Chronic Stress, Exercise and Cardiovascular Disease: Placing the Benefits and Risks of Physical Activity into Perspective
Abstract
1. Introduction
1.1. Major Life Stress
1.2. Socioeconomic Factors
1.3. Psychiatric Conditions
1.4. Pathophysiology
2. Impact of Physical Activity/Exercise on the Stress Response
2.1. Prevalence of Stress Today
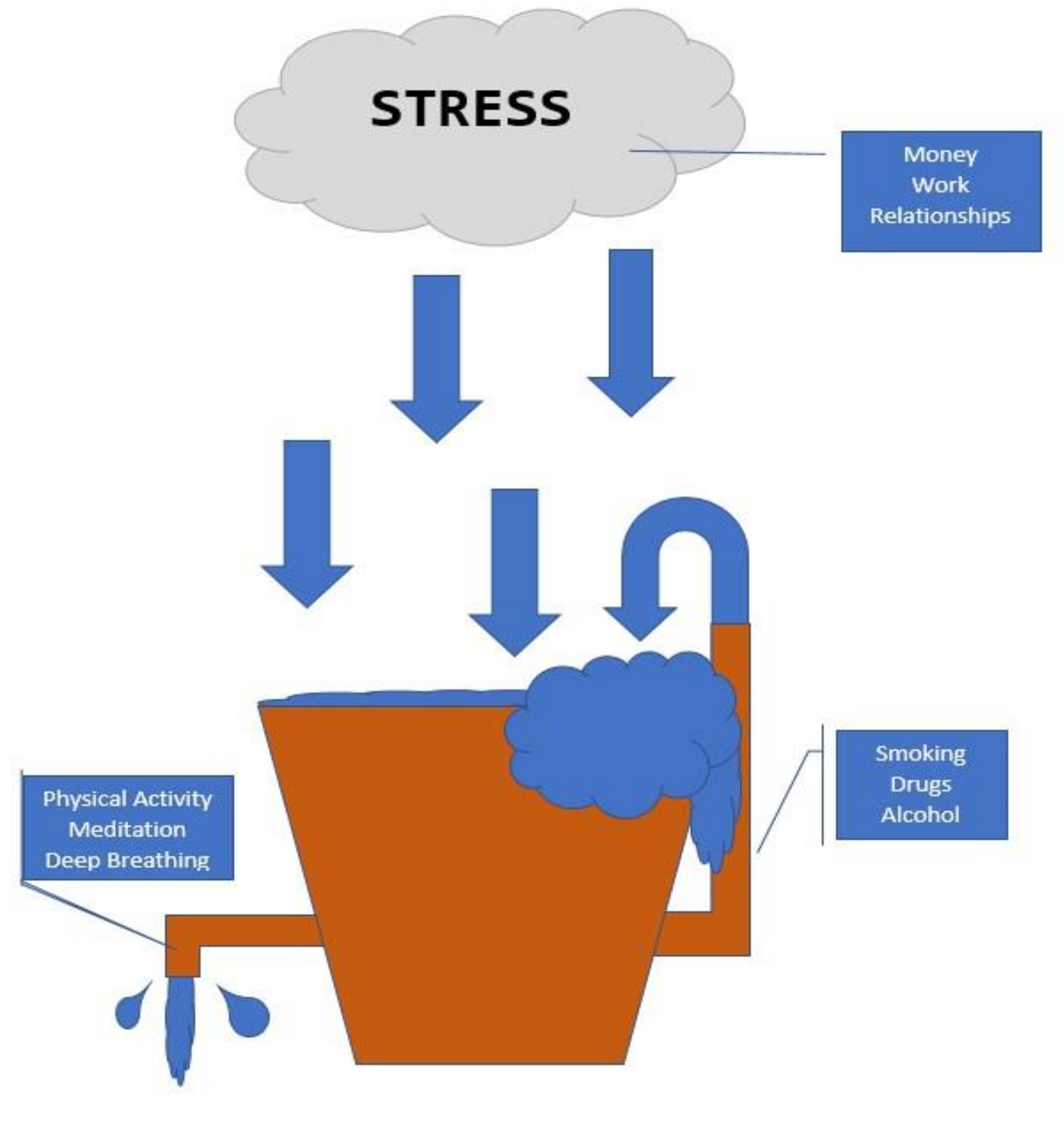
2.2. Stress Bucket Analogy
2.3. Coping Methods
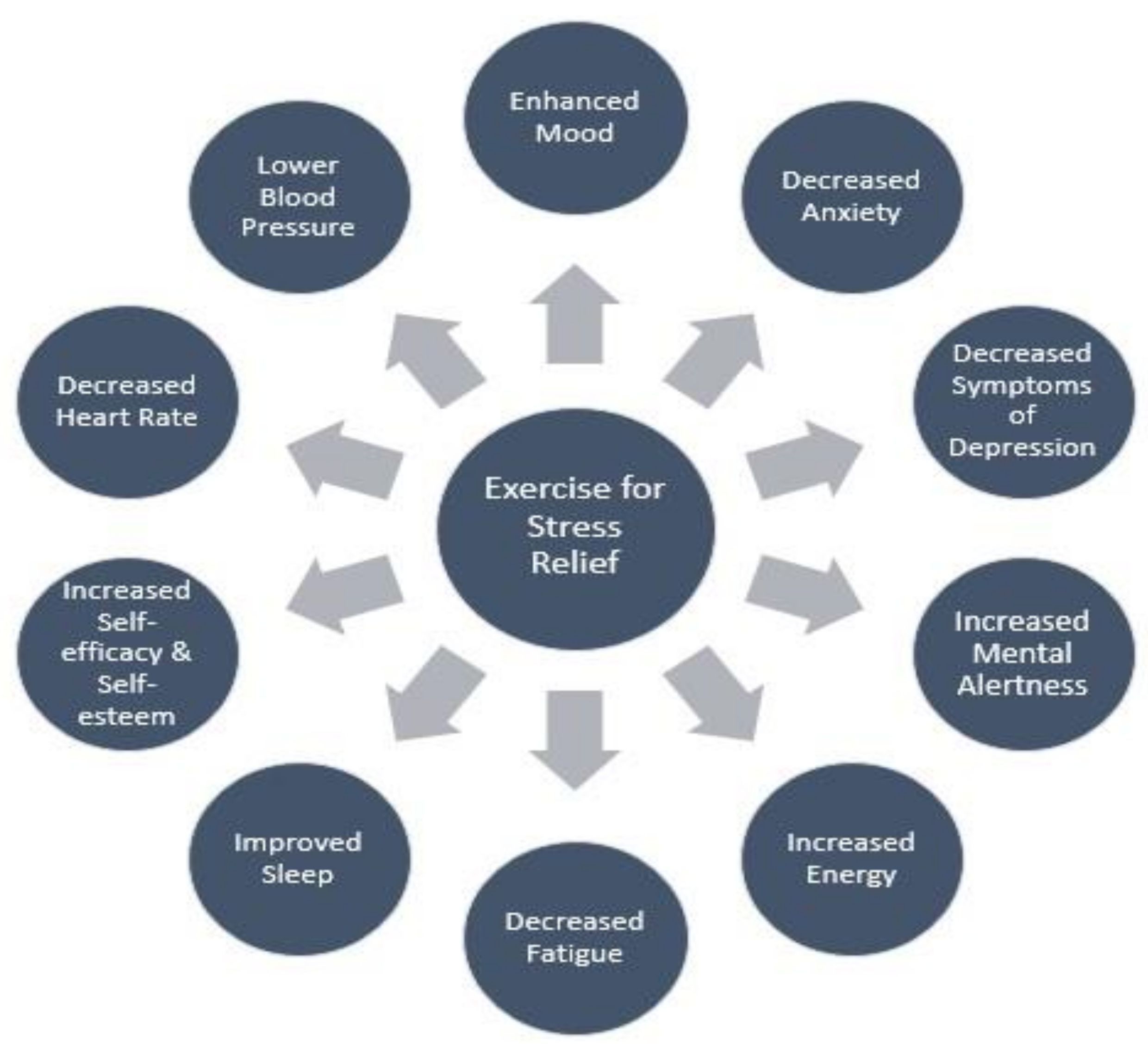
2.4. Physical Activity: Exercise Training and Stress Reduction
2.5. Physical Activity: Acute Exercise and Stress Reduction
2.6. Exercise Prescription to Reduce Stress
3. Exercise/Physical Activity: A Double-Edged Sword
4. Common Activities Associated with Acute Cardiac Events
5. High-Volume, High-Intensity Endurance Training: Too Much of a Good Thing?
5.1. Atrial Fibrillation (AF)
5.2. Accelerated Coronary Artery Calcification (CAC)
6. Prevention of Triggered Cardiac Events
Prophylactic Use of Cardioprotective Medications before Strenuous Exercise
7. Clinical Recommendations
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kemeny, M.E.; Schedlowski, M. Understanding the interaction between psychosocial stress and immune-related diseases: A stepwise progression. Brain Behav. Immun. 2007, 21, 1009–1018. [Google Scholar] [CrossRef]
- Hunter, H.J.A.; Momen, S.E.; Kleyn, C.E. The impact of psychosocial stress on healthy skin. Clin. Exp. Cermatol. 2015, 40, 540–546. [Google Scholar] [CrossRef]
- Yonas, M.A.; Lange, N.E.; Celedón, J.C. Psychosocial stress and asthma morbidity. Curr. Opin. Allergy Clin. Immunol. 2012, 12, 202–210. [Google Scholar] [CrossRef]
- Jansson, C.; Wallander, M.-A.; Johansson, S.; Johnsen, R.; Hveem, K. Stressful psychosocial factors and symptoms of gastroesophageal reflux disease: A population-based study in Norway. Scand. J. Gastroenterol. 2010, 45, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Block, J.P.; He, Y.; Zaslavsky, A.M.; Ding, L.; Ayanian, J.Z. Psychosocial stress and change in weight among US adults. Am. J. Epidemiol. 2009, 170, 181–192. [Google Scholar] [CrossRef] [PubMed]
- Yusuf, S.; Hawken, S.; Ounpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; McQueen, M.; Budaj, A.; Pais, P.; Varigos, J.; et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 937–952. [Google Scholar] [CrossRef]
- Kivimäki, M.; Leino-Arjas, P.; Luukkonen, R.; Riihimäki, H.; Kirjonen, J. Work stress and risk of cardiovascular mortality: Prospective cohort study of industrial employees. BMJ 2002, 325, 857. [Google Scholar] [CrossRef]
- Vrijkotte, T.G.M.; Van Doornen, L.J.; de Geus, E.J. Effects of work stress on ambulatory blood pressure, heart rate, and heart rate variability. Hypertension 2000, 35, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Schnall, P.L.; Pieper, C.; Schwartz, J.E.; Karasek, R.A.; Schlussel, Y.; Devereux, R.B.; Ganau, A.; Alderman, M.; Warren, K.; Pickering, T.G. The relationship between ‘job strain,’ workplace diastolic blood pressure, and left ventricular mass index: Results of a case-control study. JAMA 1990, 263, 1929–1935. [Google Scholar] [CrossRef]
- Devereux, R.B.; Pickering, T.G.; Harshfield, G.A.; Kleinert, H.D.; Denby, L.; Clark, L.; Pregibon, D.; Jason, M.; Kleiner, B.; Borer, J.S.; et al. Left ventricular hypertrophy in patients with hypertension: Importance of blood pressure response to regularly recurring stress. Circulation 1983, 68, 470–476. [Google Scholar] [CrossRef]
- Gallo, L.C.; Troxel, W.M.; Kuller, L.H.; Sutton-Tyrrell, K.; Emundowica, D. Marital status, marital quality, and atherosclerotic burden in postmenopausal women. Psychosom. Med. 2003, 65, 952–962. [Google Scholar] [CrossRef] [PubMed]
- Rohrbaugh, M.J.; Shoham, V.; Coyne, J.C. Effect of marital quality on eight-year survival of patients with heart failure. Am. J. Cardiol. 2006, 98, 1069–1072. [Google Scholar] [CrossRef] [PubMed]
- Lev-Ari, S.; Yftach, G.; Goldbourt, U. Dissatisfaction with married life in men is related to increased stroke and all-cause mortality. J. Clin. Med. 2021, 10, 1729. [Google Scholar] [CrossRef]
- Orth-Gomér, K.; Wamala, S.P.; Horsten, M.; Schenck-Gustafsson, K.; Schneiderman, N.; Mittleman, M.A. Marital stress worsens prognosis in women with coronary heart disease: The Stockholm Female Coronary Risk Study. JAMA 2000, 284, 3008–3014. [Google Scholar] [CrossRef]
- Sunmin, L.; Colditz, G.A.; Berkman, L.F.; Kawachi, I. Caregiving and risk of coronary heart disease in US women: A prospective study. Am. J. Prev. Med. 2003, 24, 113–119. [Google Scholar] [CrossRef]
- Marmot, M.G.; Smith, G.D.; Stansfeld, S.; Patel, C.; North, F.; Head, J.; White, I.; Brunner, E.; Feeney, A. Health inequalities among British civil servants: The Whitehall II study. Lancet 1991, 337, 1387–1393. [Google Scholar] [CrossRef]
- Kumari, M.; Head, J.; Marmot, M. Prospective study of social and other risk factors for incidence of type 2 diabetes in the Whitehall II study. Arch. Intern. Med. 2004, 164, 1873–1880. [Google Scholar] [CrossRef]
- Darmon, N.; Drewnowski, A. Does social class predict diet quality? Am. J. Clin. Nutr. 2008, 87, 1107–1117. [Google Scholar] [CrossRef]
- Clark, A.M.; DesMeules, M.; Luo, D.A.S.; Wielgosz, A. Socioeconomic status and cardiovascular disease: Risks and implications for care. Nat. Rev. Cardiol. 2009, 6, 712–722. [Google Scholar] [CrossRef] [PubMed]
- Kunz-Ebrecht, S.R.; Kirschbaum, C.; Steptoe, A. Work stress, socioeconomic status and neuroendocrine activation over the working day. Soc. Sci. Med. 2004, 58, 1523–1530. [Google Scholar] [CrossRef]
- Murphy, J.M.; Olivier, D.C.; Monson, R.R.; Sobol, A.M.; Federman, E.B.; Leighton, A.H. Depression and anxiety in relation to social status: A prospective epidemiologic study. Arch. Gen. Psychiatry 1991, 48, 223–229. [Google Scholar] [CrossRef]
- Winkleby, M.A.; Jatulis, D.E.; Frank, E.; Fortmann, S.P. Socioeconomic status and health: How education, income, and occupation contribute to risk factors for cardiovascular disease. Am. J. Public Health 1992, 82, 816–820. [Google Scholar] [CrossRef]
- Rodriguez, C.J.; Sciacca, R.R.; Diez-Roux, A.V.; Boden-Albala, B.; Sacco, R.L.; Homma, S.; DiTullio, M.R. Relation between socioeconomic status, race–ethnicity, and left ventricular mass: The Northern Manhattan Study. Hypertension 2004, 43, 775–779. [Google Scholar] [CrossRef]
- Cozier, Y.; Palmer, J.R.; Horton, N.J.; Fredman, L.; Wise, L.A.; Rosenberg, L. Racial discrimination and the incidence of hypertension in US black women. Ann. Epidemiol. 2006, 16, 681–687. [Google Scholar] [CrossRef]
- Sims, M.; Diez-Roux, A.V.; Dudley, A.; Gebreab, S.; Wyatt, S.B.; Bruce, M.A.; James, S.A.; Robinson, J.C.; Williams, D.R.; Taylor, H.A. Perceived discrimination and hypertension among African Americans in the Jackson Heart Study. Am. J. Public Health 2012, 102, S258–S265. [Google Scholar] [CrossRef]
- Davis, S.K.; Liu, Y.; Quarells, R.C.; Din-Dzietham, R.; Metro Atlanta Heart Disease Study Group. Stress-related racial discrimination and hypertension likelihood in a population-based sample of African Americans: The Metro Atlanta Heart Disease Study. Ethn. Dis. 2005, 15, 585–593. [Google Scholar] [PubMed]
- Dolezsar, C.M.; McGrath, J.J.; Herzig, A.J.M.; Miller, S.B. Perceived racial discrimination and hypertension: A comprehensive systematic review. Health Psychol. 2014, 33, 20–34. [Google Scholar] [CrossRef] [PubMed]
- Steptoe, A.; Brydon, L.; Kunz-Ebrecht, S. Changes in financial strain over three years, ambulatory blood pressure, and cortisol responses to awakening. Psychosom. Med. 2005, 67, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Rozanski, A.; Blumenthal, J.A.; Kaplan, J. Impact of psychological factors on the pathogenesis of cardiovascular disease and implications for therapy. Circulation 1999, 99, 2192–2217. [Google Scholar] [CrossRef] [PubMed]
- Lespérance, F.; Frasure-Smith, N.; Talajic, M.; Bourassa, M.G. Five-year risk of cardiac mortality in relation to initial severity and one-year changes in depression symptoms after myocardial infarction. Circulation 2002, 105, 1049–1053. [Google Scholar] [CrossRef] [PubMed]
- Rugulies, R. Depression as a predictor for coronary heart disease: A review and meta-analysis. Am. J. Prev. Med. 2002, 23, 51–61. [Google Scholar] [CrossRef]
- Kawachi, I.; Colditz, G.A.; Ascherio, A.; Rimm, E.B.; Giovannucci, E.; Stampfer, M.J.; Willett, W.C. Prospective study of phobic anxiety and risk of coronary heart disease in men. Circulation 1994, 89, 1992–1997. [Google Scholar] [CrossRef]
- Kubzansky, L.D.; Koenen, K.C.; Spiro, A., 3rd; Vokonas, P.S.; Sparrow, D. Prospective study of posttraumatic stress disorder symptoms and coronary heart disease in the Normative Aging Study. Arch. Gen. Psychiatry 2007, 64, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Fang, F.; Amberg, F.K.; Mataix-Cois, D.; de la Cruz, L.F.; Almqvist, C.; Fall, K.; Lichtenstein, P.; Thorgeirsson, G.; Valdimarsdóttir, U.A. Stress related disorders and risk of cardiovascular disease: Population based, sibling controlled cohort study. BMJ 2019, 365, I1255. [Google Scholar] [CrossRef] [PubMed]
- Crum-Cianflone, N.F.; Bagnell, M.E.; Schaller, E.; Boyko, E.J.; Smith, B.; Maynard, C.; Ulmer, C.S.; Vernalis, M.; Smith, T.C. Impact of combat deployment and posttraumatic stress disorder on newly reported coronary heart disease among US active duty and reserve forces. Circulation 2014, 129, 1813–1820. [Google Scholar] [CrossRef] [PubMed]
- Jordan, H.T.; Miller-Archie, S.A.; Cone, J.E.; Morabia, A.; Stellman, S.D. Heart disease among adults exposed to the September 11, 2001 World Trade Center disaster: Results from the World Trade Center Health Registry. Prev. Med. 2011, 53, 370–376. [Google Scholar] [CrossRef]
- Brown, S.; Inskip, H.; Barraclough, B. Causes of the excess mortality of schizophrenia. Br. J. Psychiatry 2000, 177, 212–217. [Google Scholar] [CrossRef]
- Goff, D.C.; Sullivan, L.M.; McEvoy, J.P.; Meyer, J.M.; Nasrallah, H.A.; Daumitt, G.L.; Lamberti, S.; D’Agostino, R.B.; Stroup, T.S.; Davis, S.; et al. A comparison of ten-year cardiac risk estimates in schizophrenia patients from the CATIE study and matched controls. Schizophr. Res. 2005, 80, 45–53. [Google Scholar] [CrossRef]
- Saha, S.; Chant, D.; McGrath, J. A systematic review of mortality in schizophrenia: Is the differential mortality gap worsening over time? Arch. Gen. Psychiatry 2007, 64, 1123–1131. [Google Scholar] [CrossRef]
- Hennekens, C.H.; Hennekens, A.R.; Hollar, D.; Casey, D.E. Schizophrenia and increased risks of cardiovascular disease. Am. Heart J. 2005, 150, 1115–1121. [Google Scholar] [CrossRef]
- De Hert, M.; Detraux, J.; van Winkel, R.; Yu, W.; Correll, C.U. Metabolic and cardiovascular adverse effects associated with antipsychotic drugs. Nat. Rev. Endocrinol. 2012, 8, 114–126. [Google Scholar] [CrossRef]
- Leucht, S.; Cipriani, A.; Spineli, L.; Mavridis, D.; Orey, D.; Richter, F.; Samara, M.; Barbui, C.; Engel, R.R.; Geddes, J.R.; et al. Comparative efficacy and tolerability of 15 antipsychotic drugs in schizophrenia: A multiple-treatments meta-analysis. Lancet 2013, 382, 951–962. [Google Scholar] [CrossRef]
- Tsigos, C.; Kyrou, I.; Kassi, E.; Chrousos, G.P. Stress: Endocrine Physiology and Pathophysiology. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dhatariya, K., Dungan, K., Grossman, A., Hershman, J.M., Hofland, J., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Fioranelli, M.; Bottaccioli, A.G.; Bottaccioli, F.; Bianchi, M.; Rovesti, M.; Roccia, M.G. Stress and inflammation in coronary artery disease: A review psychoneuroendocrineimmunology-based. Front. Immunol. 2018, 9, 2031. [Google Scholar] [CrossRef]
- Herman, J.P.; McKlveen, J.M.; Ghosal, S.; Kopp, B.; Wulsin, A.; Makinson, R.; Scheimann, J.; Myers, B. Regulation of the hypothalamic-pituitary-adrenocortical stress response. Compr. Physiol. 2016, 6, 603–621. [Google Scholar] [CrossRef]
- Le-Ha, C.; Herbison, C.E.; Beilin, L.J.; Burrows, S.; Henley, D.E.; Lye, S.J.; Matthews, S.G.; Pennell, C.E.; Mori, T.A. Hypothalamic-pituitary-adrenal axis activity under resting conditions and cardiovascular risk factors in adolescents. Psychonneurendocrinology 2016, 66, 118–124. [Google Scholar] [CrossRef]
- Gordan, R.; Gwathmey, J.K.; Xie, L.H. Autonomic and endocrine control of cardiovascular function. World J. Cardiol. 2015, 7, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Won, E.; Kim, Y.K. Stress, the autonomic nervous system, and the immune-kynurenine pathway in the etiology of depression. Curr. Neuropharmacol. 2016, 14, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Florea, V.G.; Cohn, J.N. The autonomic nervous system and heart failure. Circ. Res. 2014, 114, 1815–1826. [Google Scholar] [CrossRef] [PubMed]
- Schneiderman, N.; Ironson, G.; Siegel, S.D. Stress and health: Psychological, behavioral, and biological determinants. Annu. Rev. Clin. Psychol. 2005, 1, 607–628. [Google Scholar] [CrossRef] [PubMed]
- Woolf, S.H.; Schoomaker, H. Life expectancy and mortality rates in the United States, 1959–2017. JAMA 2019, 322, 1996–2016. [Google Scholar] [CrossRef] [PubMed]
- Cooper, S.L. Promoting physical activity for mental well-being. ACSM’s Health Fit. J. 2020, 24, 12–16. [Google Scholar] [CrossRef]
- Nichols, S. New Global Index Ranks Perceptions of Stress. 7 December 2020. Available online: https://news.gallup.com/opinion/gallup/327104/new-global-index-ranks-perceptions-stress.aspx (accessed on 8 July 2021).
- Gallup Global Emotions Report. Available online: https://www.gallup.com/analytics/349280/gallup-global-emotions-report.aspx (accessed on 8 July 2021).
- American Psychological Association. Stress in the Time of COVID-19. Stress in AmericaTM 2020. Available online: https://www.apa.org/news/press/releases/stress/2020/stress-in-america-covid.pdf (accessed on 8 July 2021).
- American Psychological Association. Stress in AmericaTM 2019. Available online: https://www.apa.org/news/press/releases/stress/2019/stress-america-2019.pdf (accessed on 8 July 2021).
- America’s State of Mind Report. Available online: https://www.express-scripts.com/corporate/americas-state-of-mind-report (accessed on 8 July 2021).
- Mental Health, UK. The Stress Bucket. Available online: https://mentalhealth-uk.org/blog/the-stress-bucket/ (accessed on 8 July 2021).
- Klaperski, S.; von Dawans, B.; Heinrichs, M.; Fuchs, R. Effects of a 12-week endurance training program on the physiological response to psychosocial stress in men: A randomized controlled trial. J. Behav. Med. 2014, 37, 1118–1133. [Google Scholar] [CrossRef]
- Rimmele, U.; Zellweger, B.C.; Marti, B.; Seiler, R.; Mohiyeddini, C.; Ehlert, U.; Heinrichs, M. Trained men show lower cortisol, heart rate and psychological responses to psychosocial stress compared with untrained men. Psychoneuroendocrinology 2007, 32, 627–635. [Google Scholar] [CrossRef]
- Norris, R.; Carroll, D.; Cochrane, R. The effects of physical activity and exercise training on psychological stress and well-being in an adolescent population. J. Psychosom. Res. 1992, 36, 55–65. [Google Scholar] [CrossRef]
- Rogers, M.W.; Probst, M.M.; Gruber, J.J.; Berger, R.; Boone, J.B., Jr. Differential effects of exercise training intensity on blood pressure and cardiovascular responses to stress in borderline hypertensive humans. J. Hypertens. 1996, 14, 1369–1375. [Google Scholar] [CrossRef]
- Wunsch, K.; Wurst, R.; von Dawans, B.; Strahler, J.; Kasten, N.; Fuchs, R. Habitual and acute exercise effects on salivary biomarkers in response to psychosocial stress. Psychoneuroendocrinology 2019, 106, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Gibbons, L.W.; Cooper, K.H.; Meyer, B.M.; Ellison, R.C. The acute cardiac risk of strenuous exercise. JAMA 1980, 244, 1799–1801. [Google Scholar] [CrossRef] [PubMed]
- Star, M. Brave heart, broken heart. Newsweek 1995, 126, 70–71. [Google Scholar]
- Thompson, P.D.; Franklin, B.A.; Balady, G.J.; Blair, S.N.; Corrado, D.; Estes, N.A.M., 3rd; Fulton, J.E.; Gordon, N.F.; Haskell, W.L.; Link, M.S.; et al. Exercise and acute cardiovascular events placing the risks into perspective: A scientific statement from the American Heart Association Council on Nutrition, Physical Activity, and Metabolism and the Council on Clinical Cardiology. Circulation 2007, 115, 2358–2368. [Google Scholar] [CrossRef]
- Thompson, P.D. The cardiovascular complications of vigorous physical activity. Arch. Intern. Med. 1996, 156, 2297–2302. [Google Scholar] [CrossRef] [PubMed]
- Franklin, B.A. Cardiovascular events associated with exercise: The risk-protection paradox. J. Cardiopulm. Rehabil. 2005, 25, 189–195. [Google Scholar] [CrossRef]
- Maron, B.J.; Gohman, T.E.; Aeppli, D. Prevalence of sudden cardiac death during competitive sports activities in Minnesota high school athletes. J. Am. Coll. Cardiol. 1998, 32, 1881–1884. [Google Scholar] [CrossRef]
- Giri, S.; Thompson, P.D.; Kiernan, F.J.; Clive, J.; Fram, D.B.; Mitchel, J.F.; Hirst, J.A.; McKay, R.G.; Waters, D.D. Clinical and angiographic characteristics of exertion-related acute myocardial infarction. JAMA 1999, 282, 1731–1736. [Google Scholar] [CrossRef]
- Tuzcu, E.M.; Kapadia, S.R.; Tutar, E.; Ziada, K.M.; Hobbs, R.E.; McCarthy, P.M.; Young, J.B.; Nissen, S.E. High prevalence of coronary atherosclerosis in asymptomatic teenagers and young adults: Evidence from intravascular ultrasound. Circulation 2001, 103, 2705–2710. [Google Scholar] [CrossRef]
- Fletcher, G.F.; Balady, G.J.; Amsterdam, E.A.; Chaitman, B.; Eckel, R.; Fleg, J.; Froelicher, V.F.; Leon, A.S.; Piña, I.L.; Rodney, R.; et al. Exercise standards for testing and training: A statement for healthcare professionals from the American Heart Association. Circulation 2001, 104, 1694–1740. [Google Scholar] [CrossRef]
- Goodman, J.M.; Burr, J.F.; Banks, L.; Thomas, S.G. The acute risks of exercise in apparently healthy adults and relevance for prevention of cardiovascular events. Can. J. Cardiol. 2016, 32, 523–532. [Google Scholar] [CrossRef]
- Mittleman, M.A.; Maclure, M.; Tofler, G.H.; Sherwood, J.B.; Goldberg, R.J.; Muller, J.E. Triggering of acute myocardial infarction by heavy physical exertion: Protection against triggering by regular exertion. N. Engl. J. Med. 1993, 329, 1677–1683. [Google Scholar] [CrossRef] [PubMed]
- Franklin, B.A. Preventing exercise-related cardiovascular events: Is a medical examination more urgent for physical activity or inactivity? Circulation 2014, 129, 1081–1084. [Google Scholar] [CrossRef]
- Powell, K.E.; Thompson, P.D.; Caspersen, C.J.; Kendrick, J.S. Physical activity and the incidence of coronary heart disease. Annu. Rev. Public Health 1987, 8, 253–287. [Google Scholar] [CrossRef] [PubMed]
- Berlin, J.A.; Colditz, G.A. A meta-analysis of physical activity in the prevention of coronary heart disease. Am. J. Epidemiol. 1990, 132, 612–628. [Google Scholar] [CrossRef] [PubMed]
- Franklin, B.A.; Lavie, C.J. Triggers of acute cardiovascular events and potential preventive strategies: Prophylactic role of regular exercise. Phys. Sportsmed. 2011, 39, 11–21. [Google Scholar] [CrossRef]
- Burtscher, M.; Pachinger, O.; Mittleman, M.A.; Ulmer, H. Prior myocardial infarction is the major risk factor associated with sudden cardiac death during downhill skiing. Int. J. Sports Med. 2000, 21, 613–615. [Google Scholar] [CrossRef]
- Northcote, R.J.; Flannigan, C.; Ballantyne, D. Sudden death and vigorous exercise—A study of 60 deaths associated with squash. Br. Heart J. 1986, 55, 198–203. [Google Scholar] [CrossRef]
- Rognmo, Ø.; Moholdt, T.; Bakken, H.; Hole, T.; Mølstad, P.; Myhr, N.E.; Grimsmo, J.; Wisløff, U. Cardiovascular risk of high-versus moderate-intensity aerobic exercise in coronary heart disease patients. Circulation 2012, 126, 1436–1440. [Google Scholar] [CrossRef]
- Link, M.S.; Estes, N.A., 3rd. Sudden cardiac death in the athlete: Bridging the gaps between evidence, policy, and practice. Circulation 2012, 125, 2511–2516. [Google Scholar] [CrossRef]
- Lown, B.; Verrier, R.L.; Rabinowitz, S.H. Neural and psychologic mechanisms and the problem of sudden cardiac death. Am. J. Cardiol. 1977, 39, 890–902. [Google Scholar] [CrossRef]
- Haapaniemi, S.; Franklin, B.A.; Wegner, J.H.; Hamar, S.; Gordon, S.; Timmis, G.C.; O’Neill, W.W. Electrocardiographic responses to deer hunting activities in men with and without coronary artery disease. Am. J. Cardiol. 2007, 100, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Franklin, B.A.; George, P.; Henry, R.; Gordon, S.; Timmis, G.C.; O’Neill, W.W. Acute myocardial infarction after manual or automated snow removal. Am. J. Cardiol. 2001, 87, 1282–1283. [Google Scholar] [CrossRef]
- Chowdhury, P.S.; Franklin, B.A.; Boura, J.A.; Dragovic, L.J.; Kanluen, S.; Spitz, W.; Hodak, J.; O’Neill, W.W. Sudden cardiac death after manual or automated snow removal. Am. J. Cardiol. 2003, 92, 833–835. [Google Scholar] [CrossRef]
- Kim, J.H.; Malhotra, R.; Chiampas, G.; d’Hemecourt, P.; Troyanos, C.; Cianca, J.; Smith, R.N.; Wang, T.J.; Roberts, W.O.; Thompson, P.D.; et al. Race Associated Cardiac Arrest Event Registry (RACER) Study Group. Cardiac arrest during long-distance running races. N. Engl. J. Med. 2012, 366, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Harris, K.M.; Creswell, L.L.; Haas, T.S.; Thomas, T.; Tung, M.; Isaacson, E.; Garberich, R.F.; Maron, B.J. Death and cardiac arrest in U.S. triathlon participants, 1985 to 2016: A case series. Ann. Intern. Med. 2017, 167, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Franklin, B.A.; Thompson, P.D.; Al-Zaiti, S.S.; Albert, C.M.; Hivert, M.-F.; Levine, B.D.; Lobelo, F.; Madan, K.; Sharrief, A.Z.; Eijsvogels, T.M.H.; et al. Exercise-related acute cardiovascular events and potential deleterious adaptations following long-term exercise training: Placing the risks into perspective—an update: A Scientific Statement from the American Heart Association. Circulation 2020, 141, e705–e736. [Google Scholar] [CrossRef] [PubMed]
- Redelmeier, D.A.; Greenwald, J.A. Competing risks of mortality with marathons: Retrospective analysis. BMJ 2007, 335, 1275–1277. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.T.; Franklin, B. Vigorous exercise and diabetic, hypertensive, and hypercholesterolemia medication use. Med. Sci. Sports Exerc. 2007, 39, 1933–1941. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Werner, C.; Fürster, T.; Widmann, T.; Pöss, J.; Roggia, C.; Hanhoun, M.; Scharhad, J.; Büchner, N.; Meyer, T.; Kindermann, W.; et al. Physical exercise prevents cellular senescence in circulating leukocytes and in the vessel wall. Circulation 2009, 120, 2438–2447. [Google Scholar] [CrossRef]
- Franklin, B.A.; Billecke, S. Putting the benefits and risks of aerobic exercise in perspective. Curr. Sports Med. Rep. 2012, 11, 201–208. [Google Scholar] [CrossRef]
- Mont, L.; Elosua, R.; Brugada, J. Endurance sport practice as a risk factor for atrial fibrillation and atrial flutter. Europace 2009, 11, 11–17. [Google Scholar] [CrossRef]
- Calvo, N.; Ramos, P.; Montserrat, S.; Guasch, E.; Coll-Vincent, B.; Domenech, M.; Bisbal, F.; Hevia, S.; Vidorreta, S.; Borras, R.; et al. Emerging risk factors and the dose-response relationship between physical activity and lone atrial fibrillation: A prospective case-control study. Europace 2016, 18, 57–63. [Google Scholar] [CrossRef]
- Ricci, C.; Gervasi, F.; Gaeta, M.; Smuts, C.M.; Schutte, A.E.; Leitzmann, M.F. Physical activity volume in relation to risk of atrial fibrillation: A non-linear meta-regression analysis. Eur. J. Prev Cardiol 2018, 25, 857–866. [Google Scholar] [CrossRef]
- Wilheim, M.; Roten, L.; Tanner, H.; Wilhelm, I.; Schmid, J.-P.; Saner, H. Atrial remodeling, autonomic tone, and lifetime training hours in nonelite athletes. Am. J. Cardiol. 2011, 108, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Möhlenkamp, S.; Lehmann, N.; Breuckmann, F.; Bröcker-Preuss, M.; Nassenstein, K.; Halle, M.; Budde, T.; Mann, K.; Barkhausen, J.; Heusch, G.; et al. Running: The risk of coronary events: Prevalence and prognostic relevance of coronary atherosclerosis in marathon runners. Eur. Heart J. 2008, 29, 1903–1910. [Google Scholar] [CrossRef] [PubMed]
- Aengevaeren, V.L.; Mosterd, A.; Braber, T.L.; Prakken, N.H.J.; Doevendans, P.A.; Grobbee, D.E.; Thompson, P.D.; Eijsvogels, T.M.H.; Velthuis, B.K. Relationship between lifelong exercise volume and coronary atherosclerosis in athletes. Circulation 2017, 136, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Merghani, A.; Maestrini, V.; Rosmini, S.; Cox, A.T.; Dhutia, H.; Bastiaenan, R.; David, S.; Yeo, T.J.; Narain, R.; Malhotra, A.; et al. Prevalence of subclinical coronary artery disease in masters endurance athletes with a low atherosclerotic risk profile. Circulation 2017, 136, 126–137. [Google Scholar] [CrossRef]
- Radford, N.B.; DeFina, L.F.; Leonard, D.; Barlow, C.E.; Willis, B.L.; Gibbons, L.W.; Gilchrist, S.C.; Khera, A.; Levine, B.D. Cardiorespiratory fitness, coronary artery calcium, and cardiovascular disease events in a cohort of generally healthy middle-age men: Results from the Cooper Center Longitudinal Study. Circulation 2018, 137, 1888–1895. [Google Scholar] [CrossRef]
- Hammoudeh, A.J.; Alhaddad, I.A. Triggers and the onset of acute myocardial infarction. Cardiol. Rev. 2009, 17, 270–274. [Google Scholar] [CrossRef]
- Iellamo, F.; Legramante, J.M.; Massaro, M.; Raimondi, G.; Galante, A. Effects of a residential exercise training on baroreflex sensitivity and heart rate variability in patients with coronary artery disease: A randomized, controlled study. Circulation 2000, 102, 2588–2592. [Google Scholar] [CrossRef]
- Thijssen, D.H.J.; Redington, A.; George, K.P.; Hopman, M.T.E.; Jones, H. Association of exercise preconditioning with immediate cardioprotection: A review. JAMA Cardiol. 2018, 3, 169–176. [Google Scholar] [CrossRef]
- Quindry, J.C.; Franklin, B.A. Exercise preconditioning as a cardioprotective phenotype. Am. J. Cardiol. 2021, 148, 8–15. [Google Scholar] [CrossRef]
- Mittleman, M.A.; Mostofsky, E. Physical, psychological and chemical triggers of acute cardiovascular events: Preventive strategies. Circulation 2011, 124, 346–354. [Google Scholar] [CrossRef]
- Smyth, A.; O’Donnell, M.; Lamelas, P.; Teo, K.; Rangarajan, S.; Yusuf, S.; INTERHEART Investigators. Physical activity and anger or emotional upset as triggers of acute myocardial infarction: The INTERHEART study. Circulation 2016, 134, 1059–1067. [Google Scholar] [CrossRef] [PubMed]
- Held, C.; Iqbal, R.; Lear, S.A.; Rosengren, A.; Islam, S.; Mathew, J.; Yusuf, S. Physical activity levels, ownership of goods promoting sedentary behaviour and risk of myocardial infarction: Results of the INTERHEART study. Eur. Heart J. 2012, 33, 452–466. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Pan, A.; Wang, D.D.; Liu, X.; Dhana, K.; Franco, O.H.; Kaptoge, S.; Di Angelantonio, E.; Stampfer, M.; Willett, W.C.; et al. Impact of healthy lifestyle factors on life expectancies in the US population. Circulation 2018, 138, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Haskell, W.L.; Lee, I.-M.; Pate, R.R.; Powell, K.E.; Blair, S.N.; Franklin, B.A.; Macera, C.A.; Heath, G.W.; American College of Sports Medicine; American Heart Association; et al. Physical activity and public health: Updated recommendation for adults from the American College of Sports Medicine and the American Heart Association. Circulation 2007, 116, 1081–1093. [Google Scholar] [CrossRef] [PubMed]
- Maron, B.J.; Zipes, D.P.; Kovacs, R.J.; American Heart Association Electrocardiography and Arrhythmias Committee of Council on Clinical Cardiology; Council on Cardiovascular Disease in Young; Council on Cardiovascular and Stroke Nursing; Council on Functional Genomics and Translational Biology; The American College of Cardiology. Eligibility and disqualification recommendations for competitive athletes with cardiovascular abnormalities: Preamble, principles, and general considerations: A scientific statement from the American Heart Association and American College of Cardiology. Circulation 2015, 132, e256–e261. [Google Scholar] [CrossRef] [PubMed]



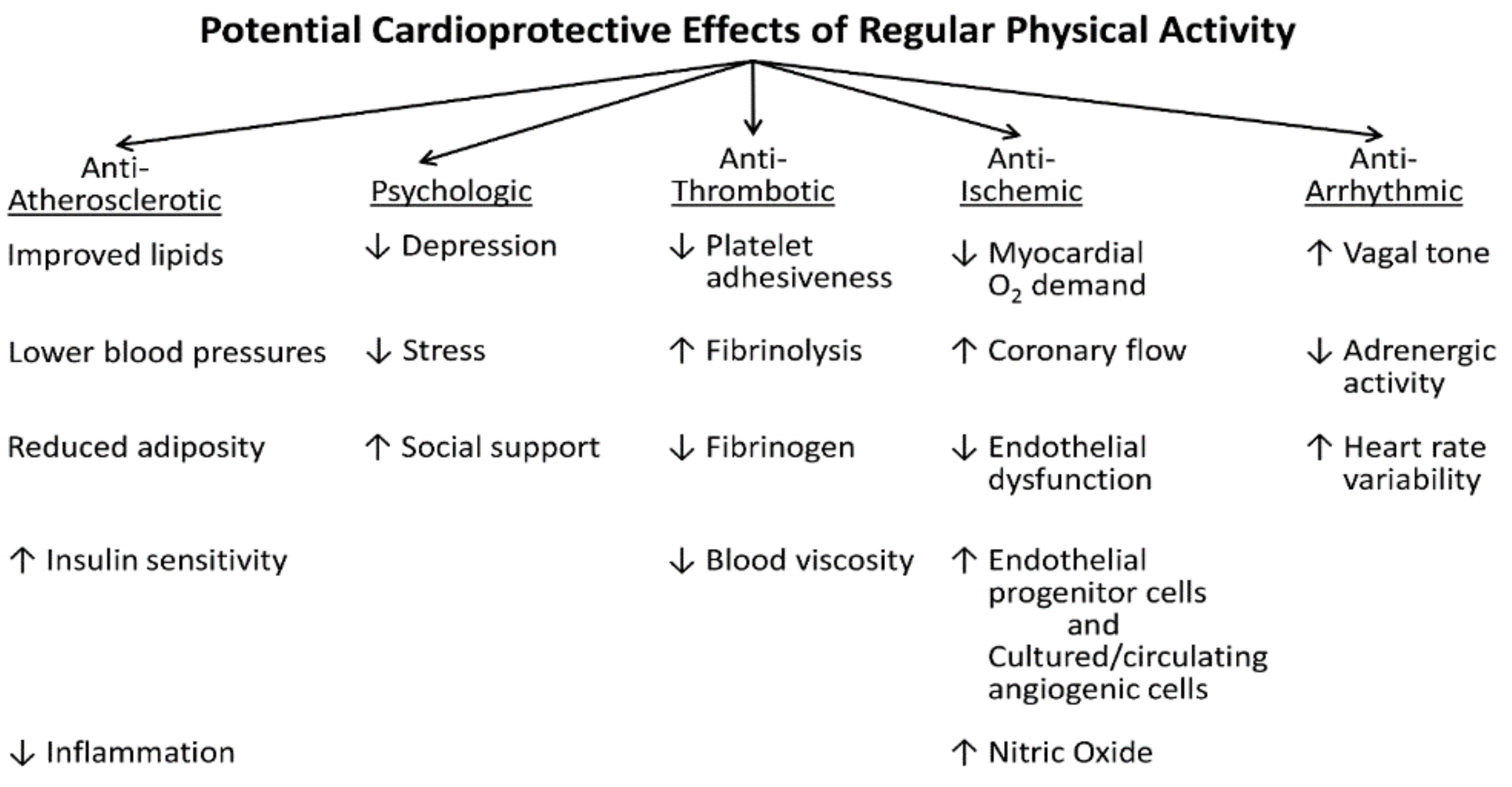
| Induces plaque rupture via: |
| • Increased heart rate, blood pressure, and shear forces |
| • Altered coronary artery dimensions |
| • Exercise-induced spasm in partially obstructed artery segments |
| Renders a fissured plaque more thrombogenic by: |
| • Deepening the fissure |
| • Increasing thrombogenicity |
| Induces thrombogenesis directly via: |
| • Catecholamine-induced platelet aggregation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franklin, B.A.; Rusia, A.; Haskin-Popp, C.; Tawney, A. Chronic Stress, Exercise and Cardiovascular Disease: Placing the Benefits and Risks of Physical Activity into Perspective. Int. J. Environ. Res. Public Health 2021, 18, 9922. https://doi.org/10.3390/ijerph18189922
Franklin BA, Rusia A, Haskin-Popp C, Tawney A. Chronic Stress, Exercise and Cardiovascular Disease: Placing the Benefits and Risks of Physical Activity into Perspective. International Journal of Environmental Research and Public Health. 2021; 18(18):9922. https://doi.org/10.3390/ijerph18189922
Chicago/Turabian StyleFranklin, Barry A., Akash Rusia, Cindy Haskin-Popp, and Adam Tawney. 2021. "Chronic Stress, Exercise and Cardiovascular Disease: Placing the Benefits and Risks of Physical Activity into Perspective" International Journal of Environmental Research and Public Health 18, no. 18: 9922. https://doi.org/10.3390/ijerph18189922
APA StyleFranklin, B. A., Rusia, A., Haskin-Popp, C., & Tawney, A. (2021). Chronic Stress, Exercise and Cardiovascular Disease: Placing the Benefits and Risks of Physical Activity into Perspective. International Journal of Environmental Research and Public Health, 18(18), 9922. https://doi.org/10.3390/ijerph18189922





