Optimization of Biomethane Production via Fermentation of Chicken Manure Using Marine Sediment: A Modeling Approach Using Response Surface Methodology
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Characterization of CM, MS, and OS
2.3. Fermentation Mixtures
2.4. Analytical Methods
2.5. Effect of Inoculum Size and NaCl Supplementation on the Biomethanation of CM Using MS or OS as the Inoculum Source
2.6. Effect of the Total Solid Content of CM on Biomethanation Using MS as an Inoculum Source
2.7. Optimization of CM Biomethanation Using MS as an Inoculum Source via the Design of Experiments and Statistical Modeling
2.8. Studying the Tolerance of Methanogenic Bacteria in MS to Increasing Ammonia Concentration
3. Results
3.1. Effect of Inoculum Size and NaCl Supplementation on Biomethanation of CM Using MS or OS as an Inoculum Source
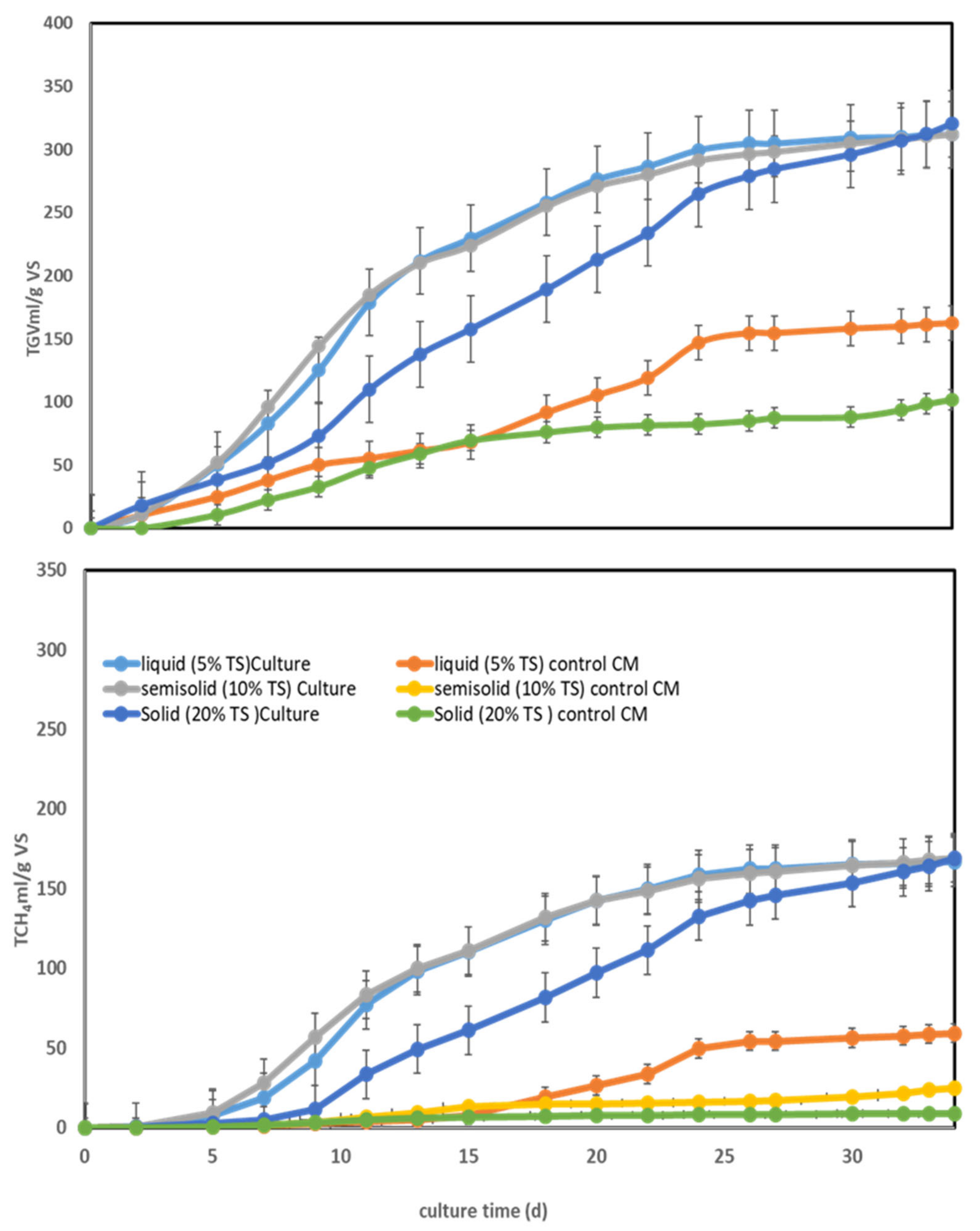
3.2. Effect of the Total Solid Content of CM on Biomethanation Using Methanogenic Consortia in MS
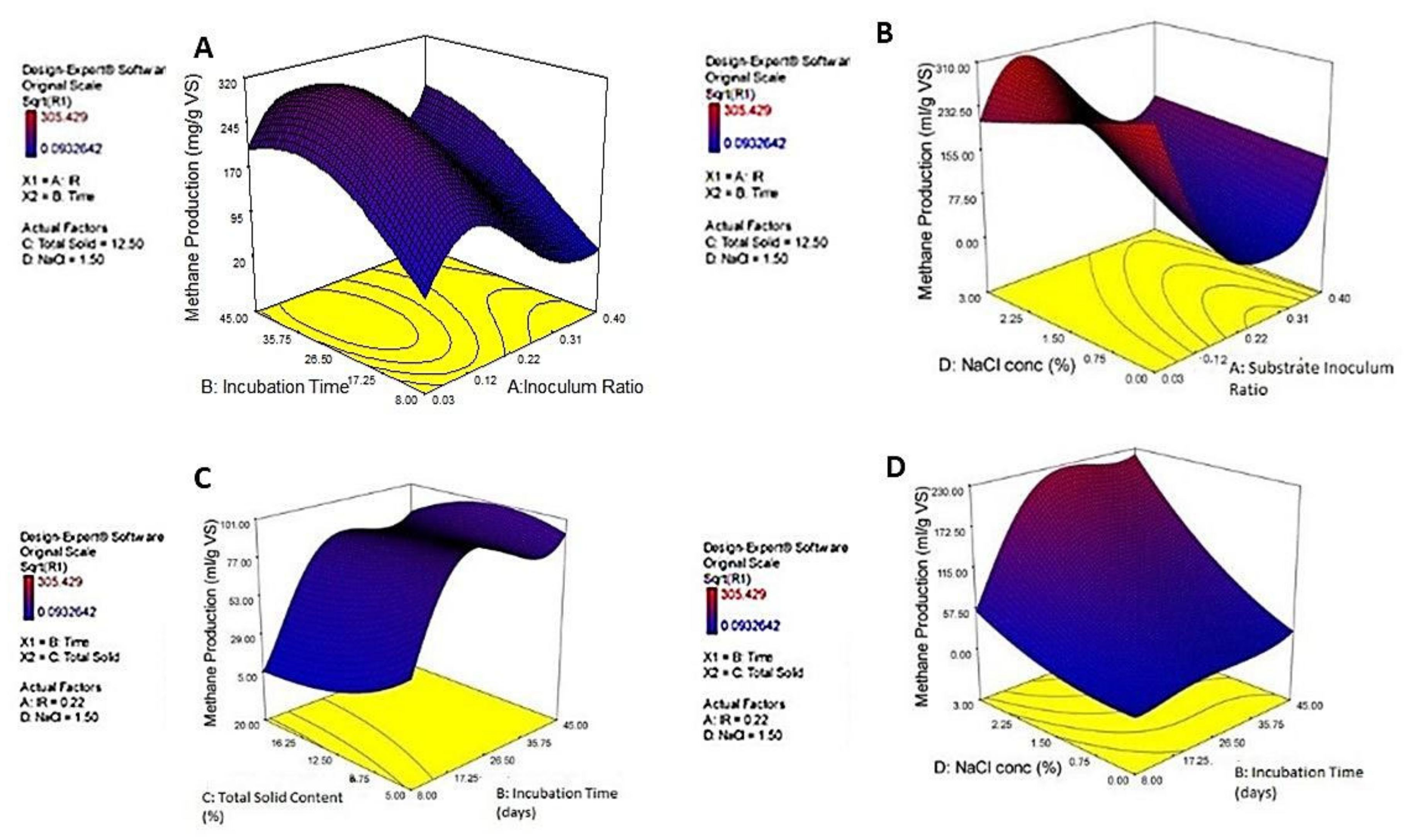
3.3. Optimization of CM Biomethanation Using MS as an Inoculum Source via the Design of Experiments and Statistical Modeling
3.4. Quality Assurance/Quality Control (QA/QC)
3.5. Studying the Tolerance of Methanogenic Bacteria in MS to Increasing Concentrations of Ammonia
4. Discussion
| Co- Substrate and/or Inoculum Source | Experimental Conditions | ||||||
|---|---|---|---|---|---|---|---|
| RetentionTime (d) | Temperature (°C) | TS% | Ratios of Substrates | Methane (CH4) or Biogas Yields | (NH3) or (NH4+) Values | References | |
| Poultry (P) and hogwastes (H) |
|
|
|
| CH4 = 0.9 mL g−1 VS | 1.66 g NH3 L−1 | [36] |
| Inoculum obtained from sludge(Ozouh) obtained after thermophilic anaerobic digestion of excess activated sludge | 254 d254 d | 35 °C55 °C | 25%25% |
|
|
| [32] |
| Agriculture wastes (AWS) | Batch I, 2, 3 and 4 = 40 d, 35, 39 and 62 d | 35 and 55 °C | 10% | CM:AWS is 7:3 (v/v) | 506 CH4 mL/gVS | 1.3 g NH3-N/kg−1 CM | [32] |
| Inoculum obtained from cattle manure under mesophilic conditions | 50 d | 35 °C | 14–32% | (1:1) to (1:7) ratio of CM and inoculum | 73.3 NmL g−1 VS biogas | - | [7] |
| Mesophilic acclimated inoculum from a lab-scale CSTR (continuous stirred-tank reactor) with swine and dairy waste water | 30 d | 37 °C | 75% | Poultry litter–inoculum ratios of 1:1; 1:1.66, and 1:3 | 183 LNbiogas.kg−1 VS and 74 L Nmethane.kg−1 VS | Below 60 mgNH3.L−1 | [30] |
| Process effluent from a bioethanol plant (BPE) | 165 d | - | 13.2–28.9% | 0%, 10%, and 20% (v/v) of CM: | 396–540 mLg−1 vs. methane | Less than 20 mgL−1 | [7] |
| Marine sediment (MS) or Ozouh sludge(OS) (thermophilic anaerobic digestion of excess activated sludge) |
|
|
|
|
|
| [46] |
| Marine sediment (MS) |
|
|
| 1:2.5 |
| 4.178 g NH3-N/kg−1 CM | |
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hafez, H.M.; Attia, Y.A. Challenges to the poultry industry: Current perspectives and strategic future after the COVID-19 outbreak. Front. Vet. Sci. 2020, 7, 516. [Google Scholar] [CrossRef]
- Burra, K.; Hussein, M.; Amano, R.; Gupta, A. Syngas evolutionary behavior during chicken manure pyrolysis and air gasification. Appl. Energy 2016, 181, 408–415. [Google Scholar] [CrossRef] [Green Version]
- Dróżdż, D.; Wystalska, K.; Malińska, K.; Grosser, A.; Grobelak, A.; Kacprzak, M. Management of poultry manure in Poland–Current state and future perspectives. J. Environ. Manag. 2020, 264, 110327. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.-H.; Park, C.-G.; Engel, B.A. Evaluating effects of poultry waste application on phosphorus loads to Lake Tenkiller. Sustainability 2015, 7, 10115–10134. [Google Scholar] [CrossRef] [Green Version]
- Wang, F.; Pei, M.; Qiu, L.; Yao, Y.; Zhang, C.; Qiang, H. Performance of anaerobic digestion of chicken manure under gradually elevated organic loading rates. Int. J. Environ. Res. Public Health 2019, 16, 2239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kreidenweis, U.; Breier, J.; Herrmann, C.; Libra, J.; Prochnow, A. Greenhouse gas emissions from broiler manure treatment options are lowest in well-managed biogas production. J. Clean. Prod. 2021, 280, 124969. [Google Scholar] [CrossRef]
- Cheong, D.-Y.; Harvey, J.T.; Kim, J.; Lee, C. Improving biomethanation of chicken manure by co-digestion with ethanol plant effluent. Int. J. Environ. Res. Public Health 2019, 16, 5023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dachs, B.; Kinkel, S.; Jäger, A.; Palčič, I. Backshoring of production activities in European manufacturing. J. Purch. Supply Manag. 2019, 25, 100531. [Google Scholar] [CrossRef] [Green Version]
- Hassanein, A.; Lansing, S.; Tikekar, R. Impact of metal nanoparticles on biogas production from poultry litter. Bioresour. Technol. 2019, 275, 200–206. [Google Scholar] [CrossRef]
- Abouelenien, F.; Fujiwara, W.; Namba, Y.; Kosseva, M.; Nishio, N.; Nakashimada, Y. Improved methane fermentation of chicken manure via ammonia removal by biogas recycle. Bioresour. Technol. 2010, 101, 6368–6373. [Google Scholar] [CrossRef]
- Abouelenien, F.; Namba, Y.; Kosseva, M.R.; Nishio, N.; Nakashimada, Y. Enhancement of methane production from co-digestion of chicken manure with agricultural wastes. Bioresour. Technol. 2014, 159, 80–87. [Google Scholar] [CrossRef]
- Li, K.; Liu, R.; Sun, C. Comparison of anaerobic digestion characteristics and kinetics of four livestock manures with different substrate concentrations. Bioresour. Technol. 2015, 198, 133–140. [Google Scholar] [CrossRef]
- Miura, T.; Kita, A.; Okamura, Y.; Aki, T.; Matsumura, Y.; Tajima, T.; Kato, J.; Nakashimada, Y. Evaluation of marine sediments as microbial sources for methane production from brown algae under high salinity. Bioresour. Technol. 2014, 169, 362–366. [Google Scholar] [CrossRef] [Green Version]
- Obata, O.; Ditchfield, A.; Hatton, A.; Akunna, J. Investigating the impact of inoculum source on anaerobic digestion of various species of marine macroalgae. Algal Res. 2020, 46, 101803. [Google Scholar] [CrossRef]
- Hoshino, T.; Doi, H.; Uramoto, G.-I.; Wörmer, L.; Adhikari, R.R.; Xiao, N.; Morono, Y.; D’Hondt, S.; Hinrichs, K.-U.; Inagaki, F. Global diversity of microbial communities in marine sediment. Proc. Natl. Acad. Sci. USA 2020, 117, 27587–27597. [Google Scholar] [CrossRef]
- Cupit, C.; Lomstein, B.A.; Kjeldsen, K.U. Contrasting community composition of endospores and vegetative Firmicutes in a marine sediment suggests both endogenous and exogenous sources of endospore accumulation. Environ. Microbiol. Rep. 2019, 11, 352–360. [Google Scholar] [CrossRef]
- Brereton, R.G.; Jansen, J.; Lopes, J.; Marini, F.; Pomerantsev, A.; Rodionova, O.; Roger, J.M.; Walczak, B.; Tauler, R. Chemometrics in analytical chemistry—part I: History, experimental design and data analysis tools. Anal. Bioanal. Chem. 2017, 409, 5891–5899. [Google Scholar] [CrossRef]
- Bystrzanowska, M.; Tobiszewski, M. Chemometrics for selection, prediction, and classification of sustainable solutions for green chemistry—A review. Symmetry 2020, 12, 2055. [Google Scholar] [CrossRef]
- Zolgharnein, J.; Shahmoradi, A.; Ghasemi, J.B. Comparative study of Box–Behnken, central composite, and Doehlert matrix for multivariate optimization of Pb (II) adsorption onto Robinia tree leaves. J. Chemom. 2013, 27, 12–20. [Google Scholar] [CrossRef]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association, , : Washington, DC, USA; American Water Work Association: Denver, CO, USA; Water Environment Federation: Alexandria, VA, USA, 1998. [Google Scholar]
- Miura, T.; Kita, A.; Okamura, Y.; Aki, T.; Matsumura, Y.; Tajima, T.; Kato, J.; Nakashimada, Y. Improved methane production from brown algae under high salinity by fed-batch acclimation. Bioresour. Technol. 2015, 187, 275–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, J.; Chen, J.P. A comprehensive review on biosorption of heavy metals by algal biomass: Materials, performances, chemistry, and modeling simulation tools. Bioresour. Technol. 2014, 160, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Aspe, E.; Marti, M.C.; Roeckel, M. Anaerobic treatment of fishery wastewater using a marine sediment inoculum. Water Res. 1997, 31, 2147–2160. [Google Scholar] [CrossRef]
- Tor, J.M.; Amend, J.P.; Lovley, D.R. Metabolism of organic compounds in anaerobic, hydrothermal sulphate-reducing marine sediments. Environ. Microbiol. 2003, 5, 583–591. [Google Scholar] [CrossRef]
- Yu, X.; Wang, Y.; Luo, Y.; Yin, L. The effects of salt on rheological properties of asphalt after long-term aging. Sci. World J. 2013, 2013, 921090. [Google Scholar] [CrossRef]
- Guo, M.; Song, W. Nutrient value of alum-treated poultry litter for land application. Poult. Sci. 2009, 88, 1782–1792. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Schaefer, D.; Guo, X.; Ren, L.; Meng, Q. Use of nitrate-nitrogen as a sole dietary nitrogen source to inhibit ruminal methanogenesis and to improve microbial nitrogen synthesis in vitro. Asian-Australas. J. Anim. Sci. 2009, 22, 542–549. [Google Scholar] [CrossRef]
- Anwar, N.; Wang, W.; Zhang, J.; Li, Y.; Chen, C.; Liu, G.; Zhang, R. Effect of sodium salt on anaerobic digestion of kitchen waste. Water Sci. Technol. 2016, 73, 1865–1871. [Google Scholar] [CrossRef]
- Mazumdar, S.; Lee, J.; Oh, M.-K. Microbial production of 2,3 butanediol from seaweed hydrolysate using metabolically engineered Escherichia coli. Bioresour. Technol. 2013, 136, 329–336. [Google Scholar] [CrossRef]
- Elasri, O.; Salem, M.; Ramdani, M.; Zaraali, O.; Lahbib, L. Effect of increasing inoculum ratio on energy recovery from chicken manure for better use in Egyptian agricultural farms. Chem. Biol. Technol. Agric. 2018, 5, 17. [Google Scholar] [CrossRef]
- Marchioro, V.; Steinmetz, R.L.; do Amaral, A.C.; Gaspareto, T.C.; Treichel, H.; Kunz, A. Poultry litter solid state anaerobic digestion: Effect of digestate recirculation intervals and substrate/inoculum ratios on process efficiency. Front. Sustain. Food Syst. 2018, 2, 46. [Google Scholar] [CrossRef]
- Abouelenien, F.; Kitamura, Y.; Nishio, N.; Nakashimada, Y. Dry anaerobic ammonia–methane production from chicken manure. Appl. Microbiol. Biotechnol. 2009, 82, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Park, S.Y.; Zhu, J. Solid-state anaerobic digestion for methane production from organic waste. Renew. Sustain. Energy Rev. 2011, 15, 821–826. [Google Scholar] [CrossRef]
- Rocamora, I.; Wagland, S.T.; Villa, R.; Simpson, E.W.; Fernández, O.; Bajón-Fernández, Y. Dry anaerobic digestion of organic waste: A review of operational parameters and their impact on process performance. Bioresour. Technol. 2020, 299, 122681. [Google Scholar] [CrossRef]
- Bujoczek, G.; Oleszkiewicz, J.; Sparling, R.; Cenkowski, S. High solid anaerobic digestion of chicken manure. J. Agric. Eng. Res. 2000, 76, 51–60. [Google Scholar] [CrossRef]
- Magbanua, B.S., Jr.; Adams, T.T.; Johnston, P. Anaerobic codigestion of hog and poultry waste. Bioresour. Technol. 2001, 76, 165–168. [Google Scholar] [CrossRef]
- Shubair, T.; Eljamal, O.; Khalil, A.; Matsunaga, N. Nitrate removal in porous media using nanoscale zero valent iron: Column experiment. In Proceedings of the International Exchange and Innovation Conference on Engineering & Sciences (IEICES); Interdisciplinary Graduate School of Engineering Sciences, Kyushu University: Fukuoka, Japan, 2017; pp. 145–148. [Google Scholar]
- Jenkins, D.G.; Quintana-Ascencio, P.F. A solution to minimum sample size for regressions. PLoS ONE 2020, 15, e0229345. [Google Scholar] [CrossRef] [Green Version]
- Frost, J. Multiple Regression Analysis: Use Adjusted R-squared and Predicted R-squared to Include the Correct Number of Variables; Minitab Blog: Hong Kong, China, 2013; Volume 13. [Google Scholar]
- Karch, J.; van Ravenzwaaij, D. Improving on Adjusted R-squared. Collabra Psychol. 2020, 6. [Google Scholar] [CrossRef]
- Shechtman, O. The coefficient of variation as an index of measurement reliability. In Methods of Clinical Epidemiology; Springer Series on Epidemiology and Public Health; Doi, S.A.R., Williams, G.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 39–49. ISBN 978-3-642-37131-8. [Google Scholar]
- Fenice, M.; Barghini, P.; Selbmann, L.; Federici, F. Combined effects of agitation and aeration on the chitinolytic enzymes production by the Antarctic fungus Lecanicillium muscarium CCFEE 5003. Microb. Cell Factories 2012, 11, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, G.-Y.; Chang, Z.-Z.; Ye, X.-M.; Du, J.; Xu, Y.-D.; Zhang, J.-Y. Methane production by anaerobic co-digestion of chicken manure and Spartina alterniflora residue after producing methane. Huan Jing Ke Xue Huanjing Kexue 2012, 33, 203–207. [Google Scholar] [PubMed]
- Elsayed, M.; Diab, A.; Soliman, M. Methane production from anaerobic co-digestion of sludge with fruit and vegetable wastes: Effect of mixing ratio and inoculum type. Biomass Convers. Biorefinery 2021, 11, 989–998. [Google Scholar] [CrossRef]
- Saad, M.F.M.; Rahman, N.A.A.; Yusoff, M.Z.M. Hydrogen and methane production from co-digestion of food waste and chicken manure. Pol. J. Environ. Stud. 2019, 28, 2805–2814. [Google Scholar] [CrossRef]
- Wei, L.; Qin, K.; Ding, J.; Xue, M.; Yang, C.; Jiang, J.; Zhao, Q. Optimization of the co-digestion of sewage sludge, maize straw and cow manure: Microbial responses and effect of fractional organic characteristics. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Parameter | Unit | Chicken Manure (CM) | Marine Sediment (MS) | Ozouh Sludge (OS) |
|---|---|---|---|---|
| TS | %W/W | 31.9 ± 0.09 | 52.42 ± 1.8 | 21.74 ± 0.6 |
| VS | %TS | 81.09 ± 1.98 | 7.385 ± 1.15 | 52.69 ± 2.61 |
| Water content | %W/W | 68.1 ± 0.09 | 47.58 ± 1.18 | 78.26 ± 0.6 |
| TOC | g-C kg-TS-1 | 385 ± 7.06 | - | 268 ± 7.9 |
| TKN | g-N kg-TS-1 | 85 ± 2.08 | - | 32 ± 0.08 |
| Salinity | % | 1.68 ± 0.26 | 1.1 ± 0.11 | 0.3 ± 0.07 |
| Condition | Substrate | Inoculum | 3% NaCl (mL) | Water (mL) | CM/MS or OS (TS/TS) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CM (g) | TS (g) | VS (g) | MS (g) | TS (g) | VS (g) | OS (g) | TS (g) | VS (g) | ||||
| Con1 a (−NaCl) | 0.505 | 0.161 | 0.131 | 9.231 | 4.839 | 0.357 | 40.26 | 1/30 | ||||
| Con1 b (+NaCl) | 0.505 | 0.161 | 0.131 | 9.231 | 4.839 | 0.357 | 40.26 | 1/30 | ||||
| Conc 1 | 0.505 | 0.161 | 0.131 | 22.26 | 4.839 | 2.55 | 27.24 | 1/30 | ||||
| Con2 a (−NaCl) | 0.746 | 0.238 | 0.193 | 9.084 | 4.762 | 0.352 | 40.17 | 1/20 | ||||
| Con2 b (+NaCl) | 0.746 | 0.238 | 0.193 | 9.084 | 4.762 | 0.352 | 40.17 | 1/20 | ||||
| Conc 2 | 0.746 | 0.238 | 0.193 | 21.9 | 4.762 | 2.509 | 27.35 | 1/20 | ||||
| Con3 a (−NaCl) | 1.423 | 0.455 | 0.369 | 8.671 | 4.545 | 0.336 | 39.91 | 1/10 | ||||
| Con3 b (+NaCl) | 1.423 | 0.455 | 0.369 | 8.671 | 4.545 | 0.336 | 39.91 | 1/10 | ||||
| Conc 3 | 1.423 | 0.455 | 0.369 | 20.91 | 4.545 | 2.395 | 27.67 | 1/10 | ||||
| Con4 a (−NaCl) | 2.609 | 0.833 | 0.676 | 7.949 | 4.167 | 0.308 | 39.44 | 1/5 | ||||
| Con4 b (+NaCl) | 2.609 | 0.833 | 0.676 | 7.949 | 4.167 | 0.308 | 39.44 | 1/5 | ||||
| Conc 4 | 2.609 | 0.833 | 0.676 | 19.17 | 4.167 | 2.195 | 28.22 | 1/5 | ||||
| Con5 a (−NaCl) | 4.473 | 1.429 | 1.158 | 6.813 | 3.571 | 0.264 | 38.71 | 1/2.5 | ||||
| Con5 b (+NaCl) | 4.473 | 1.429 | 1.158 | 6.813 | 3.571 | 0.264 | 38.71 | 1/2.5 | ||||
| Conc 5 | 4.473 | 1.429 | 1.158 | 16.43 | 3.571 | 1.882 | 29.1 | 1/2.5 | ||||
| Control a (−NaCl) | 15.66 | 5 | 4.055 | 0 | 0 | 0 | 34.34 | 1/0 | ||||
| Control b (+NaCl) | 15.66 | 5 | 4.055 | 0 | 0 | 0 | 34.34 | 1/0 | ||||
| CM/MS Ratio | CM | MS | Each TS Ratio (%) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (TS Basis) | TS (g) | TS (g) | TS of CM (%) | Wet weight (g) | VS% | VS (g) | TS (g) | TS% | Wet Weight (g) | VS% | VS (g) | NaCl (g) | Wet CM | Wet MS | |
| Liquid culture (5%TS) | 1/2.5 | 2.5 | 0.71 | 31.94 | 2.24 | 25.90 | 0.58 | 1.79 | 52.42 | 3.41 | 3.87 | 0.13 | 44.36 | 1.43 | 3.57 |
| Liquid control (5%TS) (CM) | 1/2.5 | 2.5 | 0.71 | 31.94 | 2.24 | 25.90 | 0.58 | 1.79 | 52.42 | 0.94 | 3.87 | 0.04 | 46.83 | 1.46 | - |
| Liquid control (5%TS) (MS) | 1/2.5 | 2.5 | 0.71 | 31.94 | 2.24 | 25.90 | 0.19 | 1.79 | 52.42 | 3.41 | 3.87 | 0.13 | 45.88 | 3.62 | |
| Semi-solid culture (10%TS) | 1/2.5 | 5 | 1.43 | 31.94 | 4.47 | 25.90 | 1.16 | 3.57 | 52.42 | 6.81 | 3.87 | 0.26 | 38.71 | 2.86 | 7.14 |
| Semi-solid control (10%TS) (CM) | 1/2.5 | 5 | 1.43 | 31.94 | 4.47 | 25.90 | 1.16 | 3.57 | 52.42 | 1.87 | 3.87 | 0.07 | 43.65 | 2.97 | |
| Semi-solid control (10%TS) (MS) | 1/2.5 | 5 | 1.43 | 31.94 | 1.43 | 25.90 | 0.37 | 3.57 | 52.42 | 6.81 | 3.87 | 0.07 | 41.76 | 7.35 | |
| Solid culture (20%TS) | 1/2.5 | 10 | 2.86 | 31.94 | 8.95 | 25.90 | 2.32 | 7.14 | 52.42 | 13.63 | 3.87 | 0.53 | 27.43 | 5.71 | 14.29 |
| Solid control (20%TS) (CM) | 1/2.5 | 10 | 2.86 | 31.94 | 8.95 | 25.90 | 2.32 | 7.14 | 52.42 | 3.74 | 3.87 | 0.15 | 37.31 | 6.18 | |
| Solid control (20%TS) (MS) | 1/2.5 | 10 | 2.86 | 31.94 | 2.86 | 25.90 | 0.74 | 7.14 | 52.42 | 13.63 | 3.87 | 0.53 | 33.52 | 15.15 | |
| Conditions | pH | VFAs (mM/g VS) | NH3 gN/kg CM | H mL/g VS-CM | |||||
|---|---|---|---|---|---|---|---|---|---|
| Acetate | Propionate | ||||||||
| Initial | Final | Initial | Final | Initial | Final | Initial | Final | ||
| Con1 a (−NaCl) | 7.8 ± 0 | 7.65 ± 0.07 | 0 | 0 | 0 | 0 | 0.04 ± 0.02 | 0.41 ± 0.09 | 261.88 ± 9.86 |
| Con1 b (+NaCl) | 7.85 ± 0.07 | 7.4 ± 0 | 0 | 0 | 0 | 0 | 0.09 ± 0.04 | 1.53 ± 0.06 | 195.67 ± 72.4 |
| Con1 c | 6.85 ± 0.07 | 7.6 ± 0.14 | 0 | 0 | 0 | 0 | 0.29 ± 0.14 | 1.02 ± 1 | 633.76 ± 18. 69 |
| Con2 a (−NaCl) | 7.85 ± 0.14 | 7.65 ± 0.07 | 0 | 0 | 0 | 0 | 0.07 ± 0 | 0.40 ± 0.12 | 299.14 ± 7.09 |
| Con2 b (+NaCl) | 7.7 ± 0 | 8.15 ± 0.64 | 0 | 0 | 0 | 0 | 0.10 ± 0.03 | 0.70 ± 0.35 | 0 |
| Con2 c | 6.85 ± 0.064 | 7.6 ± 0 | 0 | 0 | 0 | 0 | 0.22 ± 0.14 | 1.09 ± 0.04 | 471.41 ± 4.63 |
| Con3 a (−NaCl) | 7.75 ± 0.21 | 7.65 ± 0.07 | 0 | 0 | 0 | 0 | 0.21 ± 0.03 | 0.78 ± 0.11 | 305.43 ± 6.86 |
| Con3 b (+NaCl) | 7.85 ± 0.07 | 7.65 ± 0.07 | 0 | 0 | 0 | 0 | 0.21 ± 0.02 | 0.77 ± 0.07 | 293.8 ± 2.42 |
| Con3 c | 7.25 ± 0.07 | 7.75 ± 0.07 | 4 ± 5.65 | 0 | 0 | 0 | 0.39 ± 0.05 | 1.22 ± 0.06 | 354.12 ± 16.65 |
| Con4 a (−NaCl) | 7.3 ± 0.0 | 7.85 ± 0.07 | 23.56 ± 1.7 | 1.85 ± 0.64 | 3.94 ± 0.31 | 1.53 ± 0.54 | 0.56 ± 0.04 | 1.38 ± 0.04 | 13.12 ± 5.84 |
| Con4 b (+NaCl) | 7.4 ± 0.0 | 7.7 ± 0 | 18.25 ± 4.03 | 0.545 ± 0.70 | 3.93 ± 0.4 | 0 | 0.54 ± 0.07 | 1.09 ± 0.12 | 287.53 ± 3.15 |
| Con4 c | 7.15 ± 0.07 | 7.75 ± 0.07 | 27.88 ± 4.54 | 0.35 ± 0.49 | 2.465 ± 1.46 | 0 | 0.88 ± 0.23 | 1.25 ± 0.38 | 265.79 ± 31.55 |
| Con5 a (−NaCl) | 6.8 ± 0.07 | 7.9 ± 0 | 56.12 ± 8.41 | 3.81 ± 3.5 | 5.97 ± 0.27 | 1.6 ± 2.29 | 4.87 ± 0.5 | 8.58 ± 1.28 | 301.22 ± 1.57 |
| Con5 b (+NaCl) | 7.3 ± 0.14 | 7.85 ± 0.07 | 44.06 ± 1.75 | 2.2 ±1.82 | 7.49 ± 0.56 | 0 | 5.11 ± 1.46 | 7.95 ± 0.75 | 205.08 ± 21.18 |
| Con5 c | 6.95 ± 0.07 | 7.85 ± 0.07 | 62.61 ± 2.39 | 3.16 ± 3.5 | 8.48 ± 0.71 | 0 | 1.08 ± 0.14 | 2.36 ± 0.12 | 266.41 ± 7.9 |
| Control a (−NaCl) | 7.1 ± 0.14 | 7.2 ± 0.28 | 230.85 ± 18.45 | 310.9 ± 28.28 | 30.4 ± 1.84 | 22.38 ± 3.4 | 1.01 ± 0.03 | 3.18 ± 0.1 | 14.05 ± 1.16 |
| Control b (+NaCl) | 7.1 ± 0.42 | 7.25 ± 0.07 | 208.25 ± 31.32 | 304.7 ± 37.96 | 26.59 ± 4.57 | 27.33 ± 4.57 | 1.36 ± 0.06 | 1.90 ± 0.12 | 13.78 ± 1.92 |
| Culture Condition | pH | VFAs (mM/Kg CM) | NH3 gN/kg CM | TGV mL/g VS | CH4 mL/g VS-CM | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Acetate | Propoinate | |||||||||
| Initial | Final | Initial | Final | Initial | Final | Initial | Final | |||
| Liquid (5% TS) culture | 7.35 ± 0.07 | 7.8 ± 0 | 7.14 ± 0.54 | 0 | 0 | 0 | 0.694 ± 0 | 1.530 ± 0.32 | 311.74 ± 28.09 | 166.96 ± 17.82 |
| Liquid (5% TS) control (CM) | 7.3 ± 0 | 7.6 ± 0 | 2.055 ± 1.9 | 0 | 0 | 0 | 0.62 ± 0.04 | 0.88 ± 0.12 | 162.35 ± 53.74 | 59.25 ± 35.74 |
| Liquid (5% TS) control (MS) | 7.2 ± 0 | 7.5 ± 0 | 0 | 0 | 0 | 0 | 0.02 ± 0.01 | 0.008 ± 0.005 | 0 | 0 |
| Semi-solid (10% TS) culture | 7.35 ± 0.21 | 7.85 ± 0.07 | 17.79 ± 3.2 | 5.39 ± 4.5 | 4.115 ± 0.89 | 0 | 1.26 ± 0.22 | 1.7 ± 0.12 | 312.18 ± 36.03 | 168.79 ± 18.7 |
| Semi-solid(10% TS) control (CM) | 7.45 ± 0.21 | 7.55 ± 0.21 | 6.85 ± 4.6 | 20.98 ± 4.87 | 1.525 ± 1.15 | 4.09 ± 3.03 | 0.89 ± 0.03 | 2.15 ± 0.48 | 101.9 ± 1.83 | 24.89 ± 1.05 |
| Semi-solid(10% TS) control (MS) | 7.5 ± 0 | 7.7 ± 0.14 | 0 | 0 | 0 | 0 | 0.01 ± 0.005 | 0.009 ± 0.001 | 0 | 0 |
| Solid (20%TS) culture | 7.1 ± 0.14 | 8 ± 0.14 | 41.07 ± 6.6 | 0 | 9.51 ± 0.21 | 4.49 ± 3.06 | 2.12 ± 0.25 | 4.18 ± 1.38 | 320.46 ± 26.6 | 169.28 ± 21.11 |
| Solid (20%TS) control (CM) | 7.35 ± 0.07 | 7.2 ± 0.14 | 35.04 ± 0.8 | 80.67 ± 3.35 | 6.5 ± 0.27 | 14.9 ± 0.06 | 2.14 ± 0.04 | 3.73 ± 0.02 | 101.9 ± 0.91 | 8.62 ± 1.1 |
| Solid (20%TS) control (MS) | 7.65 ± 0.07 | 7.75 ± 0.07 | 0 | 0 | 0 | 0 | 0.001 ± 0 | 0.008 ± 0.004 | 0 | 0 |
| Source | Sum of Squares | Degrees of Freedom | Mean Square | F Value | Prob > F | |
|---|---|---|---|---|---|---|
| Model | 32,694.60 | 21 | 1556.89 | 932.94 | <0.0001 | Significant |
| Residual | 302.05 | 181 | 1.67 | |||
| Lack of fit | 229.94 | 168 | 0.25 | 0.25 | 1.0000 | Not significant |
| Pure error | 72.11 | 13 | 5.55 | |||
| Total | 32,996.65 | 202 | ||||
| R-squared = 0.9764 | ||||||
| Adjusted R-squared = 0.9741 Predicted R-squared = 0.9731 | ||||||
| Adequate precision = 36.662 CV = 17.76% | ||||||
| Source | Coefficient Estimate | Standard Error | F Value | p-Value | |
|---|---|---|---|---|---|
| A-IR | 1.29 | 1.09 | 1.40 | 0.2385 | |
| B-Time | 0.50 | 0.58 | 0.75 | 0.3884 | |
| C-Total Solid | −28.38 | 0.83 | 1161.94 | <0.0001 | Significant |
| D-NaCl | 5.67 | 0.23 | 596.43 | <0.0001 | Significant |
| AB | 1.49 | 0.19 | 58.37 | <0.0001 | Significant |
| AC | 27.94 | 0.92 | 926.62 | <0.0001 | Significant |
| AD | 0.97 | 0.13 | 58.37 | <0.0001 | Significant |
| BC | 0.81 | 0.93 | 0.77 | 0.3816 | |
| BD | 0.40 | 0.15 | 7.12 | 0.0083 | Significant |
| A2 | 4.83 | 0.26 | 332.03 | <0.0001 | Significant |
| B2 | −2.64 | 0.52 | 25.78 | <0.0001 | Significant |
| C2 | 0.21 | 0.48 | 0.20 | 0.6561 | |
| ABD | −0.94 | 0.19 | 25.47 | <0.0001 | Significant |
| A2B | 1.46 | 0.36 | 16.47 | <0.0001 | Significant |
| A2D | −6.09 | 0.25 | 612.00 | <0.0001 | Significant |
| AB2 | 0.51 | 0.32 | 2.56 | 0.1114 | |
| B2C | −0.66 | 1.38 | 0.23 | 0.6316 | |
| B2D | −1.23 | 0.24 | 25.54 | <0.0001 | Significant |
| BC2 | −0.97 | 0.84 | 1.32 | 0.2530 | |
| A3 | 6.33 | 0.93 | 45.85 | <0.0001 | Significant |
| B3 | 2.79 | 0.42 | 45.07 | <0.0001 | Significant |
| NH4 (g/L) | pH | NH3 Conc. (g/L) | Propionate (mM/L) | Acetate (mM/L) | CH4 (mL/g Glucose) | ||||
|---|---|---|---|---|---|---|---|---|---|
| Initial | Final | Initial | Final | Initial | Final | Initial | Final | ||
| 0 | 8.2 ± 0.07 | 7.55 ± 0 | 0.27 ± 0.04 | 0.27 ± 0.14 | 0 | 8 ± 1.28 | 0 | 23.48 ± 4.7 | 80.86 ± 2.73 |
| 1.5 | 8.1 ± 0.07 | 7.55 ± 0 | 0.64 ± 0.23 | 0.80 ± 0.23 | 0 | 8.48 ± 1.02 | 0 | 3.2 ± 0.5 | 130.62 ± 13.44 |
| 3 | 8.2 ± 0.02 | 7.5 ± 0.07 | 1.14 ± 0.04 | 1.17 ± 0.47 | 0 | 3.89 ± 0.12 | 0 | 13.79 ± 1.8 | 125.76 ± 8.16 |
| 4.5 | 8.15 ± 0.21 | 7.45 ± 0.07 | 1.6 ± 0.09 | 1.4 ± 0.14 | 0 | 2.82 ± 0.07 | 0 | 30.8 ± 5.6 | 47.80 ± 17 |
| 6 | 8.2 ± 0.07 | 7.4 ± 0.12 | 1.89 ± 0.12 | 2.07 ± 0.12 | 0 | 4.39 ± 0.49 | 0 | 26.54 ± 4.5 | 76.81 ± 1.14 |
| 7.5 | 8.05 ± 0.02 | 7.45 ± 0.14 | 2.42 ± 0.12 | 2.50 ± 0.02 | 0 | 4.35 ± 0.33 | 0 | 29.66 ± 1.05 | 70.42 ± 0.49 |
| 9 | 8.1 ± 0.14 | 7.45 ± 0.01 | 3.24 ± 0.1 | 3.05 ± 0.1 | 0 | 2.82 ± 0.21 | 0 | 34.59 ± 1.82 | 63.26 ± 5.05 |
| 10.5 | 7.95 ± 0 | 7.45 ± 0 | 3.52 ± 0.14 | 3.58 ± 0.03 | 0 | 2.08 ± 0.12 | 0 | 35.92 ± 1.07 | 47.25 ± 18.72 |
| 12 | 7.9 ± 0.12 | 7.45 ± 0 | 3.80 ± 0.19 | 4.18 ± 0.5 | 0 | 1.52 ± 0.05 | 0 | 30.78 ± 1.55 | 30.45 ± 11.45 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abouelenien, F.; Miura, T.; Nakashimada, Y.; Elleboudy, N.S.; Al-Harbi, M.S.; Ali, E.F.; Shukry, M. Optimization of Biomethane Production via Fermentation of Chicken Manure Using Marine Sediment: A Modeling Approach Using Response Surface Methodology. Int. J. Environ. Res. Public Health 2021, 18, 11988. https://doi.org/10.3390/ijerph182211988
Abouelenien F, Miura T, Nakashimada Y, Elleboudy NS, Al-Harbi MS, Ali EF, Shukry M. Optimization of Biomethane Production via Fermentation of Chicken Manure Using Marine Sediment: A Modeling Approach Using Response Surface Methodology. International Journal of Environmental Research and Public Health. 2021; 18(22):11988. https://doi.org/10.3390/ijerph182211988
Chicago/Turabian StyleAbouelenien, Fatma, Toyokazu Miura, Yutaka Nakashimada, Nooran S. Elleboudy, Mohammad S. Al-Harbi, Esmat F. Ali, and Mustafa Shukry. 2021. "Optimization of Biomethane Production via Fermentation of Chicken Manure Using Marine Sediment: A Modeling Approach Using Response Surface Methodology" International Journal of Environmental Research and Public Health 18, no. 22: 11988. https://doi.org/10.3390/ijerph182211988
APA StyleAbouelenien, F., Miura, T., Nakashimada, Y., Elleboudy, N. S., Al-Harbi, M. S., Ali, E. F., & Shukry, M. (2021). Optimization of Biomethane Production via Fermentation of Chicken Manure Using Marine Sediment: A Modeling Approach Using Response Surface Methodology. International Journal of Environmental Research and Public Health, 18(22), 11988. https://doi.org/10.3390/ijerph182211988








