Nutritional Composition and Bioactive Compounds in Three Different Parts of Mango Fruit
Abstract
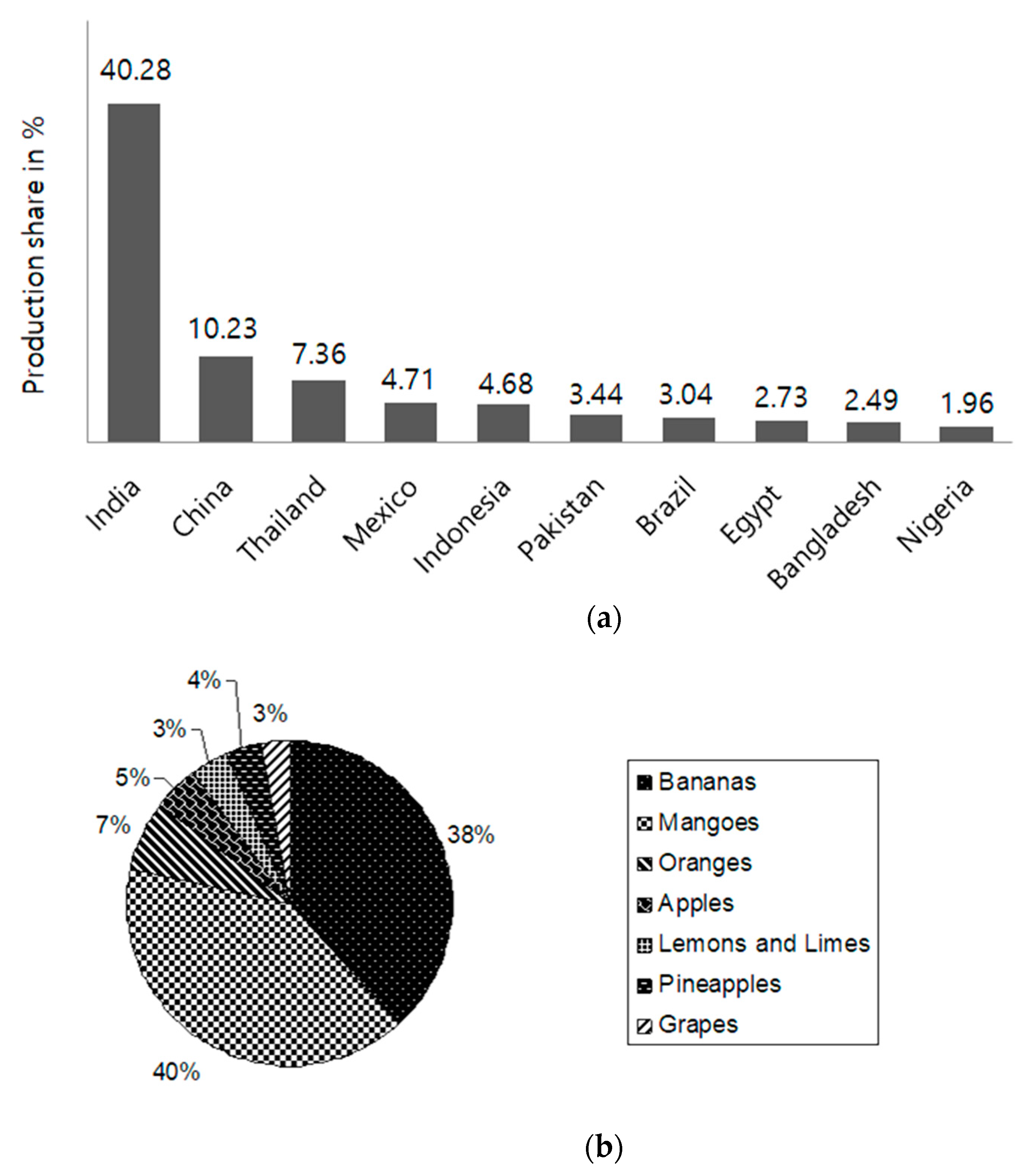
1. Introduction
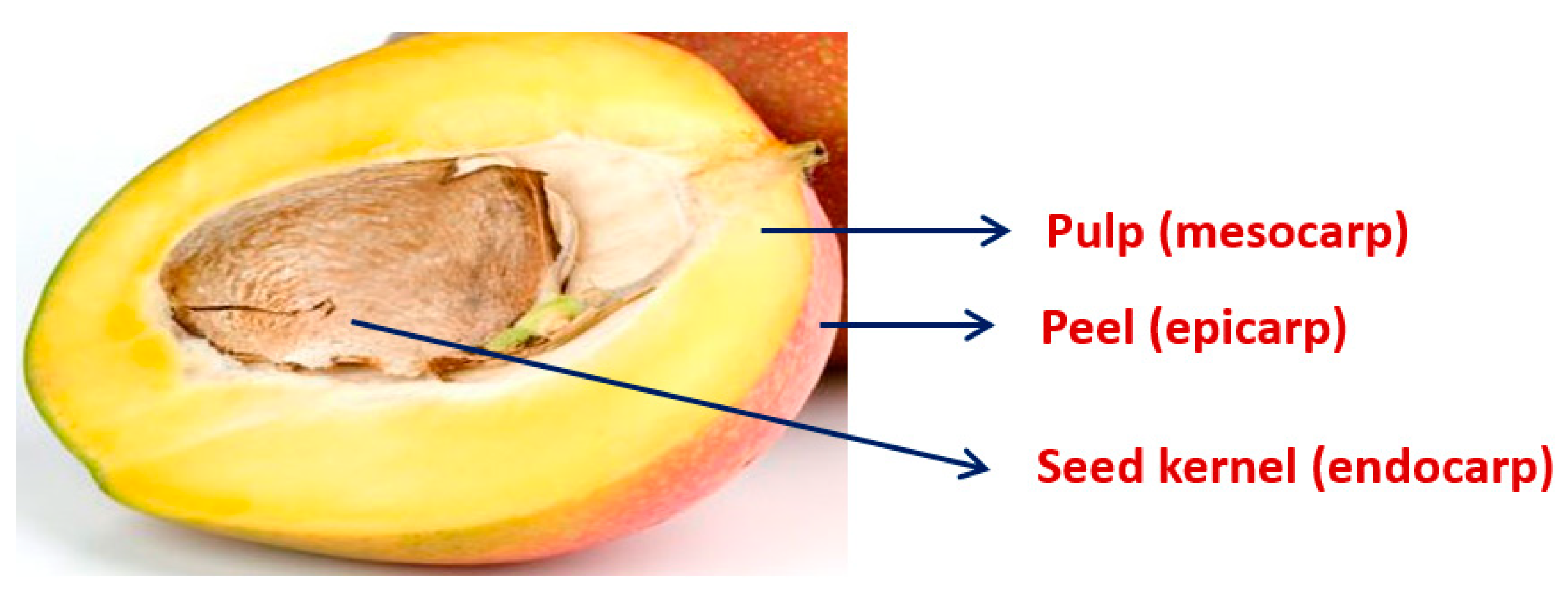
2. Mango Pulp
2.1. Nutritional Composition
2.1.1. Macronutrients
2.1.2. Minerals and Vitamins (Micronutrients)
| Compound (Per 100 g) | Pulp | Peel | Seed Kernel |
|---|---|---|---|
| Water (g) | 83.46 | 72.5 | 9.1 |
| Energy (kcal) | 60 | 327 | |
| Carbohydrate, by difference (g) | 14.98 | 28.2 | 18.2 |
| Protein (g) | 0.82 | 3.6 | 6.61 |
| Total lipid (fat) (g) | 0.38 | 2.2 | 9.4 |
| Sugars, total (g) | 25 | 70 | |
| Total dietary fiber (g) | 1.6 | 40–72.5 | 2.8 |
| Minerals (mg) | |||
| Calcium (Ca) | 11 | 150 | 450 |
| Iron (Fe) | 0.16 | 40.6 | 11.9 |
| Magnesium (Mg) | 10 | 100 | 100 |
| Phosphorus (P) | 14 | - | 140 |
| Potassium (K) | 168 | 75 | 365 |
| Sodium (Na) | 1 | 50 | 150 |
| Zinc (Zn) | 0.09 | 1.74 | 1.1 |
| Copper (Cu) | 0.04–0.32 | 10.4 | - |
| Selenium (Se) | 0–0.6 | - | - |
| Vitamins | |||
| Vitamin C (total ascorbic acid, mg) | 36.4 | 18–257 | 17 |
| Thiamin (mg) | 0.028 | 0.08 | |
| Riboflavin (mg) | 0.038 | 0.13 | |
| Niacin (mg) | 0.669 | 0.19 | |
| Pantothenic acid (mg) | 0.119 | 0.12 | |
| Folate, dietary folate equivalents (μg) | 43 | - | |
| Vitamin A, retinol activity equivalents (μg) | 54 | 100 | - |
| Vitamin E (α-tocopherol, mg) | 0.9 | 0.25–0.59 | 1.3 |
| Vitamin A (IU) | 1082 | - | 15 |
| Vitamin K (phylloquinone, μg) | 4.2 | 59 | |
| Vitamin B12 | 0.12 | ||
| Organic acids | |||
| Citric acid (%) | 0.7 | ||
| Mallic acid (%) | 0.5 | ||
| Polyphenols | |||
| Cyanidin (mg) | 0.1 | ||
| Catechin (mg) | 1.7 | ||
| Kaempferol (mg) | 0.1 | 3.6 | |
| Myricetin (mg) | 0.1 | ||
| Proanthocyanidin dimers (mg) | 1.8 | ||
| Proanthocyanidin trimers (mg) | 1.4 | ||
| Proanthocyanidin 4–6 mers (mg) | 7.2 | ||
| Gallic acid | 0.69 | 23–838 | |
| Ellagic acid | 3–156 | ||
| Coumarin | 12.7 | ||
| Caffeic acid | 7.7 | ||
| Vanillin | 202 | ||
| Cinnamic acid | 11.2 | ||
| Ferulic acid | 10.4 | ||
| Mangiferin (mg) | 169 | 4.2 | |
| Mangiferin gallate | 321 | ||
| Isomangiferin | 13.4 | ||
| Isomangiferin gallate | 82 | ||
| Quercetin | 2.2 | 6.5 | |
| Rhamnetin 3-0 galactoside/glucoside | 9.4 | ||
| Tannins | 20.7 | ||
| Flavonoids (catechin equivalent/100 g) | 0.9–9.2 | 19.91–75.35 | |
| Anthocyanins | 360–565 | ||
| Cyaniding | 22.1 | ||
| Pelargonidins | 22.73 | ||
| Delphinidins | 18.02 | ||
| Malvidins | 5.26 | ||
| Petunidins | 21.6 | ||
| Peonidins | 24.42 | ||
| Carotenoids (μg) | 3092 | ||
| β-carotene | 640 | 1310 | |
| α-carotene | 9 | ||
| β-cryptoxanthin | 10 | 600 | |
| Lycopene | 3 | ||
| Lutein and zeaxanthin | 23 | 299 | |
2.1.3. Lipids and Fatty Acids
2.1.4. Organic Acids
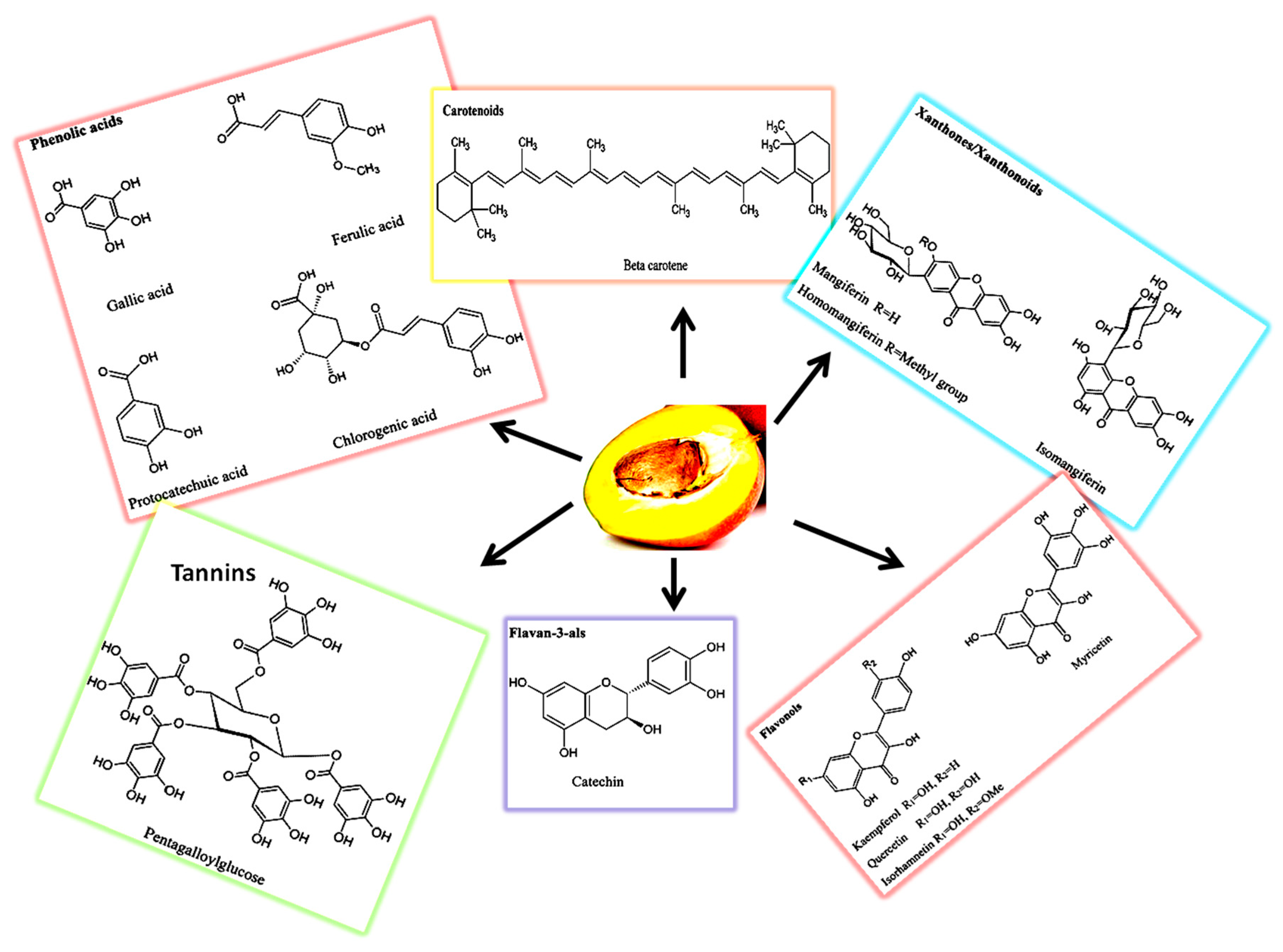
2.2. Phytochemical Composition
2.2.1. Phenolic Compounds
Phenolic Acids
2.2.2. Pigments
Chlorophylls
Carotenoids
2.2.3. Flavonoids and Flavanols
2.2.4. Phytosterols
3. Mango Peel
3.1. Nutritional Composition
3.2. Phytochemical Composition
3.2.1. Polyphenols
3.2.2. Carotenoids
4. Mango Seed Kernel (MSK)
4.1. Nutritional Composition
4.1.1. Carbohydrates
4.1.2. Proteins and Amino Acids
4.1.3. Lipids and Fatty Acids
4.1.4. Minerals and Vitamins
4.2. Phytochemical Composition
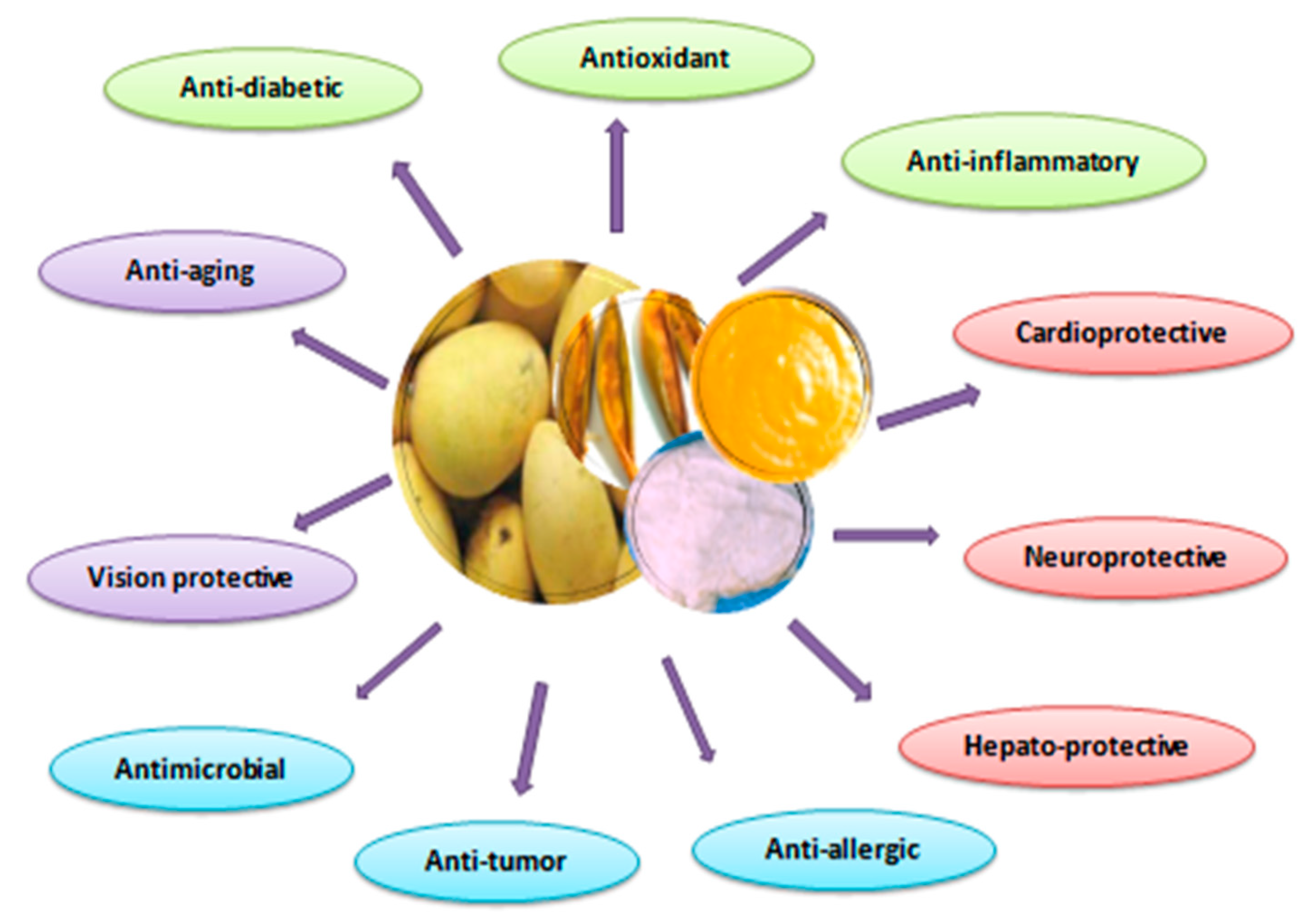
5. Therapeutic and Health-Promoting Effects of Mango Fruit
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- National Mango Database. Indian Status of Mango: Area, Production and Productivity-Growth Pattern; National Mango Database. Available online: https://mangifera.res.in/ (accessed on 6 September 2020).
- Altendorf, S. Major Tropical Fruits Market Review 2017; FAO: Rome, Italy, 2019; pp. 1–10. [Google Scholar]
- APEDA-Agricultural and Processed Food Products Export Development Authority. Products-Fresh Fruits and Vegetable: Mango; Ministry of Commerce and Industry, Government of India: New Delhi, India, 2020; Volume 2020.
- Krishnamurthi, A. The Wealth of India: Raw Materials VI. Publications and Information Directorate; CSIR: New Delhi, India, 1962. [Google Scholar]
- Schreinemachers, P.; Simmons, E.B.; Wopereis, M.C.S. Tapping the economic and nutritional power of vegetables. Glob. Food Secur. 2018, 16, 36–45. [Google Scholar] [CrossRef]
- Haque, S.; Akbar, D.; Kinnear, S. The variable impacts of extreme weather events on fruit production in subtropical Australia. Sci. Hortic. 2020, 262, 109050. [Google Scholar] [CrossRef]
- Rajan, S. Phenological Responses to Temperature and Rainfall: A Case Study of Mango. In National Agricultural; Bioversity International Office for South Asia: New Delhi, India, 2012. [Google Scholar]
- Haldankar, P.; Burondkar, M.; Singh, A.; Saitwal, Y. Sustainable mango production technology for climatic aberration in coastal agroclimate of Maharashtra. Adv. Agric. Res. Technol. J. 2020, 37, 74–87. [Google Scholar]
- Olesen, T. Late 20th century warming in a coastal horticultural region and its effects on tree phenology. N. Z. J. Crop Hortic. Sci. 2011, 39, 119–129. [Google Scholar] [CrossRef]
- Normand, F.; Lauri, P.-E.; Legave, J.M. Climate change and its probable effects on mango production and cultivation. Acta Hortic. 2015, 1075, 21–31. [Google Scholar] [CrossRef]
- Giri, K.; Krishna Murthy, D.; Narashimha Rao, P. Separation of organic acids. J. Indian Inst. Sci. A 1953, 35, 77–98. [Google Scholar]
- Shang, H.S.; Chen, C.J.; Shih, Y.L.; Peng, S.F.; Chen, Y.L.; Liu, K.C.; Huang, H.C.; Hsueh, S.C.; Chen, K.W.; Lu, H.F. Mangiferin induces immune responses and evaluates the survival rate in WEHI-3 cell generated mouse leukemia in vivo. Environ. Toxicol. 2021, 36, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Baliga, M.S.; Rao, S.; Rao, P.; Hegde, S.K.; Akbar, K.C.J.; Abraham, S.; George, T.; Palatty, P.L. Use of Indian Indigenous Fruits in Cancer Prevention and Treatment. In Anticancer Plants: Properties and Application; Springer: Berlin/Heidelberg, Germany, 2018; pp. 57–76. [Google Scholar]
- Zeng, Z.; Lin, C.; Wang, S.; Wang, P.; Xu, W.; Ma, W.; Wang, J.; Xiang, Q.; Liu, Y.; Yang, J.; et al. Suppressive activities of mangiferin on human epithelial ovarian cancer. Phytomedicine 2020, 76, 153267. [Google Scholar] [CrossRef]
- Rajendran, P.; Rengarajan, T.; Nandakumar, N.; Divya, H.; Nishigaki, I. Mangiferin in cancer chemoprevention and treatment: Pharmacokinetics and molecular targets. J. Recept. Signal Transduct. 2015, 35, 76–84. [Google Scholar] [CrossRef]
- Sánchez-Camargo, A.D.P.; Ballesteros-Vivas, D.; Buelvas-Puello, L.M.; Martinez-Correa, H.A.; Parada-Alfonso, F.; Cifuentes, A.; Ferreira, S.R.S.; Gutiérrez, L.-F. Microwave-assisted extraction of phenolic compounds with antioxidant and anti-proliferative activities from supercritical CO2 pre-extracted mango peel as valorization strategy. LWT 2021, 137, 110414. [Google Scholar] [CrossRef]
- Reddy, L.V.; Reddy, O.V.S.; Wee, Y.-J. Production of ethanol from mango (Mangifera indica L.) peel by Saccharomyces cerevisiae CFTRI101. Afr. J. Biotechnol. 2011, 10, 4183–4189. [Google Scholar]
- Burton-Freeman, B.M.; Sandhu, A.K.; Edirisinghe, I. Mangos and their bioactive components: Adding variety to the fruit plate for health. Food Funct. 2017, 8, 3010–3032. [Google Scholar] [CrossRef]
- Gerg, N. Composting of Mango Peel. Cent. Inst. Subtrop. Hortic. 2016, 1–3. Available online: https://www.cish.res.in/introduction.php (accessed on 15 January 2021).
- Reddy, L.V. Production and Characterization of Wine Like Product from Mango Fruits Mangifera indica L.; Sri Venkateswara University: Tirupati, India, 2005. [Google Scholar]
- Ramasamy, M.; Srirangarayan, R.S.; Shanmugasundaram, R.; Ramesh, P.; Shrirangasami, S.; Prasanthrajan, M.; Saravanakumar, S. Composting of Mango Wastes. Biot. Res. Today 2020, 2, 1034–1035. [Google Scholar]
- Athiappan, M.; Srinivasan, S.; Anandan, R.; Rajaram, J. Chapter 20—Novel Process of Ellagic Acid Synthesis from Waste Generated from Mango Pulp Processing Industries. In Emerging Technologies in Environmental Bioremediation; Shah, M.P., Rodriguez-Couto, S., Şengör, S.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 443–454. [Google Scholar] [CrossRef]
- World Health Organization. Report of a Join WHO/FAO Expert Consultation. In Diet Nutrition and the Prevention of Chronic Diseases: WHO Technical Report Series; WHO: Jeneva, Switzerland, 2003. [Google Scholar]
- Bartley, G.E.; Scolnik, P.A. Plant carotenoids: Pigments for photoprotection, visual attraction, and human health. Plant Cell 1995, 7, 1027. [Google Scholar]
- Maldonado-Celis, M.E.; Yahia, E.M.; Bedoya, R.; Landázuri, P.; Loango, N.; Aguillón, J.; Restrepo, B.; Ospina, J.C.G. Chemical composition of mango (Mangifera indica L.) fruit: Nutritional and phytochemical compounds. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Dar, M.S.; Oak, P.; Chidley, H.; Deshpande, A.; Giri, A.; Gupta, V. Nutrient and flavor content of mango (Mangifera indica L.) cultivars: An appurtenance to the list of staple foods. In Nutritional Composition of Fruit Cultivars; Elsevier: Amsterdam, The Netherlands, 2016; pp. 445–467. [Google Scholar]
- Ellong, E.N.; Adenet, S.; Rochefort, K. Physicochemical, nutritional, organoleptic characteristics and food applications of four mango (Mangifera indica) varieties. Food Nutr. Sci. 2015, 6, 242. [Google Scholar]
- Abbasi, A.M.; Guo, X.; Fu, X.; Zhou, L.; Chen, Y.; Zhu, Y.; Yan, H.; Liu, R.H. Comparative assessment of phenolic content and in vitro antioxidant capacity in the pulp and peel of mango cultivars. Int. J. Mol. Sci. 2015, 16, 13507–13527. [Google Scholar] [CrossRef]
- Lemmens, L.; Tchuenche, E.S.; Van Loey, A.M.; Hendrickx, M.E. Beta-carotene isomerisation in mango puree as influenced by thermal processing and high-pressure homogenisation. Eur. Food Res. Technol. 2013, 236, 155–163. [Google Scholar] [CrossRef]
- Reddy, L.; Reddy, O.V.S. Production and characterization of wine from mango fruit (Mangifera indica L.). World J. Microbiol. Biotechnol. 2005, 21, 1345–1350. [Google Scholar] [CrossRef]
- Yashoda, H.M.; Prabha, T.N.; Tharanathan, R.N. Mango ripening: Changes in cell wall constituents in relation to textural softening. J. Sci. Food Agric. 2006, 86, 713–721. [Google Scholar] [CrossRef]
- Kalra, S.; Tandon, D. Ripening-behaviour of ‘Dashehari’ mango in relation to harvest period. Sci. Hortic. 1983, 19, 263–269. [Google Scholar] [CrossRef]
- Tasneem, A. Postharvest Treatments to Reduce Chilling Injury Symptoms in Stored Mangoes. Master’s Thesis, McGill University, Ste-Anne-de-Bellevue, QC, Canada, 2004. [Google Scholar]
- Bello-Pérez, L.A.; García-Suárez, F.; Agama-Acevedo, E. Mango carbohydrates. Food 2007, 1, 36–40. [Google Scholar]
- US Department of Agriculture. USDA National Nutrient Database for Standard Reference, Release 1 April. Available online: https://ndb.nal.usda.gov/ndb (accessed on 15 June 2020).
- Tharanathan, R.; Yashoda, H.; Prabha, T. Mango (Mangifera indica L.) “The king of fruits”—An overview. Food Rev. Int. 2006, 22, 95–123. [Google Scholar] [CrossRef]
- Vallarino, J.G.; Osorio, S. Chapter 10: Organic Acids. In Postharvest Physiology and Biochemistry of Fruits and Vegetables; Yahia, E., Carrillo-López, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 207–224. [Google Scholar] [CrossRef]
- Diplock, A.; Charuleux, J.-L.; Crozier-Willi, G.; Kok, F.; Rice-Evans, C.; Roberfroid, M.; Stahl, W.; Vina-Ribes, J. Functional food science and defence against reactive oxidative species. Br. J. Nutr. 1998, 80, S77–S112. [Google Scholar] [CrossRef]
- Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: Advisory Report to the Secretary of Health and Human Services and the Secretary of Agriculture; Dietary Guidelines Advisory Committee: Washington, DC, USA, 2015.
- Shenai, J.P. Vitamin A supplementation in very low birth weight neonates: Rationale and evidence. Pediatrics 1999, 104, 1369–1374. [Google Scholar] [CrossRef]
- Muoki, P.N.; Makokha, A.O.; Onyango, C.A.; Ojijo, N.K. Potential contribution of mangoes to reduction of vitamin A deficiency in Kenya. Ecol. Food Nutr. 2009, 48, 482–498. [Google Scholar] [CrossRef]
- Matheyambath, A.C.; Subramanian, J.; Paliyath, G. Mangoes reference module in food science. In Encyclopedia of Food and Health; Paul, B.C., Told, F.F., Eds.; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar] [CrossRef]
- Singh, R.K.; Ali, S.A.; Nath, P.; Sane, V.A. Activation of ethylene-responsive p-hydroxyphenylpyruvate dioxygenase leads to increased tocopherol levels during ripening in mango. J. Exp. Bot. 2011, 62, 3375–3385. [Google Scholar] [CrossRef]
- Robles-Sánchez, R.; Islas-Osuna, M.; Astiazarán-García, H.; Vázquez-Ortiz, F.; Martín-Belloso, O.; Gorinstein, S.; González-Aguilar, G. Quality index, consumer acceptability, bioactive compounds, and antioxidant activity of fresh-cut “Ataulfo” mangoes (Mangifera indica L.) as affected by low-temperature storage. J. Food Sci. 2009, 74, S126–S134. [Google Scholar]
- Mène-Saffrané, L. Vitamin E biosynthesis and its regulation in plants. Antioxidants 2018, 7, 2. [Google Scholar] [CrossRef]
- Pathak, S.; Sarada, R. Lipids of mango (Mangifera indica). Curr. Sci. 1974, 43, 716–717. [Google Scholar]
- Deshpande, A.B.; Anamika, K.; Jha, V.; Chidley, H.G.; Oak, P.S.; Kadoo, N.Y.; Pujari, K.H.; Giri, A.P.; Gupta, V.S. Transcriptional transitions in Alphonso mango (Mangifera indica L.) during fruit development and ripening explain its distinct aroma and shelf life characteristics. Sci. Rep. 2017, 7, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, C.; Gholap, A.S. Changes in fatty acids in ripening mango pulp (var Alphonso). J. Agric. Food Chem. 1973, 21, 496–497. [Google Scholar] [CrossRef]
- Shashirekha, M.; Patwardhan, M. Changes in amino acids, sugars and nonvolatile organic acids in a ripening mango fruit (Mangifera indica, Badami variety). LWT Lebensmitt Wissensch Technol. 1976, 9, 369–370. [Google Scholar]
- Robineau, L.; Soejarto, D.D. TRAMIL: A research project on the medicinal plant resources of the Caribbean. Med. Resour. Trop. For. Biodivers. Importance Hum. Health 1996, 1, 317–325. [Google Scholar]
- Palafox-Carlos, H.; Yahia, E.; González-Aguilar, G. Identification and quantification of major phenolic compounds from mango (Mangifera indica, cv. Ataulfo) fruit by HPLC–DAD–MS/MS-ESI and their individual contribution to the antioxidant activity during ripening. Food Chem. 2012, 135, 105–111. [Google Scholar] [CrossRef]
- Hu, K.; Dars, A.G.; Liu, Q.; Xie, B.; Sun, Z. Phytochemical profiling of the ripening of Chinese mango (Mangifera indica L.) cultivars by real-time monitoring using UPLC-ESI-QTOF-MS and its potential benefits as prebiotic ingredients. Food Chem. 2018, 256, 171–180. [Google Scholar] [CrossRef]
- Ramirez, J.E.; Zambrano, R.; Sepúlveda, B.; Simirgiotis, M.J. Antioxidant properties and hyphenated HPLC-PDA-MS profiling of Chilean Pica mango fruits (Mangifera indica L. cv. piqueño). Molecules 2014, 19, 438–458. [Google Scholar] [CrossRef]
- Berardini, N.; Fezer, R.; Conrad, J.; Beifuss, U.; Carle, R.; Schieber, A. Screening of mango (Mangifera indica L.) cultivars for their contents of flavonol O-and xanthone C-glycosides, anthocyanins, and pectin. J. Agric. Food Chem. 2005, 53, 1563–1570. [Google Scholar] [CrossRef]
- Ribeiro, S.; Barbosa, L.; Queiroz, J.; Knödler, M.; Schieber, A. Phenolic compounds and antioxidant capacity of Brazilian mango (Mangifera indica L.) varieties. Food Chem. 2008, 110, 620–626. [Google Scholar] [CrossRef]
- Ediriweera, M.K.; Tennekoon, K.H.; Samarakoon, S.R. A review on ethnopharmacological applications, pharmacological activities, and bioactive compounds of Mangifera indica (mango). Evid.-Based Complement. Altern. Med. 2017, 2017. [Google Scholar] [CrossRef]
- Imran, M.; Arshad, M.S.; Butt, M.S.; Kwon, J.-H.; Arshad, M.U.; Sultan, M.T. Mangiferin: A natural miracle bioactive compound against lifestyle related disorders. Lipids Health Dis. 2017, 16, 84. [Google Scholar] [PubMed]
- Sudhakar, P.; Latha, P.; Reddy, P. Phenotyping Crop Plants for Physiological and Biochemical Traits; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Lee, J.; Schwartz, S. Pigments in Plant Foods: Handbook of Food Science, Technology and Engineering; CRC Taylor: New York, NY, USA, 2006. [Google Scholar]
- Choo, W.S. Fruit Pigment Changes during Ripening; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–7. [Google Scholar]
- Varakumar, S.; Kumar, Y.S.; Reddy, O.V.S. Carotenoid composition of mango (Mangifera indica L.) wine and its antioxidant activity. J. Food Biochem. 2011, 35, 1538–1547. [Google Scholar] [CrossRef]
- Manthey, J.A.; Perkins-Veazie, P. Influences of harvest date and location on the levels of β-carotene, ascorbic acid, total phenols, the in vitro antioxidant capacity, and phenolic profiles of five commercial varieties of mango (Mangifera indica L.). J. Agric. Food Chem. 2009, 57, 10825–10830. [Google Scholar] [CrossRef] [PubMed]
- Barreto, J.C.; Trevisan, M.T.; Hull, W.E.; Erben, G.; De Brito, E.S.; Pfundstein, B.; Würtele, G.; Spiegelhalder, B.; Owen, R.W. Characterization and quantitation of polyphenolic compounds in bark, kernel, leaves, and peel of mango (Mangifera indica L.). J. Agric. Food Chem. 2008, 56, 5599–5610. [Google Scholar] [CrossRef] [PubMed]
- Berardini, N.; Carle, R.; Schieber, A. Characterization of gallotannins and benzophenone derivatives from mango (Mangifera indica L. cv. ‘Tommy Atkins’) peels, pulp and kernels by high-performance liquid chromatography/electrospray ionization mass spectrometry. Rapid Commun. Mass Spectrom. 2004, 18, 2208–2216. [Google Scholar]
- Vilela, C.; Santos, S.A.; Oliveira, L.; Camacho, J.F.; Cordeiro, N.; Freire, C.S.; Silvestre, A.J. The ripe pulp of Mangifera indica L.: A rich source of phytosterols and other lipophilic phytochemicals. Food Res. Int. 2013, 54, 1535–1540. [Google Scholar] [CrossRef]
- Beerh, O.; Raghuramaiah, B.; Krishnamurthy, G.; Giridhar, N. Utilization of mango waste: Recovery of juice from waste pulp and peel. J. Food Sci. Technol. 1976, 13, 138–141. [Google Scholar]
- Larrauri, J. New approaches in the preparation of high dietary fibre powders from fruit by-products. Trends Food Sci. Technol. 1999, 10, 3–8. [Google Scholar] [CrossRef]
- Tokas, J.; Punia, H.; Baloda, S.; Sheokand, R.N. Mango Peel: A Potential Source of Bioactive Compounds and Phytochemicals. Austin Food Sci. 2020, 5, 1035. [Google Scholar]
- Ajila, C.; Rao, U.P. Mango peel dietary fibre: Composition and associated bound phenolics. J. Funct. Foods 2013, 5, 444–450. [Google Scholar] [CrossRef]
- Njiru, M.; Nawiri, P.; Wanjau, R.; Odundo, J. Residues of Mangifera indica L. as alternative animal feed in Embu county, Kenya. Green Chem. 2014, 16, 1–10. [Google Scholar]
- Lakshminarayana, S.; Subhadra, N.; Subramanyam, H. Some aspects of developmental physiology of the mango fruit. J. Hortic. Sci. 1970, 45, 133–142. [Google Scholar] [CrossRef]
- Ajila, C.; Bhat, S.; Rao, U.P. Valuable components of raw and ripe peels from two Indian mango varieties. Food Chem. 2007, 102, 1006–1011. [Google Scholar] [CrossRef]
- Schieber, A.; Berardini, N.; Carle, R. Identification of flavonol and xanthone glycosides from mango (Mangifera indica L. cv. “Tommy Atkins”) peels by high-performance liquid chromatography-electrospray ionization mass spectrometry. J. Agric. Food Chem. 2003, 51, 5006–5011. [Google Scholar] [CrossRef]
- Medlicott, A.; Bhogal, M.; Reynolds, S. Changes in peel pigmentation during ripening of mango fruit (Mangifera indica var. Tommy Atkins). Ann. Appl. Biol. 1986, 109, 651–656. [Google Scholar] [CrossRef]
- Ranganath, K.G.; Shivashankara, K.S.; Roy, T.K.; Dinesh, M.R.; Geetha, G.A.; Pavithra, K.C.; Ravishankar, K.V. Profiling of anthocyanins and carotenoids in fruit peel of different colored mango cultivars. J. Food Sci. Technol. 2018, 55, 4566–4577. [Google Scholar] [CrossRef]
- Kayesh, E.; Shangguan, L.; Korir, N.K.; Sun, X.; Bilkish, N.; Zhang, Y.; Han, J.; Song, C.; Cheng, Z.-M.; Fang, J. Fruit skin color and the role of anthocyanin. Acta Physiol. Plant. 2013, 35, 2879–2890. [Google Scholar] [CrossRef]
- Rahman, M.M.; Ichiyanagi, T.; Komiyama, T.; Sato, S.; Konishi, T. Effects of anthocyanins on psychological stress-induced oxidative stress and neurotransmitter status. J. Agric. Food Chem. 2008, 56, 7545–7550. [Google Scholar] [CrossRef]
- Giampieri, F.; Alvarez-Suarez, J.M.; Mazzoni, L.; Forbes-Hernandez, T.Y.; Gasparrini, M.; Gonzàlez-Paramàs, A.M.; Santos-Buelga, C.; Quiles, J.L.; Bompadre, S.; Mezzetti, B. An anthocyanin-rich strawberry extract protects against oxidative stress damage and improves mitochondrial functionality in human dermal fibroblasts exposed to an oxidizing agent. Food Funct. 2014, 5, 1939–1948. [Google Scholar] [CrossRef]
- Yang, L.; Ling, W.; Du, Z.; Chen, Y.; Li, D.; Deng, S.; Liu, Z.; Yang, L. Effects of anthocyanins on cardiometabolic health: A systematic review and meta-analysis of randomized controlled trials. Adv. Nutr. 2017, 8, 684–693. [Google Scholar] [CrossRef]
- Skates, E.; Overall, J.; DeZego, K.; Wilson, M.; Esposito, D.; Lila, M.A.; Komarnytsky, S. Berries containing anthocyanins with enhanced methylation profiles are more effective at ameliorating high fat diet-induced metabolic damage. Food Chem. Toxicol. 2018, 111, 445–453. [Google Scholar] [CrossRef]
- Puravankara, D.; Boghra, V.; Sharma, R.S. Effect of antioxidant principles isolated from mango (Mangifera indica L) seed kernels on oxidative stability of buffalo ghee (butter-fat). J. Sci. Food Agric. 2000, 80, 522–526. [Google Scholar]
- Bhalerao, S.; Mulmuley, G.; Anathakrishna, S.; Potty, V. Wash and waste water management in food industry. Fruit and vegetable processing. Indian Food Pack. 1989, 43, 5–11. [Google Scholar]
- Odunsi, A. Response of laying hens and growing broilers to the dietary inclusion of mango (Mangifera indica L.) seed kernel meal. Trop. Anim. Health Prod. 2005, 37, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Yadav, K.; Garg, N.; Verma, A.; Kumar, S.; Trivedi, M. Optimization and extraction of oil from mango seed kernel (Mangifera Indica). Indian J. Agric. Sci. 2017, 87, 943–946. [Google Scholar]
- Hemavathy, J.; Prabhakar, J.; Sen, D. Drying and storage behaviour of mango (Mangifera indica) seeds and composition of kernel fat. Asean Food J. (Malays.) 1988, 4, 59–63. [Google Scholar]
- Muchiri, D.R.; Mahungu, S.M.; Gituanja, S.N. Studies on mango (Mangifera indica, L.) kernel fat of some Kenyan varieties in Meru. J. Am. Oil Chem. Soc. 2012, 89, 1567–1575. [Google Scholar]
- Jahurul, M.; Zaidul, I.; Norulaini, N.; Sahena, F.; Jaffri, J.; Omar, A.M. Supercritical carbon dioxide extraction and studies of mango seed kernel for cocoa butter analogy fats. Cyta. J. Food 2014, 12, 97–103. [Google Scholar]
- Ribeiro, S.M.R.; Schieber, A. Bioactive compounds in mango (Mangifera indica L.). In Bioactive Foods in Promoting Health; Elsevier: Amsterdam, The Netherlands, 2010; pp. 507–523. [Google Scholar]
- O’Shea, N.; Arendt, E.K.; Gallagher, E. Dietary fibre and phytochemical characteristics of fruit and vegetable by-products and their recent applications as novel ingredients in food products. Innov. Food Sci. Emerg. Technol. 2012, 16, 1–10. [Google Scholar]
- Diarra, S.S. Potential of mango (Mangifera indica L.) seed kernel as a feed ingredient for poultry: A review. Worlds Poult. Sci. J. 2014, 70, 279–288. [Google Scholar]
- Lakshminarayana, G.; Chandrasekhara, R.T.; Ramalingaswamy, P. Varietal variations in content, characteristics and composition of mango seeds and fat. J. Am. Oil Chem. Soc. 1983, 60, 88–89. [Google Scholar] [CrossRef]
- Sandhu, K.S.; Lim, S.-T. Structural characteristics and in vitro digestibility of mango kernel starches (Mangifera indica L.). Food Chem. 2008, 107, 92–97. [Google Scholar] [CrossRef]
- Abdalla, A.E.; Darwish, S.M.; Ayad, E.H.; El-Hamahmy, R.M. Egyptian mango by-product 1. Compositional quality of mango seed kernel. Food Chem. 2007, 103, 1134–1140. [Google Scholar] [CrossRef]
- Arogba, S.S. The performance of processed mango (Mangifera indica) kernel flour in a model food system. Bioresour. Technol. 1999, 70, 277–281. [Google Scholar] [CrossRef]
- Dhingra, S.; Kapoor, A.C. Nutritive value of mango seed kernel. J. Sci. Food Agric. 1985, 36, 752–756. [Google Scholar] [CrossRef]
- Solís-Fuentes, J.A.; del Carmen Durán-de-Bazúa, M. Mango (Mangifera indica L.) seed and its fats. In Nuts and Seeds in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2011; pp. 741–748. [Google Scholar]
- Youssef, A. Utilization of the seeds of mango processing wastes as a secondary source of oil and protein. Alex. J. Agric. Res. 1999, 44, 149–166. [Google Scholar]
- Nadeem, M.; Imran, M.; Khalique, A. Promising features of mango (Mangifera indica L.) kernel oil: A review. J. Food Sci. Technol. 2016, 53, 2185–2195. [Google Scholar] [CrossRef]
- Abdel-Razik, M.; Ashoush, I.; Yassin, N. Characteristics of mango seed kernel butter and its effects on quality attributes of muffins. Alex J. Food Sci. Technol. 2012, 9, 1–9. [Google Scholar]
- Azeem, W.; Nadeem, M.; Ahmad, S. Stabilization of winterized cottonseed oil with chia (Salvia hispanica l.) seed extract at ambient temperature. J. Food Sci. Technol. 2015, 52, 7191–7199. [Google Scholar] [CrossRef]
- Dhara, R.; Bhattacharyya, D.K.; Ghosh, M. Analysis of sterol and other components present in unsaponifiable matters of mahua, sal and mango kernel oil. J. Oleo Sci. 2010, 59, 169–176. [Google Scholar] [CrossRef]
- Fowomola, M. Some nutrients and antinutrients contents of mango (Magnifera indica) seed. Afr. J. Food Sci. 2010, 4, 472–476. [Google Scholar]
- Soong, Y.-Y.; Barlow, P.J. Quantification of gallic acid and ellagic acid from longan (Dimocarpus longan Lour.) seed and mango (Mangifera indica L.) kernel and their effects on antioxidant activity. Food Chem. 2006, 97, 524–530. [Google Scholar] [CrossRef]
- Kittiphoom, S.; Sutasinee, S. Mango seed kernel oil and its physicochemical properties. Int. Food Res. J. 2013, 20, 1145. [Google Scholar]
- Abdel-Aty, A.M.; Salama, W.H.; Hamed, M.B.; Fahmy, A.S.; Mohamed, S.A. Phenolic-antioxidant capacity of mango seed kernels: Therapeutic effect against viper venoms. Rev. Bras. Farmacogn. 2018, 28, 594–601. [Google Scholar] [CrossRef]
- Dorta, E.; González, M.; Lobo, M.G.; Sánchez-Moreno, C.; de Ancos, B. Screening of phenolic compounds in by-product extracts from mangoes (Mangifera indica L.) by HPLC-ESI-QTOF-MS and multivariate analysis for use as a food ingredient. Food Res. Int. 2014, 57, 51–60. [Google Scholar] [CrossRef]
- Dorta, E.; Lobo, M.G.; González, M. Using drying treatments to stabilise mango peel and seed: Effect on antioxidant activity. LWT Food Sci. Technol. 2012, 45, 261–268. [Google Scholar] [CrossRef]
- Lauricella, M.; Emanuele, S.; Calvaruso, G.; Giuliano, M.; D’Anneo, A. Multifaceted health benefits of Mangifera indica L. (Mango): The inestimable value of orchards recently planted in Sicilian rural areas. Nutrients 2017, 9, 525. [Google Scholar] [CrossRef] [PubMed]
- Du, Z.; Fanshi, F.; Lai, Y.-H.; Chen, J.-R.; Hao, E.; Deng, J.; Hsiao, C.-D. Mechanism of anti-dementia effects of mangiferin in a senescence accelerated mouse (SAMP8) model. Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef]
- Gelabert-Rebato, M.; Wiebe, J.C.; Martin-Rincon, M.; Galvan-Alvarez, V.; Curtelin, D.; Perez-Valera, M.; Juan Habib, J.; Pérez-López, A.; Vega, T.; Morales-Alamo, D. Enhancement of exercise performance by 48 hours, and 15-day supplementation with mangiferin and luteolin in men. Nutrients 2019, 11, 344. [Google Scholar] [CrossRef]
- Kim, H.; Venancio, V.P.; Fang, C.; Dupont, A.W.; Talcott, S.T.; Mertens-Talcott, S.U. Mango (Mangifera indica L.) polyphenols reduce IL-8, GRO, and GM-SCF plasma levels and increase Lactobacillus species in a pilot study in patients with inflammatory bowel disease. Nutr. Res. 2020, 75, 85–94. [Google Scholar] [CrossRef]
- Fam, V.W.; Holt, R.R.; Keen, C.L.; Sivamani, R.K.; Hackman, R.M. Prospective Evaluation of Mango Fruit Intake on Facial Wrinkles and Erythema in Postmenopausal Women: A Randomized Clinical Pilot Study. Nutrients 2020, 12, 3381. [Google Scholar] [CrossRef]
- Calderon-Montano, J.M.; Burgos-Morón, E.; Pérez-Guerrero, C.; López-Lázaro, M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Munafo, J.P., Jr.; Didzbalis, J.; Schnell, R.J.; Schieberle, P.; Steinhaus, M. Characterization of the major aroma-active compounds in mango (Mangifera indica L.) cultivars Haden, White Alfonso, Praya Sowoy, Royal Special, and Malindi by application of a comparative aroma extract dilution analysis. J. Agric. Food Chem. 2014, 62, 4544–4551. [Google Scholar] [CrossRef]
- Khare, P.; Shanker, K. Mangiferin: A review of sources and interventions for biological activities. BioFactors 2016, 42, 504–514. [Google Scholar]
- Formica, J.; Regelson, W. Review of the biology of quercetin and related bioflavonoids. Food Chem. Toxicol. 1995, 33, 1061–1080. [Google Scholar] [CrossRef]
- Croft, K.D. The chemistry and biological effects of flavonoids and phenolic acids a. Ann. N. Y. Acad. Sci. 1998, 854, 435–442. [Google Scholar] [CrossRef]
- Moskaug, J.Ø.; Carlsen, H.; Myhrstad, M.; Blomhoff, R. Molecular imaging of the biological effects of quercetin and quercetin-rich foods. Mech. Ageing Dev. 2004, 125, 315–324. [Google Scholar] [CrossRef]
- Scartezzini, P.; Speroni, E. Review on some plants of Indian traditional medicine with antioxidant activity. J. Ethnopharmacol. 2000, 71, 23–43. [Google Scholar] [CrossRef]
- Abbasi, A.M.; Liu, F.; Guo, X.; Fu, X.; Li, T.; Liu, R.H. Phytochemical composition, cellular antioxidant capacity and antiproliferative activity in mango (Mangifera indica L.) pulp and peel. Int. J. Food Sci. Technol. 2017, 52, 817–826. [Google Scholar] [CrossRef]
- Knoedler, M.; Wenzig, E.; Bauer, R.; Conrad, J.; Beifuss, U.; Carle, R.; Schieber, A. Cyclooxygenase inhibitory 5-alkenylresorcinols isolated from mango (Mangifera indica L.) peels. Planta Med. 2007, 73, P_031. [Google Scholar] [CrossRef]
- Wauthoz, N.; Balde, A.; Balde, E.S.; Van Damme, M.; Duez, P. Ethnopharmacology of Mangifera indica L. bark and pharmacological studies of its main C-glucosylxanthone, mangiferin. Int. J. Biomed. Pharm. Sci. 2007, 1, 112–119. [Google Scholar]
- Telang, M.; Dhulap, S.; Mandhare, A.; Hirwani, R. Therapeutic and cosmetic applications of mangiferin: A patent review. Expert Opin. Ther. Pat. 2013, 23, 1561–1580. [Google Scholar] [CrossRef]
- Garrido, G.; González, D.; Delporte, C.; Backhouse, N.; Quintero, G.; Núñez-Sellés, A.J.; Morales, M.A. Analgesic and anti-inflammatory effects of Mangifera indica L. extract (Vimang). Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2001, 15, 18–21. [Google Scholar]
- Park, S.-N.; Lim, Y.K.; Freire, M.O.; Cho, E.; Jin, D.; Kook, J.-K. Antimicrobial effect of linalool and α-terpineol against periodontopathic and cariogenic bacteria. Anaerobe 2012, 18, 369–372. [Google Scholar] [CrossRef]
- González-Aguilar, G.; Wang, C.; Buta, J.; Krizek, D. Use of UV-C irradiation to prevent decay and maintain postharvest quality of ripe ‘Tommy Atkins’ mangoes. Int. J. Food Sci. Technol. 2001, 36, 767–773. [Google Scholar] [CrossRef]
| Characteristic/Composition | Content |
|---|---|
| Oil (%) | 11.5 |
| Free fatty acid (FFA%) | 0.22 |
| Moisture (%) | 0.21 |
| Iodine Value | 54.6 |
| Refractive Index | 1.457 |
| Melting Point (°C) | 35.2 |
| Saponification | 193 |
| Unsaponifiable Matter (%) | 1.68 |
| Peroxide Value (meq O2/kg) | 0.65 |
| Color | R2.8 + 30Y |
| C16:0 | 7.43 |
| C18:0 | 37.5 |
| C18:1 | 45.59 |
| C18:2 | 5.48 |
| C18:3 | 0.40 |
| C20:0 | 2.48 |
| C22:0 | 0.45 |
| C24:0 | 0.40 |
| Campesterol | 0.07% |
| Stigma Sterol | 10.66 |
| β-Sitosterol | 58.63 |
| Δ5-Avenasterol | 10.19 |
| Δ7-Stigmasterol | 4.34 |
| Δ7-Avenasterol | 19.10 |
| Biochemical Property and Name of Part Used | Name of the Compound | Reference |
|---|---|---|
| 1. Antioxidant Activity | ||
| Peel | 3,4-Dihydroxybenzoic acid (protocatechuic acid) | [63] |
| Fruit pulp | Kaempferol | [73,113] |
| Fruit pulp | Linalool | [114] |
| Fruit pulp | Mangiferin | [115] |
| Fruit pulp | Quercetin | [116,117,118] |
| Fruit pulp | β-Carotene | [119] |
| Peel | β-Carotene | [120] |
| 2. Anti-Inflammatory Activity | ||
| Peel | 5-(11Z-Heptadecenyl)-resorcinol and | [121] |
| 5-(8,Z,11,Z-Heptadecadienyl)-resorcinol | ||
| Fruit pulp | Kaempferol | [55,73,113] |
| Fruit pulp | Mangiferin | [115,122,123] |
| Fruit pulp | Shikimic acid | [124] |
| 3. Antimicrobial Activity | ||
| Fruit pulp | Kaempferol | [55,73,113] |
| Fruit pulp | Mangiferin | [115,122,123] |
| Fruit pulp | Quercetin | [116,117,118] |
| Peel | 3,4-Dihydroxybenzoic acid | [63] |
| (protocatechuic acid) | ||
| 4. Anti-Diabetic and Anti-Obesity Activity | ||
| Fruit pulp | Kaempferol | [55,73,113] |
| Peel | 3,4-Dihydroxybenzoic acid (protocatechuic acid) | [63] |
| Fruit pulp | Mangiferin | [115,122,123] |
| Fruit pulp | β-Carotene | [119] |
| Fruit pulp | Quercetin | [116,117,118] |
| 5. Cytotoxic and Apoptotic Activity | ||
| Fruit pulp | Kaempferol | [55,73,113] |
| Fruit pulp | Linalool | [114] |
| Fruit pulp | Mangiferin | [115,122,123] |
| Fruit pulp | Quercetin | [116,117,118] |
| Fruit pulp | β-Carotene | [119] |
| Peel | 3,4-Dihydroxybenzoic acid | [63] |
| (protocatechuic acid) | ||
| Seed | Mangiferin | [15] |
| Seed | Gallic acid | [103] |
| 6. Neuroprotective Activity | ||
| Fruit pulp | Linalool | [125] |
| Fruit pulp | Mangiferin | [115,122,123] |
| 7. Cardio-Protective | ||
| Fruit pulp | Mangiferin | [115,122,123] |
| 8. Anticoagulant/Antithrombotic | ||
| Fruit pulp | Shikimic acid | [126] |
| 9. Blood-Pressure-Lowering Activity | ||
| Fruit pulp | Quercetin | [116,117,118] |
| 10. Gastro-Protective | ||
| Seed | Mangiferin | [103] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lebaka, V.R.; Wee, Y.-J.; Ye, W.; Korivi, M. Nutritional Composition and Bioactive Compounds in Three Different Parts of Mango Fruit. Int. J. Environ. Res. Public Health 2021, 18, 741. https://doi.org/10.3390/ijerph18020741
Lebaka VR, Wee Y-J, Ye W, Korivi M. Nutritional Composition and Bioactive Compounds in Three Different Parts of Mango Fruit. International Journal of Environmental Research and Public Health. 2021; 18(2):741. https://doi.org/10.3390/ijerph18020741
Chicago/Turabian StyleLebaka, Veeranjaneya Reddy, Young-Jung Wee, Weibing Ye, and Mallikarjuna Korivi. 2021. "Nutritional Composition and Bioactive Compounds in Three Different Parts of Mango Fruit" International Journal of Environmental Research and Public Health 18, no. 2: 741. https://doi.org/10.3390/ijerph18020741
APA StyleLebaka, V. R., Wee, Y.-J., Ye, W., & Korivi, M. (2021). Nutritional Composition and Bioactive Compounds in Three Different Parts of Mango Fruit. International Journal of Environmental Research and Public Health, 18(2), 741. https://doi.org/10.3390/ijerph18020741








