The Impact of COVID-19 on the Preparation for the Tokyo Olympics: A Comprehensive Performance Assessment of Top Swimmers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Procedures
2.3.1. Sports Cardiology Screening Protocol
2.3.2. Post-COVID Return-to-Play Examinations
2.3.3. Time-Ranking Points Calculation
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.2. Findings of the Sports Cardiology Screening
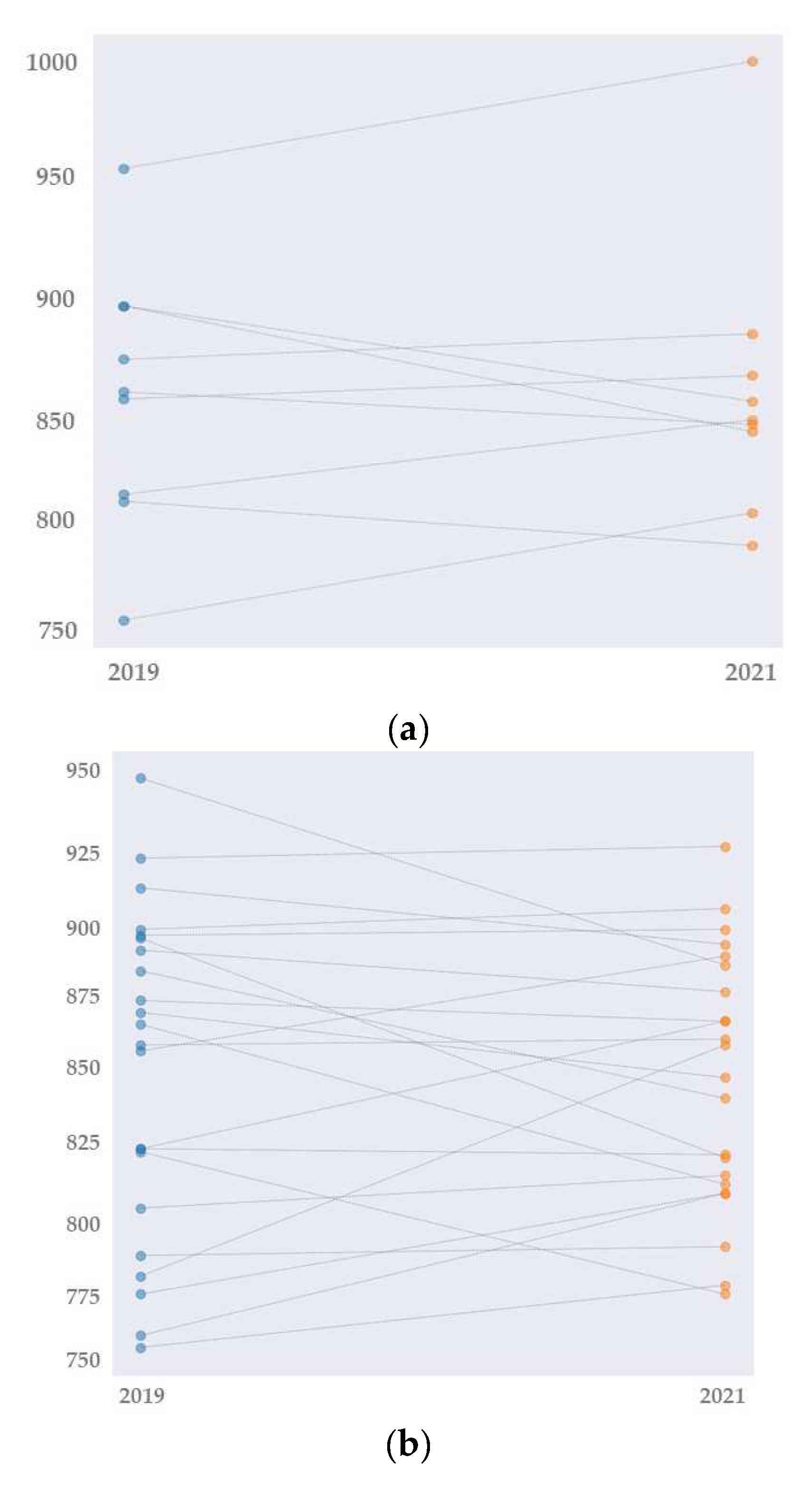
3.3. Performance Analyses by Time-Ranking Points
3.4. Findings of Return-to-PLAY Examination
4. Discussion
4.1. Practical Implications
4.2. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thompson, P.D.; Levine, B.D. Protecting athletes from sudden cardiac death. JAMA 2006, 296, 1648–1650. [Google Scholar] [CrossRef] [PubMed]
- Corrado, D.; Pelliccia, A.; Bjørnstad, H.H.; Vanhees, L.; Biffi, A.; Borjesson, M.; Panhuyzen-Goedkoop, N.; Deligiannis, A.; Solberg, E.; Dugmore, D.; et al. Cardiovascular pre-participation screening of young competitive athletes for prevention of sudden death: Proposal for a common European protocol. Consensus Statement of the Study Group of Sport Cardiology of the Working Group of Cardiac Rehabilitation and Exercise Physiology and the Working Group of Myocardial and Pericardial Diseases of the European Society of Cardiology. Eur. Heart J. 2005, 26, 516–524. [Google Scholar]
- Gautret, P.; Al-Tawfiq, J.A.; Hoang, V.T. COVID 19: Will the 2020 Hajj pilgrimage and Tokyo Olympic Games be cancelled? Travel Med. Infect. Dis. 2020, 34, 101622. [Google Scholar] [CrossRef]
- Håkansson, A.; Moesch, K.; Jönsson, C.; Kenttä, G. Potentially Prolonged Psychological Distress from Postponed Olympic and Paralympic Games during COVID-19—Career Uncertainty in Elite Athletes. Int. J. Environ. Res. Public Health 2021, 18, 2. [Google Scholar] [CrossRef]
- Mon-López, D.; de la Rubia Riaza, A.; Hontoria Galán, M.; Refoyo Roman, I. The Impact of Covid-19 and the Effect of Psychological Factors on Training Conditions of Handball Players. Int. J. Environ. Res. Public Health 2020, 17, 6471. [Google Scholar] [CrossRef]
- Mehrsafar, A.H.; Gazerani, P.; Moghadam Zadeh, A.; Jaenes Sánchez, J.C. Addressing potential impact of COVID-19 pandemic on physical and mental health of elite athletes. Brain Behav. Immun. 2020, 87, 147–148. [Google Scholar] [CrossRef]
- Kaneda, K.; Maeda, N.; Suzuki, Y.; Fukui, K.; Urabe, Y. Impact of the COVID-19 Pandemic on Life Space Extent and Apathy: A Comparison of Competitive Japanese Swimmers with and without Disabilities. Int. J. Environ. Res. Public Health 2021, 18, 5106. [Google Scholar] [CrossRef]
- Haddad, M.; Abbes, Z.; Mujika, I.; Chamari, K. Impact of COVID-19 on Swimming Training: Practical Recommendations during Home Confinement/Isolation. Int. J. Environ. Res. Public Health 2021, 18, 4767. [Google Scholar] [CrossRef] [PubMed]
- Romano Spica, V.; Gallè, F.; Baldelli, G.; Valeriani, F.; Di Rosa, E.; Liguori, G.; Brandi, G. Swimming Pool safety and prevention at the time of Covid-19: A consensus document from GSMS-SItI. Ann. Ig. 2020, 32, 439–448. [Google Scholar]
- Swimming World Magazine. Available online: https://www.swimmingworldmagazine.com/news/will-2020-be-the-first-year-without-world-swimming-records-since-1896/ (accessed on 18 May 2021).
- Yeo, T.J. Sport and exercise during and beyond the COVID-19 pandemic. Eur. J. Prev. Cardiol. 2020, 27, 1239–1241. [Google Scholar] [CrossRef] [PubMed]
- Madjid, M.; Safavi-Naeini, P.; Solomon, S.D.; Vardeny, O. Potential Effects of Coronaviruses on the Cardiovascular System: A Review. JAMA Cardiol. 2020, 5, 831–840. [Google Scholar] [CrossRef] [Green Version]
- Dores, H.; Cardim, N. Return to play after COVID-19: A sport cardiologist’s view. Br. J. Sports Med. 2020, 54, 1132–1133. [Google Scholar] [CrossRef]
- Costello, F.E.; Duggan, K.; Lang, E.; Norris, S. A safe return to sport and the right to play during COVID-19. CMAJ 2021, 193, E291. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Levine, B.D.; Phelan, D.; Emery, M.S.; Martinez, M.W.; Chung, E.H.; Thompson, P.D.; Baggish, A.L. Coronavirus Disease 2019 and the Athletic Heart: Emerging Perspectives on Pathology, Risks, and Return to Play. JAMA Cardiol. 2021, 6, 219–227. [Google Scholar] [CrossRef]
- Udelson, J.E.; Curtis, M.A.; Rowin, E.J. Return to Play for Athletes After Coronavirus Disease 2019 Infection-Making High-Stakes Recommendations as Data Evolve. JAMA Cardiol. 2021, 6, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Phelan, D.; Kim, J.H.; Chung, E.H. A Game Plan for the Resumption of Sport and Exercise After Coronavirus Disease 2019 (COVID-19) Infection. JAMA Cardiol. 2020, 5, 1085–1086. [Google Scholar] [CrossRef]
- American College of Cardiology. Available online: https://www.acc.org/latest-in-cardiology/articles/2020/07/13/13/37/returning-to-play-after-coronavirus-infection (accessed on 1 June 2021).
- Counsilman, J.E. The Science of Swimming, 1st ed.; Prentice Hall: Upper Saddle River, NJ, USA, 1968; pp. 23–27. [Google Scholar]
- Maglischo, E.W. Season planning. In Swimming Fastest; Barnard, M., Hambly, L., Brachtesende, D., Thoren, K., Seyfert, S., Replinger, B., Bihler, D.K., Eds.; Human Kinetics: Champaign, IL, USA, 2003; pp. 593–595. [Google Scholar]
- Sharma, S.; Drezner, J.A.; Baggish, A.; Papadakis, M.; Wilson, M.G.; Prutkin, J.M.; La Gerche, A.; Ackerman, M.J.; Borjesson, M.; Salerno, J.C.; et al. International recommendations for electrocardiographic interpretation in athletes. Eur. Heart J. 2018, 39, 1466–1480. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015, 16, 233–271. [Google Scholar]
- Shetler, K.; Marcus, R.; Froelicher, V.F.; Vora, S.; Kalisetti, D.; Prakash, M.; Do, D.; Myers, J. Heart rate recovery: Validation and methodologic issues. J. Am. Coll. Cardiol. 2001, 38, 1980–1987. [Google Scholar] [CrossRef] [Green Version]
- Fédération Internationale de Natation. Available online: https://www.fina.org/swimming/points (accessed on 17 May 2021).
- Cavigli, L.; Frascaro, F.; Turchini, F.; Mochi, N.; Sarto, P.; Bianchi, S.; Parri, A.; Carraro, N.; Valente, S.; Focardi, M.; et al. A prospective study on the consequences of SARS-CoV-2 infection on the heart of young adult competitive athletes: Implications for a safe return-to-play. Int. J. Cardiol. 2021, 336, 130–136. [Google Scholar] [CrossRef]
- Grant, W.B.; Lahore, H.; Rockwell, M.S. The Benefits of Vitamin D Supplementation for Athletes: Better Performance and Reduced Risk of COVID-19. Nutrients 2020, 12, 3741. [Google Scholar] [CrossRef] [PubMed]
- Mercola, J.; Grant, W.B.; Wagner, C.L. Evidence Regarding Vitamin D and Risk of COVID-19 and Its Severity. Nutrients 2020, 12, 3361. [Google Scholar] [CrossRef] [PubMed]
- Cannell, J.J.; Hollis, B.W.; Sorenson, M.B.; Taft, T.N.; Anderson, J.J. Athletic performance and vitamin D. Med. Sci. Sports Exerc. 2009, 41, 1102–1110. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.; Cheung, C.; Currie, K.; King, T.; Mountjoy, M.; Burr, J. Cardiac Remodeling in Elite Aquatic Sport Athletes. SportRxiv 2021. [Google Scholar] [CrossRef]
- Pluim, B.M.; Zwinderman, A.H.; van der Laarse, A.; van der Wall, E.E. The athlete’s heart. A meta-analysis of cardiac structure and function. Circulation 2000, 101, 336–344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mujika, I.; Padilla, S. Detraining: Loss of training-induced physiological and performance adaptations. Part I: Short term insufficient training stimulus. Sports Med. 2000, 30, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Mujika, I.; Padilla, S. Detraining: Loss of training-induced physiological and performance adaptations. Part II: Long term insufficient training stimulus. Sports Med. 2000, 30, 145–154. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, B.T.; De Lima-Junior, D.; Da Silva Filho, E.M. The impact of COVID-19 on sporting events and high-performance athletes. J. Sports Med. Phys. Fit. 2020, 60, 1507–1508. [Google Scholar] [CrossRef] [PubMed]
- Milovancev, A.; Avakumovic, J.; Lakicevic, N.; Stajer, V.; Korovljev, D.; Todorovic, N.; Bianco, A.; Maksimovic, N.; Ostojic, S.; Drid, P. Cardiorespiratory Fitness in Volleyball Athletes Following a COVID-19 Infection: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2021, 18, 4059. [Google Scholar] [CrossRef]
- Fikenzer, S.; Fikenzer, K.; Laufs, U.; Falz, R.; Pietrek, H.; Hepp, P. Impact of COVID-19 lockdown on endurance capacity of elite handball players. J. Sports Med. Phys. Fit. 2021, 61, 977–982. [Google Scholar]
- Dauty, M.; Menu, P.; Fouasson-Chailloux, A. Effects of the COVID-19 confinement period on physical conditions in young elite soccer players. J. Sports Med. Phys. Fit. 2020. online ahead of print. [Google Scholar] [CrossRef]
- de Albuquerque Freire, L.; Tannure, M.; Sampaio, M.; Slimani, M.; Znazen, H.; Bragazzi, N.L.; Aedo-Muñoz, E.; Sobarzo Soto, D.A.; Brito, C.J.; Miarka, B. COVID-19-Related Restrictions and Quarantine COVID-19: Effects on Cardiovascular and Yo-Yo Test Performance in Professional Soccer Players. Front. Psychol. 2020, 11, 589543. [Google Scholar] [CrossRef] [PubMed]


| Non-COVID Swimmers (n = 32) | COVID Swimmers (n = 14) | P | |
|---|---|---|---|
| Age (years) | 24.2 ± 4.4 | 23 ± 3.8 | 0.40 |
| Female (n, %) | 14 (43.8%) | 7 (50%) | 0.70 |
| Sports experience (years) | 17.8 ± 4.5 | 16.8 ± 3.9 | 0.44 |
| Pre-COVID training hours/week | 24 ± 4.5 | 24.5 ± 3.9 | 0.71 |
| Post-COVID training hours/week | 24 ± 4.5 | 24.5 ± 3.9 | 0.71 |
| Missed training days | 18.5 ± 3.4 | 19 ± 6 | 0.76 |
| Non-COVID Swimmers (n = 32) | COVID Swimmers (n = 14) | 2019 vs. 2019 | 2021 vs. 2021 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 2019 | 2021 | p | 2019 | 2021 | p | p | p | ||
| Ejection fraction (%) | M | 55.8 ± 2.1 | 58.8 ± 4.1 | 0.02 * | 59.7 ± 8.0 | 59.3 ± 4.8 | 0.93 | 0.1 | 0.81 |
| F | 59.1 ± 2.6 | 60.0 ± 3.3 | 0.48 | 56.9 ± 3.3 | 60.0 ± 4.7 | 0.17 | 0.097 | 1 | |
| Septal wall thickness (mm) | M | 11 ± 1.2 | 10.8 ± 1.1 | 0.62 | 11.3 ± 2.5 | 11.4 ± 1.3 | 0.94 | 0.72 | 0.25 |
| F | 9.2 ± 0.8 | 9.2 ± 1.3 | 0.97 | 9.9 ± 0.9 | 10.1 ± 0.7 | 0.52 | 0.11 | 0.15 | |
| Posterior wall thickness (mm) | M | 9.6 ± 0.99 | 10.7 ± 1.1 | 0.009 * | 9 ± 0 | 10 (10–11) | 0.002 ** | 0.32 | 0.36 |
| F | 8.4 ± 0.85 | 9.9 ± 1.3 | 0.003 * | 8.4 ± 1 | 11 ± 1.4 | 0.002 ** | 1 | 0.12 | |
| LV end-diastolic diameter (mm) | M | 55.7 ± 4.1 | 51.4 ± 2.9 | 0.003 * | 54.7 ± 4.7 | 50.1 ± 3.5 | 0.14 | 0.71 | 0.02 * |
| F | 48.4 ± 2.3 | 44.4 ± 2.8 | 0.001 * | 49 ± 2.5 | 46.4 ± 2 | 0.046 * | 0.48 | 0.12 | |
| RV diameter (mm) | M | 35.5 ± 3.3 | 37.9 ± 3.2 | 0.06 | 35.3 ± 4.2 | 39 (38–41) | 0.24 | 0.93 | 0.20 |
| F | 31.8 ± 4 | 34.6 ± 4.4 | 0.11 | 32.9 ± 3 | 37.6 ± 2.3 | 0.006 * | 0.54 | 0.12 | |
| Non-COVID Swimmers (n = 32) | Covid Swimmers (n = 14) | 2019 vs. 2019 | 2021 vs. 2021 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 2019 | 2021 | p | 2019 | 2021 | p | p | p | ||
| Resting HR (bpm) | 68.4 ± 13.4 | 62.0 ± 11 | 0.06 | 69.0 ± 15 | 72.4 ± 17 | 0.61 | 0.90 | 0.024 * | |
| Peak HR (bpm) | 190.0 ± 10.3 | 190.5 ± 11.5 | 0.88 | 191.2 ± 9.1 | 188.0 ± 11 | 0.44 | 0.74 | 0.53 | |
| HR recovery (1/min) | 32.3 ± 10.8 | 22.0 (20.0–32.0) | 0.03 * | 33.1 ± 13.9 | 23.5 (19.7–31.2) | 0.32 | 0.85 | 0.78 | |
| RER | 1.15 ± 0.05 | 1.17 ± 0.08 | 0.28 | 1.15 ± 0.07 | 1.17 ± 0.07 | 0.38 | 0.81 | 0.94 | |
| Treadmill time (min) | M | 16.0 (13.0–16.5) | 14.5 ± 2.7 | 0.12 | 14.5 ± 1.3 | 15.0 (13.7–15.0) | 1.0 | 0.73 | 0.40 |
| F | 13.2 ± 2.8 | 12.9 ± 1.6 | 0.80 | 14.4 ± 2.0 | 13.5 ± 2.0 | 0.45 | 0.35 | 0.55 | |
| Max load (Watt) | M | 432.6 ± 74.0 | 402.9 ± 60.8 | 0.25 | 464.3 ± 25.8 | 458.0 ± 31.0 | 0.75 | 0.42 | 0.052 |
| F | 294.0 ± 50.4 | 270.4 ± 41.3 | 0.32 | 311.4 ± 61.7 | 283.3 ± 41.5 | 0.37 | 0.52 | 0.59 | |
| VO2 max (L/min) | M | 4.7 (4.5–5) | 4.6 ± 0.7 | 0.89 | 4.7 ± 0.4 | 5.2 ± 0.6 | 0.14 | 0.89 | 0.08 |
| F | 2.9 ± 0.3 | 3.1 ± 0.4 | 0.48 | 3.3 ± 0.5 | 3.2 ± 0.4 | 0.82 | 0.10 | 0.47 | |
| VO2 max (mL/min/kg) | M | 56.7 ± 4.7 | 55.5 ± 4.5 | 0.49 | 55 ± 3.8 | 56.5 ± 4.9 | 0.53 | 0.41 | 0.20 |
| F | 49.6 ± 3 | 50.7 ± 2.6 | 0.47 | 53.1 ± 5.5 | 52.9 ± 4.1 | 0.97 | 0.12 | 0.76 | |
| O2 pulse (mL/bpm) | M | 25.0 ± 1.9 | 24.0 ± 1.8 | 0.23 | 25.6 ± 2.1 | 26.4 ± 2.4 | 0.58 | 0.58 | 0.03 * |
| F | 15.0 (13.7–17.9) | 16.2 ± 2.1 | 0.64 | 17.8 ± 2.8 | 18.2 ± 2.7 | 0.82 | 0.06 | 0.18 | |
| VE (L/min) | M | 158.0 ± 33.0 | 159.0 ± 31.5 | 0.95 | 153.0 ± 9.5 | 178.0 ± 16.6 | 0.03 * | 0.74 | 0.19 |
| F | 114.0 ± 17.0 | 109.0 ± 25.0 | 0.64 | 118.0 ± 10.4 | 111.0 ± 12.4 | 0.28 | 0.60 | 0.90 | |
| VE/VCO2 | M | 25.0 ± 2.2 | 29.4 ± 3.9 | 0.70 | 28.5 ± 2.2 | 28.9 ± 3.5 | 0.86 | 0.88 | 0.78 |
| F | 32.9 ± 2 | 31.0 ± 2.9 | 0.12 | 31.5 ± 2.2 | 30.2 ± 1.6 | 0.32 | 0.17 | 0.58 | |
| Peak lactate (mmol/L) | M | 7.6 ± 2.5 | 7.8 ± 3.1 | 0.88 | 9.7 ± 4.1 | 9.5 ± 2.1 | 0.95 | 0.22 | 0.23 |
| F | 8.5 ± 2.5 | 9.1 ± 2.2 | 0.58 | 8.4 ± 1.9 | 6.6 ± 1.7 | 0.09 | 0.99 | 0.04 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Csulak, E.; Petrov, Á.; Kováts, T.; Tokodi, M.; Lakatos, B.; Kovács, A.; Staub, L.; Suhai, F.I.; Szabó, E.L.; Dohy, Z.; et al. The Impact of COVID-19 on the Preparation for the Tokyo Olympics: A Comprehensive Performance Assessment of Top Swimmers. Int. J. Environ. Res. Public Health 2021, 18, 9770. https://doi.org/10.3390/ijerph18189770
Csulak E, Petrov Á, Kováts T, Tokodi M, Lakatos B, Kovács A, Staub L, Suhai FI, Szabó EL, Dohy Z, et al. The Impact of COVID-19 on the Preparation for the Tokyo Olympics: A Comprehensive Performance Assessment of Top Swimmers. International Journal of Environmental Research and Public Health. 2021; 18(18):9770. https://doi.org/10.3390/ijerph18189770
Chicago/Turabian StyleCsulak, Emese, Árpád Petrov, Tímea Kováts, Márton Tokodi, Bálint Lakatos, Attila Kovács, Levente Staub, Ferenc Imre Suhai, Erzsébet Liliána Szabó, Zsófia Dohy, and et al. 2021. "The Impact of COVID-19 on the Preparation for the Tokyo Olympics: A Comprehensive Performance Assessment of Top Swimmers" International Journal of Environmental Research and Public Health 18, no. 18: 9770. https://doi.org/10.3390/ijerph18189770
APA StyleCsulak, E., Petrov, Á., Kováts, T., Tokodi, M., Lakatos, B., Kovács, A., Staub, L., Suhai, F. I., Szabó, E. L., Dohy, Z., Vágó, H., Becker, D., Müller, V., Sydó, N., & Merkely, B. (2021). The Impact of COVID-19 on the Preparation for the Tokyo Olympics: A Comprehensive Performance Assessment of Top Swimmers. International Journal of Environmental Research and Public Health, 18(18), 9770. https://doi.org/10.3390/ijerph18189770






