The Pandemic beyond the Pandemic: A Scoping Review on the Social Relationships between COVID-19 and Antimicrobial Resistance
Abstract
:1. Introduction
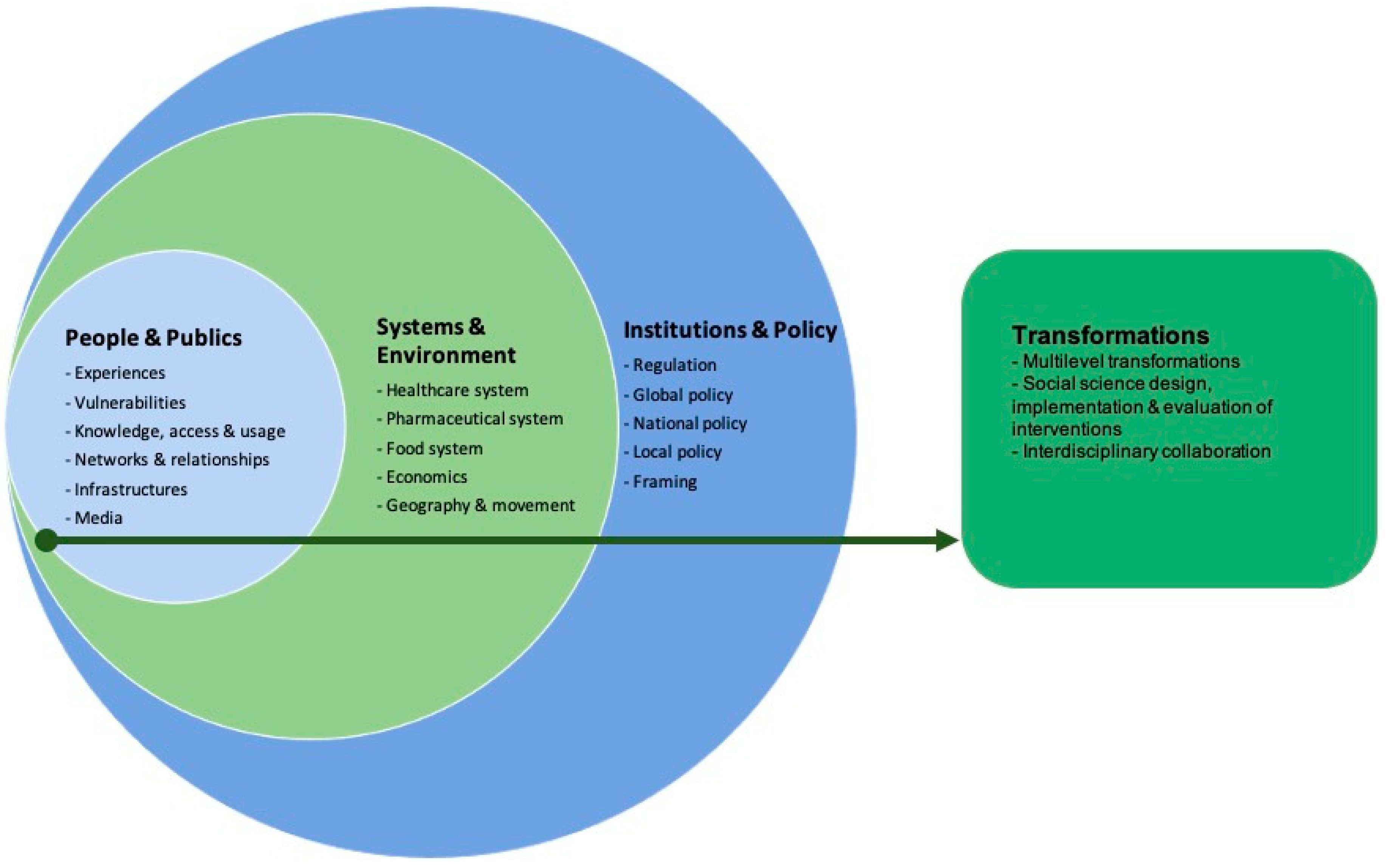
- People and publics: portraying prescription practices, knowledge about antibiotics, behavioural practices of consumption and access to antibiotics, its impact on vulnerable groups, the social networks and relationships (including user-prescriber relationship), and the role of media on the prescribing and consumption of antibiotics during the pandemic.
- Systems and environments: framing the dynamics and interactions between AMR and the healthcare, pharmaceutical and food systems. It also includes the economic impact and the role of geography and movement on AMR.
- Institutions and policies: exploring the AMR policy-making from the local (hospital stewardship) to the national and the global level.
- Transformations: integrating the mitigation strategies advised and developed to tackle AMR during pandemic times, the social sciences’ role, and the resulting interventions. These transformations also cover the collaboration between social scientists and non-social scientists.
- What evidence is available in peer-reviewed research that addresses the social dimensions of the relationship between AMR and COVID-19?
- What actions are suggested and generated to deal with these social dimensions?
- What is the role of the social sciences as a transformation agent during the COVID-19 pandemic?
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
- The article addresses both topics (AMR and COVID-19) throughout all its content;
- The article is published in a peer-review journal, including commentaries, review articles, editorials, and viewpoints;
- The content of the article includes a social sciences perspective.
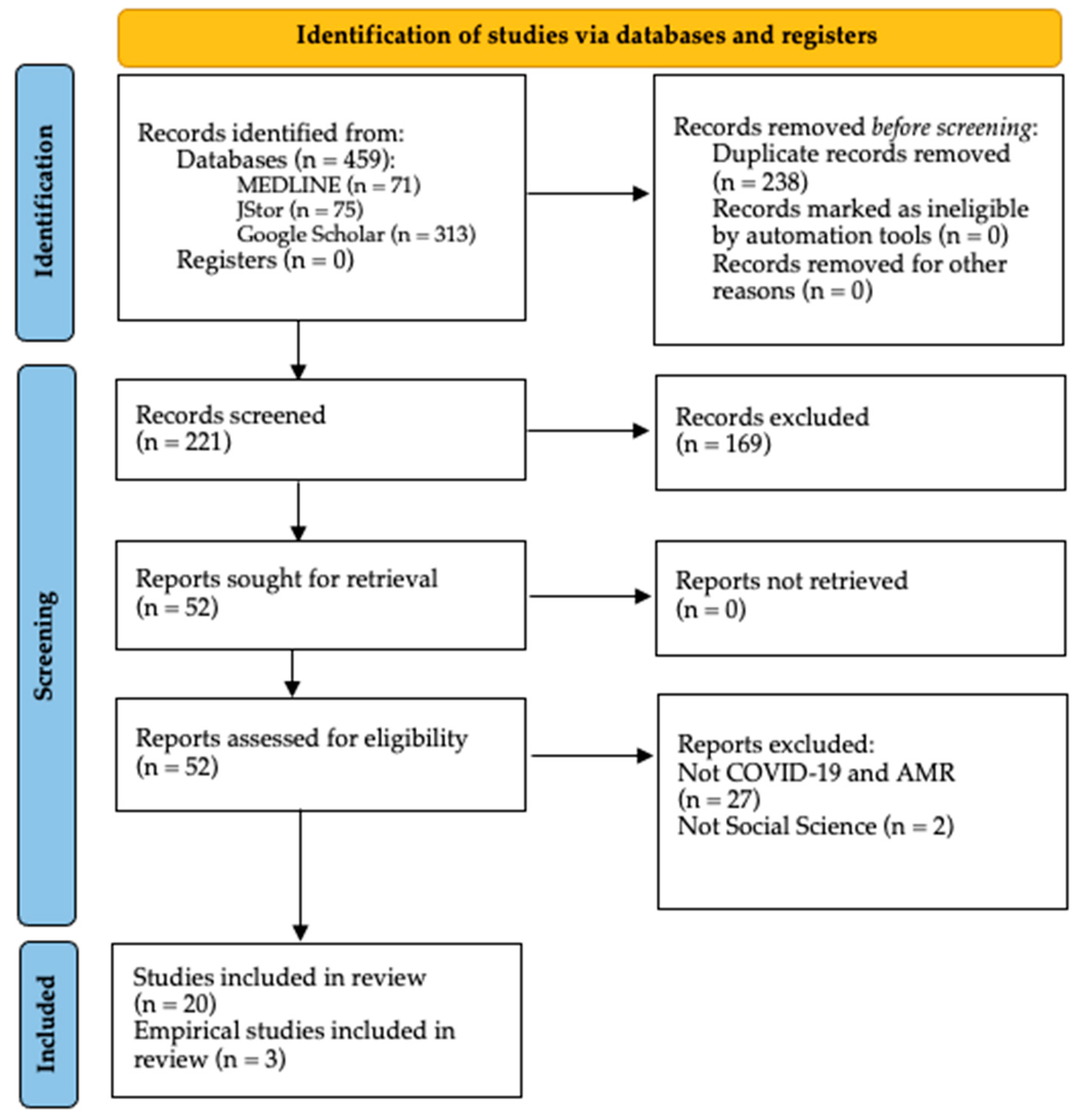
2.3. Study Selection
3. Results
3.1. People and Publics
3.1.1. Antibiotic Prescription Behaviour, Knowledge and Access
3.1.2. Vulnerable Populations
3.1.3. Social Relationships and Networks
3.1.4. Media and Misinformation
3.2. Systems and Environments
3.2.1. Healthcare System
3.2.2. Pharmaceutical System
3.2.3. Economics
3.2.4. Environment
3.3. Institutions and Policies
3.3.1. Local Policies (Hospital Stewardship Programmes)
3.3.2. National and Global Policies
3.4. Transformations
3.4.1. Social Engagement and Sensitisation
3.4.2. Misinformation Control
3.4.3. Health Systems Strengthening
3.4.4. Infection Prevention and Control
3.4.5. Environmental Protection
3.4.6. Antimicrobial Stewardship
3.4.7. AMR and Infectious Disease Governance
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Author | Social Engagement and Sensitisation (Awareness, Behaviour Change) | Misinformation Control | Health Systems Strengthening | Infection Prevention and Control Measures | Environmental Protection | AMR Surveillance and Antimicrobial Stewardship Programmes | AMR and Infectious Diseases Governance During COVID-19 Pandemic |
|---|---|---|---|---|---|---|---|
| Rawson, Ming, et al., 2020 [12] | Evaluate the impact of the COVID-19 pandemic on antimicrobial use, antimicrobial resistance and access to effective antimicrobial treatments. Keep and promote routine surveillance and AMS principles on AMR during COVID-19 times. Strengthen and prioritise antimicrobial stewardship programmes during pandemic times. | Review national policies that do not neglect essential public health programmes in TB and immunisation delivery. | |||||
| Usman, Farooq and Hanna, 2020 [19] | Educate the public about the unwanted effects of antimicrobial/antibacterial products during the pandemic. | AMS should continue to be applied and promoted during COVID-19 times. | Development of an antimicrobial policy specific for COVID-19, with coordinated strategies at the individual, healthcare and policy levels. | ||||
| Strathdee, Davies and Marcelin, 2020 [28] | Leverage infection control principles from COVID-19 experience to control AMR. | Prioritise antimicrobial stewardship programmes during the pandemic. | |||||
| Khor et al., 2020 [21] | Patient education on the appropriate use of antimicrobials and the lack of evidence that antibiotics can be used as a treatment for viral infections, including COVID-19. | Adherence to guidelines recommendations to prevent over- and inappropriate prescribing of antimicrobials during the pandemic. | |||||
| Iwu et al., 2020 [31] | Integrate antimicrobial stewardship into the pandemic response will help to minimise the emergence of AMR during the pandemic. Local guidelines should incorporate the WHO guidance on the use of antimicrobials in the treatment of COVID-19. | ||||||
| Getahun et al., 2020 [24] | Targeted training to increase clinical competence among health workers treating COVID-19 patients. Ensure the continuity of essential health services and regular supply of antimicrobials, including retroviral and tuberculosis drugs and vaccines. | Prioritise biocidal agents without or with a low selection pressure for AMR. | |||||
| Hsu, 2020 [17] | Collect data on how healthcare responses to the pandemic may be affecting AMR. | ||||||
| Chibabhai et al., 2020 [25] | Development of COVID-19 management protocols by AMS teams Continue AMS activities in non-COVID-19 sections of healthcare facilities. | ||||||
| Arshad et al., 2020 [18] | Development of digital platforms to correct antimicrobial misinformation showing the ineffectiveness of antimicrobials as a treatment for COVID-19 infection. | ||||||
| Wilson et al., 2020 [7] | Prepare and strengthen health systems to the rising burden of AMR after pandemic by strength health systems through investments in capacity building, adequate training for healthcare personnel, adequate supply of antimicrobials and PPE. | ||||||
| Miranda et al., 2020 [32] | Increase societal sensitisation towards infectious diseases and good sanitary practices during the pandemic to diminish the potential impact on rates and transmission of AMR. | ||||||
| Nieuwlaat et al., 2020 [9] | The behavioural changes implemented to deal with the COVID-19 pandemic would also be beneficial in dealing with AMR, as both face similar paths. | ||||||
| Yam, 2020 [22] | A globally coordinated establishment of a framework of governance, surveillance and reporting of AMR to deal with AMR during and after COVID-19. | ||||||
| Monnet and Harbarth, 2020 [11] | Compliance with IPC measures is essential for controlling the spread of COVID-19 infections and AMR bacteria, as well. | ||||||
| Heydargoy, 2020 [20] | Expand virtual consultations systems with reduced cost to decrease antibiotic use in self-medication practices in people who cannot leave home because of the pandemic. | ||||||
| Zhu et al., 2021 [13] | Monitoring of consultations, antibiotics prescribing and AMR should continue during and beyond the COVID-19 pandemic to determine the long-term impact on prescribing behaviour among clinicians. | ||||||
| Ashiru-Oredope et al., 2021 [8] | Increased awareness of antimicrobial guidelines and improvements on infection prevention and control. | Technology as a facilitator for AMS activities. Better use of technology (virtual platforms and remote working). |
References
- De Vries, D.; Hofstraat, K.; Spaan, V. COVID-19 as analogy for antimicrobial resistance. Available online: http://somatosphere.net/2020/covid-19-analogy-amr.html/?format=pdf (accessed on 8 March 2021).
- Frid-Nielsen, S.S.; Rubin, O.; Baekkeskov, E. The State of Social Science Research on Antimicrobial Resistance. Soc. Sci. Med. 2019, 242, 112596. [Google Scholar] [CrossRef]
- Arksey, H.; O’Malley, L. Scoping Studies: Towards a Methodological Framework. Int. J. Soc. Res. Methodol. Theory Pract. 2005, 8, 19–32. [Google Scholar] [CrossRef] [Green Version]
- De Vries, D.; Hofstraat, K.; Begemann, S.; Chandler, C.; Fortané, N.; Lambert, H.; Munkholm, L.; Rodrigues, C.; Schultz, C. SPECIAL SOC AMR Curriculum: Training Social Scientists on Social Dimensions of AMR; AIGHD: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Léger, A.; Lambraki, I.; Graells, T.; Cousins, M.; Henriksson, P.J.G.; Harbarth, S.; Carson, C.; Majowicz, S.; Troell, M.; Parmley, E.J.; et al. AMR-Intervene: A Social-Ecological Framework to Capture the Diversity of Actions to Tackle Antimicrobial Resistance from a One Health Perspective. J. Antimicrob. Chemother. 2021, 76, 1–21. [Google Scholar] [CrossRef]
- Noyes, N.R.; Slizovskiy, I.B.; Singer, R.S. Beyond Antimicrobial Use: A Framework for Prioritizing Antimicrobial Resistance Interventions. Annu. Rev. Animal Biosci. 2021, 9, 313–332. [Google Scholar] [CrossRef]
- Wilson, L.A.; Rogers Van Katwyk, S.; Fafard, P.; Viens, A.M.; Hoffman, S.J. Lessons Learned from COVID-19 for the Post-Antibiotic Future. Glob. Health 2020, 16, 10–12. [Google Scholar] [CrossRef]
- Ashiru-Oredope, D.; Kerr, F.; Hughes, S.; Urch, J.; Lanzman, M.; Yau, T.; Cockburn, A.; Patel, R.; Sheikh, A.; Gormley, C.; et al. Assessing the Impact of COVID-19 on Antimicrobial Stewardship Activities/Programs in the United Kingdom. Antibiotics 2021, 10, 110. [Google Scholar] [CrossRef]
- Nieuwlaat, R.; Mbuagbaw, L.; Mertz, D.; Burrows, L.L.; Bowdish, D.M.E.; Moja, L.; Wright, G.D.; Schünemann, H.J. Coronavirus Disease 2019 and Antimicrobial Resistance: Parallel and Interacting Health Emergencies. Clin. Infect. Dis. 2020, 2, 2019–2021. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Álvarez, M.; López-Vidal, Y.; Soto-Hernández, J.L.; Miranda-Novales, M.G.; Flores-Moreno, K.; Ponce de León-Rosales, S. COVID-19: Clouds Over the Antimicrobial Resistance Landscape. Arch. Med. Res. 2021, 52, 123–126. [Google Scholar] [CrossRef]
- Monnet, D.L.; Harbarth, S. Will Coronavirus Disease (COVID-19) Have an Impact on Antimicrobial Resistance? Eurosurveillance 2020, 25, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Rawson, T.M.; Moore, L.S.P.; Castro-Sanchez, E.; Charani, E.; Davies, F.; Satta, G.; Ellington, M.J.; Holmes, A.H. COVID-19 and the Potential Long-Term Impact on Antimicrobial Resistance. J. Antimicrob. Chemother. 2020, 75, 1681–1684. [Google Scholar] [CrossRef] [PubMed]
- Zhu, N.J.; McLeod, M.; McNulty, C.A.M.; Lecky, D.M.; Holmes, A.H.; Ahmad, R. Trends in Antibiotic Prescribing in Out-of-Hours Primary Care in England from January 2016 to June 2020 to Understand Behaviours during the Firstwave of COVID-19. Antibiotics 2021, 10, 32. [Google Scholar] [CrossRef]
- Abelenda-Alonso, G.; Padullés, A.; Rombauts, A.; Gudiol, C.; Pujol, M.; Alvarez-Pouso, C.; Jodar, R.; Carratalà, J. Antibiotic Prescription during the COVID-19 Pandemic: A Biphasic Pattern. Infect. Control. Hosp. Epidemiol. 2020, 41, 1371–1372. [Google Scholar] [CrossRef] [PubMed]
- Rawson, T.M.; Ming, D.; Ahmad, R.; Moore, L.S.P.; Holmes, A.H. Antimicrobial Use, Drug-Resistant Infections and COVID-19. Nat. Rev. Microbiol. 2020, 18, 409–410. [Google Scholar] [CrossRef] [PubMed]
- Pulia, M.S.; Wolf, I.; Schulz, L.T.; Pop-Vicas, A.; Schwei, R.J.; Lindenauer, P.K. COVID-19: An Emerging Threat to Antibiotic Stewardship in the Emergency Department. West. J. Emerg. Med. 2020, 21, 1283–1286. [Google Scholar] [CrossRef]
- Hsu, J. How COVID-19 Is Accelerating the Threat of Antimicrobial Resistance. BMJ 2020, 369, 18–19. [Google Scholar] [CrossRef]
- Arshad, M.; Mahmood, S.F.; Khan, M.; Hasan, R. COVID-19, Misinformation, and Antimicrobial Resistance. BMJ 2020, 371, 1–2. [Google Scholar] [CrossRef]
- Usman, M.; Farooq, M.; Hanna, K. Environmental Side Effects of the Injudicious Use of Antimicrobials in the Era of COVID-19. Sci. Total Environ. 2020, 745, 141053. [Google Scholar] [CrossRef]
- Heydargoy, M.H. The Effect of the Prevalence of COVID-19 on Arbitrary Use of Antibiotics. Iran. J. Med. Microbiol. 2020, 14, 374–378. [Google Scholar]
- Khor, W.P.; Olaoye, O.; D’arcy, N.; Krockow, E.M.; Elshenawy, R.A.; Rutter, V.; Ashiru-Oredope, D. The Need for Ongoing Antimicrobial Stewardship during the COVID-19 Pandemic and Actionable Recommendations. Antibiotics 2020, 9, 904. [Google Scholar] [CrossRef]
- Yam, E.L.Y. COVID-19 Will Further Exacerbate Global Antimicrobial Resistance. J. Travel Med. 2020, 27, 1–2. [Google Scholar] [CrossRef]
- Murray, A.K. The Novel Coronavirus COVID-19 Outbreak: Global Implications for Antimicrobial Resistance. Front. Microbiol. 2020, 11, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Getahun, H.; Smith, I.; Trivedi, K.; Paulin, S.; Balkhy, H.H. Tackling Antimicrobial Resistance in the COVID-19 Pandemic. Bull. World Health Organ. 2020, 98, 19–20. [Google Scholar] [CrossRef] [PubMed]
- Chibabhai, V.; Duse, A.G.; Perovic, O.; Richards, G.A. Collateral Damage of the COVID-19 Pandemic: Exacerbation of Antimicrobial Resistance and Disruptions to Antimicrobial Stewardship Programmes? S. Afr. Med. J. 2020, 110, 572–573. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Impact of the COVID-19 Pandemic on TB Detection and Mortality in 2020; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- Silver, L.L. Challenges of Antibacterial Discovery. Clin. Microbiol. Rev. 2011, 24, 71–109. [Google Scholar] [CrossRef] [Green Version]
- Strathdee, S.A.; Davies, S.C.; Marcelin, J.R. Confronting Antimicrobial Resistance beyond the COVID-19 Pandemic and the 2020 US Election. Lancet 2020, 396, 1050–1053. [Google Scholar] [CrossRef]
- Ukuhor, H.O. The Interrelationships between Antimicrobial Resistance, COVID-19, Past, and Future Pandemics. J. Infect. Public Health 2021, 14, 53–60. [Google Scholar] [CrossRef]
- Comber, S.D.W.; Upton, M.; Lewin, S.; Powell, N.; Hutchinson, T.H. COVID-19, Antibiotics and One Health: A UK Environmental Risk Assessment. J. Antimicrob. Chemother. 2020, 75, 3411–3412. [Google Scholar] [CrossRef]
- Iwu, C.J.; Jordan, P.; Jaja, I.F.; Iwu, C.D.; Wiysonge, C.S. Treatment of COVID-19: Implications for Antimicrobial Resistance in Africa. Pan Afr. Med. J. 2020, 35 (Suppl. S2), 119. [Google Scholar] [CrossRef]
- Miranda, C.; Silva, V.; Capita, R.; Alonso-Calleja, C.; Igrejas, G.; Poeta, P. Implications of Antibiotics Use during the COVID-19 Pandemic: Present and Future. J. Antimicrob. Chemother. 2020, 75, 3413–3416. [Google Scholar] [CrossRef]
- Lynch, C.; Mahida, N.; Gray, J. Antimicrobial Stewardship: A COVID Casualty? J. Hosp. Infect. 2020, 106, 401–403. [Google Scholar] [CrossRef]
- Knight, G.M.; Glover, R.E.; McQuaid, C.F.; Olaru, I.D.; Gallandat, K.; Leclerc, Q.J.; Fuller, N.M.; Willcocks, S.J.; Hasan, R.; van Kleef, E.; et al. Antimicrobial Resistance and COVID-19: Intersections and Implications. eLife 2021, 10, 1–27. [Google Scholar] [CrossRef]
- Rodríguez-Baño, J.; Rossolini, G.M.; Schultsz, C.; Tacconelli, E.; Murthy, S.; Ohmagari, N.; Holmes, A.; Bachmann, T.; Goossens, H.; Canton, R.; et al. Key Considerations on the Potential Impacts of the COVID-19 Pandemic on Antimicrobial Resistance Research and Surveillance. Trans. R. Soc. Trop. Med. Hyg. 2021, 1–8. [Google Scholar] [CrossRef]
- Du Yan, L.; Dean, K.; Park, D.; Thompson, J.; Tong, I.; Liu, C.; Hamdy, R.F. Education vs Clinician Feedback on Antibiotic Prescriptions for Acute Respiratory Infections in Telemedicine: A Randomized Controlled Trial. J. Gen. Intern. Med. 2021, 36, 305–312. [Google Scholar] [CrossRef]
- Hoffman, L. Shedding Light on Telemedicine & Online Prescribing: The Need to Balance Access to Health Care and Quality of Care. Am. J. Law Med. 2020, 46, 237–251. [Google Scholar] [CrossRef]
- Torres, T.S.; Hoagland, B.; Bezerra, D.R.B.; Garner, A.; Jalil, E.M.; Coelho, L.E.; Benedetti, M.; Pimenta, C.; Grinsztejn, B.; Veloso, V.G. Impact of COVID-19 Pandemic on Sexual Minority Populations in Brazil: An Analysis of Social/Racial Disparities in Maintaining Social Distancing and a Description of Sexual Behavior. AIDS Behav. 2021, 25, 73–84. [Google Scholar] [CrossRef]
- Karaye, I.M.; Horney, J.A. The Impact of Social Vulnerability on COVID-19 in the U.S.: An Analysis of Spatially Varying Relationships. Am. J. Prev. Med. 2020, 59, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Gaynor, T.S.; Wilson, M.E. Social Vulnerability and Equity: The Disproportionate Impact of COVID-19. Public Adm. Rev. 2020, 80, 832–838. [Google Scholar] [CrossRef]
- Freitas, C.M.D.; Silva, I.V.D.M.; Cidade, N.D.C. COVID-19 AS A GLOBAL DISASTER: Challenges to Risk Governance and Social Vulnerability in Brazil. Ambiente Soc. 2020, 23, 1–12. [Google Scholar] [CrossRef]
- Person, B.; Sy, F.; Holton, K.; Govert, B.; Liang, A.; The NCID, SARS Community Outreach Team. Fear and Stigma: The Epidemic within the SARS Outbreak. Emerg. Infect. Dis. 2004, 10, 358–363. [Google Scholar] [CrossRef]
- Eaton, L.A.; Kalichman, S.C. Social and Behavioral Health Responses to COVID-19: Lessons Learned from Four Decades of an HIV Pandemic. J. Behav. Med. 2020, 43, 341–345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubin, H.; Saidel, N. Global Governance Structure for Infectious Disease: An Enforceable Strategy. Orbis 2016, 60, 279–295. [Google Scholar] [CrossRef]
| Framework Level | Framework Sublevel | Results |
|---|---|---|
| People and Publics | Antibiotic prescription behaviour, knowledge and access |
|
| Vulnerable populations |
| |
| Social relationships and networks |
| |
| Media and misinformation |
| |
| Systems and Environments | Healthcare system |
|
| Pharmaceutical system |
| |
| Economics |
| |
| Environment |
| |
| Institutions and Policies | Local, National and Global policies |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toro-Alzate, L.; Hofstraat, K.; de Vries, D.H. The Pandemic beyond the Pandemic: A Scoping Review on the Social Relationships between COVID-19 and Antimicrobial Resistance. Int. J. Environ. Res. Public Health 2021, 18, 8766. https://doi.org/10.3390/ijerph18168766
Toro-Alzate L, Hofstraat K, de Vries DH. The Pandemic beyond the Pandemic: A Scoping Review on the Social Relationships between COVID-19 and Antimicrobial Resistance. International Journal of Environmental Research and Public Health. 2021; 18(16):8766. https://doi.org/10.3390/ijerph18168766
Chicago/Turabian StyleToro-Alzate, Luisa, Karlijn Hofstraat, and Daniel H. de Vries. 2021. "The Pandemic beyond the Pandemic: A Scoping Review on the Social Relationships between COVID-19 and Antimicrobial Resistance" International Journal of Environmental Research and Public Health 18, no. 16: 8766. https://doi.org/10.3390/ijerph18168766
APA StyleToro-Alzate, L., Hofstraat, K., & de Vries, D. H. (2021). The Pandemic beyond the Pandemic: A Scoping Review on the Social Relationships between COVID-19 and Antimicrobial Resistance. International Journal of Environmental Research and Public Health, 18(16), 8766. https://doi.org/10.3390/ijerph18168766





