A New Paired Associative Stimulation Protocol with High-Frequency Peripheral Component and High-Intensity 20 Hz Repetitive Transcranial Magnetic Stimulation—A Pilot Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Transcranial Magnetic Stimulation
2.3. Peripheral Nerve Stimulation
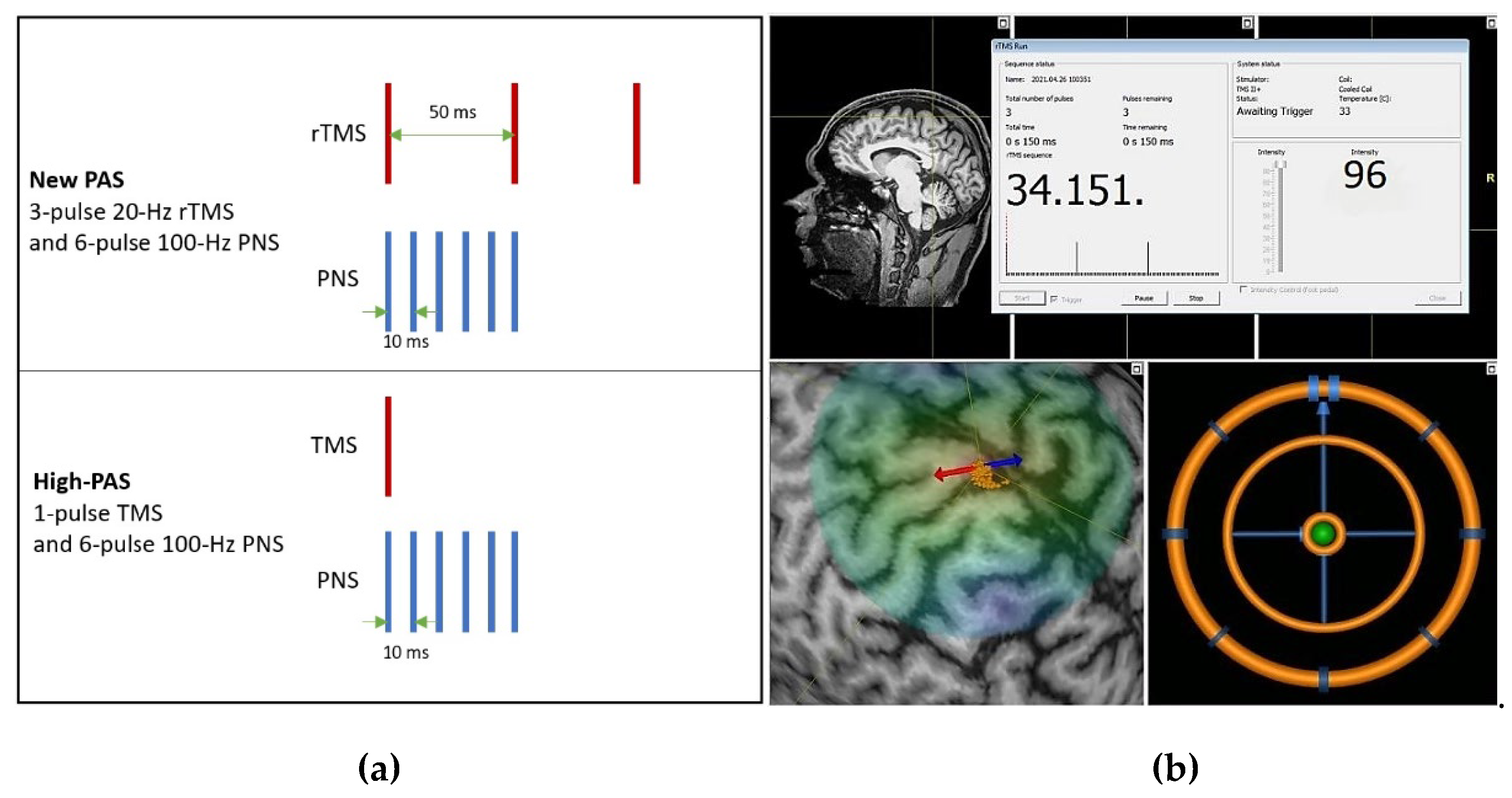
2.4. Paired Associative Stimulation Protocols
2.5. Long Intracortical Inhibition Protocol
2.6. Statistical Analysis
3. Results
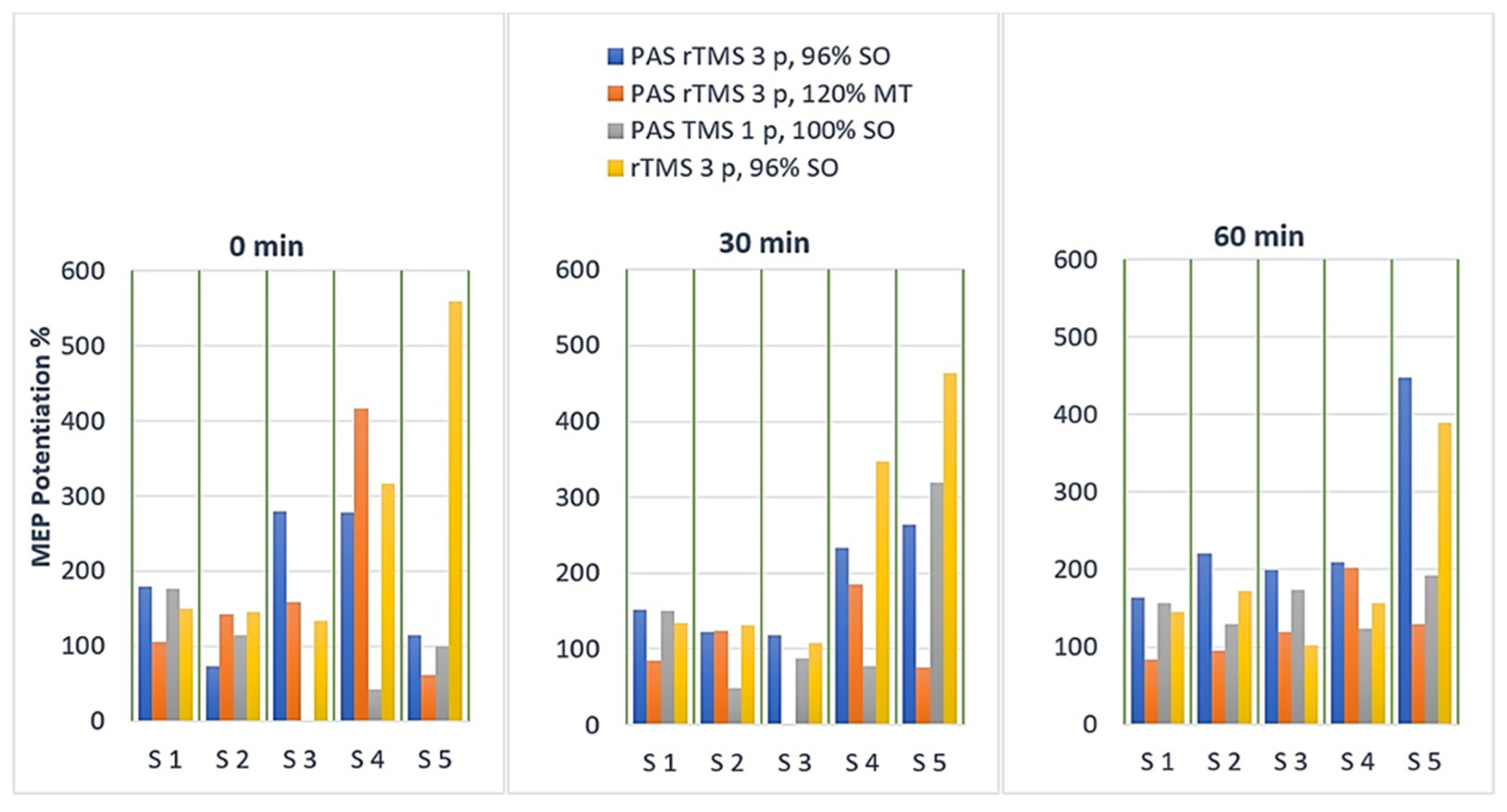
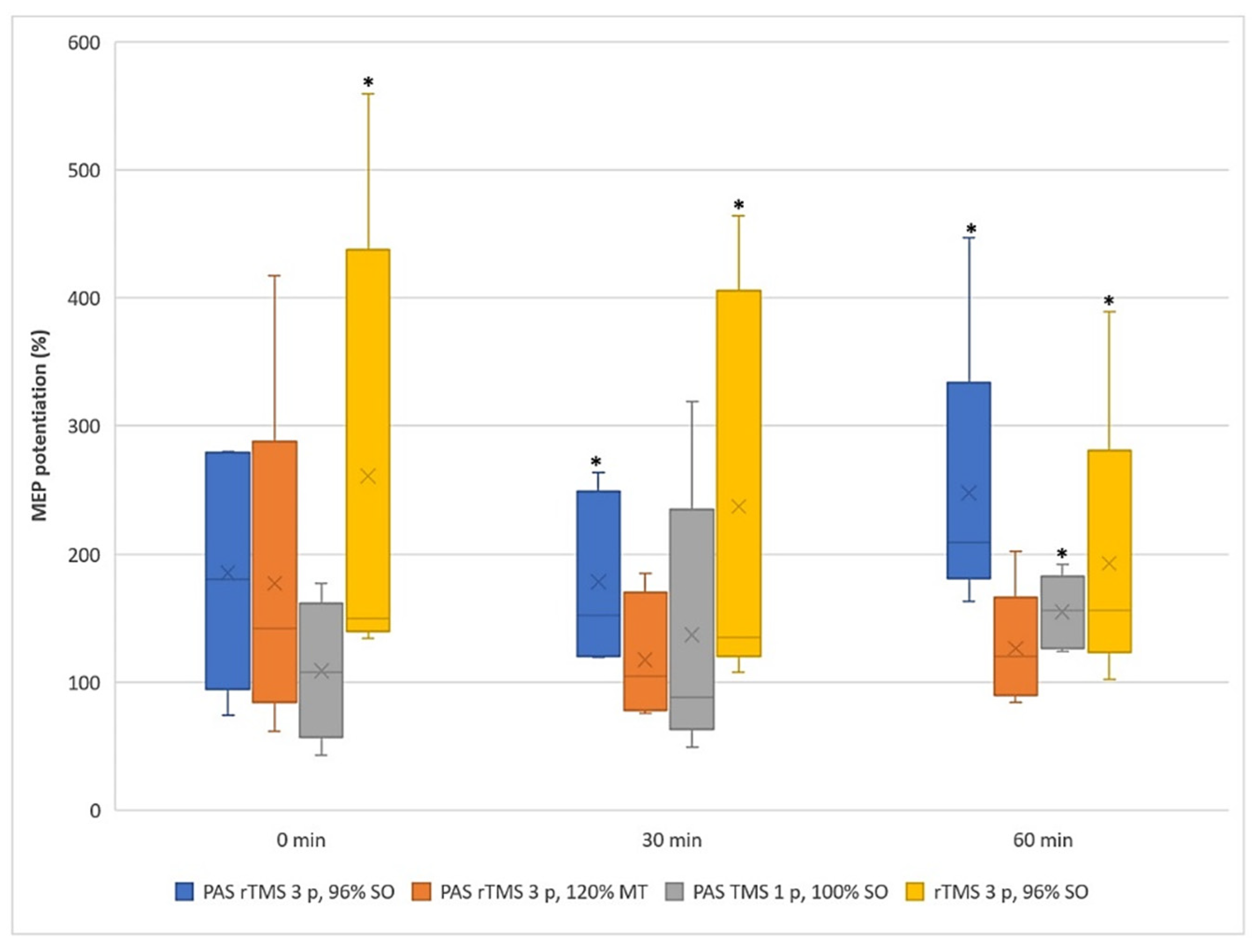
3.1. Motor Evoked Potentials
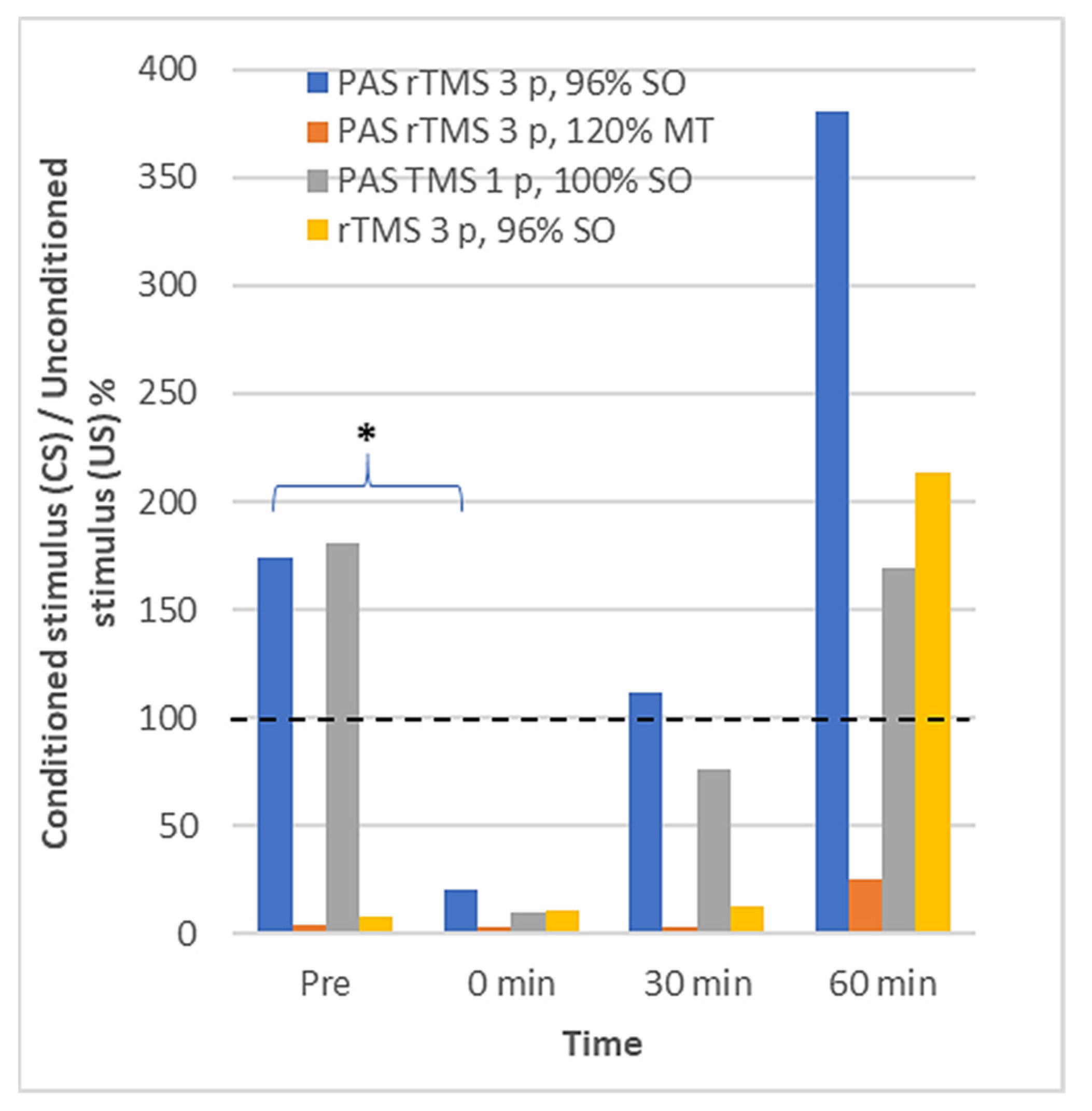
3.2. Long Interval Cortical Inhibition
3.3. Safety and Tolerability of the New Protocol
4. Discussion
4.1. Efficacy and Safety of the Proposed PAS Paradigm
4.2. Effects on LICI
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stefan, K.; Kunesch, E.; Cohen, L.G.; Benecke, R.; Classen, J. Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 2000, 123, 572–584. [Google Scholar] [CrossRef] [Green Version]
- Bi, G.; Poo, M. Synaptic modifications in cultured hippocampal neurons: Dependence on spike timing, synaptic strength, and postsynaptic cell type. J. Neurosci. 1998, 18, 10464–10472. [Google Scholar] [CrossRef] [PubMed]
- Dan, Y.; Poo, M.M. Spike timing-dependent plasticity of neural circuits. Neuron 2004, 44, 23–30. [Google Scholar] [CrossRef] [Green Version]
- Wolters, A.; Sandbrink, F.; Schlottmann, A.; Kunesch, E.; Stefan, K.; Cohen, L.G.; Classen, J. A Temporally Asymmetric Hebbian Rule Governing Plasticity in the Human Motor Cortex. J. Neurophysiol. 2003, 89, 2339–2345. [Google Scholar] [CrossRef]
- Carson, R.G.; Kennedy, N.C. Modulation of human corticospinal excitability by paired associative stimulation. Front. Hum. Neurosci. 2013, 7, 823. [Google Scholar] [CrossRef] [Green Version]
- Taylor, J.L.; Martin, P.G. Voluntary motor output is altered by spike-timing-dependent changes in the human corticospinal pathway. J. Neurosci. 2009, 29, 11708–11716. [Google Scholar] [CrossRef]
- Cortes, M.; Thickbroom, G.W.; Valls-Sole, J.; Pascual-Leone, A.; Edwards, D.J. Spinal associative stimulation: A non-invasive stimulation paradigm to modulate spinal excitability. Clin. Neurophysiol. 2011, 122, 2254–2259. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bunday, K.L.; Perez, M.A. Motor recovery after spinal cord injury enhanced by strengthening corticospinal synaptic transmission. Curr. Biol. 2012, 22, 2355–2361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leukel, C.; Taube, W.; Beck, S.; Schubert, M. Pathway-specific plasticity in the human spinal cord. Eur. J. Neurosci. 2012, 35, 1622–1629. [Google Scholar] [CrossRef] [Green Version]
- Shulga, A.; Lioumis, P.; Kirveskari, E.; Savolainen, S.; Mäkelä, J.P.; Ylinen, A. The use of F-response in defining interstimulus intervals appropriate for LTP-like plasticity induction in lower limb spinal paired associative stimulation. J. Neurosci. Methods 2015, 242, 112–117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Knikou, M. Spinal excitability changes after transspinal and transcortical paired associative stimulation in humans. Neural Plast. 2017, 6751810. [Google Scholar] [CrossRef] [Green Version]
- Ridding, M.C.; Taylor, J.L. Mechanisms of motor-evoked potential facilitation following prolonged dual peripheral and central stimulation in humans. J. Physiol. 2001, 537, 623–631. [Google Scholar] [CrossRef]
- Ridding, M.C.; Uy, J. Changes in motor cortical excitability induced by paired associative stimulation. Clin. Neurophysiol. 2003, 114, 1437–1444. [Google Scholar] [CrossRef]
- Fratello, F.; Veniero, D.; Curcio, G.; Ferrara, M.; Marzano, C.; Moroni, F.; De Gennaro, L. Modulation of corticospinal excitability by paired associative stimulation: Reproducibility of effects and intraindividual reliability. Clin. Neurophysiol. 2006, 117, 2667–2674. [Google Scholar] [CrossRef]
- Kujirai, K.; Kujirai, T.; Sinkjaer, T.; Rothwell, J.C. Associative plasticity in human motor cortex during voluntary muscle contraction. J. Neurophysiol. 2006, 96, 1337–1346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quartarone, A.; Rizzo, V.; Bagnato, S.; Morgante, F.; Sant’Angelo, A.; Girlanda, P.; Siebner, H.R. Rapid rate paired associative stimulation of the median nerve and motor cortex can produce long-lasting changes in motor cortical excitability in humans. J. Physiol. 2006, 575, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Ridding, M.; Flavel, S. Induction of plasticity in the dominant and non-dominant motor cortices of humans. Exp. Brain Res. 2006, 171, 551–557. [Google Scholar] [CrossRef]
- Rosenkranz, K.; Rothwell, J.C. Differences between the effects of three plasticity inducing protocols on the organization of the human motor cortex. Eur. J. Neurosci. 2006, 23, 822–829. [Google Scholar] [CrossRef]
- Roy, F.D.; Norton, J.A.; Gorassini, M.A. Role of sustained excitability of the leg motor cortex after transcranial magnetic stimulation in associative plasticity. J. Neurophysiol. 2007, 98, 657–667. [Google Scholar] [CrossRef]
- Stinear, J.W.; Hornby, T.G. Stimulation-induced changes in lower limb corticomotor excitability during treadmill walking in humans. J. Physiol. 2005, 567, 701–711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prior, M.M.; Stinear, J.W. Phasic spike-timing-dependent plasticity of human motor cortex during walking. Brain Res. 2006, 1110, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Mrachacz-Kersting, N.; Fong, M.; Murphy, B.A.; Sinkjaer, T. Changes in excitability of the cortical projections to the human tibialis anterior after paired associative stimulation. J. Neurophysiol. 2007, 97, 1951–1958. [Google Scholar] [CrossRef]
- Kumpulainen, S.; Mrachacz-Kersting, N.; Peltonen, J.; Voigt, M.; Avela, J. The optimal interstimulus interval and repeatability of paired associative stimulation when the soleus muscle is targeted. Exp. Brain Res. 2012, 221, 241–249. [Google Scholar] [CrossRef]
- Mrachacz-Kersting, N. Paired-Associative Stimulation, an Introduction to Neural Engineering for Motor Rehabilitation; IEEE Press Series in Biomedical Engineering; Farina, D., Jensen, W., Akay, M., Eds.; Wiley-IEEE Press: Hoboken, NJ, USA, 2013; pp. 529–547. [Google Scholar]
- Roy, F.D.; Yang, J.F.; Gorassini, M.A. Afferent Regulation of Leg Motor Cortex Excitability After Incomplete Spinal Cord Injury. J. Neurophysiol. 2010, 103, 2222–2233. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, M.; Suda, K.; Safdar, N.; Goldstein, B.; Jones, M.; Poggensee, L.; Evans, C. Unique Risks and Clinical Outcomes Associated with Extended-Spectrum β-Lactamase Enterobacteriaceae in Veterans with Spinal Cord Injury or Disorder: A Case-Case-Control Study. Infect. Control. Hosp. Epidemiol. 2016, 37, 768–776. [Google Scholar] [CrossRef]
- Urbin, M.A.; Ozdemir, R.A.; Tazoe, T.; Perez, M.A. Spike-timing-dependent plasticity in lower-limb motoneurons after human spinal cord. J. Neurophysiol. 2017, 118, 2171–2180. [Google Scholar] [CrossRef] [PubMed]
- D’Amico, J.M.; Dongés, S.C.; Taylor, J.L. High-intensity, low-frequency repetitive transcranial magnetic stimulation enhances excitability of the human corticospinal pathway. J. Neurophysiol. 2020, 123, 1969–1978. [Google Scholar] [CrossRef] [PubMed]
- Shulga, A.; Lioumis, P.; Zubareva, A.; Brandstack, N.; Kuusela, L.; Kirveskari, E.; Savolainen, S.; Ylinen, A.; Mäkelä, J.P. Long-term paired associative stimulation can restore voluntary control over paralyzed muscles in incomplete chronic spinal cord injury patients. Spinal Cord Ser Cases 2016, 2, 16016. [Google Scholar] [CrossRef] [Green Version]
- Tolmacheva, A.; Savolainen, S.; Kirveskari, E.; Lioumis, P.; Kuusela, L.; Brandstack, N.; Shulga, A.; Ylinen, A.; Mäkelä, J.P. Long-Term Paired Associative Stimulation Enhances Motor Output of the Tetraplegic Hand. J. Neurotrauma 2017, 915, 2668–2674. [Google Scholar] [CrossRef]
- Rodionov, A.; Savolainen, S.; Kirveskari, E.; Makela, J.P.; Shulga, A. Restoration of hand function with long-term paired associative stimulation after chronic incomplete tetraplegia: A case study. Spinal Cord Ser Cases 2019, 5, 1–8. [Google Scholar] [CrossRef]
- Suppa, A.; Quartarone, A.; Siebner, H.; Chen, R.; Di Lazzaro, V.; Del Giudice, P.; Paulus, W.; Rothwell, J.C.; Ziemann, U.; Classen, J. The associative brain at work: Evidence from paired associative stimulation studies in humans. Clin. Neurophysiol. 2017, 128, 2140–2164. [Google Scholar] [CrossRef]
- Mezes, M.; Havu, R.; Tolmacheva, A.; Lioumis, P.; Mäkelä, J.P.; Shulga, A. The impact of TMS and PNS frequencies on MEP potentiation in PAS with high-frequency peripheral component. PLoS ONE 2020, 15, 0233999. [Google Scholar] [CrossRef]
- Oudega, M. Molecular and cellular mechanisms underlying the role of blood vessels in spinal cord injury and repair. Cell Tissue Res. 2012, 349, 269–288. [Google Scholar] [CrossRef] [PubMed]
- Tolmacheva, A.; Savolainen, S.; Kirveskari, E.; Brandstack, N.; Makela, J.P.; Shulga, A. Paired associative stimulation improves hand function after non-traumatic spinal cord injury: A case series. Clin. Neurophysiol. Pract. 2019, 4, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Shulga, A.; Lioumis, P.; Kirveskari, E.; Savolainen, S.; Mäkelä, J.P. A novel paired associative stimulation protocol with a high-frequency peripheral component: A review on results in spinal cord injury rehabilitation. Eur. J. Neurosci. 2021, 53, 3242–3257. [Google Scholar] [CrossRef] [PubMed]
- Tolmacheva, A.; Mäkelä, J.P.; Shulga, A. Increasing the frequency of peripheral component in paired associative stimulation strengthens its efficacy. Sci. Rep. 2019, 9, 3849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kujirai, T.; Caramia, M.D.; Rothwell, J.C.; Day, B.L.; Thompson, P.D.; Ferbert, A.; Wroe, S.; Asselman, P.; Marsden, C.D. Corticocortical inhibition in human motor cortex. J. Physiol. 1993, 471, 501–519. [Google Scholar] [CrossRef]
- Reis, J.; Swayne, O.B.; Vandermeeren, Y.; Camus, M.; Dimyan, M.A.; Harris-Love, M.; Perez, M.A..; Ragert, P.; Rothwell, J.C.; Cohen, L.G. Contribution of transcranial magnetic stimulation to the understanding of cortical mechanisms involved in motor control. J. Physiol. 2008, 586, 325–351. [Google Scholar] [CrossRef] [PubMed]
- Valls-Solé, J.; Pascual-Leone, A.; Wassermann, E.M.; Hallett, M. Human motor evoked responses to paired transcranial magnetic stimuli. Electroencephalogr. Clin. Neurophysiol. 1992, 85, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Ilic, T.V.; Meintzschel, F.; Cleff, U.; Ruge, D.; Kessler, K.R.; Ziemann, U. Short-interval paired-pulse inhibition and facilitation of human motor cortex: The dimension of stimulus intensity. J Physiol. 2002, 545 Pt 1, 153–167. [Google Scholar] [CrossRef] [PubMed]
- Rossi, S.; Antal, A.; Bestmann, S.; Bikson, M.; Brewer, C.; Brockmöller, J.; Hallett, M. Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: Expert Guidelines. Clin. Neurophysiol. 2021, 132, 269–306. [Google Scholar] [CrossRef]
- McDonnell, M.N.; Orekhov, Y.; Ziemann, U. The role of GABA(B) receptors in intracortical inhibition in the human motor cortex. Exp Brain Res. 2006, 173, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Magistris, M.R.; Rösler, K.M.; Truffert, A.; Myers, J.P. Transcranial stimulation excites virtually all motor neurons supplying the target muscle. A demonstration and a method improving the study of motor evoked potentials. Brain 1998, 121 Pt 3, 437–450. [Google Scholar] [CrossRef] [Green Version]
- Magistris, M.R.; Rösler, K.M.; Truffert, A.; Landis, T.; Hess, C.W. A clinical study of motor evoked potentials using a triple stimulation technique. Brain 1999, 122 Pt 2, 265–279. [Google Scholar] [CrossRef] [Green Version]
- Rösler, K.M.; Magistris, M.R. The size of motor-evoked potentials: Influencing parameters and quantification. In Oxford Handbook of Transcranial Stimulation, 1st ed.; Epsein, C.M., Wassermann, E.M., Ziemann, U., Eds.; Oxford University Press: Oxford, UK, 2008; Volume 1, Chapter 9; pp. 77–90. [Google Scholar]
- Bühler, R.; Magistris, M.R.; Truffert, A.; Hess, C.W.; Rösler, K.M. The triple stimulation technique to study central motor conduction to the lower limbs. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2001, 112, 938–949. [Google Scholar] [CrossRef]
- Humm, A.M.; Z’Graggen, W.J.; von Hornstein, N.E.; Magistris, M.R.; Rosler, K.M. Assessment of central motor conduction to intrinsic hand muscles using the triple stimulation technique: Normal values and repeatability. Clin. Neurophysiol. 2004, 115, 2558–2566. [Google Scholar] [CrossRef]
- Krogh, S.; Aagaard, P.; Jønsson, A.B.; Figlewski, K.; Kasch, H. Effects of repetitive transcranial magnetic stimulation on recovery in lower limb muscle strength and gait function following spinal cord injury: A randomized controlled trial. Spinal Cord 2021. [Google Scholar] [CrossRef]
- Shulga, A.; Zubareva, A.; Lioumis, P.; Mäkelä, J.P. Paired associative stimulation with high-frequency peripheral component leads to enhancement of corticospinal transmission at wide range of interstimulus intervals. Front. Hum. Neurosci. 2016, 10, 470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McDonnell, M.N.; Orekhov, Y.; Ziemann, U. Suppression of LTP-like plasticity in human motor cortex by the GABAB receptor agonist baclofen. Exp. Brain Res. 2007, 180, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Claus, D.; Weis, M.; Jahnke, U.; Plewe, A.; Brunhölzl, C. Corticospinal conduction studied with magnetic double stimulation in the intact human. J. Neurol. Sci. 1992, 111, 180–188. [Google Scholar] [CrossRef]
| Subjects | RMT (% SO) | PNS Intensity (mA) | F Latency (ms) | MEP Latency (ms) | ISI (ms) |
|---|---|---|---|---|---|
| S1 | 60 | 7 | 48.3 | 44.8 | 4 |
| S2 | 55 | 9 | 54.5 | 42.7 | 12 |
| S3 | 77 | 16 | 55.0 | 48.0 | 7 |
| S4 | 64 | 13 | 54.0 | 43.6 | 10 |
| S5 | 75 | 7 | 58.0 | 42.3 | 16 |
| Measurement Time | PAS rTMS 3p 96% SO | PAS rTMS 3p1 20% MT | PAS TMS 1p 100% SO | rTMS 3p 96% SO |
|---|---|---|---|---|
| Post 60 min | p = 0.043, SE = ±52% | p = 0.500, SE = ±21% | p = 0.043, SE = ±13% | p = 0.043, SE = ±50% |
| Post 30 min | p = 0.043, SE = ±30% | p = 0.465, SE = ±25% | p = 0.893, SE = ±49% | p = 0.043, SE = ±71% |
| Post 0 min | p = 0.345, SE = ±42% | p = 0.138, SE = ±62% | p = 0.285, SE = ±28% | p = 0.043, SE = ±82% |
| Measurement Time | PAS rTMS 3p 96% SO | PAS rTMS 3p 120% MT | PAS TMS 1p 100% SO | rTMS 3p 96% SO |
|---|---|---|---|---|
| Post 60 min | p = 0.345 | p = 0.144 | p = 0. 465 | p = 0.5 |
| Post 30 min | p = 0.225 | p = 0.068 | p = 0. 465 | p = 0.5 |
| Post 0 min | p = 0.043* | p = 0.465 | p = 0. 465 | p = 0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sathyan, S.; Tolmacheva, A.; Tugin, S.; Mäkelä, J.P.; Shulga, A.; Lioumis, P. A New Paired Associative Stimulation Protocol with High-Frequency Peripheral Component and High-Intensity 20 Hz Repetitive Transcranial Magnetic Stimulation—A Pilot Study. Int. J. Environ. Res. Public Health 2021, 18, 11224. https://doi.org/10.3390/ijerph182111224
Sathyan S, Tolmacheva A, Tugin S, Mäkelä JP, Shulga A, Lioumis P. A New Paired Associative Stimulation Protocol with High-Frequency Peripheral Component and High-Intensity 20 Hz Repetitive Transcranial Magnetic Stimulation—A Pilot Study. International Journal of Environmental Research and Public Health. 2021; 18(21):11224. https://doi.org/10.3390/ijerph182111224
Chicago/Turabian StyleSathyan, Sabin, Aleksandra Tolmacheva, Sergei Tugin, Jyrki P. Mäkelä, Anastasia Shulga, and Pantelis Lioumis. 2021. "A New Paired Associative Stimulation Protocol with High-Frequency Peripheral Component and High-Intensity 20 Hz Repetitive Transcranial Magnetic Stimulation—A Pilot Study" International Journal of Environmental Research and Public Health 18, no. 21: 11224. https://doi.org/10.3390/ijerph182111224
APA StyleSathyan, S., Tolmacheva, A., Tugin, S., Mäkelä, J. P., Shulga, A., & Lioumis, P. (2021). A New Paired Associative Stimulation Protocol with High-Frequency Peripheral Component and High-Intensity 20 Hz Repetitive Transcranial Magnetic Stimulation—A Pilot Study. International Journal of Environmental Research and Public Health, 18(21), 11224. https://doi.org/10.3390/ijerph182111224






