The Effects of Reduced Physical Activity on the Lipid Profile in Patients with High Cardiovascular Risk during COVID-19 Lockdown
Abstract
:1. Introduction
2. Methods
Statistical Analysis
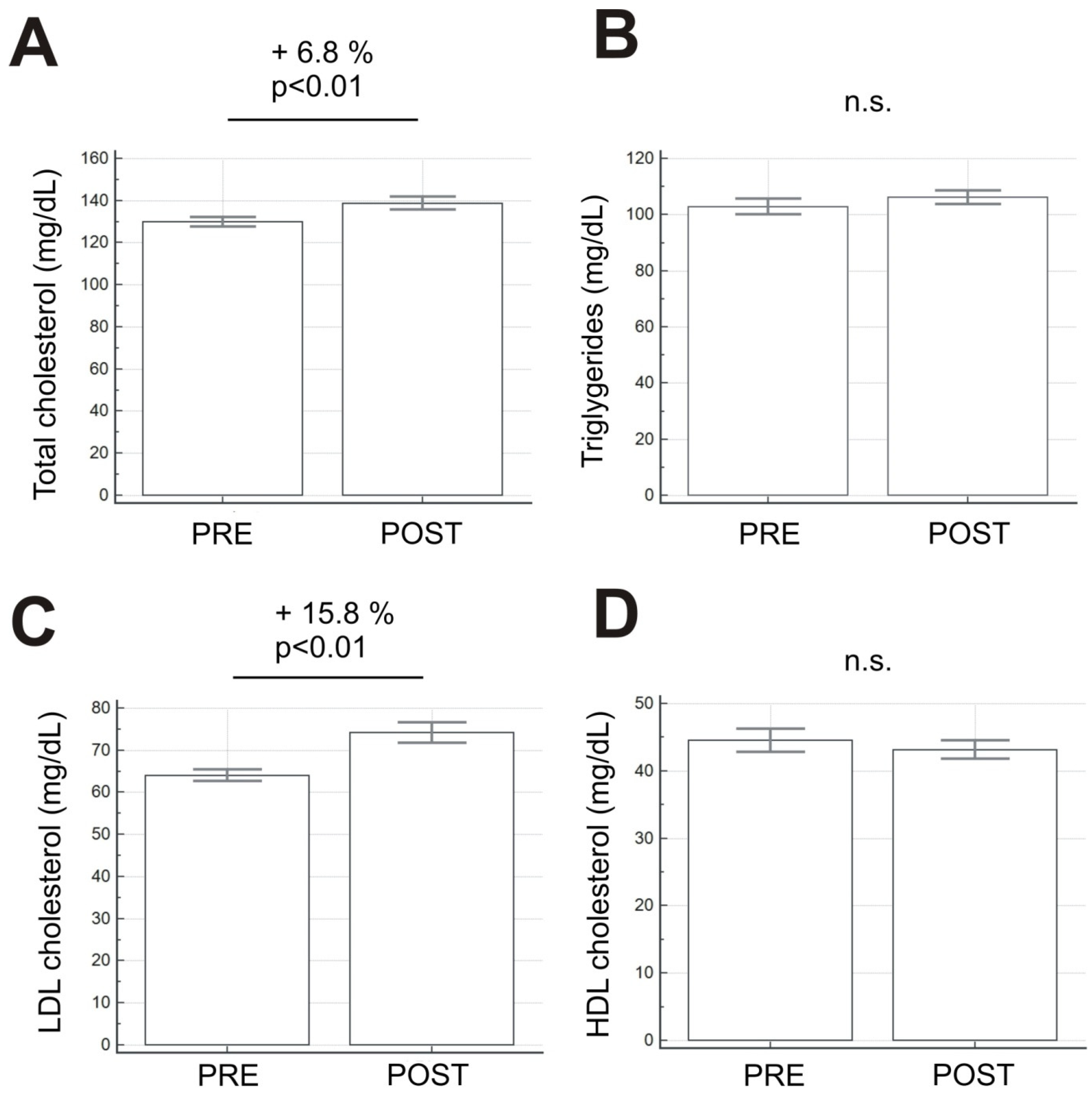
3. Results
4. Discussion
Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- World Health Organization. Coronavirus Disease 2019 (COVID-19) Situation Report-44. Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/ (accessed on 20 May 2020).
- Chakraborty, C.; Sharma, A.R.; Sharma, G.; Bhattacharya, M.; Lee, S.S. SARS-CoV-2 causing pneumonia-associated respiratory disorder (COVID-19): Diagnostic and proposed therapeutic options. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 4016–4026. [Google Scholar]
- Italian Ministry of Health. COVID-19 Report. Available online: http://www.salute.gov.it/portale/nuovocoronavirus/ (accessed on 16 June 2020).
- Peçanha, T.; Goessler, K.F.; Roschel, H.; Gualano, B. Social isolation during the COVID-19 pandemic can increase physical inactivity and the global burden of cardiovascular disease. Am. J. Physiol. Heart Circ. Physiol. 2020, 318, H1441–H1446. [Google Scholar] [CrossRef]
- Wilke, J.; Mohr, L.; Tenforde, A.; Edouard, P.; Fossati, C.; González-Gross, M.; Sánchez Ramírez, C.; Laiño, F.; Tan, B.; Pillay, J.D.; et al. A Pandemic within the Pandemic? Physical Activity Levels Substantially Decreased in Countries Affected by COVID-19. Int. J. Environ. Res. Public Health 2021, 18, 2235. [Google Scholar] [CrossRef] [PubMed]
- Shiroma, E.J.; Lee, I.M. Physical activity and cardiovascular health: Lessons learned from epidemiological studies across age, gender, and race/ethnicity. Circulation 2010, 122, 743–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eijsvogels, T.M.H.; Fernandez, A.B.; Thompson, P.D. Are there deleterious cardiac effects of acute and chronic endurance exercise? Physiol. Rev. 2016, 96, 99–125. [Google Scholar] [CrossRef]
- Aakre, K.M.; Omland, T. Physical activity, exercise and cardiac troponins: Clinical implications. Prog. Cardiovasc. Dis. 2019, 62, 108–115. [Google Scholar] [CrossRef]
- Christensen, R.; Arneja, J.; St. Cyr, K.; Sturrock, S.; Brooks, J. The association of estimated cardiorespiratory fitness with COVID-19 incidence and mortality: A cohort study. PLoS ONE 2021, 16, e0250508. [Google Scholar] [CrossRef] [PubMed]
- Perrone, M.A.; Dasari, J.B.; Intorcia, A.; Gualtieri, P.; Marche, M.; Di Luozzo, M.; Merra, G.; Bernardini, S.; Romeo, F.; Sergi, D. Phenotypic classification and biochemical profile of obesity for cardiovascular prevention. Gazz. Med. Ital. Arch. Sci. Med. 2020, 179, 356–371. [Google Scholar] [CrossRef]
- Perrone, M.A.; Donatucci, B.; Salvati, A.; Gualtieri, P.; De Lorenzo, A.; Romeo, F.; Bernardini, S. Inflammation, oxidative stress and gene expression: The postprandial approach in professional soccer players to reduce the risk of muscle injuries and early atherosclerosis. Med. Sport 2019, 72, 234–243. [Google Scholar] [CrossRef]
- Khosravani, M.; Azarbayjani, M.A.; Abolmaesoomi, M.; Yusof, A.; Abidin, Z.N.; Rahimi, E.; Feizolahi, F.; Akbari, M.; Seyedjalali, S.; Dehghan, F. Ginger extract and aerobic training reduces lipid profile in high-fat fed diet rats. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1617–1622. [Google Scholar] [PubMed]
- Colica, C.; Merra, G.; Gasbarrini, A.; De Lorenzo, A.; Cioccoloni, G.; Gualtieri, P.; Perrone, M.A.; Bernardini, S.; Bernardo, V.; Di Renzo, L.; et al. Efficacy and safety of very-low-calorie ketogenic diet: A double blind randomized crossover study. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2274–2289. [Google Scholar]
- Di Renzo, L.; Merra, G.; Botta, R.; Gualtieri, P.; Manzo, A.; Perrone, M.A.; Mazza, M.; Cascapera, S.; De Lorenzo, A. Post-prandial effects of hazelnut-enriched high fat meal on LDL oxidative status, oxidative and inflammatory gene expression of healthy subjects: A randomized trial. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1610–1626. [Google Scholar]
- De Lorenzo, A.; Bernardini, S.; Gualtieri, P.; Cabibbo, A.; Perrone, M.A.; Giambini, I.; Di Renzo, L. Mediterranean meal versus Western meal effects on postprandial ox-LDL, oxidative and inflammatory gene expression in healthy subjects: A randomized controlled trial for nutrigenomic approach in cardiometabolic risk. Acta Diabetol. 2017, 54, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Giraldi, L.; Miele, L.; Aleksovska, K.; Manca, F.; Leoncini, E.; Biolato, M.; Arzani, D.; Pirro, M.A.; Marrone, G.; Cefalo, C.; et al. Mediterranean diet and the prevention of non-alcoholic fatty liver disease: Results from a case-control study. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 7391–7398. [Google Scholar] [PubMed]
- Perrone, M.A.; Gualtieri, P.; Gratteri, S.; Ali, W.; Sergi, D.; Muscoli, S.; Cammarano, A.; Bernardini, S.; Di Renzo, L.; Romeo, F. Effects of postprandial hydroxytyrosol and derivates on oxidation of LDL, cardiometabolic state and gene expression: A nutrigenomic approach for cardiovascular prevention. J. Cardiovasc. Med. 2019, 20, 419–426. [Google Scholar] [CrossRef]
- Mitra, S.; Goyal, T.; Mehta, J.L.; Oxidized, L.D.L. LOX-1 and atherosclerosis. Cardiovasc. Drugs Ther. 2011, 25, 419–429. [Google Scholar] [CrossRef]
- Fitó, M.; Guxens, M.; Corella, D.; Sáez, G.; Estruch, R.; de la Torre, R.; Francés, F.; Cabezas, C.; López-Sabater, M.D.C.; Marrugat, J.; et al. PREDIMED Study Investigators. Arch. Intern. Med. 2007, 167, 1195–1203. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. ESC Scientific Document Group. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2020, 41, 111–188. [Google Scholar] [CrossRef] [PubMed]
- Navarese, E.P.; Robinson, J.G.; Kowalewski, M.; Kolodziejczak, M.; Andreotti, F.; Bliden, K.; Tantry, U.; Kubica, J.; Raggi, P.; Gurbel, P.A. Association Between Baseline LDL-C Level and Total and Cardiovascular Mortality After LDL-C Lowering: A Systematic Review and Meta-analysis. J. Am. Med. Assoc. 2018, 319, 1566–1579. [Google Scholar] [CrossRef] [PubMed]
- Romano, L.; Marchetti, M.; Gualtieri, P.; Di Renzo, L.; Belcastro, M.; De Santis, G.L.; Perrone, M.A.; De Lorenzo, A. Effects of a Personalized VLCKD on Body Composition and Resting Energy Expenditure in the Reversal of Diabetes to Prevent Complications. Nutrients 2019, 11, 1526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravnskov, U.; Diamond, D.M.; Hama, R.; Hamazaki, T.; Hammarskjöld, B.; Hynes, N.; Kendrick, M.; Langsjoen, P.H.; Malhotra, A.; Mascitelli, L.; et al. Lack of an association or an inverse association between low-density-lipoprotein cholesterol and mortality in the elderly: A systematic review. Br. Med. J. Open 2016, 6, e010401. [Google Scholar] [CrossRef]
- Clifton, P.M. Diet, exercise and weight loss and dyslipidaemia. Pathology 2019, 51, 222–226. [Google Scholar] [CrossRef]
- Mann, S.; Beedie, C.; Jimenez, A. Differential effects of aerobic exercise, resistance training and combined exercise modalities on cholesterol and the lipid profile: Review, synthesis and recommendations. Sports Med. 2014, 44, 211–221. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Forouzesh, M.; Rahimi, A.; Valizadeh, R.; Dadashzadeh, N.; Mirzazadeh, A. Clinical display, diagnostics and genetic implication of novel Coronavirus (COVID-19) epidemic. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 4607–4615. [Google Scholar]
- Le Roux, E.; De Jong, N.P.; Blanc, S.; Simon, C.; Bessesen, D.H.; Bergouignan, A. Physiology of physical inactivity, sedentary behaviors and non-exercise activity: Insights from space bedrest model. J. Physiol. 2021. [Google Scholar] [CrossRef]
- Hussain, A.; Mahawar, K.; Xia, Z.; Yang, W.; El-Hasani, S. Obesity and mortality of COVID-19. Meta-analysis. Obes. Res. Clin. Pract. 2020, 14, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Sung, Y.-C.; Liao, Y.-H.; Chen, C.-Y.; Chen, Y.-L.; Chou, C.-C. Acute changes in blood lipid profiles and metabolic risk factors in collegiate elite taekwondo athletes after short-term de-training: A prospective insight for athletic health management. Lipids Health Dis. 2017, 16, 143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goethals, L.; Barth, N.; Guyot, J.; Hupin, D.; Celarier, T.; Bongue, B. Impact of Home Quarantine on Physical Activity Among Older Adults Living at Home During the COVID-19 Pandemic: Qualitative Interview Study. J. Med. Internet Res. Aging 2020, 3, e19007. [Google Scholar] [CrossRef]
- Martinez-Ferran, M.; de la Guía-Galipienso, F.; Sanchis-Gomar, F.; Pareja-Galeano, H. Metabolic Impacts of Confinement during the COVID-19 Pandemic Due to Modified Diet and Physical Activity Habits. Nutrients 2020, 12, 1549. [Google Scholar] [CrossRef]
- Carvalho, V.O.; Gois, C.O. COVID-19 pandemic and home-based physical activity. J. Allergy Clin. Immunol. Pract. 2020, 8, 2833–2834. [Google Scholar] [CrossRef]
- Mann, D.; Chen, J.; Chunara, R.; Testa, P.; Nov, O. COVID-19 transforms health care through telemedicine: Evidence from the field. J. Am. Med. Inform. Assoc. 2020, 27, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
- Casasco, M.; Pigozzi, F.; Porcelli, S.; Beltrami, G.; Agnello, L.; Parisi, A.; Pecci, C.; Pecorelli, G.; Scorcu, M.; Pecorelli, S. FMSI guidelines for return to physical activity after coronavirus pandemics lockdown. Med. Sport 2020, 73, 167–175. [Google Scholar] [CrossRef]
- Calabrese, M.; Garofano, M.; Palumbo, R.; Di Pietro, P.; Izzo, C.; Damato, A.; Venturini, E.; Iesu, S.; Virtuoso, N.; Strianese, A.; et al. Exercise Training and Cardiac Rehabilitation in COVID-19 Patients with Cardiovascular Complications: State of Art. Life 2021, 11, 259. [Google Scholar] [CrossRef] [PubMed]
- Mattioli, A.V.; Sciomer, S.; Cocchi, C.; Maffei, S.; Gallina, S. Quarantine during COVID-19 outbreak: Changes in diet and physical activity increase the risk of cardiovascular disease. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 1409–1417. [Google Scholar] [CrossRef] [PubMed]
- Clerico, A.; Masotti, S.; Musetti, V.; Ripoli, A.; Aloe, R.; Di Pietro, M.; Rizzardi, S.; Dittadi, R.; Carrozza, C.; Belloni, L.; et al. Evaluation of 99th percentile and reference change values of the hs-cTnI method using ADVIA Centaur XPT platform: A multicenter study. Clin. Chim. Acta 2019, 495, 161–166. [Google Scholar] [CrossRef]
- Pinto, A.J.; Dunstan, D.W.; Owen, N.; Bonfá, E.; Gualano, B. Combating physical inactivity during the COVID-19 pandemic. Nat. Rev. Rheumatol. 2020, 16, 347–348. [Google Scholar] [CrossRef]
- Shurlock, J.; Muniz-Pardos, B.; Tucker, R.; Norbert Bachl, N.; Papadopoulou, T.; Holloway, G.; Jones, N.; Bigard, X.; Vonbank, K.; Niederseer, D.; et al. Recommendations for Face Coverings While Exercising During the COVID-19 Pandemic. Sports Med. Open 2021, 7, 19. [Google Scholar] [CrossRef]
- Müller-Riemenschneider, F.; Petrunoff, N.; Sia, A.; Ramiah, A.; Ng, A.; Han, J.; Wong, M.; Bee Choo, T.; Uijtdewilligen, L. Prescribing Physical Activity in Parks to Improve Health and Wellbeing: Protocol of the Park Prescription Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2018, 15, 1154. [Google Scholar] [CrossRef] [Green Version]
- Kim, K.; Choi, S.; Hwang, S.E.; Son, J.S.; Lee, J.K.; Oh, J.; Park, S.M. Changes in exercise frequency and cardiovascular outcomes in older adults. Eur. Heart J. 2020, 41, 1490–1499. [Google Scholar] [CrossRef] [Green Version]
| Age (years) | 54,6 ± 5,1 |
| Weight (kg) | 74,9 ± 6,3 |
| Height (cm) | 166,9 ± 5,0 |
| BMI (kg/m2) | 26,9 ± 2,1 |
| Dyslipidemia (%) | 100 |
| Hypertension (%) | 57,1 |
| Diabetes (%) | 14,3 |
| Cigarette smoke (%) | 28,6 |
| Sex (M:F) | 9:5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perrone, M.A.; Feola, A.; Pieri, M.; Donatucci, B.; Salimei, C.; Lombardo, M.; Perrone, A.; Parisi, A. The Effects of Reduced Physical Activity on the Lipid Profile in Patients with High Cardiovascular Risk during COVID-19 Lockdown. Int. J. Environ. Res. Public Health 2021, 18, 8858. https://doi.org/10.3390/ijerph18168858
Perrone MA, Feola A, Pieri M, Donatucci B, Salimei C, Lombardo M, Perrone A, Parisi A. The Effects of Reduced Physical Activity on the Lipid Profile in Patients with High Cardiovascular Risk during COVID-19 Lockdown. International Journal of Environmental Research and Public Health. 2021; 18(16):8858. https://doi.org/10.3390/ijerph18168858
Chicago/Turabian StylePerrone, Marco Alfonso, Alessandro Feola, Massimo Pieri, Bruno Donatucci, Chiara Salimei, Mauro Lombardo, Andrea Perrone, and Attilio Parisi. 2021. "The Effects of Reduced Physical Activity on the Lipid Profile in Patients with High Cardiovascular Risk during COVID-19 Lockdown" International Journal of Environmental Research and Public Health 18, no. 16: 8858. https://doi.org/10.3390/ijerph18168858
APA StylePerrone, M. A., Feola, A., Pieri, M., Donatucci, B., Salimei, C., Lombardo, M., Perrone, A., & Parisi, A. (2021). The Effects of Reduced Physical Activity on the Lipid Profile in Patients with High Cardiovascular Risk during COVID-19 Lockdown. International Journal of Environmental Research and Public Health, 18(16), 8858. https://doi.org/10.3390/ijerph18168858











