychophysiology 2008, 45, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, J.K.; Nordin, S.; Sequeira, H.; Polich, J. Affective picture processing: An integrative review of ERP findings. Biol. Psychol. 2008, 77, 247–265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Madsen, J.; Margulis, E.H.; Simchy-Gross, R.; Parra, L.C. Music synchronizes brainwaves across listeners with strong effects of repetition, familiarity and training. Sci. Rep. 2019, 9, 3576. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.J.; Andrade, E.B.; Palmer, S.E. Interpersonal Relationships and Preferences for Mood-Congruency in Aesthetic Experiences. J. Consum. Res. 2013, 40, 382–391. [Google Scholar] [CrossRef]
| Variables | HTG (M ± SD) | LTG (M ± SD) | t | |
|---|---|---|---|---|
| n = 31 | n = 31 | |||
| Age | 20.45 (2.46) | 20.03 (1.47) | 0.81 | |
| Sex | Male = 9, female = 21 | Male = 6, female = 25 | ||
| PANAS | PA | 2.55 (0.77) | 2.32 (0.77) | 1.29 |
| NA | 1.24 (0.34) | 1.48 (0.54) | 1.06 | |
| TMS *** | pre-test | 33.77 (5.59) | 33.94 (3.95) | 0.13 |
| post-test | 36.06 (5.13) | 35.03 (4.69) | 0.83 | |
| MAAS *** | pre-test | 67.84 (6.08) | 49.35 (6.62) | 11.45 |
| post-test | 68.35 (8.33) | 51.61 (11.11) | 6.71 | |
| Variables | HTG (M ± SD) | LTG (M ± SD) | |||||
|---|---|---|---|---|---|---|---|
| n = 31 | n = 31 | ||||||
| MELs | CM | HM | SM | CM | HM | SM * | |
| FR | ACC (pre-test) | 0.85 (0.13) | 0.87 (0.16) | 0.84 (0.14) | 0.84 (0.13) * | 0.81 (0.20) * | 0.81 (0.15) * |
| ACC (post-test) | 0.88 (0.16) | 0.84 (0.27) | 0.83 (0.20) | 0.89 (0.08) * | 0.84 (0.22) * | 0.85 (0.14) * | |
| RTs (pre-test) | 1249.35 (98.53) | 990.24 (146.33) | 1260.40 (140.16) | 1244.06 (180.83) | 969.95 (161.59) | 1249.87 (178.15) | |
| RTs (post-test) *** | 1182.51 (120.15) | 933.15 (159.44) | 1142.08 (113.97) | 1098.00 (162.02) | 804.39 (174.47) | 1063.99 (165.88) | |
| EE | Arousal (pre-test) | 4.94 (1.49) * | 6.26 (1.06) * | 6.29 (1.44) * | 4.91 (1.18) | 5.98 (1.19) | 5.93 (1.27) |
| Arousal (post-test) | 4.78 (0.68) * | 5.94 (1.24) * | 6.04 (1.45) * | 4.98 (1.24) | 5.98 (1.28) | 6.10 (1.55) | |
| RTs (pre-test) | 879.71 (217.31) | 783.92 (193.73) | 839.47 (225.83) | 834.12 (244.99) | 781.94 (249.18) | 930.89 (270.46) | |
| RTs (post-test) ** | 723.24 (226.59) | 687.23 (232.43) | 731.09 (222.66) | 604.07 (197.26) | 550.67 (186.84) | 597.28 (231.15) | |
| Variables | HTG (M ± SD) | LTG (M ± SD) | ||||
|---|---|---|---|---|---|---|
| n = 31 | n = 31 | |||||
| MELs | CM | HM * | SM | CM | HM * | SM |
| PAEs (pre-test) | 2.47 (0.74) * | 2.21 (0.78) | 2.36 (0.71) | 2.35 (0.57) | 2.27 (0.66) | 2.31 (0.60) |
| PAEs (post-test) | 2.13 (0.67) * | 2.51 (0.83) | 2.24 (0.81) | 2.10 (0.51) | 2.47 (0.65) | 2.32 (0.74) |
| PEs (pre-test) | 2.44 (0.66) ** | 2.27 (0.81) | 3.22 (0.81) *** | 2.29 (0.67) | 2.14 (0.61) | 2.92 (0.72) *** |
| PEs (post-test) | 3.08 (0.92) ** | 1.82 (0.68) | 1.81 (0.54) *** | 2.72 (0.82) | 1.89 (0.74) | 1.85 (0.54) *** |
| EEs (pre-test) | 2.30 (0.69) | 2.14 (0.83) | 2.70 (0.75) *** | 2.04 (0.56) | 2.20 (0.69) | 2.51 (0.61) *** |
| EEs (post-test) | 2.39 (0.78) | 2.38 (0.69) | 2.10 (0.64) *** | 2.26 (0.62) | 2.35 (0.69) | 2.16 (0.73) *** |
| NEs (pre-test) | 1.46 (0.48) ** | 1.31 (0.36) | 1.24 (0.33) *** | 1.58 (0.42) | 1.49 (0.58) | 1.41 (0.54) *** |
| NEs (post-test) | 1.24 (0.38) ** | 1.95 (0.66) | 1.74 (0.46) *** | 1.38 (0.47) | 1.86 (0.54) | 1.87 (0.51) *** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Shi, H.; Liu, Y.; Yuan, H.; Zheng, M. Mindfulness Meditation Improves Musical Aesthetic Emotion Processing in Young Adults. Int. J. Environ. Res. Public Health 2021, 18, 13045. https://doi.org/10.3390/ijerph182413045
Liu X, Shi H, Liu Y, Yuan H, Zheng M. Mindfulness Meditation Improves Musical Aesthetic Emotion Processing in Young Adults. International Journal of Environmental Research and Public Health. 2021; 18(24):13045. https://doi.org/10.3390/ijerph182413045
Chicago/Turabian StyleLiu, Xiaolin, Huijuan Shi, Yong Liu, Hong Yuan, and Maoping Zheng. 2021. "Mindfulness Meditation Improves Musical Aesthetic Emotion Processing in Young Adults" International Journal of Environmental Research and Public Health 18, no. 24: 13045. https://doi.org/10.3390/ijerph182413045
APA StyleLiu, X., Shi, H., Liu, Y., Yuan, H., & Zheng, M. (2021). Mindfulness Meditation Improves Musical Aesthetic Emotion Processing in Young Adults. International Journal of Environmental Research and Public Health, 18(24), 13045. https://doi.org/10.3390/ijerph182413045







