Potential Protection of Pre-Existent Antibodies to Human Coronavirus 229E on COVID-19 Severity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design, Subjects, and Samples Collection
2.2. Collection of Clinical Information and Severity Classification
2.3. ELISA for The Detection of IgG of Seasonal HCoVs
2.4. Statistical Analysis
3. Results
3.1. Study Population
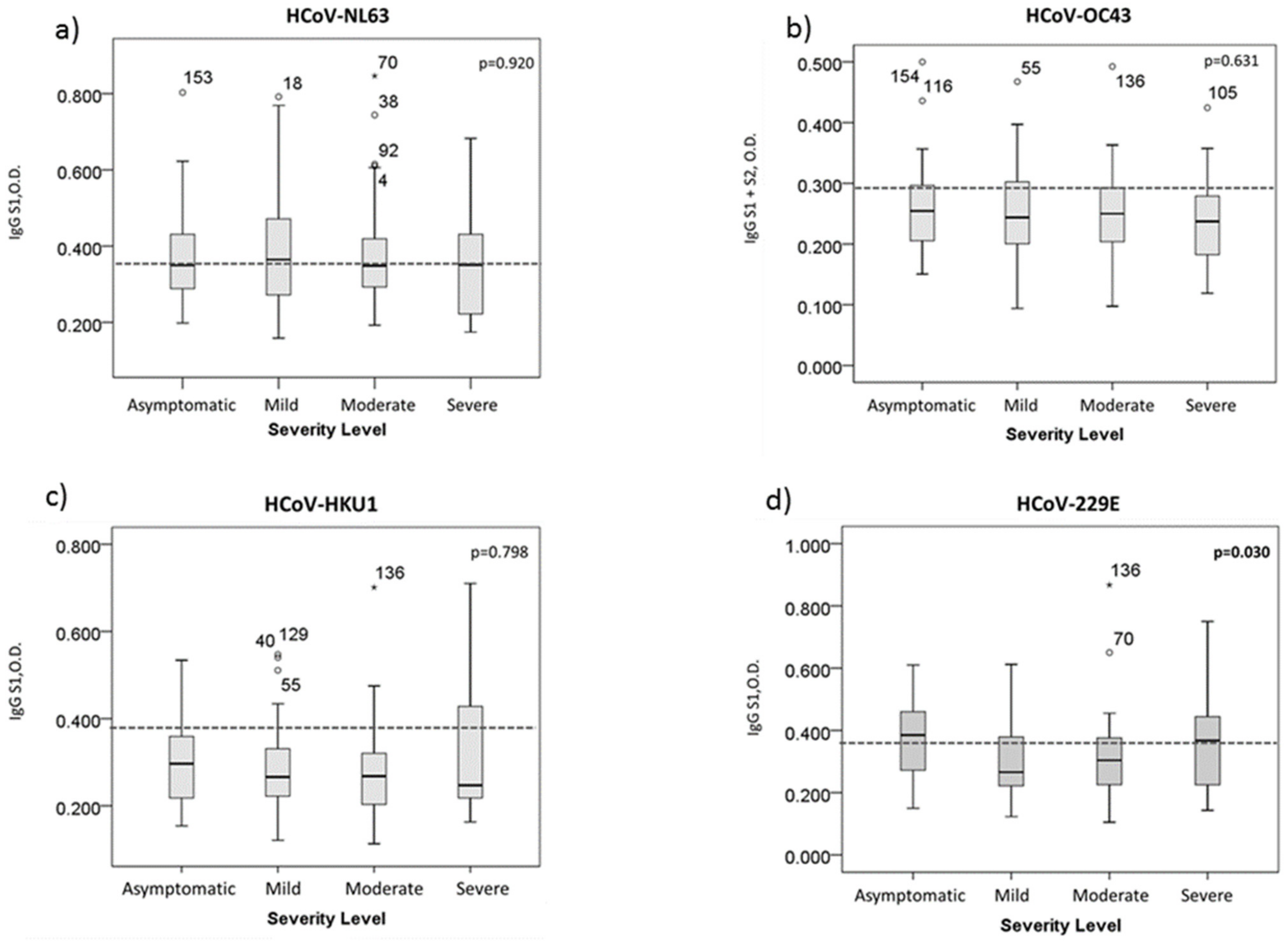
3.2. HCoV-229E Antibody Titer Is Related to The Severity Level of COVID-19
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Coronavirus Disease (COVID-19) Dashboard: World Health Organization. 2021. Available online: https://covid19.who.int/ (accessed on 8 July 2021).
- Alluwaimi, A.M.; Alshubaith, I.H.; Al-Ali, A.M.; Abohelaika, S. The Coronaviruses of Animals and Birds: Their Zoonosis, Vaccines, and Models for SARS-CoV and SARS-CoV2. Front. Vet. Sci. 2020, 7, 1–12. [Google Scholar] [CrossRef]
- Malik, Y.A. Properties of Coronavirus and SARS-CoV-2. Malays. J. Pathol. 2020, 42, 3–11. [Google Scholar]
- Oran, D.P.; Topol, E.J. Prevalence of Asymptomatic SARS-CoV-2 Infection: A Narrative Review. Ann. Intern. Med. 2020, 173, 362–367. [Google Scholar] [CrossRef]
- Lai, C.C.; Liu, Y.H.; Wang, C.Y.; Wang, Y.H.; Hsueh, S.C.; Yen, M.Y.; Ko, W.C.; Hsueh, P.R. Asymptomatic carrier state, acute respiratory disease, and pneumonia due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): Facts and myths. J. Microbiol. Immunol. Infect. 2020, 53, 404–412. [Google Scholar] [CrossRef]
- Roltgen, K.; Powell, A.E.; Wirz, O.F.; Stevens, B.A.; Hogan, C.A.; Najeeb, J.; Hunter, M.; Wang, H.; Sahoo, M.K.; Huang, C.; et al. Defining the features and duration of antibody responses to SARS-CoV-2 infection associated with disease severity and outcome. Sci. Immunol. 2020, 5, 1–19. [Google Scholar] [CrossRef]
- Barek, M.A.; Aziz, M.A.; Islam, M.S. Impact of age, sex, comorbidities and clinical symptoms on the severity of COVID-19 cases: A meta-analysis with 55 studies and 10014 cases. Heliyon 2020, 6, e05684. [Google Scholar] [CrossRef]
- Mahase, E. Covid-19: Why are age and obesity risk factors for serious disease? BMJ 2020, 371, 1–2. [Google Scholar]
- Garcia, L.F. Immune Response, Inflammation, and the Clinical Spectrum of COVID-19. Front. Immunol. 2020, 11, 1441. [Google Scholar] [CrossRef] [PubMed]
- Miyara, M.; Sterlin, D. Pre-COVID-19 humoral immunity to common coronaviruses does not confer crossprotection against SARS-CoV-2. medRxiv 2020. [Google Scholar] [CrossRef]
- Sagar, M.; Reifler, K.; Rossi, M.; Miller, N.S.; Sinha, P.; White, L.; Mizgerd, J.P. Recent endemic coronavirus infection is associated with less severe COVID-19. J. Clin. Investig. 2020, 131, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Sekine, T.; Perez-Potti, A.; Rivera-Ballesteros, O.; Stralin, K.; Gorin, J.B.; Olsson, A.; Llewellyn-Lacey, S.; Kamal, H.; Bogdanovic, G.; Muschiol, S.; et al. Robust T Cell Immunity in Convalescent Individuals with Asymptomatic or Mild COVID-19. Cell 2020, 183, 158–168. [Google Scholar] [CrossRef]
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E. Remdesivir for the Treatment of Covid-19—Preliminary Report. Reply N. Engl. J. Med. 2020, 383, 1–12. [Google Scholar]
- Espinosa, R.; Lopez, T.; Bogdanoff, W.A.; Espinoza, M.A.; Lopez, S.; DuBois, R.M.; Arias, C.F. Isolation of Neutralizing Monoclonal Antibodies to Human Astrovirus and Characterization of Virus Variants That Escape Neutralization. J. Virol. 2019, 93, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stadlbauer, D.; Amanat, F.; Chromikova, V.; Jiang, K.; Strohmeier, S.; Arunkumar, G.A.; Tan, J.; Bhavsar, D.; Capuano, C.; Kirkpatrick, E.; et al. SARS-CoV-2 Seroconversion in Humans: A Detailed Protocol for a Serological Assay, Antigen Production, and Test Setup. Curr. Protoc. Microbiol. 2020, 57, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Shah, K.; Maghsoudlou, P. Enzyme-linked immunosorbent assay (ELISA): The basics. Br. J. Hosp. Med. 2016, 77, 98–101. [Google Scholar] [CrossRef] [PubMed]
- Nicola, M.; Alsafi, Z.; Sohrabi, C.; Kerwan, A.; Al-Jabir, A.; Iosifidis, C.; Agha, M.; Agha, R. The socio-economic implications of the coronavirus pandemic (COVID-19): A review. Int. J. Surg. 2020, 78, 185–193. [Google Scholar] [CrossRef]
- Dijkman, R.; Jebbink, M.F.; Gaunt, E.; Rossen, J.W.; Templeton, K.E.; Kuijpers, T.W.; van der Hoek, L. The dominance of human coronavirus OC43 and NL63 infections in infants. J. Clin. Virol. 2012, 53, 135–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choe, P.G.; Perera, R.; Park, W.B.; Song, K.H.; Bang, J.H.; Kim, E.S.; Kim, H.B.; Ko, L.W.R.; Park, S.W.; Kim, N.J.; et al. MERS-CoV Antibody Responses 1 Year after Symptom Onset, South Korea, 2015. Emerg. Infect. Dis. 2017, 23, 1079–1084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Okba, N.M.A.; Raj, V.S.; Widjaja, I.; GeurtsvanKessel, C.H.; de Bruin, E.; Chandler, F.D.; Park, W.B.; Kim, N.J.; Farag, E.; Al-Hajri, M.; et al. Sensitive and Specific Detection of Low-Level Antibody Responses in Mild Middle East Respiratory Syndrome Coronavirus Infections. Emerg. Infect. Dis. 2019, 25, 1868–1877. [Google Scholar] [CrossRef]
- Song, G.; He, W.T.; Callaghan, S.; Anzanello, F.; Huang, D.; Ricketts, J.; Torres, J.L.; Beutler, N.; Peng, L.; Vargas, S.; et al. Cross-reactive serum and memory B-cell responses to spike protein in SARS-CoV-2 and endemic coronavirus infection. Nat. Commun. 2021, 12, 2938. [Google Scholar] [CrossRef]
- Ju, B.; Zhang, Q.; Ge, J.; Wang, R.; Sun, J.; Ge, X.; Yu, J.; Shan, S.; Zhou, B.; Song, S.; et al. Human neutralizing antibodies elicited by SARS-CoV-2 infection. Nature 2020, 584, 115–119. [Google Scholar] [CrossRef]
- Lipsky, M.S.; Hung, M. Men and COVID-19: A Pathophysiologic Review. Am. J. Men’s Health 2020, 14, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kraaijeveld, C.A.; Reed, S.E.; Macnaughton, M.R. Enzyme-linked immunosorbent assay for detection of antibody in volunteers experimentally infected with human coronavirus strain 229 E. J. Clin. Microbiol. 1980, 12, 493–497. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, J.H.; Muller, M.A.; Seok, H.; Park, G.E.; Lee, J.Y.; Cho, S.Y.; Ha, Y.E.; Baek, J.Y.; Kim, S.H.; Kang, J.M.; et al. Serologic responses of 42 MERS-coronavirus-infected patients according to the disease severity. Diagn. Microbiol. Infect. Dis. 2017, 89, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Birra, D.; Benucci, M.; Landolfi, L.; Merchionda, A.; Loi, G.; Amato, P.; Licata, G.; Quartuccio, L.; Triggiani, M.; Moscato, P. COVID 19: A clue from innate immunity. Immunol. Res. 2020, 68, 161–168. [Google Scholar] [CrossRef]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; et al. Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell 2020, 181, 1489–1501. [Google Scholar] [CrossRef]
| Characteristic | Classification of Severity | |||||
|---|---|---|---|---|---|---|
| Asymptomatic | Mild | Moderate | Severe | Total | p Value | |
| n = 26 | n = 74 | n = 47 | n = 18 | n = 165 | ||
| General characteristics | ||||||
| Patient gender | <0.001 | |||||
| Female | 19 (23.2) | 47 (57.3) | 13 (15.9) | 3 (3.7) | 82 (100) | |
| Male | 7 (8.4) | 27 (32.5) | 34 (41) | 15 (18.1) | 83 (100) | |
| Age | 0.045 | |||||
| 42 years old and under | 12 (14.8) | 45 (55.6) | 17 (21) | 7 (8.6) | 81 (100) | |
| More than 42 years old | 14 (16.7) | 29 (34.5) | 30 (35.7) | 11 (13.1) | 84 (100) | |
| Comorbidities | ||||||
| Diabetes | 2 (9.5) | 7 (33.3) | 12 (57.1) | 0 (0) | 21 (100) | 0.012 |
| Cardiovascular problems | 3 (9.4) | 10 (31.3) | 15 (46.9) | 4 (12.5) | 32 (100) | 0.058 |
| Allergic diseases | 2 (33.3) | 3 (50) | 1 (16.7) | 0 (0) | 6 (100) | 0.531 |
| Neoplasic lesions | 0 (0) | 1 (100) | 0 (0) | 0 (0) | 1 (100) | 0.747 |
| Autoimmune diseases | 1 (100) | 0 (0) | 0 (0) | 0 (0) | 1 (100) | 0.257 |
| Smoking | 0 (0) | 0 (0) | 1 (100) | 0 (0) | 1 (100) | 0.461 |
| Positive Results | Classification of Severity | |||||
|---|---|---|---|---|---|---|
| Asymptomatic | Mild | Moderate | Severe | Total | p Value | |
| n = 26 | n = 74 | n = 47 | n = 18 | n = 165 | ||
| HCoV-NL63 | 12 (15.0) | 38 (47.5) | 22 (27.5) | 8 (100) | 80 (100) | 0.925 |
| HCoV-229E | 15 (25.0) | 21 (35.0) | 14 (23.0) | 10 (17.0) | 60 (100) | 0.012 |
| HCoV-HKU1 | 11 (26.8) | 17 (41.5) | 8 (19.5) | 5 (12.2) | 41 (100) | 0.111 |
| HCoV-OC43 | 7 (17.1) | 19 (46.3) | 13 (31.7) | 2 (4.9) | 41 (100) | 0.551 |
| α y β HCoV | 2 (16.7) | 6 (50.0) | 3 (25.0) | 1 (8.3) | 12 (100) | 0.975 |
| αHCoV | 8 (20.0) | 15 (37.5) | 11 (27.5) | 6 (15.0) | 40 (100) | 0.559 |
| βHCoV | 5 (29.4) | 7 (41.2) | 4 (23.5) | 1 (5.9) | 17 (100) | 0.407 |
| Seasonal | Classification of Severity | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Asymptomatic | Mild | Moderate | ||||||||||
| HCoV | OR | CI 95% | p | OR | CI 95% | p | OR | CI 95% | p | |||
| LL | UL | value | LL | UL | value | LL | UL | value | ||||
| Female | 11.285 | 2.212 | 57.569 | 0.004 | 9.792 | 2.250 | 42.611 | 0.002 | 2.079 | 0.455 | 9.504 | 0.345 |
| >42 years old | 1.459 | 0.322 | 6.623 | 0.624 | 3.052 | 0.789 | 11.807 | 0.106 | 0.781 | 0.199 | 3.068 | 0.724 |
| HCoV-NL63 | 0.818 | 0.163 | 4.107 | 0.807 | 0.342 | 0.081 | 1.450 | 0.146 | 0.488 | 0.114 | 2.093 | 0.334 |
| HCoV-229E | 1.336 | 0.264 | 6.757 | 0.726 | 8.641 | 2.011 | 37.135 | 0.004 | 4.754 | 1.135 | 19.914 | 0.033 |
| HCoV-HKU1 | 0.497 | 0.091 | 2.709 | 0.419 | 0.569 | 0.116 | 2.796 | 0.488 | 2.321 | 0.415 | 12.992 | 0.338 |
| HCoV-OC43 | 0.147 | 0.014 | 1.554 | 0.111 | 0.138 | 0.015 | 1.305 | 0.084 | 0.163 | 0.017 | 1.548 | 0.114 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guzmán-Martínez, O.; Guardado, K.; Varela-Cardoso, M.; Trujillo-Rivera, A.; Gómez-Ñañez, I.; Ortiz-León, M.C.; Espinosa, R.; Ramos, C.; Pérez-Carreón, J.I.; López-Guerrero, D.V.; et al. Potential Protection of Pre-Existent Antibodies to Human Coronavirus 229E on COVID-19 Severity. Int. J. Environ. Res. Public Health 2021, 18, 9058. https://doi.org/10.3390/ijerph18179058
Guzmán-Martínez O, Guardado K, Varela-Cardoso M, Trujillo-Rivera A, Gómez-Ñañez I, Ortiz-León MC, Espinosa R, Ramos C, Pérez-Carreón JI, López-Guerrero DV, et al. Potential Protection of Pre-Existent Antibodies to Human Coronavirus 229E on COVID-19 Severity. International Journal of Environmental Research and Public Health. 2021; 18(17):9058. https://doi.org/10.3390/ijerph18179058
Chicago/Turabian StyleGuzmán-Martínez, Oscar, Kathia Guardado, Miguel Varela-Cardoso, Alejandro Trujillo-Rivera, Iván Gómez-Ñañez, María Cristina Ortiz-León, Rafaela Espinosa, Celso Ramos, Julio Isael Pérez-Carreón, Delia Vanesa López-Guerrero, and et al. 2021. "Potential Protection of Pre-Existent Antibodies to Human Coronavirus 229E on COVID-19 Severity" International Journal of Environmental Research and Public Health 18, no. 17: 9058. https://doi.org/10.3390/ijerph18179058
APA StyleGuzmán-Martínez, O., Guardado, K., Varela-Cardoso, M., Trujillo-Rivera, A., Gómez-Ñañez, I., Ortiz-León, M. C., Espinosa, R., Ramos, C., Pérez-Carreón, J. I., López-Guerrero, D. V., Sampieri, C. L., Alanís-García, A. B., Rojas-Durán, F., Zenteno-Cuevas, R., Gutiérrez, M., & Montero, H. (2021). Potential Protection of Pre-Existent Antibodies to Human Coronavirus 229E on COVID-19 Severity. International Journal of Environmental Research and Public Health, 18(17), 9058. https://doi.org/10.3390/ijerph18179058






