Differences in Incidence of Acute Viral Hepatitis between Foreigners and Autochthonous Population in Italy
Abstract
:1. Introduction
2. Materials and Methods
- acute hepatitis A: positive hepatitis A virus (HAV) specific IgM antibodies, regardless of other viral markers;
- acute hepatitis B: positive IgM anti-HBc and negative IgM anti-HAV, regardless of other viral markers;
- acute hepatitis C: negative IgM anti-HAV and IgM anti-HBc and positive anti-HCV or HCV-RNA;
- acute nonA-nonC: negative IgM anti-HAV, IgM anti-HBc, anti-HCV/HCV-RNA; IgM anti-HDV (hepatitis Delta virus) and IgM anti-HEV assays were used to further discriminate within nonA-nonC hepatitis cases;
- Unknown: a differential diagnosis was not possible due to missing/inconsistent data on hepatitis markers.
3. Results
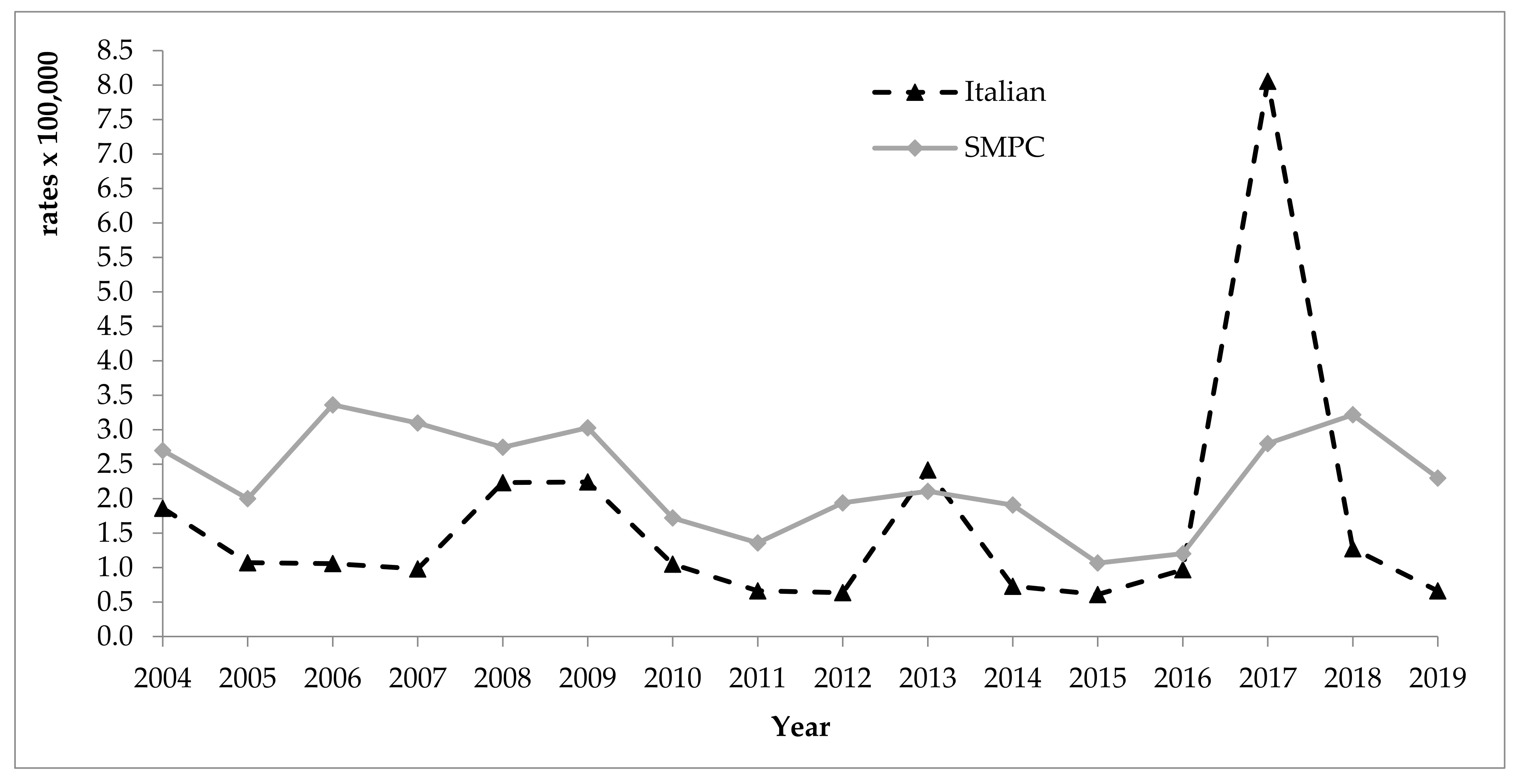
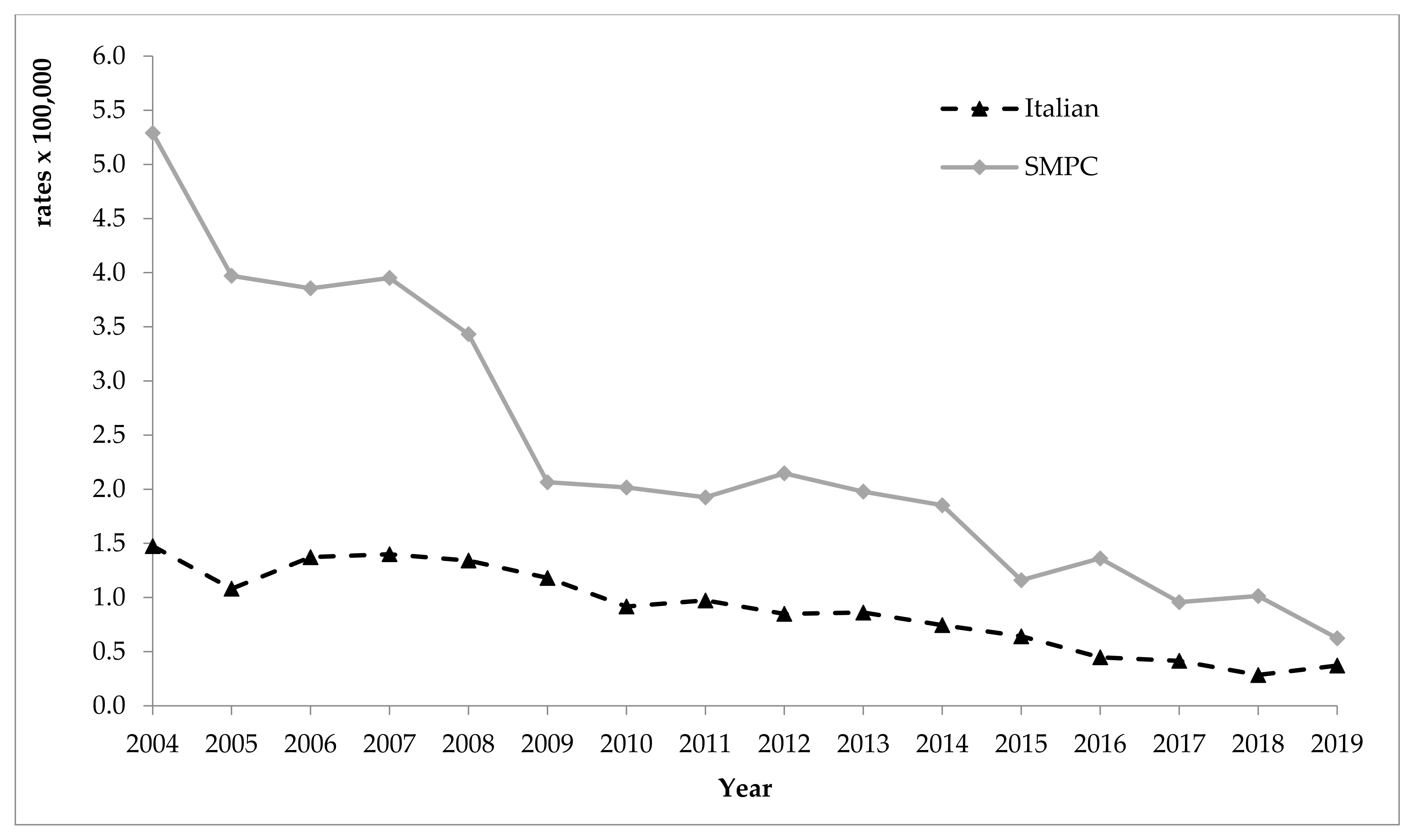
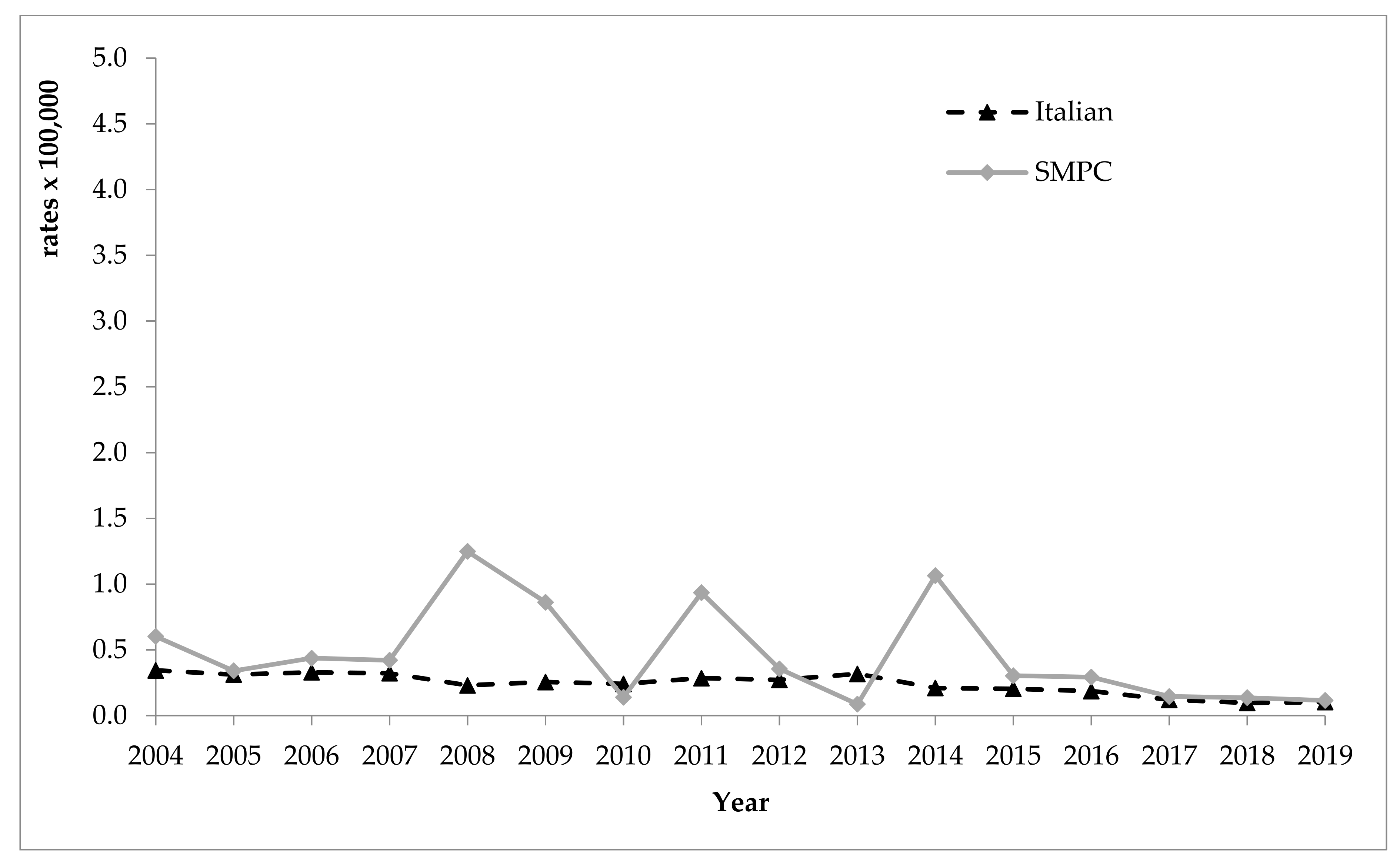
3.1. Hepatitis A
3.2. Hepatitis B
3.3. Hepatitis C
3.4. NonA-nonC and of Unknown Origin Hepatitis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- IOM International Organization for Migration—UN Migration. World Migration Report 2020; IOM International Organization for Migration—UN Migration: Geneva, Switzerland, 2019. [Google Scholar]
- Greenaway, C.; Castelli, F. Infectious diseases at different stages of migration: An expert review. J. Travel Med. 2019, 26, 1–10. [Google Scholar] [CrossRef]
- UNHCR—The UN Refugee Agency. Figure at a Glance 2020. Available online: https://www.unhcr.org/figures-at-a-glance.html (accessed on 14 June 2021).
- ISTAT. Censimento Permanente Della Popolazione e Delle Abitazioni and Infografica Sugli Stranieri in Italia. 2020. Available online: https://www.istat.it/it/archivio/251687 (accessed on 14 June 2021).
- Fondazione ISMU. Venticinquesimo Rapporto Sulle Migrazioni 2019; Fondazione ISMU: Milano, Italy, 2020. [Google Scholar]
- Centro Studi e Ricerche IDOS. Dossier Statistico Immigrazione 2020; Centro Studi e Ricerche IDOS: Roma, Italy, 2020. [Google Scholar]
- Jefferies, M.; Rauff, B.; Rashid, H.; Lam, T.; Rafiq, S. Update on global epidemiology of viral hepatitis and preventive strategies. World J. Clin. Cases 2018, 6, 589–599. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Epidemiological Assessment of Hepatitis B and C among Migrants in the EU/EEA; ECDC: Stockholm, Sweden, 2016. [Google Scholar]
- World Health Organzation Europe. Report on the Health of Refugees and Migrants in the WHO European Region: No Public Health without Refugee and Migrant Health; World Health Organzation Europe: Geneva, Switzerland, 2018. [Google Scholar]
- McNaughton, A.L.; Lourenço, J.; Bester, P.A.; Mokaya, J.; Lumley, S.F.; Obolski, U.; Forde, D.; Maponga, T.G.; Katumba, K.R.; Goedhals, D.; et al. Hepatitis B virus seroepidemiology data for Africa: Modelling intervention strategies based on a systematic review and meta-analysis. PLoS Med. 2020, 17, e1003068. [Google Scholar] [CrossRef] [Green Version]
- Coppola, N.; Alessio, L.; Gualdieri, L.; Pisaturo, M.; Sagnelli, C.; Minichini, C.; Di Caprio, G.; Starace, M.; Onorato, L.; Signoriello, G.; et al. Hepatitis B virus infection in undocumented immigrants and refugees in Southern Italy: Demographic, virological, and clinical features. Infect Dis. Poverty 2017, 6, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cuomo, G.; Franconi, I.; Riva, N.; Bianchi, A.; Digaetano, M.; Santoro, A.; Codeluppi, M.; Bedini, A.; Guaraldi, G.; Mussini, C. Migration and health: A retrospective study about the prevalence of HBV, HIV, HCV, tuberculosis and syphilis infections amongst newly arrived migrants screened at the Infectious Diseases Unit of Modena, Italy. J. Infect Public Health 2019, 12, 200–204. [Google Scholar] [CrossRef]
- Scotto, G.; Fazio, V.; Lo Muzio, L.; Coppola, N. Screening for infectious diseases in newly arrived asymptomatic immigrants in southern Italy. East Mediterr. Health J. 2019, 25, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Scotto, G.; Armignacco, O.; Starnini, G.; Francavilla, R.; Foti, G.; Portelli, V.; Mazzeo, M.; Minerva, N.; Carretta, V. Hepatitis C and immigration: A multicentre study. Infez. Med. 2016, 24, 210–216. [Google Scholar]
- European Centre for Disease Prevention and Control. Hepatitis B and C Epidemiology in Selected Population Groups in the EU/EEA; ECDC: Stockholm, Sweden, 2018. [Google Scholar]
- Andriulli, A.; Stroffolini, T.; Mariano, A.; Valvano, M.R.; Grattagliano, I.; Ippolito, A.M.; Grossi, A.; Brancaccio, G.; Coco, C.; Russello, M.; et al. Declining prevalence and increasing awareness of HCV infection in Italy: A population-based survey in five metropolitan areas. Eur. J. Intern Med. 2018, 53, 79–84. [Google Scholar] [CrossRef]
- Lembo, T.; Saffioti, F.; Chiofalo, B.; Granese, R.; Filomia, R.; Grasso, R.; Triolo, O.; Raimondo, G. Low prevalence of hepatitis B and hepatitis C virus serum markers in a cohort of pregnant women from Southern Italy. Dig. Liver Dis. 2017, 49, 1368–1372. [Google Scholar] [CrossRef]
- Piffer, S.; Mazza, A.; Dell’Anna, L. Serological screening for hepatitis C during pregnancy: Seroprevalence and maternal and neonatal outcomes in 45,000 pregnant women. Eur. J. Obstet. Gynecol. Reprod. Biol. 2020, 254, 195–199. [Google Scholar] [CrossRef]
- Tosti, M.E.; Ferrigno, L.; Mele, A.; Alfonsi, V.; Iantosca, G.; Crateri, S.; Franca D’Angelo, F.; Andreozzi, S. Epidemiologia Delle Epatiti Virali Acute in Italia, Bollettino SEIEVA N. 6 March 2020. Available online: https://www.epicentro.iss.it/epatite/bollettino/Bollettino-n-6-marzo-2020.pdf (accessed on 14 June 2021).
- Tosti, M.E.; Alfonsi, V.; Lacorte, E.; Mele, A.; Galli, C.; Zanetti, A.R.; Romanò, L.; SEIEVACollaborating Group Ferrigno, L.; Crateri, S.; Iantosca, G. Acute Hepatitis B after the implementation of universal vaccination in Italy: Results from 22 years of surveillance (1993–2014). CID 2016, 62, 1412–1418. [Google Scholar] [CrossRef] [Green Version]
- Tosti, M.E.; Longhi, S.; De Waure, C.; Mele, A.; Franco, E.; Ricciardi, W.; Filia, A. Assessment of timeliness, representativeness and quality of data reported to Italy’s national integrated surveillance system for acute viral hepatitis (SEIEVA). Public Health 2015, 129, 561–568. [Google Scholar] [CrossRef]
- Carletti, P. La Salute Della Popolazione Immigrata: Metodologia di Analisi; Promozione della Salute della Popolazione Immigrata in Italia Accordo Ministero della Salute/CCM—Regione Marche (Direzione Generale Prevenzione Sanitaria, Ufficio I, n DG/PREV/I 3488/P/F 3 ad, 2007). 2009. Available online: http://www.ccm-network.it/progetto.jsp?id=node/74&idP=740 (accessed on 14 June 2021).
- Giraudo, M.; Bena, A.; Costa, G. Migrant workers in Italy: An analysis of injury risk taking into account occupational characteristics and job tenure. BMC Public Health 2017, 17, 351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leonardsson, H.; Hreinsson, J.P.; Löve, A.; Björnsson, E.S. Hepatitis due to Epstein-Barr virus and cytomegalovirus: Clinical features and outcomes. Scand. J. Gastroenterol. 2017, 52, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Ndumbi, P.; Freidl, G.S.; Williams, C.J.; Mårdh, O.; Varela, C.; Avellón, A.; Friesema, I.; Vennema, H.; Beebeejaun, K.; Ngui, S.L.; et al. Hepatitis A outbreak disproportionately affecting men who have sex with men (MSM) in the European Union and European Economic Area, June 2016 to May 2017. Euro Surveill. 2018, 23, 1700641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Severi, E.; Verhoef, L.; Thornton, L.; Guzman-Herrador, B.R.; Faber, M.; Sundqvist, L.; Rimhanen-Finne, R.; Roque-Afonso, A.M.; Ngui, S.L.; Allerberger, F.; et al. Large and prolonged food-borne multistate hepatitis A outbreak in Europe associated with consumption of frozen berries, 2013 to 2014. Euro Surveill. 2015, 20, 21192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Visalli, G.; Facciolà, A.; Carnuccio, S.M.; Cristiano, P.; D’Andrea, G.; Picerno, I.; Di Pietro, A. Health conditions of migrants landed in north-eastern Sicily and perception of health risks of the resident population. Public Health 2020, 185, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Eiset, A.H.; Wejse, C. Review of infectious diseases in refugees and asylum seekers-current status and going forward. Public Health Rev. 2017, 38, 22. [Google Scholar] [CrossRef] [Green Version]
- European Centre for Disease Prevention and Control. Infectious Diseases of Specific Relevance to Newly-Arrived Migrants in the EU/EEA; ECDC: Stockholm, Sweden, 2015. [Google Scholar]
- Eonomopoulou, A.; Pavli, A.; Stasinopoulou, P.; Giannopoulos, L.A.; Tsiodras, S. Migrant screening: Lessons learned from the migrant holding level at the Greek-Turkish borders. J. Infect Public Health 2017, 10, 177–184. [Google Scholar] [CrossRef] [Green Version]
- Mele, A.; Ferrigno, L.; Romanò, L.; Alfonsi, V.; D’Angelo, F.; Crateri, S.; Tosti, M.E. An update on the epidemiology of hepatitis A in Italy 2015-2019. Data from the surveillance of acute viral hepatitis SEIEVA. Epidemiol. Prev. 2021, 45, 173–180. [Google Scholar] [PubMed]
- Gassowski, M.; Michaelis, K.; Wenzel, J.J.; Faber, M.; Figoni, J.; Mouna, L.; Friesema, I.H.; Vennema, H.; Avellon, A.; Varela, C.; et al. Two concurrent outbreaks of hepatitis A highlight the risk of infection for non-immune travellers to Morocco, January to June 2018. Euro Surveill. 2018, 23, 1800329. [Google Scholar] [CrossRef]
- Miglietta, A.; Quinten, C.; Lopalco, P.L.; Duffell, E. Impact of hepatitis B vaccination on acute hepatitis B epidemiology in European Union/European Economic Area countries, 2006 to 2014. Euro Surveill. 2018, 23, 17–00278. [Google Scholar] [CrossRef]
- World Health Organzation. Hepatitis B: Key Facts; World Health Organzation: Geneva, Switzerland, 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/hepatitis-b (accessed on 14 June 2021).
- Del Pinto, R.; Pietropaoli, D.; Russomando, U.; Evangelista, P.; Ferri, C. Health status of Afro-Asian refugees in an Italian urban area: A cross-sectional monocentric study. Public Health 2018, 158, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Marrone, R.; Baglio, G.; Bruscino, G.; Costanzo, G.; Cavani, A.; Mirisola, C. Prevalence of latent tuberculosis infection, hepatitis B, hepatitis C, and syphilis among newly arrived unaccompanied minors living in reception centers in Rome. Int. J. Infect Dis. 2020, 101, 126–130. [Google Scholar] [CrossRef] [PubMed]
- Boccalini, S.; Pellegrino, E.; Tiscione, E.; Pesavento, G.; Bechini, A.; Levi, M.; Rapi, S.; Mercurio, S.; Mannelli, F.; Peruzzi, M.; et al. Sero-epidemiology of hepatitis B markers in the population of Tuscany, Central Italy, 20 years after the implementation of universal vaccination. Hum. Vaccines Immunother. 2013, 9, 636–641. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alfonsi, V.; Romanò, L.; Ciccaglione, A.R.; La Rosa, G.; Bruni, R.; Zanetti, A.; Della Libera, S.; Iaconelli, M.; Bagnarelli, P.; Capobianchi, M.R.; et al. Hepatitis E in Italy: 5 years of national epidemiological, virological and environmental surveillance, 2012 to 2016. Euro Surveill. 2018, 23, 1700517. [Google Scholar] [CrossRef] [PubMed]
- World Health Organzation. Hepatitis E: Key Facts; World Health Organzation: Geneva, Switzerland, 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/hepatitis-e (accessed on 14 June 2021).
- Tosti, M.E.; Ferrigno, L.; Mele, A.; Romanò, L.; Fiacchini, D.; Bagnarelli, P.; Zotti, C.; Chironna, M.; Prato, R.; Giordani, M.T.; et al. Epidemiology and surveillance of hepatitis E in Italy. Data from the SEIEVA surveillance system 2007–2019. Epidemiol. Prev. 2021, 45, 46–53. [Google Scholar] [CrossRef]
- Stroffolini, T.; Ciancio, A.; Furlan, C.; Vinci, M.; Fontana, R.; Russello, M.; Colloredo, G.; Morisco, F.; Coppola, N.; Babudieri, S.; et al. Migratory flow and hepatitis delta infection in Italy: A new challenge at the beginning of the third millennium. J. Viral Hepat. 2020, 27, 941–947. [Google Scholar] [CrossRef]
- Coppola, N.; Alessio, L.; Onorato, L.; Sagnelli, C.; Macera, M.; Sagnelli, E.; Pisaturo, M. Epidemiology and management of hepatitis C virus infections in immigrant populations. Infect Dis. Poverty 2019, 8, 17. [Google Scholar] [CrossRef] [Green Version]
- Tosti, M.E.; Marceca, M.; Eugeni, E.; D’Angelo, F.; Geraci, S.; Declich, S.; Della Seta, M.; Ferrigno, L.; Marrone, R.; Pajno, C.; et al. Health assessment for migrants and asylum seekers upon arrival and while hosted in reception centres: Italian guidelines. Health Policy 2020, 125, 393–405. [Google Scholar] [CrossRef]
- Seedat, F.; Hargreaves, S.; Nellums, L.B.; Ouyang, J.; Brown, M.; Friedland, J.S. How effective are approaches to migrant screening for infectious diseases in Europe? A systematic review. Lancet Infect Dis. 2018, 18, e259–e271. [Google Scholar] [CrossRef]
- Whelan, J.; Sonder, G.; van den Hoek, A. Declining incidence of hepatitis A in Amsterdam (The Netherlands), 1996–2011: Second generation migrants still an important risk group for virus importation. Vaccine 2013, 31, 1806–1811. [Google Scholar] [CrossRef] [PubMed]
| Foreigners/N Cases (%) | |||||
|---|---|---|---|---|---|
| Year | Total Cases | Hepatitis A | Hepatitis B | Hepatitis C | Hepatitis nonA-nonC/Unknown |
| 2004 | 153/1384 (11.0) | 58/651 (8.9) | 63/498 (12.6) | 7/112 (6.2) | 25/123 (20.3) |
| 2005 | 127/969 (13.1) | 46/386 (11.9) | 57/374 (15.2) | 4/101 (4.0) | 20/108 (18.5) |
| 2006 | 197/1114 (17.7) | 84/407 (20.6) | 69/473 (14.6) | 7/109 (6.4) | 37/125 (29.6) |
| 2007 | 169/1040 (16.2) | 56/353 (15.9) | 85/487 (17.4) | 6/102 (5.9) | 22/98 (22.4) |
| 2008 | 167/1372 (12.2) | 71/742 (9.6) | 68/453 (15.0) | 6/74 (8.1) | 22/103 (21.4) |
| 2009 | 164/1303 (12.6) | 84/753 (11.2) | 52/390 (13.3) | 8/83 (9.6) | 20/77 (26.0) |
| 2010 | 148/826 (17.9) | 62/371 (16.7) | 63/326 (19.3) | 5/75 (6.7) | 18/54 (33.3) |
| 2011 | 153/744 (20.6) | 58/244 (23.8) | 61/334 (18.3) | 10/88 (11.4) | 24/78 (30.8) |
| 2012 | 161/712 (22.6) | 70/251 (27.9) | 63/304 (20.7) | 9/86 (10.5) | 19/71 (26.8) |
| 2013 | 170/1260 (13.5) | 90/781 (11.5) | 65/313 (20.8) | 3/94 (3.2) | 12/72 (16.7) |
| 2014 | 146/666 (21.9)) | 73/281 (26.0) | 54/260 (20.8) | 9/69 (13.0) | 10/56 (17.9) |
| 2015 | 104/587 (17.7) | 41/214 (19.2) | 40/225 (17.8) | 5/61 (8.2) | 18/87 (20.7) |
| 2016 | 91/544 (16.7) | 39/282 (13.8) | 39/168 (23.2) | 3/37 (8.1) | 10/57 (17.5) |
| 2017 | 139/2254 (6.2) | 98/2011 (4.9) | 27/144 (18.7) | 4/24 (16.7) | 10/75 (13.3) |
| 2018 | 142/590 (24.1) | 101/407 (24.8) | 27/109 (24.8) | 3/16 (18.7) | 11/58 (19.0) |
| 2019 | 121/507 (23.9) | 85/274 (31.0) | 16/123 (13.0) | 4/30 (13.3) | 16/80 (20.0) |
| Total | 2352/15,872 (14.8) | 1116/8408 (13.3) | 849/4981 (17.0) | 93/1161 (8.0) | 294/1322 (22.2) |
| Origin | Total Cases N (%) | Hepatitis A N (%) | Hepatitis B N (%) | Hepatitis C N (%) | Hepatitis nonA-nonC/Unknown N (%) |
|---|---|---|---|---|---|
| Level of development | |||||
| SMPC | 2278 (96.9) | 1061 (95.1) | 834 (98,2) | 92 (98.9) | 291 (99.0) |
| HIC | 74 (3.1) | 55 (4.9) | 15 (1,8) | 1 (1.1) | 3 (1.0) |
| Geographic area | |||||
| Africa | 773 (32.9) | 533 (47.8) | 183 (21.6) | 23 (24.7) | 34 (11.6) |
| Central/South America | 212 (9.0) | 108 (9.7) | 76 (8.9) | 6 (6.4) | 22 (7.5) |
| Asia | 417 (17.7) | 140 (12.5) | 124 (14.6) | 8 (8.6) | 145 (49.3) |
| East Europe | 886 (37.7) | 288 (25.8) | 453 (53.4) | 55 (59.1) | 90 (30.6) |
| West Europe | 56 (2.4) | 42 (3.8) | 11 (1.3) | 1. (1.1) | 2 (0.7) |
| North America | 6 (0.3) | 4 (0.4) | 1 (0.1) | 0 (0.0) | 1 (0.3) |
| Oceania | 2 (0.1) | 1 (0.1) | 1 (0.1) | 0 (0.0) | 0 (0.0) |
| Sex | |||||
| Males | 1426 (60.8) | 610 (54.9) | 544 (64,2) | 56 (60.2) | 216 (74.0) |
| Females | 918 (39.2) | 501 (45.1) | 304 (35.8) | 37 (39.8) | 76 (26.1) |
| Age | |||||
| 0–14 | 681 (29.1) | 646 (58.4) | 12 (1.4) | 4 (4.3) | 19 (6.5) |
| 15–24 | 453 (19.4) | 186 (16.8) | 192 (22.7) | 12 (12.9) | 63 (21.5) |
| 25–34 | 608 (26.0) | 129 (11.7) | 335 (39.6) | 30 (32.3) | 114 (38.9) |
| 35–54 | 540 (23.1) | 128 (11.6) | 284 (33.6) | 40 (43.0) | 88 (30.0) |
| ≥55 | 56 (2.4) | 17 (1.5) | 23 (2.7) | 7 (7.5) | 9 (3.1) |
| Total | 2352 (100) | 1116 (100) | 849 (100) | 93 (100) | 294 (100) |
| Etiologic Agent | N. of Cases (%) | Area of Origin (No. of Cases) |
|---|---|---|
| Delta | 11 (3.7) | Eastern Europe (10), Africa (1) |
| E | 94 (32.0) | Asia (79) Eastern Europe (6), Central/South America (5), Africa (3), West Europe (1) |
| nonA-nonC * | 57 (19.4) | Asia (24), Eastern Europe (21), Africa (8) and Central/South America (4) |
| nonA-nonE ** | 16 (5.4) | Africa (5), Asia (5), Central/South America (4), Eastern Europe (2) |
| Unknown | 116 (39.5) | Eastern Europe (51), Asia (37), Africa (17), Central/South America (9), West Europe (1), North America (1) |
| Total | 294 (100) | Asia (145), Eastern Europe (90), Africa (34), Central/South America (22), West Europe (2) and North America (1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Angelo, F.; Ferrigno, L.; Mele, A.; Alfonsi, V.; Declich, S.; De Ponte, G.; Crateri, S.; Burgio, A.; Caminada, S.; Tosti, M.E.; et al. Differences in Incidence of Acute Viral Hepatitis between Foreigners and Autochthonous Population in Italy. Int. J. Environ. Res. Public Health 2021, 18, 7944. https://doi.org/10.3390/ijerph18157944
D’Angelo F, Ferrigno L, Mele A, Alfonsi V, Declich S, De Ponte G, Crateri S, Burgio A, Caminada S, Tosti ME, et al. Differences in Incidence of Acute Viral Hepatitis between Foreigners and Autochthonous Population in Italy. International Journal of Environmental Research and Public Health. 2021; 18(15):7944. https://doi.org/10.3390/ijerph18157944
Chicago/Turabian StyleD’Angelo, Franca, Luigina Ferrigno, Annamaria Mele, Valeria Alfonsi, Silvia Declich, Giulia De Ponte, Simonetta Crateri, Alessandra Burgio, Susanna Caminada, Maria Elena Tosti, and et al. 2021. "Differences in Incidence of Acute Viral Hepatitis between Foreigners and Autochthonous Population in Italy" International Journal of Environmental Research and Public Health 18, no. 15: 7944. https://doi.org/10.3390/ijerph18157944
APA StyleD’Angelo, F., Ferrigno, L., Mele, A., Alfonsi, V., Declich, S., De Ponte, G., Crateri, S., Burgio, A., Caminada, S., Tosti, M. E., & on behalf of The SEIEVA Collaborating Group. (2021). Differences in Incidence of Acute Viral Hepatitis between Foreigners and Autochthonous Population in Italy. International Journal of Environmental Research and Public Health, 18(15), 7944. https://doi.org/10.3390/ijerph18157944






