Comparison of Interventional Strategies to Improve Recovery after Eccentric Exercise-Induced Muscle Fatigue
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
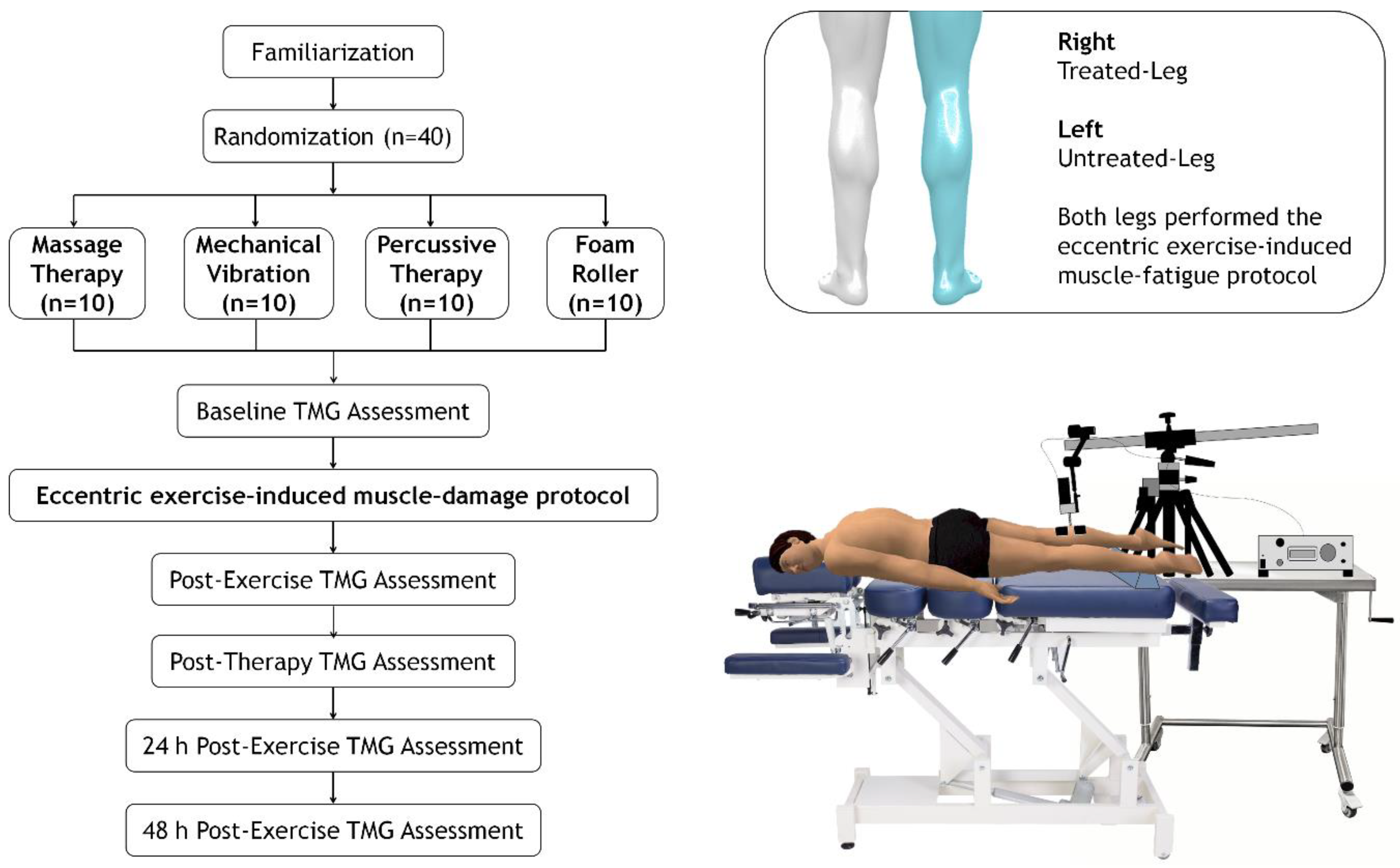
2.2. Trial Design
2.3. Procedures
2.3.1. Eccentric Exercise-Induced Muscle Fatigue Protocol
2.3.2. Manual Therapy
2.3.3. Mechanical Vibration
2.3.4. Percussion Therapy
2.3.5. Foam Roller
2.4. Outcomes
2.5. Statistical Analysis
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malm, C.; Jakobsson, J.; Isaksson, A. Physical Activity and Sports-Real Health Benefits: A Review with Insight into the Public Health of Sweden. Sports 2019, 7, 127. [Google Scholar] [CrossRef] [PubMed]
- Kellmann, M.; Bertollo, M.; Bosquet, L.; Brink, M.; Coutts, A.J.; Duffield, R.; Erlacher, D.; Halson, S.L.; Hecksteden, A.; Heidari, J.; et al. Recovery and Performance in Sport: Consensus Statement. Int. J. Sports Physiol. Perform. 2018, 13, 240–245. [Google Scholar] [CrossRef]
- Fuller, J.T.; Thomson, R.L.; Howe, P.R.; Buckley, J.D. Vibration Therapy is No More Effective than the Standard Practice of Massage and Stretching for Promoting Recovery From Muscle Damage After Eccentric Exercise. Clin. J. Sport Med. 2015, 25, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wang, Y.; Lu, J.; You, Y.; Zhang, L.; Zhu, D.; Yao, F. Does vibration benefit delayed-onset muscle soreness? A meta-analysis and systematic review. J. Int. Med. Res. 2019, 47, 3–18. [Google Scholar] [CrossRef] [PubMed]
- Romero-Moraleda, B.; Gonzalez-Garcia, J.; Cuellar-Rayo, A.; Balsalobre-Fernandez, C.; Munoz-Garcia, D.; Morencos, E. Effects of Vibration and Non-Vibration Foam Rolling on Recovery after Exercise with Induced Muscle Damage. J. Sports Sci. Med. 2019, 18, 172–180. [Google Scholar]
- Weerapong, P.; Hume, P.A.; Kolt, G.S. The mechanisms of massage and effects on performance, muscle recovery and injury prevention. Sports Med. 2005, 35, 235–256. [Google Scholar] [CrossRef]
- Zainuddin, Z.; Newton, M.; Sacco, P.; Nosaka, K. Effects of massage on delayed-onset muscle soreness, swelling, and recovery of muscle function. J. Athl. Train. 2005, 40, 174–180. [Google Scholar]
- Veqar, Z.; Imtiyaz, S. Vibration Therapy in Management of Delayed Onset Muscle Soreness (DOMS). J. Clin. Diagn. Res. 2014, 8, LE01–LE04. [Google Scholar] [CrossRef]
- Comeaux, Z. Dynamic fascial release and the role of mechanical/vibrational assist devices in manual therapies. J. Bodyw. Mov. Ther. 2011, 15, 35–41. [Google Scholar] [CrossRef][Green Version]
- Cerciello, S.; Rossi, S.; Visona, E.; Corona, K.; Oliva, F. Clinical applications of vibration therapy in orthopaedic practice. Muscles Ligaments Tendons J. 2016, 6, 147–156. [Google Scholar] [CrossRef]
- Martinez Rodriguez, R.; Galan del Rio, F. Mechanistic basis of manual therapy in myofascial injuries. Sonoelastographic evolution control. J. Bodyw. Mov. Ther. 2013, 17, 221–234. [Google Scholar] [CrossRef] [PubMed]
- Schleip, R.; Muller, D.G. Training principles for fascial connective tissues: Scientific foundation and suggested practical applications. J. Bodyw. Mov. Ther. 2013, 17, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Romero-Moraleda, B.; La Touche, R.; Lerma-Lara, S.; Ferrer-Pena, R.; Paredes, V.; Peinado, A.B.; Munoz-Garcia, D. Neurodynamic mobilization and foam rolling improved delayed-onset muscle soreness in a healthy adult population: A randomized controlled clinical trial. PeerJ 2017, 5, e3908. [Google Scholar] [CrossRef] [PubMed]
- Pearcey, G.E.; Bradbury-Squires, D.J.; Kawamoto, J.E.; Drinkwater, E.J.; Behm, D.G.; Button, D.C. Foam rolling for delayed-onset muscle soreness and recovery of dynamic performance measures. J. Athl. Train. 2015, 50, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Garcia, O.; Cuba-Dorado, A.; Alvarez-Yates, T.; Carballo-Lopez, J.; Iglesias-Caamano, M. Clinical utility of tensiomyography for muscle function analysis in athletes. Open Access J. Sports Med. 2019, 10, 49–69. [Google Scholar] [CrossRef]
- Dahmane, R.; Djordjevic, S.; Simunic, B.; Valencic, V. Spatial fiber type distribution in normal human muscle Histochemical and tensiomyographical evaluation. J. Biomech. 2005, 38, 2451–2459. [Google Scholar] [CrossRef]
- Beato, M.; Madruga-Parera, M.; Piqueras-Sanchiz, F.; Moreno-Perez, V.; Romero-Rodriguez, D. Acute Effect of Eccentric Overload Exercises on Change of Direction Performance and Lower-Limb Muscle Contractile Function. J. Strength Cond. Res. 2019. [Google Scholar] [CrossRef]
- Bollinger, L.M.; Brantley, J.T.; Tarlton, J.K.; Baker, P.A.; Seay, R.F.; Abel, M.G. Construct Validity, Test-Retest Reliability, and Repeatability of Performance Variables Using a Flywheel Resistance Training Device. J. Strength Cond. Res. 2018, 34, 3149–3156. [Google Scholar] [CrossRef]
- Smith, L.L.; Keating, M.N.; Holbert, D.; Spratt, D.J.; McCammon, M.R.; Smith, S.S.; Israel, R.G. The effects of athletic massage on delayed onset muscle soreness, creatine kinase, and neutrophil count: A preliminary report. J. Orthop. Sports Phys. Ther. 1994, 19, 93–99. [Google Scholar] [CrossRef]
- Aminian-Far, A.; Hadian, M.R.; Olyaei, G.; Talebian, S.; Bakhtiary, A.H. Whole-body vibration and the prevention and treatment of delayed-onset muscle soreness. J. Athl. Train. 2011, 46, 43–49. [Google Scholar] [CrossRef]
- Baumgart, C.; Freiwald, J.; Kuhnemann, M.; Hotfiel, T.; Huttel, M.; Hoppe, M.W. Foam Rolling of the Calf and Anterior Thigh: Biomechanical Loads and Acute Effects on Vertical Jump Height and Muscle Stiffness. Sports 2019, 7, 27. [Google Scholar] [CrossRef]
- Rey, E.; Lago-Penas, C.; Lago-Ballesteros, J. Tensiomyography of selected lower-limb muscles in professional soccer players. J. Electromyogr. Kinesiol. 2012, 22, 866–872. [Google Scholar] [CrossRef]
- de Paula Simola, R.A.; Harms, N.; Raeder, C.; Kellmann, M.; Meyer, T.; Pfeiffer, M.; Ferrauti, A. Assessment of neuromuscular function after different strength training protocols using tensiomyography. J. Strength Cond. Res. 2015, 29, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.J.; Rogers, E.L.; Granata, K.P. Active trunk stiffness increases with co-contraction. J. Electromyogr. Kinesiol. 2006, 16, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Simunic, B.; Degens, H.; Rittweger, J.; Narici, M.; Mekjavic, I.B.; Pisot, R. Noninvasive estimation of myosin heavy chain composition in human skeletal muscle. Med. Sci. Sports Exerc. 2011, 43, 1619–1625. [Google Scholar] [CrossRef] [PubMed]
- Rusu, L.D.; Cosma, G.G.; Cernaianu, S.M.; Marin, M.N.; Rusu, P.F.; Ciocanescu, D.P.; Neferu, F.N. Tensiomyography method used for neuromuscular assessment of muscle training. J. Neuroeng. Rehabil. 2013, 10, 67. [Google Scholar] [CrossRef]
- Whitehead, N.P.; Weerakkody, N.S.; Gregory, J.E.; Morgan, D.L.; Proske, U. Changes in passive tension of muscle in humans and animals after eccentric exercise. J. Physiol. 2001, 533, 593–604. [Google Scholar] [CrossRef]
- Macgregor, L.J.; Hunter, A.M.; Orizio, C.; Fairweather, M.M.; Ditroilo, M. Assessment of Skeletal Muscle Contractile Properties by Radial Displacement: The Case for Tensiomyography. Sports Med. 2018, 48, 1607–1620. [Google Scholar] [CrossRef]
- Simunic, B.; Pisot, R.; Rittweger, J.; Degens, H. Age-Related Slowing of Contractile Properties Differs between Power, Endurance, and Nonathletes: A Tensiomyographic Assessment. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 1602–1608. [Google Scholar] [CrossRef]
- Simunic, B.; Koren, K.; Rittweger, J.; Lazzer, S.; Reggiani, C.; Rejc, E.; Pisot, R.; Narici, M.; Degens, H. Tensiomyography detects early hallmarks of bed-rest-induced atrophy before changes in muscle architecture. J. Appl. Physiol. (1985) 2019, 126, 815–822. [Google Scholar] [CrossRef]
- Harmsen, J.F.; Franz, A.; Mayer, C.; Zilkens, C.; Buhren, B.A.; Schrumpf, H.; Krauspe, R.; Behringer, M. Tensiomyography parameters and serum biomarkers after eccentric exercise of the elbow flexors. Eur. J. Appl. Physiol. 2019, 119, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Chai, J.H.; Bae, S.W. Evaluation of Calf Muscular Function during the Recovery Phase after the Repair of an Achilles Tendon Rupture. Front. Surg. 2019, 6, 57. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.T.; Ryan, A.M.F.; Vallance, S.R.; Dias-Dougan, A.; Dugdale, J.H.; Hunter, A.M.; Hamilton, D.L.; Macgregor, L.J. Tensiomyography Derived Parameters Reflect Skeletal Muscle Architectural Adaptations Following 6-Weeks of Lower Body Resistance Training. Front. Physiol. 2019, 10, 1493. [Google Scholar] [CrossRef] [PubMed]
- Piqueras-Sanchiz, F.; Martin-Rodriguez, S.; Martinez-Aranda, L.M.; Lopes, T.R.; Raya-Gonzalez, J.; Garcia-Garcia, O.; Nakamura, F.Y. Effects of moderate vs. high iso-inertial loads on power, velocity, work and hamstring contractile function after flywheel resistance exercise. PLoS ONE 2019, 14, e0211700. [Google Scholar]
- Giovanelli, N.; Taboga, P.; Rejc, E.; Simunic, B.; Antonutto, G.; Lazzer, S. Effects of an Uphill Marathon on Running Mechanics and Lower-Limb Muscle Fatigue. Int. J. Sports Physiol. Perform. 2016, 11, 522–529. [Google Scholar] [CrossRef]
- Dupuy, O.; Douzi, W.; Theurot, D.; Bosquet, L.; Dugué, B. An Evidence-Based Approach for Choosing Post-exercise Recovery Techniques to Reduce Markers of Muscle Damage, Soreness, Fatigue, and Inflammation: A Systematic Review With Meta-Analysis. Front. Physiol. 2018, 9, 403. [Google Scholar] [CrossRef]
- Fernandez-Gonzalo, R.; Lundberg, T.R.; Alvarez-Alvarez, L.; de Paz, J.A. Muscle damage responses and adaptations to eccentric-overload resistance exercise in men and women. Eur. J. Appl. Physiol. 2014, 114, 1075–1084. [Google Scholar] [CrossRef]
- Pierrynowski, M.R.; Morrison, J.B. A physiological model for the evaluation of muscular forces in human locomotion: Theoretical aspects. Math. Biosci. 1985, 75, 69–101. [Google Scholar] [CrossRef]
- de Benito, A.M.; Valldecabres, R.; Ceca, D.; Richards, J.; Barrachina Igual, J.; Pablos, A. Effect of vibration vs non-vibration foam rolling techniques on flexibility, dynamic balance and perceived joint stability after fatigue. PeerJ 2019, 7, e8000. [Google Scholar] [CrossRef]
- Laffaye, G.; Da Silva, D.T.; Delafontaine, A. Self-Myofascial Release Effect with Foam Rolling on Recovery after High-Intensity Interval Training. Front. Physiol. 2019, 10, 1287. [Google Scholar] [CrossRef]
- Wilke, J.; Vleeming, A.; Wearing, S. Overuse Injury: The Result of Pathologically Altered Myofascial Force Transmission? Exerc. Sport Sci. Rev. 2019, 47, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.L.; Alabed, S.; Chico, T.J.A. Effect of sports massage on performance and recovery: A systematic review and meta-analysis. BMJ Open Sport Exerc. Med. 2020, 6, e000614. [Google Scholar] [CrossRef] [PubMed]
| MT | MV | PT | FR | ||
|---|---|---|---|---|---|
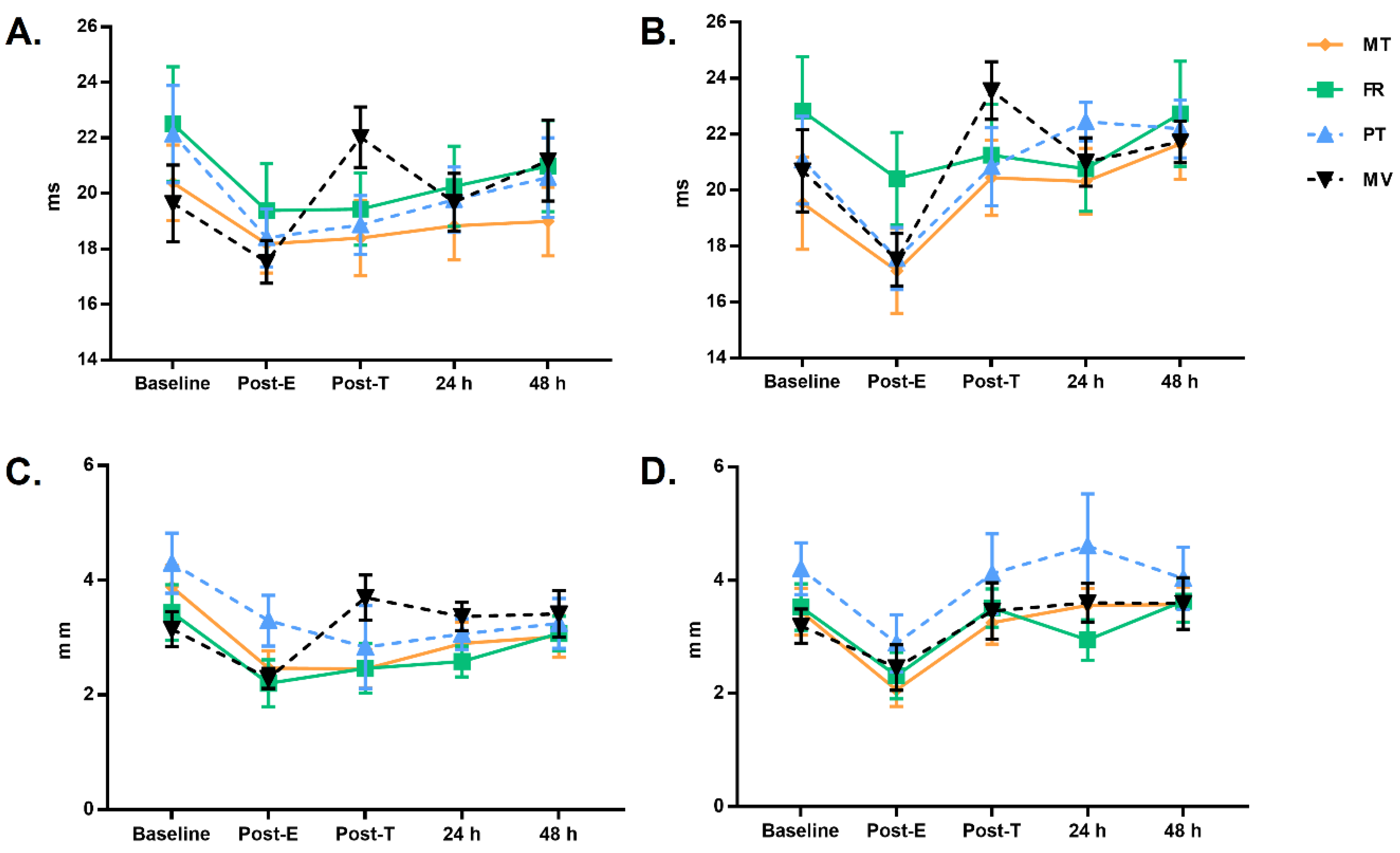
| Tc (ms) | |||||
| Pre-Fatigue | UT | 20.4 ± 0.7 (18.9–21.9) | 19.6 ± 0.7 (18.2–21.1) | 22.1 ± 0.7 (20.6–23.6) | 22.5 ± 0.7 (21.0–24.0) |
| T | 19.5 ± 0.7 (18.0–21.0) | 20.7 ± 0.7 (19.2–22.2) | 21.1 ± 0.7 (19.6–22.6) | 22.8 ± 0.7 (21.3–24.3) | |
| Post-Fatigue | UT | 18.2 ± 0.5 (17.1–19.2) | 17.5 ± 0.5 (16.5–18.6) | 18.4 ± 0.5 (17.3–19.5) | 19.4 ± 0.5 (18.3–20.5) |
| T | 17.1 ± 0.6 (15.9–18.3) | 17.5 ± 0.6 (16.3–18.7) | 17.6 ± 0.6 (16.4–18.8) | 20.4 ± 0.6 (19.2–21.6) | |
| Post-0 | UT | 18.4 ± 0.5 (17.3–19.5) | 22.0 ± 0.5 (20.9–23.1) | 18.9 ± 0.5 (17.8–19.9) | 19.4 ± 0.5 (18.4–20.5) |
| T | 20.4 ± 0.6 (19.2–21.7) | 23.6 ± 0.6 (22.3–24.8) | 20.8 ± 0.6 (19.6–22.1) | 21.3 ± 0.6 (20.0–22.5) | |
| Post-24 h | UT | 18.8 ± 0.5 (17.7–19.9) | 19.7 ± 0.5 (18.6–20.8) | 19.8 ± 0.5 (18.7–20.9) | 20.3 ± 0.5 (19.2–21.4) |
| T | 20.3 ± 0.5 (19.3–21.3) | 21.0 ± 0.5 (20.0–22.0) | 22.5 ± 0.5 (21.5–23.4) | 20.8 ± 0.5 (19.8–21.8) | |
| Post-48 h | UT | 19.0 ± 0.6 (17.7–20.3) | 21.2 ± 0.6 (19.9–22.5) | 20.6 ± 0.6 (19.3–21.9) | 21.0 ± 0.6 (19.7–22.3) |
| T | 21.7 ± 0.6 (20.5–22.8) | 21.7 ± 0.6 (20.6–22.9) | 22.2 ± 0.6 (21.0–23.4) | 22.7 ± 0.6 (21.6–23.9) | |
| Dm (mm) | |||||
| Pre-Fatigue | UT | 3.9 ± 0.2 (3.5–4.3) | 3.1 ± 0.2 (2.8–3.5) | 4.3 ± 0.2 (3.9–4.7) | 3.4 ± 0.2 (3.0–3.8) |
| T | 3.4 ± 0.2 (3.1–3.8) | 3.2 ± 0.2 (2.8–3.6) | 4.2 ± 0.2 (3.8–4.6) | 3.5 ± 0.2 (3.2–3.9) | |
| Post-Fatigue | UT | 2.5 ± 0.2 (2.2–2.8) | 2.3 ± 0.2 (2.0–2.6) | 3.3 ± 0.2 (3.0–3.6) | 2.2 ± 0.2 (1.9–2.5) |
| T | 2.1 ± 0.2 (1.7–2.4) | 2.5 ± 0.2 (2.1–2.8) | 2.9 ± 0.2 (2.5–3.3) | 2.3 ± 0.2 (1.9–2.7) | |
| Post-0 | UT | 2.5 ± 0.2 (2.0–2.9) | 3.7 ± 0.2 (3.3–4.1) | 2.8 ± 0.2 (2.4–3.3) | 2.5 ± 0.2 (2.0–2.9) |
| T | 3.3 ± 0.2 (2.8–3.7) | 3.5 ± 0.2 (3.0–3.9) | 4.1 ± 0.2 (3.7–4.6) | 3.5 ± 0.2 (3.1–4.0) | |
| Post-24 h | UT | 2.9 ± 0.1 (2.6–3.2) | 3.4 ± 0.1 (3.1–3.6) | 3.1 ± 0.1 (2.8–3.3) | 2.6 ± 0.1 (2.3–2.8) |
| T | 3.6 ± 0.2 (3.1–4.0) | 3.6 ± 0.2 (3.1–4.1) | 4.6 ± 0.2 (4.1–5.1) | 2.9 ± 0.2 (2.5–3.4) | |
| Post-48 h | UT | 3.0 ± 0.2 (2.7–3.4) | 3.4 ± 0.2 (3.1–3.8) | 3.2 ± 0.2 (2.9–3.6) | 3.1 ± 0.2 (2.7–3.4) |
| T | 3.6 ± 0.2 (3.2–4.0) | 3.6 ± 0.2 (3.2–4.0) | 4.0 ± 0.2 (3.6–4.4) | 3.6 ± 0.2 (3.2–4.0) | |
| Origin | Measure | F | Sig. | η2p |
|---|---|---|---|---|
| Time | Tc | 61.65 | <0.01 | 0.63 |
| Dm | 64.12 | <0.01 | 0.64 | |
| Therapy | Tc | 1.96 | 0.137 | 0.14 |
| Dm | 5.82 | 0.002 | 0.33 | |
| Leg | Tc | 50.01 | <0.01 | 0.58 |
| Dm | 27.58 | <0.01 | 0.43 | |
| Time × Leg | Tc | 27.71 | <0.01 | 0.43 |
| Dm | 23.16 | <0.01 | 0.39 | |
| Time × Therapy | Tc | 9.47 | <0.01 | 0.44 |
| Dm | 5.33 | <0.01 | 0.31 | |
| Leg × Therapy | Tc | 0.17 | 0.917 | 0.01 |
| Dm | 3.30 | 0.031 | 0.22 | |
| Time × Leg × Therapy | Tc | 5.76 | <0.01 | 0.32 |
| Dm | 5.93 | <0.01 | 0.33 |
| Variable | Time | Protocols | Mean Diff * | Sig.b | Lower Limit | Upper Limit |
|---|---|---|---|---|---|---|
| Untreated leg | ||||||
| Tc | Post-T | MV vs. MT | 3.6 | 0.000 | 1.5 | 5.7 |
| Post-T | MV vs. PT | 3.2 | 0.001 | 1.1 | 5.3 | |
| Post-T | MV vs. FR | 2.6 | 0.009 | 0.5 | 4.7 | |
| Dm | Baseline | PT vs. MV | 1.2 | 0.001 | 0.4 | 1.9 |
| Baseline | PT vs. FR | 0.9 | 0.018 | 0.1 | 1.6 | |
| Post-E | PT vs. FR | 1.1 | 0.000 | 0.5 | 1.7 | |
| Post-E | PT vs. MV | 1.0 | 0.000 | 0.4 | 1.6 | |
| Post-E | PT vs. MT | 0.8 | 0.003 | 0.2 | 1.4 | |
| Post-T | MT vs. MV | −1.2 | 0.002 | −2.1 | −0.4 | |
| Post-T | MV vs. FR | 1.2 | 0.002 | 0.4 | 2.1 | |
| Post-T | PT vs. MV | −0.9 | 0.048 | −1.7 | 0.0 | |
| 24 h | MV vs. FR | 0.8 | 0.001 | 0.3 | 1.3 | |
| Treated leg | ||||||
| Tc | Basal | FR vs. MT | 3.3 | 0.019 | 0.4 | 6.2 |
| Post-E | FR vs. PT | 2.9 | 0.009 | 0.5 | 5.2 | |
| Post-E | FR vs. MV | 2.9 | 0.008 | 0.6 | 5.2 | |
| Post-E | FR vs. MT | 3.3 | 0.002 | 1.0 | 5.6 | |
| Post-T | PT vs. MV | −2.7 | 0.025 | −5.2 | −0.2 | |
| Post-T | MV vs. MT | 3.1 | 0.008 | 0.6 | 5.6 | |
| 24 h | PT vs. MT | 2.1 | 0.023 | 0.2 | 4.1 | |
| Dm | Baseline | PT vs. MT | 0.8 | 0.027 | 0.1 | 1.5 |
| Baseline | PT vs. MV | 1.0 | 0.002 | 0.3 | 1.7 | |
| Post | PT vs. MT | 0.8 | 0.017 | 0.1 | 1.5 | |
| 24 h | PT vs. MV | 1.0 | 0.033 | 0.1 | 2.0 | |
| 24 h | PT vs. MT | −1.0 | 0.023 | 0.1 | 2.0 | |
| 24 h | PT vs. FR | 3.3 | 0.000 | 0.7 | 2.6 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Sillero, M.; Benítez-Porres, J.; García-Romero, J.; Bonilla, D.A.; Petro, J.L.; Vargas-Molina, S. Comparison of Interventional Strategies to Improve Recovery after Eccentric Exercise-Induced Muscle Fatigue. Int. J. Environ. Res. Public Health 2021, 18, 647. https://doi.org/10.3390/ijerph18020647
García-Sillero M, Benítez-Porres J, García-Romero J, Bonilla DA, Petro JL, Vargas-Molina S. Comparison of Interventional Strategies to Improve Recovery after Eccentric Exercise-Induced Muscle Fatigue. International Journal of Environmental Research and Public Health. 2021; 18(2):647. https://doi.org/10.3390/ijerph18020647
Chicago/Turabian StyleGarcía-Sillero, Manuel, Javier Benítez-Porres, Jerónimo García-Romero, Diego A. Bonilla, Jorge L. Petro, and Salvador Vargas-Molina. 2021. "Comparison of Interventional Strategies to Improve Recovery after Eccentric Exercise-Induced Muscle Fatigue" International Journal of Environmental Research and Public Health 18, no. 2: 647. https://doi.org/10.3390/ijerph18020647
APA StyleGarcía-Sillero, M., Benítez-Porres, J., García-Romero, J., Bonilla, D. A., Petro, J. L., & Vargas-Molina, S. (2021). Comparison of Interventional Strategies to Improve Recovery after Eccentric Exercise-Induced Muscle Fatigue. International Journal of Environmental Research and Public Health, 18(2), 647. https://doi.org/10.3390/ijerph18020647










