Sex-Specific Differences in the Toxic Effects of Heavy Fuel Oil on Sea Urchin (Strongylocentrotus intermedius)
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Water-Accommodated Fractions (WAF) of 380# Heavy Fuel Oil (HFO)
2.2. Sea Urchin Maintenance and Toxicity Tests
2.3. Chemicals Analysis
2.4. Biochemical Analysis
2.5. Statistical Analysis
3. Results
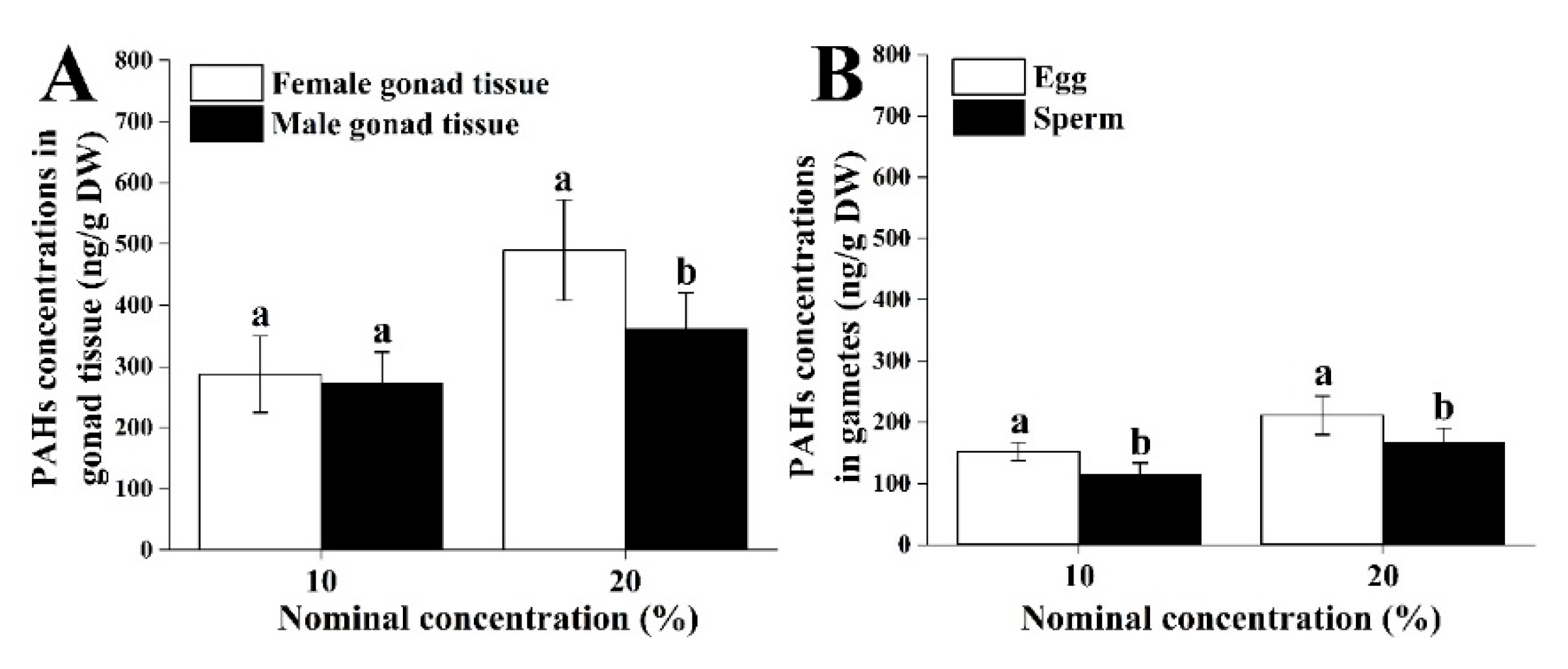
3.1. Analytical Chemistry
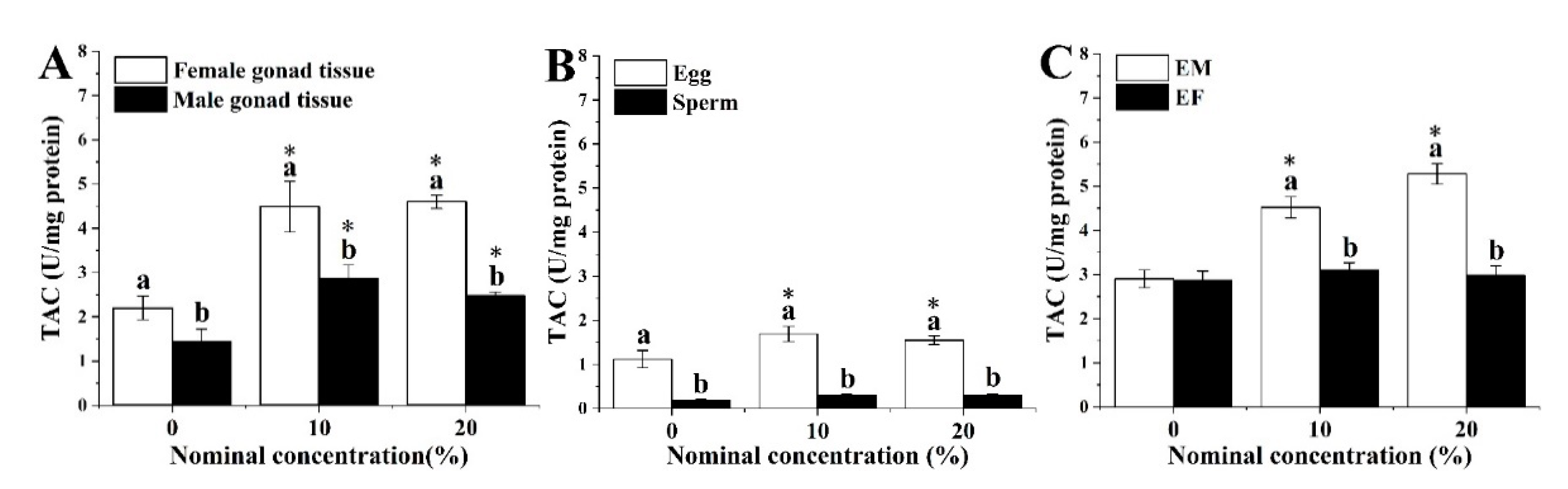
3.2. Total Antioxidant Capacity Level
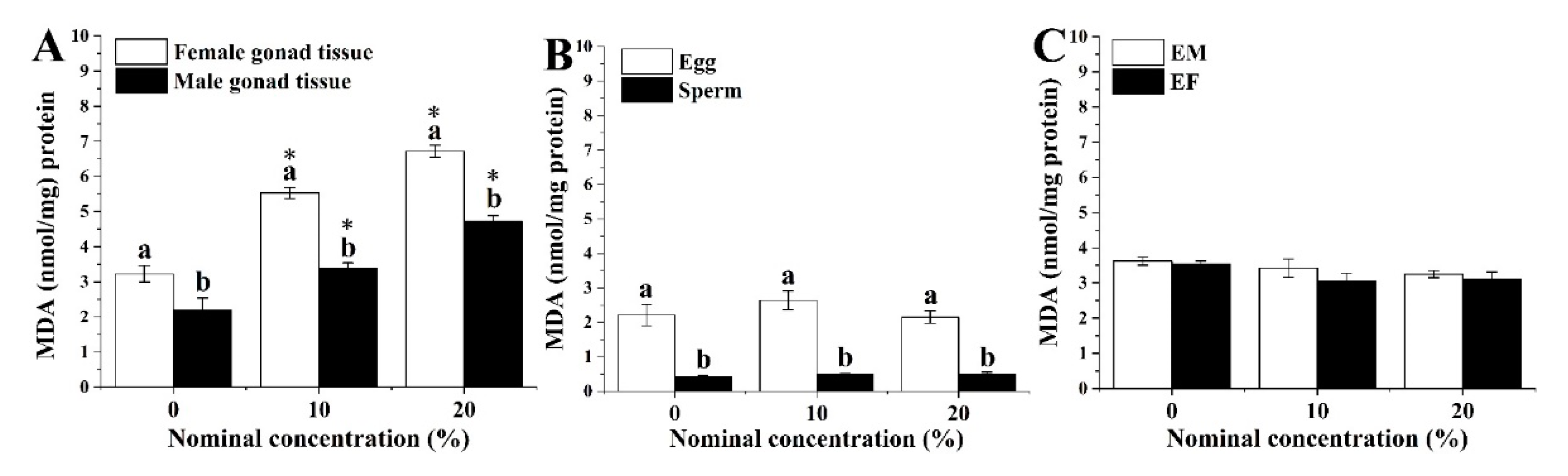
3.3. Lipid Peroxidation Level
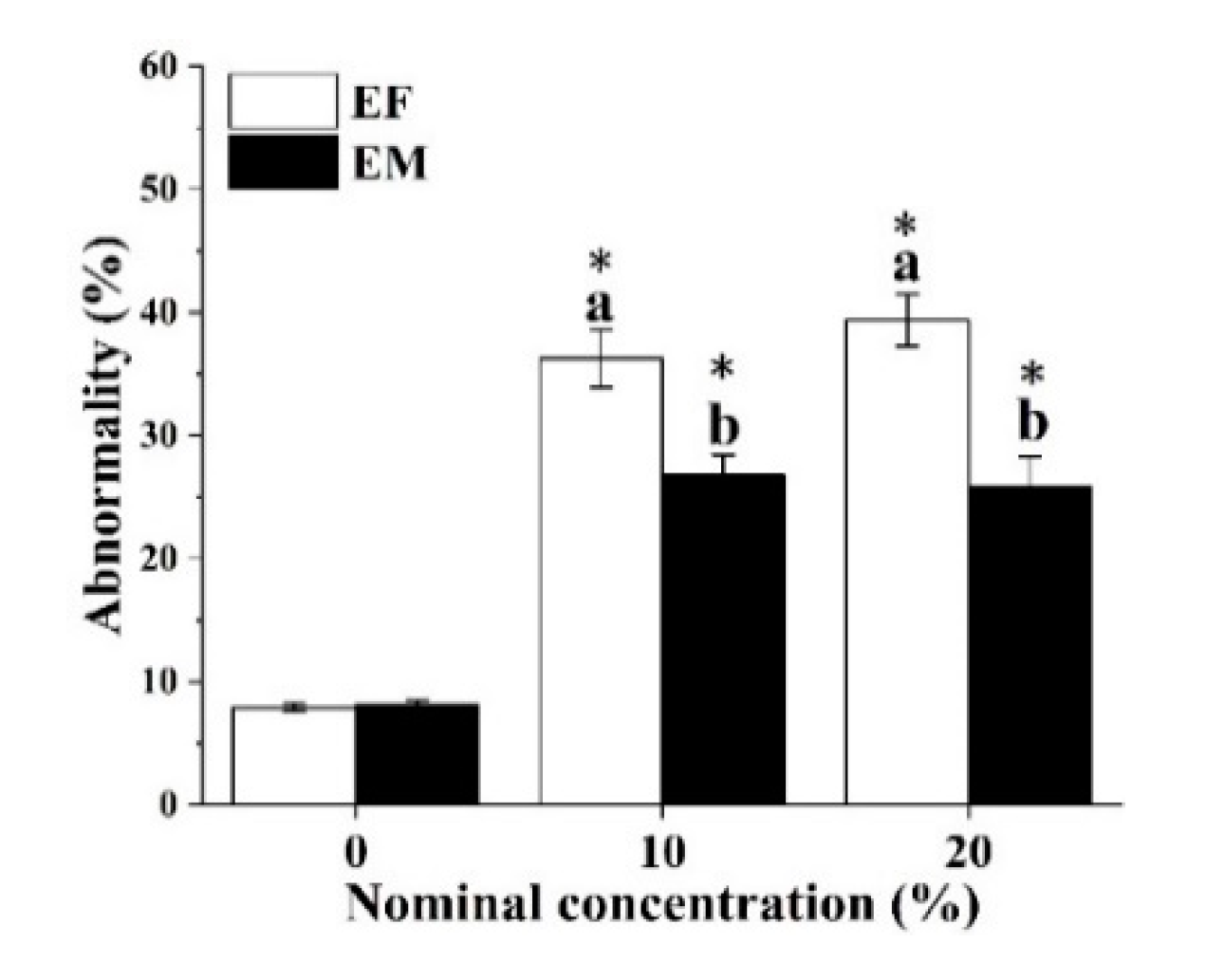
3.4. Malformed Offspring
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gutierrez, T.; Berry, D.; Teske, A.; Aitken, M.D. Enrichment of Fusobacteria in sea surface oil slicks from the deepwater horizon oil spill. Microorganisms 2016, 4, 24. [Google Scholar] [CrossRef] [PubMed]
- Lewis, J.P.; Tarnecki, J.H.; Garner, S.B.; Chagaris, D.D.; Patterson, W.F. Changes in reef fish community structure following the deepwater horizon oil spill. Sci. Rep. UK 2020, 10, 11459. [Google Scholar] [CrossRef] [PubMed]
- Sekiguchi, T.; Akitaya, H.; Nakayama, S.; Yazawa, T.; Ogasawara, M.; Suzuki, N.; Hayakawa, K.; Wada, S. Effect of polycyclic aromatic hydrocarbons on development of the Ascidian Ciona intestinalis type A. Int. J. Environ. Res. Public Health 2020, 17, 1340. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.L.; Lin, Q.; Gu, Y.G.; Du, F.Y.; Wang, X.H.; Shi, F.Q.; Ke, C.L.; Xiang, M.D.; Yu, Y.J. Bioaccumulation of polycyclic aromatic hydrocarbons (PAHs) in wild marine fish from the coastal waters of the northern South China Sea: Risk assessment for human health. Ecotoxicol. Environ. Saf. 2019, 180, 742–748. [Google Scholar] [CrossRef]
- Fernandez, C.; Pergent, G. Effect of different formulated diets and rearing conditions on growth parameters in the sea urchin Paracentrotus lividus. J. Shellfish. Res. 1998, 17, 1571–1581. [Google Scholar]
- Lukyanova, O.N.; Zhuravel, E.V.; Chulchekov, D.N.; Mazur, A.A. Sea urchin embryogenesis as bioindicators of marine pollution in impact areas of the sea of Japan/East Sea and the sea of okhotsk. Arch. Environ. Contam. Toxicol. 2017, 73, 322–333. [Google Scholar] [CrossRef]
- Stefansson, E.S.; Langdon, C.J.; Pargee, S.M.; Blunt, S.M.; Gage, S.J.; Stubblefield, W.A. Acute effects of non-weathered and weathered crude oil and dispersant associated with the deepwater horizon incident on the development of marine bivalve and echinoderm larvae. Environ. Toxicol. Chem. 2016, 35, 2016–2028. [Google Scholar] [CrossRef]
- Shimada Borges, J.C.; Branco, P.C.; Pressinotti, L.N.; Severino, D.; Machado Cunha da Silva, J.R. Intranuclear crystalloids of Antarctic sea urchins as a biomarker for oil contamination. Polar Biol. 2010, 33, 843–849. [Google Scholar] [CrossRef]
- Yang, B.L.; Xiong, D.Q. Bioaccumulation and subacute toxicity of mechanically and chemically dispersed heavy fuel oil in sea urchin (Glyptocidaris crenulari). Sci. Mar. 2015, 79, 497–504. [Google Scholar] [CrossRef]
- Dupont, S.; Dorey, N.; Stumpp, M.; Melzner, F.; Thorndyke, M. Long-term and trans-life-cycle effects of exposure to ocean acidification in the green sea urchin Strongylocentrotus droebachiensis. Mar. Biol. 2013, 160, 1835–1843. [Google Scholar] [CrossRef]
- Barbosa, D.B.; Mello, A.d.A.; Allodi, S.; de Barros, C.M. Acute exposure to water-soluble fractions of marine diesel oil: Evaluation of apoptosis and oxidative stress in an ascidian. Chemosphere 2018, 211, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Solé, M.; Lima, D.; Reis-Henriques, M.A.; Santos, M.M. Stress biomarkers in juvenile senegal sole, solea senegalensis, exposed to the water-accommodated fraction of the “Prestige” fuel oil. Bull. Environ. Contam. Toxicol. 2008, 80, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Hylland, K. Polycyclic aromatic hydrocarbon (PAH) ecotoxicology in marine ecosystems. J. Toxicol. Environ. Health A 2006, 69, 109–123. [Google Scholar] [CrossRef] [PubMed]
- Tanwir, K.; Javed, M.T.; Shahid, M.; Akram, M.S.; Ali, Q. Chapter 32—Antioxidant defense systems in bioremediation of organic pollutants. In Handbook of Bioremediation; Hasanuzzaman, M., Prasad, M.N.V., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 505–521. [Google Scholar]
- Wang, H.D.; Pan, L.Q.; Zhang, X.; Ji, R.W.; Si, L.; Cao, Y.H. The molecular mechanism of AhR-ARNT-XREs signaling pathway in the detoxification response induced by polycyclic aromatic hydrocarbons (PAHs) in clam Ruditapes philippinarum. Environ. Res. 2020, 183, 109165. [Google Scholar] [CrossRef] [PubMed]
- Cunha, I.; García, L.M.; Guilhermino, L. Sea urchin (Paracentrotus lividus) glutathione S-transferases and cholinesterase activities as biomarkers of environmental contamination. J. Environ. Monitor. 2005, 7, 288–294. [Google Scholar] [CrossRef]
- Milito, A.; Murano, C.; Castellano, I.; Romano, G.; Palumbo, A. Antioxidant and immune response of the sea urchin Paracentrotus lividus to different re-suspension patterns of highly polluted marine sediments. Mar. Environ. Res. 2020, 160, 104978. [Google Scholar] [CrossRef]
- Lister, K.N.; Lamare, M.D.; Burritt, D.J. Dietary pollutants induce oxidative stress, altering maternal antioxidant provisioning and reproductive output in the temperate sea urchin Evechinus chloroticus. Aquat. Toxicol. 2016, 177, 106–115. [Google Scholar] [CrossRef]
- Lister, K.N.; Lamare, M.D.; Burritt, D.J. Maternal antioxidant provisioning mitigates pollutant-induced oxidative damage in embryos of the temperate sea urchin Evechinus chloroticus. Sci. Rep. UK 2017, 7, 1954. [Google Scholar] [CrossRef]
- Singer, M.M.; Aurand, D.; Bragin, G.E.; Clark, J.R.; Coelho, G.M.; Sowby, M.L.; Tjeerdema, R.S. Standardization of the preparation and quantitation of water-accommodated fractions of petroleum for toxicity testing. Mar. Pollut. Bull. 2000, 40, 1007–1016. [Google Scholar] [CrossRef]
- Duan, M.N.; Xiong, D.Q.; Yang, M.Y.; Xiong, Y.J.; Ding, G.H. Parental exposure to heavy fuel oil induces developmental toxicity in offspring of the sea urchin Strongylocentrotus intermedius. Ecotoxicol. Environ. Saf. 2018, 159, 109–119. [Google Scholar] [CrossRef]
- Duan, M.N.; Xiong, D.Q.; Bai, X.; Gao, Y.L.; Xiong, Y.J.; Gao, X.; Ding, G.H. Transgenerational effects of heavy fuel oil on the sea urchin Strongylocentrotus intermedius considering oxidative stress biomarkers. Mar. Environ. Res. 2018, 141, 138–147. [Google Scholar] [CrossRef] [PubMed]
- US Environmental Protection Agency (EPA). Method 3510C: Separatory Funnel Liquid-Liquid Extraction; U.S. Environmental Protection Agency: Washington, DC, USA, 1996; p. 8.
- US Environmental Protection Agency (EPA). Method 3630C: Silica gel Cleanup, Part of Test Methods for Evaluating Solid Waste, Physical/Chemical Methods; U.S. Environmental Protection Agency: Washington, DC, USA, 1996; p. 15.
- Burritt, D.J. The polycyclic aromatic hydrocarbon phenanthrene causes oxidative stress and alters polyamine metabolism in the aquatic liverwort Riccia fluitans L. Plant Cell Environ. 2008, 31, 1416–1431. [Google Scholar] [CrossRef] [PubMed]
- Lovenberg, T.W.; Baron, B.M.; de Lecea, L.; Miller, J.D.; Prosser, R.A.; Rea, M.A.; Foye, P.E.; Racke, M.; Slone, A.L.; Siegel, B.W. A novel adenylyl cyclase-activating serotonin receptor (5-HT7) implicated in the regulation of mammalian circadian rhythms. Neuron 1993, 11, 449–458. [Google Scholar] [CrossRef]
- Esterbauer, H.; Cheeseman, K.H. Determination of aldehydic lipid peroxidation products: Malonaldehyde and 4-hydroxynonenal. Methods Enzymol. 1990, 186, 407–421. [Google Scholar]
- Rocha Monteiro, P.R.; Reis-Henriques, M.A.; Coimbra, J. Polycyclic aromatic hydrocarbons inhibit in vitro ovarian steroidogenesis in the flounder (Platichthys flesus L.). Aquat. Toxicol. 2000, 48, 549–559. [Google Scholar] [CrossRef]
- Kalachev, A.V. An ultrastructural study of testes permeability in sea urchins, Strongylocentrotus intermedius. Micron 2015, 68, 66–69. [Google Scholar] [CrossRef]
- Schaefer, S.; Koehler, A. Gonadal lesions of female sea urchin (Psammechinus miliaris) after exposure to the polycyclic aromatic hydrocarbon phenanthrene. Mar. Environ. Res. 2009, 68, 128–136. [Google Scholar] [CrossRef]
- Nam, S.-E.; Saravanan, M.; Rhee, J.-S. Benzo[a]pyrene constrains embryo development via oxidative stress induction and modulates the transcriptional responses of molecular biomarkers in the marine medaka Oryzias javanicus. J. Environ. Sci. Health A 2020, 55, 1–9. [Google Scholar] [CrossRef]
- Yazdani, M. Comparative toxicity of selected PAHs in rainbow trout hepatocytes: Genotoxicity, oxidative stress and cytotoxicity. Drug Chem. Toxicol. 2020, 43, 71–78. [Google Scholar] [CrossRef]
- Schaefer, S.; Abele, D.; Weihe, E.; Koehler, A. Sex-specific biochemical and histological differences in gonads of sea urchins (Psammechinus miliaris) and their response to phenanthrene exposure. Mar. Environ. Res. 2011, 71, 70–78. [Google Scholar] [CrossRef][Green Version]
- Aitken, R.J.; Koopman, P.; Lewis, S.E.M. Seeds of concern. Nature 2004, 432, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Xue, X.H.; Liu, Q.; Zhang, S.Z.; Peng, M.L.; Zhou, J.; Chen, L.J.; Fang, F.G. Effects of duration of thermal stress on growth performance, serum oxidative stress indices, the expression and localization of ABCG2 and mitochondria ROS production of skeletal muscle, small intestine and immune organs in broilers. J. Therm. Biol. 2019, 85, 102420. [Google Scholar] [CrossRef] [PubMed]
- Kozhina, V.P.; Terekhova, T.A.; Svetashev, V.I. Lipid composition of gametes and embryos of the sea urchin Strongylocentrotus intermedius at early stages of development. Dev. Biol. 1978, 62, 512–517. [Google Scholar] [CrossRef]
- Isely, N.; Lamare, M.; Marshall, C.; Barker, M. Expression of the DNA repair enzyme, photolyase, in developmental tissues and larvae, and in response to ambient UV-R in the Antarctic sea urchin Sterechinus neumayeri. Photochem. Photobiol. 2009, 85, 1168–1176. [Google Scholar] [CrossRef]
- Metcalfe, N.B.; Alonso-Alvarez, C. Oxidative stress as a life-history constraint: The role of reactive oxygen species in shaping phenotypes from conception to death. Funct. Ecol. 2010, 24, 984–996. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Ren, H.; Li, X.; Chen, H.; Ju, Z.; Xiong, D. Sex-Specific Differences in the Toxic Effects of Heavy Fuel Oil on Sea Urchin (Strongylocentrotus intermedius). Int. J. Environ. Res. Public Health 2021, 18, 499. https://doi.org/10.3390/ijerph18020499
Wang X, Ren H, Li X, Chen H, Ju Z, Xiong D. Sex-Specific Differences in the Toxic Effects of Heavy Fuel Oil on Sea Urchin (Strongylocentrotus intermedius). International Journal of Environmental Research and Public Health. 2021; 18(2):499. https://doi.org/10.3390/ijerph18020499
Chicago/Turabian StyleWang, Xuanbo, Hang Ren, Xishan Li, Huishu Chen, Zhonglei Ju, and Deqi Xiong. 2021. "Sex-Specific Differences in the Toxic Effects of Heavy Fuel Oil on Sea Urchin (Strongylocentrotus intermedius)" International Journal of Environmental Research and Public Health 18, no. 2: 499. https://doi.org/10.3390/ijerph18020499
APA StyleWang, X., Ren, H., Li, X., Chen, H., Ju, Z., & Xiong, D. (2021). Sex-Specific Differences in the Toxic Effects of Heavy Fuel Oil on Sea Urchin (Strongylocentrotus intermedius). International Journal of Environmental Research and Public Health, 18(2), 499. https://doi.org/10.3390/ijerph18020499




