Towards a Comprehensive Strategy for the Management of Rare Diseases in Slovenia: Outlining an IT-Enabled Ecosystemic Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Research Design
2.2. Sample
2.3. Data Collection
2.4. Data Analysis
3. Results
3.1. A Cross Section of the Critical Aspects in the Field
3.1.1. Policy and Normative Aspects
3.1.2. Institutional and Organizational Aspects
3.1.3. Digitalization and Data Management Aspects
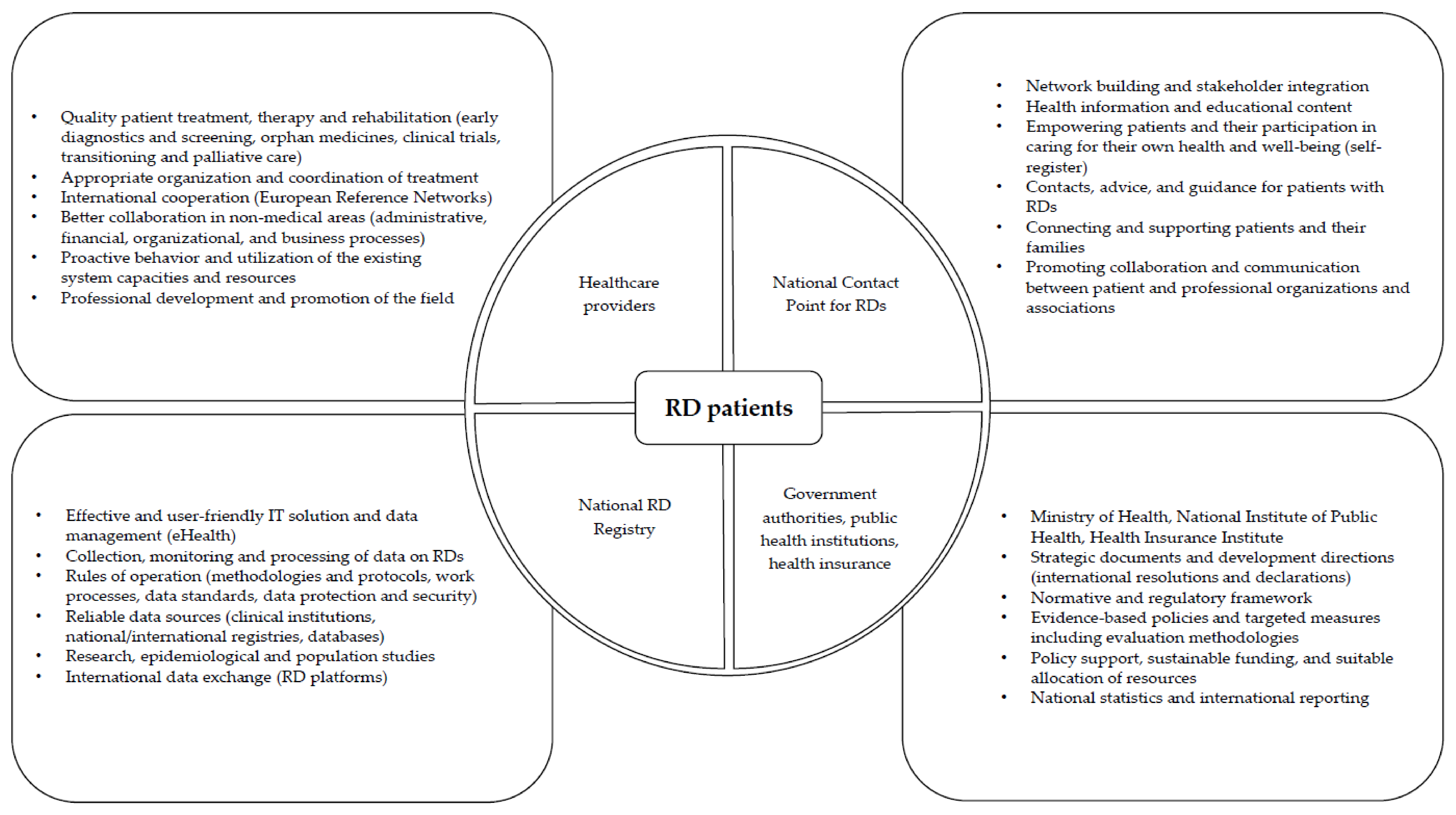
3.2. Outlining an IT-Enabled Ecosystemic Approach
3.2.1. The National RD Registry
3.2.2. NCP for RDs
3.2.3. Health Insurance and Orphan Medicines for RDs
3.2.4. ERNs and Patient Organizations for RDs
3.2.5. Early and Specialized Diagnostics of RDs
3.2.6. Transitioning Care and Palliative Care in RDs
4. Discussion
- The absence of an up-to-date policy framework, strategic documents, action plan, and evaluation metrics, including public health objectives;
- Normative deficiencies and systemic/institutional non-compliance with regulations in the field;
- Non-optimal institutional collaboration and coordination, and rather unsettled organizational and processual matters;
- A substantial lack of resources (material, human, IT, organizational);
- Inconsistent clinical and institutional practice in certain segments of the coding procedures; and
- Isolated, non-user-friendly, and ineffective IT tools and platforms in the entire field of RDs.
Methodological Limitations and Forthcoming Research Orientations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Ministry of Health. Work Plan in the Field of Rare Diseases in the Republic of Slovenia; Ministry of Health of the Republic of Slovenia: Ljubljana, Slovenia, 2011.
- Groselj, U. Analysis and Development in the Field of Rare Diseases in Slovenia; 6th Edition of Rare Diseases; Society of Patients with Blood Diseases of Slovenia: Ljubljana, Slovenia, 2016. [Google Scholar]
- Organisation for Economic Co-operation and Development (OECD). Health at a Glance 2019: OECD Indicators; OECD Publishing: Paris, France, 2019. [Google Scholar] [CrossRef]
- Albreht, T.; Pribakovic, R.; Josar, D.; Poldrugovac, M.; Kostnapfel, T.; Zaletel, M.; Panteli, D.; Maresso, A. Slovenia: Health System Review; World Health Organization (WHO), Regional Office for Europe, European Observatory on Health Systems and Policies: Brussels, Belgium, 2016. [Google Scholar]
- Ministry of Health. Resolution on the National Health Care Plan 2016–2025; Ministry of Health of the Republic of Slovenia: Ljubljana, Slovenia, 2016.
- Valdez, R.; Ouyang, L.; Bolen, J. Public health and rare diseases: Oxymoron no more. Prev. Chronic Dis. 2016, 13, E05. [Google Scholar] [CrossRef]
- Kodra, Y.; Cavazza, M.; de Santis, M.; Guala, A.; Liverani, M.-E.; Armeni, P.; Masini, M.; Taruscio, D. Social Economic Costs, Health-Related Quality of Life and Disability in Patients with Cri Du Chat Syndrome. Int. J. Environ. Res. Public Health 2020, 17, 5951. [Google Scholar] [CrossRef]
- Stanimirovic, D.; Murko, E.; Battelino, T.; Groselj, U. Development of a pilot rare disease registry: A focus group study of initial steps towards the establishment of a rare disease ecosystem in Slovenia. Orphanet. J. Rare Dis. 2019, 14, 172. [Google Scholar] [CrossRef]
- Stanimirovic, D. A framework for information and communication technology induced transformation of the healthcare business model in Slovenia. J. Glob. Inf. Technol. Manag. 2015, 18, 29–47. [Google Scholar] [CrossRef]
- Witte, A.-K. A Review on Digital Healthcare Ecosystem Structure: Identifying Elements and Characteristics. In Proceedings of the Pacific Asia Conference on Information Systems (PACIS) 2020, 22–24 June 2020; Association for Information Systems: Dubai, United Arab Emirates, 2020. [Google Scholar]
- Sklyar, A.; Kowalkowski, C.; Tronvoll, B.; Sörhammar, D. Organizing for digital servitization: A service ecosystem perspective. J. Bus. Res. 2019, 104, 450–460. [Google Scholar] [CrossRef]
- Thomas, D.R. A general inductive approach for analyzing qualitative evaluation data. Am. J. Eval. 2006, 27, 237–246. [Google Scholar] [CrossRef]
- Yin, R.K. Case Study Research and Applications: Design and Methods; Sage Publications: Thousand Oaks, CA, USA, 2017. [Google Scholar]
- Hsieh, H.F.; Shannon, S.E. Three approaches to qualitative content analysis. Qual. Health Res. 2005, 15, 1277–1288. [Google Scholar] [CrossRef]
- The Council of the European Union. Council Recommendation of 8 June 2009 on an Action in the Field of Rare Diseases (2009/C 151/02). Official Journal of the European Union; C 151/7; European Commission: Brussels, Belgium, 2009. [Google Scholar]
- Wakap, S.N.; Lambert, D.M.; Olry, A.; Rodwell, C.; Gueydan, C.; Lanneau, V.; Murphy, D.; Le Cam, Y.; Rath, A. Estimating cumulative point prevalence of rare diseases: Analysis of the Orphanet database. Eur. J. Hum. Genet. 2020, 28, 165–173. [Google Scholar] [CrossRef]
- Taruscio, D.; Mollo, E.; Gainotti, S.; de la Paz, M.P.; Bianchi, F.; Vittozzi, L. The EPIRARE proposal of a set of indicators and common data elements for the European platform for rare disease registration. Arch. Public Health 2014, 72, 35. [Google Scholar] [CrossRef]
- White, M.D.; Marsh, E.E. Content Analysis: A Flexible Methodology. Libr. Trends 2006, 55, 22–45. [Google Scholar] [CrossRef]
- Krippendorff, K. Content Analysis: An Introduction to Its Methodology; Sage Publications: Thousand Oaks, CA, USA, 2018. [Google Scholar]
- Harwood, T.G.; Garry, T. An overview of content analysis. Mark. Rev. 2003, 3, 479–498. [Google Scholar] [CrossRef]
- Neuendorf, K.A. The Content Analysis Guidebook; Sage Publications: Los Angeles, CA, USA, 2017. [Google Scholar]
- ZZPPZ. Healthcare Databases Act; No. 65/00, 47/15, 31/18; Official Gazette of the Republic of Slovenia: Ljubljana, Slovenia, 2021. [Google Scholar]
- Ayme, S.; Bellet, B.; Rath, A. Rare diseases in ICD11: Making rare diseases visible in health information systems through appropriate coding. Orphanet J. Rare Dis. 2015, 10, 35. [Google Scholar] [CrossRef]
- Garcelon, N.; Burgun, A.; Salomon, R.; Neuraz, A. Electronic health records for the diagnosis of rare diseases. Kidney Int. 2020, 97, 676–686. [Google Scholar] [CrossRef]
- Boulanger, V.; Schlemmer, M.; Rossov, S.; Seebald, A.; Gavin, P. Establishing Patient Registries for Rare Diseases: Rationale and Challenges. Pharm. Med. 2020, 25, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kodra, Y.; Weinbach, J.; Posada-de-la-Paz, M.; Coi, A.; Lemonnier, S.L.; Van Enckevort, D.; Roos, M.; Jacobsen, A.; Cornet, R.; Ahmed, S.F.; et al. Recommendations for Improving the Quality of Rare Disease Registries. Int. J. Environ. Res. Public Health 2018, 15, 1644. [Google Scholar] [CrossRef]
- European Union Committee of Experts on Rare Diseases (EUCERD). Recommendations on Rare Disease Patient Registration and Data Collection; European Commission: Brussels, Belgium, 2013. [Google Scholar]
- Haendel, M.; Vasilevsky, N.; Unni, D.; Bologa, C.; Harris, N.; Rehm, H.; Hamosh, A.; Baynam, G.; Groza, T.; McMurry, J.; et al. How many rare diseases are there? Nat. Rev. Drug Discov. 2019, 19, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Lapeyre-Mestre, M. The challenges of pharmacoepidemiology of orphan drugs in rare diseases. Therapies 2020, 75, 215–220. [Google Scholar] [CrossRef]
- Blumenrath, S.H.; Lee, B.Y.; Low, L.; Prithviraj, R.; Tagle, D. Tackling rare diseases: Clinical trials on chips. Exp. Biol. Med. 2020, 245, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Tambuyzer, E.; Vandendriessche, B.; Austin, C.P.; Brooks, P.J.; Larsson, K.; Needleman, K.I.M.; Valentine, J.; Davies, K.; Groft, S.C.; Preti, R.; et al. Therapies for rare diseases: Therapeutic modalities, progress and challenges ahead. Nat. Rev. Drug Discov. 2019, 19, 93–111. [Google Scholar] [CrossRef]
- EPIRARE Project. European Platform for Rare Disease Registries. Specific Objectives. Available online: http://www.epirare.eu/ (accessed on 15 September 2021).
- European Platform on Rare Disease Registration (EU RD Platform). Aim of the Platform. Available online: https://eu-rd-platform.jrc.ec.europa.eu/aim-of-the-platform_en (accessed on 2 September 2021).
- National Contact Point for Rare Diseases (NCP). Rare Diseases and Treatment. Available online: https://www.redkebolezni.si/ (accessed on 9 August 2021).
- Houyez, F.; Sanchez de Vega, R.; Brignol, T.N.; Mazzucato, M.; Polizzi, A. A European Network of Email and Telephone Help Lines Providing Information and Support on Rare Diseases: Results from a 1-Month Activity Survey. Interact. J. Med. Res. 2014, 3, e9. [Google Scholar] [CrossRef][Green Version]
- ZZVZZ. Health Care and Health Insurance Act; No. 72/06, 114/06, 91/07, 76/08, 62/10, 87/11, 40/12, 21/13, 91/13, 99/13, 99/13, 111/13, 95/14, 47/15, 61/17, 64/17, 36/19; Official Gazette of the Republic of Slovenia: Ljubljana, Slovenia, 2021. [Google Scholar]
- Heard, J.M.; Vrinten, C.; Schlander, M.; Bellettato, C.M.; van Lingen, C.; Scarpa, M.; MetabERN Collaboration Group. Availability, accessibility and delivery to patients of the 28 orphan medicines approved by the European Medicine Agency for hereditary metabolic diseases in the MetabERN network. Orphanet. J. Rare Dis. 2020, 15, 3. [Google Scholar] [CrossRef] [PubMed]
- Rare Disease Day 2020. 6th National Conference. 3 March 2020. Available online: http://www.drustvo-bkb.si/media/moddoc_20_ca20df1b1a321f5c1c4608.pdf (accessed on 11 August 2021).
- Clinical Patient Management System (CPMS). Available online: https://cpms.ern-net.eu/login/ (accessed on 1 August 2021).
- Heon-Klin, V. European Reference networks for rare diseases: What is the conceptual framework? Orphanet. J. Rare Dis. 2017, 12, 137. [Google Scholar] [CrossRef] [PubMed]
- Association for Rare Diseases of Slovenia. Available online: https://zrbs.si/ (accessed on 13 August 2021).
- Bryson, B.A.; Bogart, K.R. Social support, stress, and life satisfaction among adults with rare diseases. Health Psychol. 2020, 39, 912–920. [Google Scholar] [CrossRef] [PubMed]
- Repic Lampret, B.; Remec, Z.I.; Drole Torkar, A.; Zerjav Tansek, M.; Smon, A.; Koracin, V.; Cuk, V.; Perko, D.; Ulaga, B.; Jelovsek, A.M.; et al. Expanded newborn screening program in Slovenia using tandem mass spectrometry and confirmatory next generation sequencing genetic testing. Slov. J. Public Health 2020, 59, 256–263. [Google Scholar] [CrossRef]
- Smon, A.; Repic Lampret, B.; Groselj, U.; Zerjav Tansek, M.; Kovac, J.; Perko, D.; Bertok, S.; Battelino, T.; Trebusak Podkrajsek, K. Next generation sequencing as a follow-up test in an expanded newborn screening programme. Clin. Biochem. 2018, 52, 48–55. [Google Scholar] [CrossRef]
- Singh, S.P.; Anderson, B.; Liabo, K.; Ganeshamoorthy, T. Supporting young people in their transition to adults’ services: Summary of NICE guidance. BMJ 2016, 353, i2225. [Google Scholar] [CrossRef]
- Lyon, M.E.; Thompkins, J.D.; Fratantoni, K.; Fraser, J.L.; Schellinger, S.E.; Briggs, L.; Friebert, S.; Aoun, S.; Cheng, Y.I.; Wang, J. Family caregivers of children and adolescents with rare diseases: A novel palliative care intervention. BMJ Support Palliat Care 2019. [Google Scholar] [CrossRef]
- Ferreira, C.R. The burden of rare diseases. Am. J. Med Genet. Part A 2019, 179, 885–892. [Google Scholar] [CrossRef]
- Crowe, A.L.; McKnight, A.J.; McAneney, H. Communication Needs for Individuals with Rare Diseases within and Around the Healthcare System of Northern Ireland. Front. Public Health 2019, 7, 236. [Google Scholar] [CrossRef]
- Hirsch, M.C.; Ronicke, S.; Krusche, M.; Wagner, A.D. Rare diseases 2030: How augmented AI will support diagnosis and treatment of rare diseases in the future. Ann. Rheum. Dis. 2020, 79, 740–743. [Google Scholar] [CrossRef]
- Elo, S.; Kyngäs, H. The qualitative content analysis process. J. Adv. Nurs. 2008, 62, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Franzosi, R.; Doyle, S.; McClelland, L.E.; Putnam Rankin, C.; Vicari, S. Quantitative narrative analysis software options compared: PC-ACE and CAQDAS (ATLAS.ti, MAXqda, and NVivo). Qual. Quant. 2013, 47, 3219–3247. [Google Scholar] [CrossRef]
- Duriau, V.J.; Reger, R.K.; Pfarrer, M.D. A content analysis of the content analysis literature in organization studies: Research themes, data sources, and methodological refinements. Organ. Res. Methods 2007, 10, 5–34. [Google Scholar] [CrossRef]
- Shapiro, G.; Markoff, G. Text Analysis for the Social Sciences: Methods for Drawing Statistical Inferences from Text and Transcripts; Roberts, C.W., Ed.; Lawrence Erlbaum Associates: Mahwah, NJ, USA, 1997; pp. 9–31. [Google Scholar]
- Roberts, C.W. A conceptual framework for quantitative text analysis. Qual. Quant. 2000, 34, 259–274. [Google Scholar] [CrossRef]
| Institution | Number of Participating Experts | Areas of Work and Research |
|---|---|---|
| University Medical Center Ljubljana (UMCL)/University of Ljubljana, Faculty of Medicine | 11 | Pediatric and adult endocrinology and diabetology, pediatric immunology, epidemiology, molecular genetics, inherited metabolic diseases, clinical genetics, human reproduction, neurogenetics, dysmorphology, neurology, pharmacy, biochemistry and molecular biology, clinical biochemistry, laboratory medicine |
| National Institute of Public Health | 3 | Public health, the healthcare system, healthcare system organization and resources, health insurance and reimbursements, healthcare data, data management, methodologies, statistics, and data analytics |
| University Medical Center Maribor/University of Maribor, Faculty of Medicine | 6 | Microbiology and immunology, human reproduction, medical genetics and cytogenetics, biochemistry and molecular biology, embryogenesis and dysmorphology, blood cancer, atherosclerosis and rare cardiovascular diseases, molecular genetics, molecular oncology |
| Slovenj Gradec General Hospital | 2 | The cardiovascular system, metabolic and hormonal disorders |
| IT companies | 2 | Systems and cybernetics, computer science and informatics, IT architecture, electronic health records, registries, health information systems, IT tools, web apps and platforms for patients, IT system infrastructure, designing and building IT solutions, data standards, data security and privacy (according to the General Data Protection Regulation (GDPR) and safe-by-design principle) |
| Critical Aspects | Position of the Focus Group Participants |
|---|---|
| Policy and normative aspects | 24/24 respondents agreed |
| Institutional and organizational aspects | 24/24 respondents agreed |
| Digitalization and data management aspects | 24/24 respondents agreed |
| Questions Identified by the Focus Group Participants | |
|---|---|
| Q1 | How is the strategic (policy) and regulatory framework currently regulated and does it offer all the necessary bases for further development in the field of RDs? |
| Q2 | Who are the main stakeholders in the field, what are their roles, and what competences and powers should they have? |
| Q3 | What should the starting points be for the preparation of the relevant strategic and development orientations, including sectoral policies, operational measures, and the evaluation framework? |
| Q4 | What actions should be taken by the government to ensure continuous development in the field and the best patient treatment under the given circumstances? |
| Q5 | What are the main benefits regarding the treatment of patients that we want to ensure by introducing an IT-enabled ecosystemic approach in the field of RDs in Slovenia? |
| Q6 | How to facilitate the appropriate arrangement and mobilization of all necessary experts and resources in Slovenia and abroad for the treatment of patients with RDs? |
| Q7 | What activities and systemic measures are additionally needed by healthcare providers for more efficient management of RDs in Slovenia? |
| Q8 | How to ensure the use of available systemic and institutional potentials to improve patient treatment and enable more effective management of RDs? |
| Q9 | How to establish a comprehensive IT infrastructure that will connect stakeholders and provide adequate support for working with patients? |
| Q10 | Which local or national IT solutions need to be established and which functionalities and operations should these IT solutions enable? |
| Q11 | What procedural, methodological, organizational and other preconditions must be put in place for the successful use of the potentials offered by IT solutions? |
| Q12 | What conditions must the designed IT solutions meet and what goals should they serve in terms of the secondary use of data and international cooperation? |
| Q13 | What information and services should the NCP provide to patients (and their families) on a personal level and what information and services to the general public? |
| Q14 | What information and services should the NCP provide to healthcare professionals and what information and services to patient associations and government institutions? |
| Q15 | What are the longstanding goals of the NCP in terms of its development and use? |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stanimirovic, D.; Murko, E.; Battelino, T.; Groselj, U.; Zerjav Tansek, M. Towards a Comprehensive Strategy for the Management of Rare Diseases in Slovenia: Outlining an IT-Enabled Ecosystemic Approach. Int. J. Environ. Res. Public Health 2021, 18, 12395. https://doi.org/10.3390/ijerph182312395
Stanimirovic D, Murko E, Battelino T, Groselj U, Zerjav Tansek M. Towards a Comprehensive Strategy for the Management of Rare Diseases in Slovenia: Outlining an IT-Enabled Ecosystemic Approach. International Journal of Environmental Research and Public Health. 2021; 18(23):12395. https://doi.org/10.3390/ijerph182312395
Chicago/Turabian StyleStanimirovic, Dalibor, Eva Murko, Tadej Battelino, Urh Groselj, and Mojca Zerjav Tansek. 2021. "Towards a Comprehensive Strategy for the Management of Rare Diseases in Slovenia: Outlining an IT-Enabled Ecosystemic Approach" International Journal of Environmental Research and Public Health 18, no. 23: 12395. https://doi.org/10.3390/ijerph182312395
APA StyleStanimirovic, D., Murko, E., Battelino, T., Groselj, U., & Zerjav Tansek, M. (2021). Towards a Comprehensive Strategy for the Management of Rare Diseases in Slovenia: Outlining an IT-Enabled Ecosystemic Approach. International Journal of Environmental Research and Public Health, 18(23), 12395. https://doi.org/10.3390/ijerph182312395







