COVID-19 Prevention and Control Measures in Workplace Settings: A Rapid Review and Meta-Analysis
Abstract
:1. Introduction
2. Methods
2.1. Research Questions
- What COVID-19 IPC measures are used in workplace settings?
- What IPC measures reduce COVID-19 infections in the workplace?
2.2. Workplace COVID-19 IPC Measures
- Surveillance measures—COVID-19 symptom monitoring, strategies to screen or test individuals, such as symptomatic or asymptomatic testing;
- Outbreak investigations and response—contact tracing and testing of close contacts, quarantine of potentially infected individuals or groups, self-isolation of confirmed cases;
- PPE—masks, full PPE (i.e., masks, goggles, gloves, work clothes) in medium/high-risk jobs;
- Environmental adjustments—improving airflow and ventilation, adding physical barriers to help employees avoid physical contact, environmental cleaning;
- Education initiatives—training on IPC measures, communication and signage, ongoing education and support;
- Changes in work arrangements—social distancing, facility zoning, entrance restrictions, changes in assignments for high-risk workers (i.e., individuals with medical conditions, pregnant women, over-60 population), facility shutdown, paid sick leave;
- Combined measures—approaches that combine measures from two or more categories.
2.3. Defining Effectiveness
2.4. Search Strategy
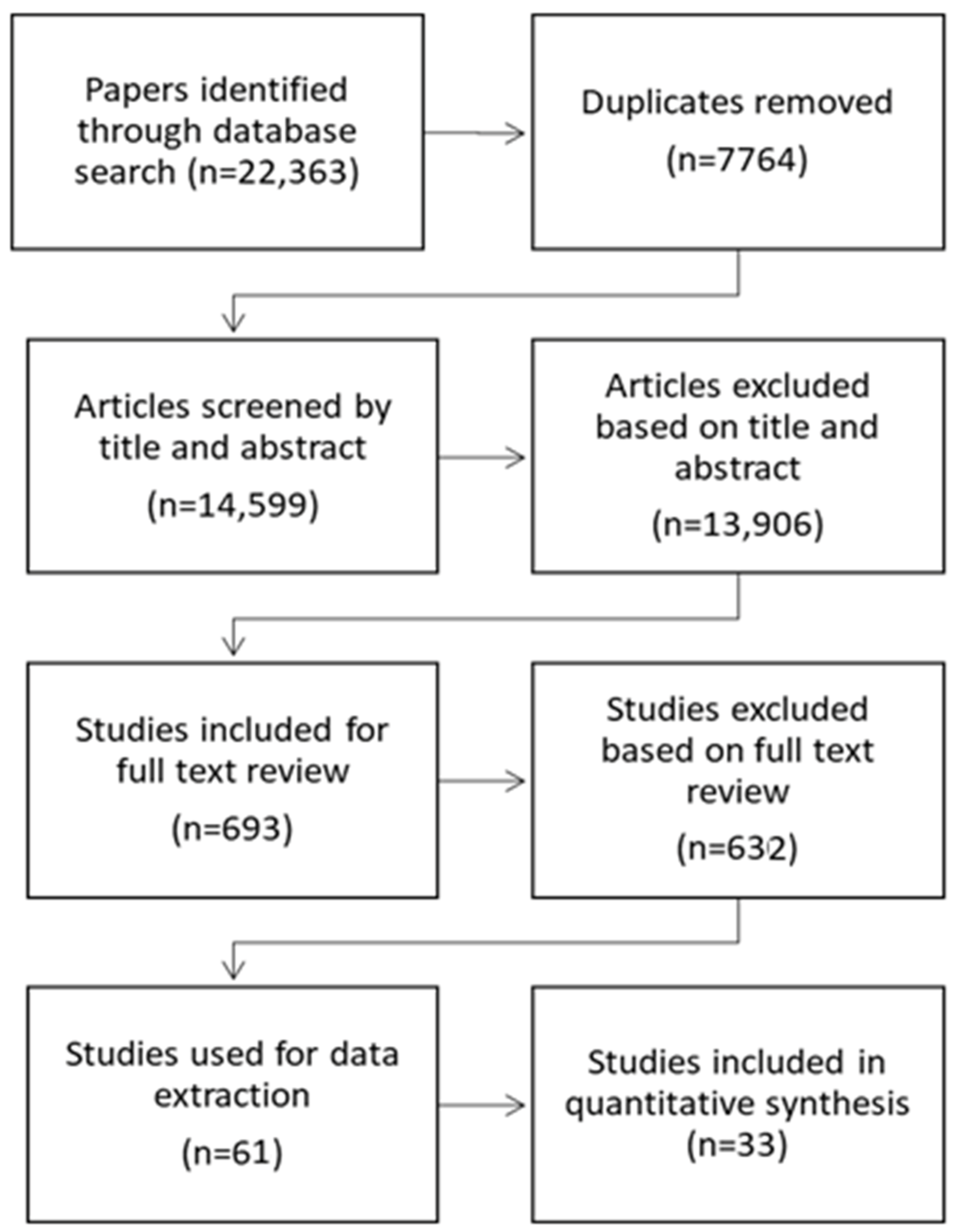
2.5. Study Selection (Identification, Screening, and Inclusion)
2.6. Data Extraction
2.7. Quality Assessment
2.8. Statistical Analysis
3. Results
3.1. Study Characteristics
3.2. Study Quality
3.3. Effectiveness of Workplace COVID-19 IPC Measures
3.3.1. Surveillance Measures
3.3.2. Outbreak Investigation and Response
3.3.3. PPE
3.3.4. Combined Measures
3.3.5. Key Findings from Modelling Studies
3.3.6. Key Findings from Studies Not Included in Quantitative Synthesis
3.3.7. Summary of Findings
4. Discussion
4.1. Completeness and Applicability of Evidence
4.2. Limitations and Strengths
4.3. Implications for Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gorbalenya, A.E.; Baker, S.C.; Baric, R.S.; de Groot, R.J.; Drosten, C.; Gulyaeva, A.A.; Ziebuhr, J. The species Severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat. Microbiol. 2020, 5, 536–544. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Ning, Z.; Chen, Y.; Guo, M.; Liu, Y.; Gali, N.K.; Sun, L.; Duan, Y.; Cai, J.; Westerdahl, D.; et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature 2020, 582, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, T.; Jimenez, J.L.; Prather, K.A.; Tufekci, Z.; Fisman, D.; Schooley, R. Ten scientific reasons in support of airborne transmission of SARS-CoV-2. Lancet 2021, 397, 1603–1605. [Google Scholar] [CrossRef]
- Buitrago-Garcia, D.; Egli-Gany, D.; Counotte, M.J.; Hossmann, S.; Imeri, H.; Ipekci, A.M.; Salanti, G.; Low, N. Occurrence and transmission potential of asymptomatic and presymptomatic SARS-CoV-2 infections: A living systematic review and meta-analysis. PLoS Med. 2020, 17, e1003346. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Smith, J.O.; Schreiber, S.J.; Kopp, P.E.; Getz, W. Superspreading and the effect of individual variation on disease emergence. Nat. Cell Biol. 2005, 438, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, N.; Kuroda, R.; Tsuno, K.; Kawakami, N. Workplace responses to COVID-19 associated with mental health and work performance of employees in Japan. J. Occup. Health 2020, 62, e12134. [Google Scholar] [CrossRef]
- Michaels, D.; Wagner, G.R. Occupational Safety and Health Administration (OSHA) and Worker Safety During the COVID-19 Pandemic. JAMA 2020, 324, 1389. [Google Scholar] [CrossRef]
- Meßerschmidt, K. COVID-19 legislation in the light of the precautionary principle. Theory Pr. Legis. 2020, 8, 267–292. [Google Scholar] [CrossRef]
- Houghton, C.; Meskell, P.; Delaney, H.; Smalle, M.; Glenton, C.; Booth, A.; Chan, X.H.; Devane, D.; Biesty, L.M. Barriers and facilitators to healthcare workers’ adherence with infection prevention and control (IPC) guidelines for respiratory infectious diseases: A rapid qualitative evidence synthesis. Cochrane Database Syst. Rev. 2020, 2020, 3582. [Google Scholar] [CrossRef]
- Risko, N.; Werner, K.; Offorjebe, O.A.; Vecino-Ortiz, A.I.; Wallis, L.A.; Razzak, J. Cost-effectiveness and return on investment of protecting health workers in low- and middle-income countries during the COVID-19 pandemic. PLoS ONE 2020, 15, e0240503. [Google Scholar] [CrossRef]
- Singu, S.; Acharya, A.; Challagundla, K.; Byrareddy, S.N. Impact of Social Determinants of Health on the Emerging COVID-19 Pandemic in the United States. Front. Public Health 2020, 8, 406. [Google Scholar] [CrossRef]
- Huang, X.; Lin, J.; Demner-Fushman, D. Evaluation of PICO as a Knowledge Representation for Clinical Questions. AMIA. Annu. Symp. Proc. 2006, 2006, 359–363. [Google Scholar]
- World Health Organization. Coronavirus Disease (COVID-19): Health and Safety in the Workplace. Available online: https://www.who.int/news-room/q-a-detail/coronavirus-disease-covid-19-health-and-safety-in-the-workplace (accessed on 6 May 2021).
- Hopkins, J.; Coronavirus Resource Center. ‘COVID-19 Map’, Johns Hopkins Coronavirus Resource Center. Available online: https://coronavirus.jhu.edu/map.html (accessed on 6 May 2021).
- Gulumian, M.; Verbeek, J.; Andraos, C.; Sanabria, N.; De Jager, P. Systematic Review of Screening and Surveillance Programs to Protect Workers from Nanomaterials. PLoS ONE 2016, 11, e0166071. [Google Scholar] [CrossRef]
- McNair, P.; Lewis, G. Levels of evidence in medicine. Int. J. Sports Phys. Ther. 2012, 7, 474–481. [Google Scholar] [PubMed]
- Borenstein, M.; Hedges, L.V.; Higgins, J.P.T.; Rothstein, H.R. A basic introduction to fixed-effect and random-effects models for meta-analysis. Res. Synth. Methods 2010, 1, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Xu, C. Arcsine-based transformations for meta-analysis of proportions: Pros, cons, and alternatives. Health Sci. Rep. 2020, 3, e178. [Google Scholar] [CrossRef]
- Barendregt, J.J.; Doi, S.; Lee, Y.Y.; Norman, R.E.; Vos, T. Meta-analysis of prevalence. J. Epidemiol. Community Health 2013, 67, 974–978. [Google Scholar] [CrossRef] [PubMed]
- Wang, N. How to Conduct a Meta-Analysis of Proportions in R: A Comprehensive Tutorial. 2018; Preprint. [Google Scholar] [CrossRef]
- Buntinx, F.; Claes, P.; Gulikers, M.; Verbakel, J.; Jan, D.L.; Van der Elst, M.; Van Elslande, J.; Van Ranst, M.; Vermeersch, P. Added value of anti-SARS-CoV-2 antibody testing in a Flemish nursing home during an acute COVID-19 outbreak in April 2020. Acta Clin. Belg. 2020, 1–6. [Google Scholar] [CrossRef]
- Bontadi, D.; Bergamo, L.; Torri, P.; Patanè, P.A.; Bertoldi, A.; Lonardi, U. Efficacia delle misure volte a contenere la diffusione del virus Sars-Cov-2 negli ambienti di lavoro: Un’indagine in nove aziende con sede nella regione Veneto. Med. Lav. Work Environ. Health 2020, 111, 404–410. [Google Scholar]
- Krastinova, E.; Garrait, V.; Coste, A.; Varon, E.; Delacroix, I.; Siali, A.; Jung, C.; Cherbit, M.; Pairon, J.; Andujar, P. SARS-CoV2 positive RT-PCR incidence in symptomatic healthcare workers, clinical evolution, outcome and household transmission, experience from a French hospital. Med. Mal. Infect. 2020, 50, S61–S62. [Google Scholar] [CrossRef]
- Marshall, D.L.; Bois, F.; Jensen, S.K.; Linde, S.A.; Higby, R.; Rémy-McCort, Y.; Murray, S.; Dieckelman, B.; Sudradjat, F.; Martin, G.G. Sentinel Coronavirus environmental monitoring can contribute to detecting asymptomatic SARS-CoV-2 virus spreaders and can verify effectiveness of workplace COVID-19 controls. Microb. Risk Anal. 2020, 16, 100137. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, L.M.A.; Sanmark, E.; Oksanen, S.; Anttila, V.J.; Paterno, J.J.; Lappalainen, M.; Lehtonen, L.; Geneid, A. Healthcare workers’ high COVID-19 infection rate: The source of infections and potential for respirators and surgical masks to reduce occupational infections. medRxiv 2020, Preprint. [Google Scholar] [CrossRef]
- Pérez-García, F.; Pérez-Zapata, A.; Arcos, N.; De la Mata, M.; Ortiz, M.; Simón, E.; Fernández, I.H.; Ventosa, V.G.; Monte, M.M.; Arroyo, J.G.; et al. Severe acute respiratory coronavirus virus 2 (SARS-CoV-2) infection among hospital workers in a severely affected institution in Madrid, Spain: A surveillance cross-sectional study. Infect. Control Hosp. Epidemiol. 2020, 42, 1–7. [Google Scholar] [CrossRef]
- Lentz, R.J.; Colt, H.; Chen, H.; Cordovilla, R.; Popevic, S.; Tahura, S.; Candoli, P.; Tomassetti, S.; Meachery, G.J.; Cohen, B.P.; et al. Assessing coronavirus disease 2019 (COVID-19) transmission to healthcare personnel: The global ACT-HCP case-control study. Infect. Control Hosp. Epidemiol. 2021, 42, 381–387. [Google Scholar] [CrossRef]
- Roxby, A.C.; Greninger, A.L.; Hatfield, K.M.; Lynch, J.B.; Dellit, T.H.; James, A.; Taylor, J.; Page, L.C.; Kimball, A.; Arons, M.; et al. Outbreak Investigation of COVID-19 Among Residents and Staff of an Independent and Assisted Living Community for Older Adults in Seattle, Washington. JAMA Intern. Med. 2020, 180, 1101–1105. [Google Scholar] [CrossRef]
- Benea, C.; Rendon, L.; Papenburg, J.; Frenette, C.; Imacoudene, A.; McDonald, E.G.; Nguyen, Q.D.; Rajda, E.; Tran, E.; Vameghestahbanati, M.; et al. Evaluation of a home-based 7-day infection control strategy for healthcare workers following high-risk exposure to severe acute respiratory coronavirus virus 2 (SARS-CoV-2): A cohort study. Infect. Control Hosp. Epidemiology 2020, 16, 1–4. [Google Scholar] [CrossRef]
- Chang, E.; Choi, J.-S.; Park, T.Y.; Bin Kim, S.; Ko, S.; Kwon, Y.S.; Kim, E.J.; Song, H.; Noh, H.K.; Park, S.-W. A Universal Screening Strategy for SARS-CoV-2 Infection in Intensive Care Units: Korean Experience in a Single Hospital. Infect. Chemother. 2020, 52, 352–359. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.; Kim, E.O.; Kim, S.-H.; Jung, J. Risk of COVID-19 Transmission from Infected Outpatients to Healthcare Workers in an Outpatient Clinic. J. Korean Med. Sci. 2020, 35, e431. [Google Scholar] [CrossRef]
- Dubey, A.; Bansal, A.; Sonkar, S.C.; Goswami, B.; Makwane, N.; Manchanda, V.; Koner, B.C. In-house assembled protective devices in laboratory safety against SARS-nCoV-2 in clinical biochemistry laboratory of a COVID dedicated hospital. medRxiv 2020, 5713, Preprint. [Google Scholar] [CrossRef]
- Erber, J.; Kappler, V.; Haller, B.; Mijocevic, H.; Galhoz, A.; da Costa, C.P.; Gebhardt, F.; Graf, N.; Hoffmann, D.; Thaler, M.; et al. Strategies for infection control and prevalence of anti-SARS-CoV-2 IgG in 4,554 employees of a university hospital in Munich, Germany. medRxiv 2020, 6136. [Google Scholar] [CrossRef]
- Huy, M.D.N.; My, N.N.; Minh, H.D.; Hoa, T.N.; Phuong, T.D.T.; Ty, H.V.T.; Van, T.L.; Quynh, N.N.T.; Thanh, D.N.; Manh, H.L.; et al. Preventing laboratory-associated infections in the COVID-19 era: Experience from a tertiary care infectious disease hospital in Southern Vietnam. Res. Sq. 2021. Preprint. [Google Scholar] [CrossRef]
- Jones, N.K.; Rivett, L.; Sparkes, D.; Forrest, S.; Sridhar, S.; Young, J.; Pereira-Dias, J.; Cormie, C.; Gill, H.; Reynolds, N.; et al. Effective control of SARS-CoV-2 transmission between healthcare workers during a period of diminished community prevalence of COVID-19. eLife 2020, 9, 59391. [Google Scholar] [CrossRef]
- Karmarkar, E.N.; Blanco, I.; Amornkul, P.N.; DuBois, A.; Deng, X.; Moonan, P.K.; Rubenstein, B.L.; Miller, D.A.; Kennedy, I.; Yu, J.; et al. Timely intervention and control of a novel coronavirus (COVID-19) outbreak at a large skilled nursing facility—San Francisco, California, 2020. Infect. Control Hosp. Epidemiol. 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Menting, T.; Krause, K.; Benz-Tettey, F.; Boehringer, R.; Laufer, D.; Gruber, B.; Crump, M.; Schieferdecker, R.; Reuhl, S.; Kaeferstein, A.; et al. Low-threshold SARS-CoV-2 testing facility for hospital staff: Prevention of COVID-19 outbreaks? Int. J. Hyg. Environ. Health 2021, 231, 113653. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, S.; Lakkireddy, D.; Trivedi, C.; MacDonald, B.; Mayedo, A.Q.; Rocca, D.G.D.; Atkins, D.; Park, P.; Shah, A.; Gopiathannair, R.; et al. Creating a safe workplace by universal testing of SARS-CoV-2 infection in patients and healthcare workers in the electrophysiology unit having no symptoms of COVID-19: A multi-center experience. medRxiv 2020, 3494. [Google Scholar] [CrossRef]
- Mortgat, L.; Barbezange, C.; Fischer, N.; Heyndrickx, L.; Hutse, V.; Thomas, I.; Vuylsteke, B.; Arien, K.; Desombere, I.; Duysburgh, E. SARS-CoV-2 Prevalence and Seroprevalence among Healthcare Workers in Belgian Hospitals: Baseline Results of a Prospective Cohort Study. medRxiv 2020, 4545, Preprint. [Google Scholar] [CrossRef]
- Rivett, L.; Sridhar, S.; Sparkes, D.; Routledge, M.; Jones, N.K.; Forrest, S.; Young, J.; Pereira-Dias, J.; Hamilton, W.L.; Ferris, M.; et al. Screening of healthcare workers for SARS-CoV-2 highlights the role of asymptomatic carriage in COVID-19 transmission. eLife 2020, 9, e58728. [Google Scholar] [CrossRef]
- Seidelman, J.L.; Lewis, S.S.; Advani, S.D.; Akinboyo, I.C.; Epling, C.; Case, M.; Said, K.; Yancey, W.; Stiegel, M.; Schwartz, A.; et al. Universal masking is an effective strategy to flatten the severe acute respiratory coronavirus virus 2 (SARS-CoV-2) healthcare worker epidemiologic curve. Infect. Control Hosp. Epidemiol. 2020, 41, 1466–1467. [Google Scholar] [CrossRef] [PubMed]
- Squeri, R.; Levita, A.; Intelisano, R.; Costa, G.B.; Mancuso, G.; Grasso, L.; D’Amato, S.; Mazzitelli, F.; Squeri, A.; Midiri, A.; et al. Correct management and low rate of contagiousness of healthcare workers in a University Hospital in Southern Italy: From contact tracing to serological investigation. Acta Bio. Med. Atenei Parm. 2020, 91, 79–86. [Google Scholar]
- Suzuki, T.; Hayakawa, K.; Ainai, A.; Iwata-Yoshikawa, N.; Sano, K.; Nagata, N.; Suzuki, T.; Wakimoto, Y.; Akiyama, Y.; Miyazato, Y.; et al. Effectiveness of personal protective equipment in preventing severe acute respiratory syndrome coronavirus 2 infection among healthcare workers. J. Infect. Chemother. 2021, 27, 120–122. [Google Scholar] [CrossRef] [PubMed]
- Vimercati, L.; Dell’Erba, A.; Migliore, G.; De Maria, L.; Caputi, A.; Quarato, M.; Stefanizzi, P.; Cavone, D.; Ferorelli, D.; Sponselli, S.; et al. Prevention and protection measures of healthcare workers exposed to SARS-CoV-2 in a university hospital in Bari, Apulia, Southern Italy. J. Hosp. Infect. 2020, 105, 454–458. [Google Scholar] [CrossRef]
- Wang, D.D.; O’Neill, W.W.; Zervos, M.J.; McKinnon, J.E.; Allard, D.; Alangaden, G.J.; Schultz, L.R.; Poisson, L.M.; Chu, B.S.; Kalkanis, S.N.; et al. Association Between Implementation of a Universal Face Mask Policy for Healthcare Workers in a Health Care System and SARS-CoV-2 Positivity Testing Rate in Healthcare Workers. J. Occup. Environ. Med. 2021, 63, 476–481. [Google Scholar] [CrossRef]
- Woon, Y.L.; Lee, Y.L.; Chong, Y.M.; Ayub, N.A.; Krishnabahawan, S.L.; Lau, J.F.W.; Goh, P.P. Serology surveillance of anti-SARS-CoV-2 antibodies among asymptomatic healthcare workers in Malaysian healthcare facilities designated for COVID-19 care. Res. Sq. 2021, Preprint. [Google Scholar] [CrossRef]
- Wee, L.E.; Sim, X.Y.J.; Conceicao, E.P.; Aung, M.K.; Goh, J.Q.; Yeo, D.W.T.; Gan, W.H.; Chua, Y.Y.; Wijaya, L.; Tan, T.T.; et al. Containment of COVID-19 cases among healthcare workers: The role of surveillance, early detection, and outbreak management. Infect. Control Hosp. Epidemiol. 2020, 41, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.; Yan, L. Prevention and Control Strategies for Coronavirus Disease-2019 in a Tertiary Hospital in the Middle East of China. Risk Manag. Healthc. Policy 2020, 13, 1563–1569. [Google Scholar] [CrossRef] [PubMed]
- Gunawardana, M.; Breslin, J.; Cortez, J.M.; Rivera, S.; Webster, S.; Ibarrondo, F.J.; Yang, O.O.; Pyles, R.B.; Ramirez, C.M.; Adler, A.P.; et al. Longitudinal COVID-19 Surveillance and Characterization in the Workplace with Public Health and Diagnostic Endpoints. mSphere 2021, e0054221. [Google Scholar] [CrossRef]
- Miller, S.L.; Mukherjee, D.; Wilson, J.; Clements, N.; Steiner, C. Implementing a Negative Pressure Isolation Space within a Skilled Nursing Facility to Control SARS-CoV-2 Transmission. Am. J. Infect. Control 2020, 49, 438–446. [Google Scholar] [CrossRef]
- Lipsitz, L.A.; Lujan, A.M.; Dufour, A.; Abrahams, G.; Magliozzi, H.; Herndon, L.; Dar, M. Stemming the Tide of COVID -19 Infections in Massachusetts Nursing Homes. J. Am. Geriatr. Soc. 2020, 68, 2447–2453. [Google Scholar] [CrossRef] [PubMed]
- Herstein, J.J.; Degarege, A.; Stover, D.; Austin, C.; Schwedhelm, M.M.; Lawler, J.V.; Lowe, J.J.; Ramos, A.K.; Donahue, M. Characteristics of SARS-CoV-2 Transmission among Meat Processing Workers in Nebraska, USA, and Effectiveness of Risk Mitigation Measures. Emerg. Infect. Dis. 2021, 27, 1032–1038. [Google Scholar] [CrossRef]
- Ladhani, S.N.; Chow, J.Y.; Janarthanan, R.; Fok, J.; Crawley-Boevey, E.; Vusirikala, A.; Fernandez, E.; Perez, M.S.; Tang, S.; Dun-Campbell, K.; et al. Increased risk of SARS-CoV-2 infection in staff working across different care homes: Enhanced CoVID-19 outbreak investigations in London care Homes. J. Infect. 2020, 81, 621–624. [Google Scholar] [CrossRef] [PubMed]
- Rath, S.; Tripathy, D.; Roy, A.K.; Khanna, R.C.; Jalali, S.; Panigrahy, B.; Parija, D.C. Point-of-care rapid antigen testing for COVID-19 at a tertiary eye care facility: Role in commencement of elective surgeries, contact tracing and implementation of back-to-work policy. Indian J. Ophthalmol. 2021, 69, 964–970. [Google Scholar] [CrossRef]
- Wang, X.; Ferro, E.G.; Zhou, G.; Hashimoto, D.; Bhatt, D.L. Association Between Universal Masking in a Health Care System and SARS-CoV-2 Positivity Among Health Care Workers. JAMA 2020, 324, 703–704. [Google Scholar] [CrossRef]
- Kim, S.W.; Jo, S.J.; Lee, H.; Oh, J.H.; Lim, J.; Lee, S.H.; Choi, J.H.; Lee, J. Containment of a healthcare-associated COVID-19 outbreak in a university hospital in Seoul, Korea: A single-center experience. PLoS ONE 2020, 15, e0237692. [Google Scholar] [CrossRef] [PubMed]
- Canetti, D.; Dell’Acqua, R.; Riccardi, N.; Della Torre, L.; Bigoloni, A.; Muccini, C.; Lazzarin, A. SARS-CoV-2 IgG/IgM Rapid Test as a Diagnostic Tool in Hospitalized Patients and Healthcare Workers, at a large Teaching Hospital in northern Italy, during the 2020 COVID-19 Pandemic. New Microbiol. 2020, 43, 161–165. [Google Scholar] [PubMed]
- Brainard, J.; Rushton, S.; Winters, T.; Hunter, P.R. Introduction to and spread of COVID-19-like illness in care homes in Norfolk, UK. J. Public Health 2021, 43, 228–235. [Google Scholar] [CrossRef]
- Krone, M.; Noffz, A.; Richter, E.; Vogel, U.; Schwab, M. Control of a COVID-19 outbreak in a nursing home by general screening and cohort isolation in Germany, March to May 2020. Eurosurveillance 2021, 26, 2001365. [Google Scholar] [CrossRef]
- Mandic-Rajcevic, S.; Masci, F.; Crespi, E.; Franchetti, S.; Longo, A.; Bollina, I.; Boselli, L. Contact tracing and isolation of asymptomatic spreaders to successfully control the COVID-19 epidemic among healthcare workers in Milan (Italy). medRxiv 2020, 2818, Preprint. [Google Scholar] [CrossRef]
- Wee, L.E.; Venkatachalam, I.; Sim, X.Y.J.; Tan, K.B.-K.; Wen, R.; Tham, C.K.; Gan, W.H.; Ko, K.K.K.; Ho, W.Q.; Kwek, G.T.C.; et al. Containment of COVID-19 and reduction in healthcare-associated respiratory viral infections through a multi-tiered infection control strategy. Infect. Dis. Health 2021, 26, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Cattelan, A.M.; Sasset, L.; Di Meco, E.; Cocchio, S.; Barbaro, F.; Cavinato, S.; Gardin, S.; Carretta, G.; Donato, D.; Crisanti, A.; et al. An Integrated Strategy for the Prevention of SARS-CoV-2 Infection in Healthcare Workers: A Prospective Observational Study. Int. J. Environ. Res. Public Health 2020, 17, 5785. [Google Scholar] [CrossRef]
- Kumar, D.; Ferreira, V.H.; Chruscinski, A.; Kulasingam, V.; Pugh, T.J.; Dus, T.; Humar, A. Prospective Observational Study of Screening Asymptomatic Healthcare Workers for SARS-CoV-2 at a Canadian Tertiary Care Center. medRxiv 2020, 9053, Preprint. [Google Scholar] [CrossRef]
- Wee, L.E.I.; Sim, X.Y.J.; Conceicao, E.P.; Aung, M.K.; Tan, K.Y.; Ko, K.K.K.; Wong, H.M.; Wijaya, L.; Tan, B.H.; Venkatachalam, I.; et al. Containing COVID-19 outside the isolation ward: The impact of an infection control bundle on environmental contamination and transmission in a cohorted general ward. Am. J. Infect. Control 2020, 48, 1056–1061. [Google Scholar] [CrossRef]
- Eyre, D.W.; Lumley, S.; O’Donnell, D.; Campbell, M.; Sims, E.; Lawson, E.; Warren, F.; James, T.; Cox, S.; Howarth, A.; et al. Differential occupational risks to healthcare workers from SARS-CoV-2 observed during a prospective observational study. eLife 2020, 9, 60675. [Google Scholar] [CrossRef] [PubMed]
- Reyné, B.; Selinger, C.; Sofonea, M.T.; Miot, S.; Pisoni, A.; Tuaillon, E.; Alizon, S. Wearing masks and establishing COVID-19 areas reduces secondary attack risk in nursing homes. medRxiv 2020, 9913, Preprint. [Google Scholar] [CrossRef]
- Kabesch, M.; Roth, S.; Brandstetter, S.; Häusler, S.; Juraschko, E.; Weigl, M.; Wellmann, S.; Lang, T.; Schmidt, B.; Salzberger, B.; et al. Successful containment of Covid-19 outbreak in a large maternity and perinatal center while continuing clinical service. Pediatr. Allergy Immunol. 2020, 31, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Knoll, R.L.; Klopp, J.; Bonewitz, G.; Gröndahl, B.; Hilbert, K.; Kohnen, W.; Weise, K.; Plachter, B.; Hitzler, W.; Kowalzik, F.; et al. Containment of a Large SARS-CoV-2 Outbreak Among Healthcare Workers in a Pediatric Intensive Care Unit. Pediatr. Infect. Dis. J. 2020, 39, e336–e339. [Google Scholar] [CrossRef]
- Sanchez, G.V.; Biedron, C.; Fink, L.R.; Hatfield, K.M.; Polistico, J.M.F.; Meyer, M.P.; Noe, R.S.; Copen, C.E.; Lyons, A.K.; Gonzalez, G.; et al. Initial and Repeated Point Prevalence Surveys to Inform SARS-CoV-2 Infection Prevention in 26 Skilled Nursing Facilities—Detroit, Michigan, March–May 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 882–886. [Google Scholar] [CrossRef]
- MacIntyre, C.R.; Dung, T.C.; Chughtai, A.A.; Seale, H.; Rahman, B. Contamination and washing of cloth masks and risk of infection among hospital health workers in Vietnam: A post hoc analysis of a randomised controlled trial. BMJ Open 2020, 10, e042045. [Google Scholar] [CrossRef] [PubMed]
- Brehm, T.T.; Schwinge, D.; Lampalzer, S.; Schlicker, V.; Kuechen, J.; Thompson, M.; Lohse, A.W. High effectiveness of multimodal infection control interventions in preventing SARS-CoV-2 infections in healthcare professionals: A prospective longitudinal seroconversion study. medRxiv 2020, 5936, Preprint. [Google Scholar] [CrossRef]
- Rolland, Y.; Lacoste, M.-H.; De Mauleon, A.; Ghisolfi, A.; Barreto, P.D.S.; Blain, H.; Villars, H. Guidance for the Prevention of the COVID-19 Epidemic in Long-Term Care Facilities: A Short-Term Prospective Study. J. Nutr. Health Aging 2020, 24, 812–816. [Google Scholar] [CrossRef]
- Chi Chen, H.; Hsing Chen, M.; Shen, C.; Hsieh, M.H.; Wu, L.K.; Chin Chen, L.; Hsiao, S.H. Secure Healthcare Workers’ Health and Safety: Active Surveillance, Early Detection and Outbreak Management during COVID-19 Epidemic in Taiwan; SSRN Scholarly Paper ID 3622408; Social Science Research Network: Rochester, NY, USA, 2020. [Google Scholar] [CrossRef]
- Porru, S.; Carta, A.; Monaco, M.G.L.; Verlato, G.; Battaggia, A.; Parpaiola, M.; Cascio, G.L.; Pegoraro, M.; Militello, V.; Moretti, F.; et al. Health Surveillance and Response to SARS-CoV-2 Mass Testing in Health Workers of a Large Italian Hospital in Verona, Veneto. Int. J. Environ. Res. Public Health 2020, 17, 5104. [Google Scholar] [CrossRef] [PubMed]
- Grassly, N.C.; Pons-Salort, M.; Parker, E.P.K.; White, P.J.; Ferguson, N.M.; Ainslie, K.; Baguelin, M.; Bhatt, S.; Boonyasiri, A.; Brazeau, N.; et al. Comparison of molecular testing strategies for COVID-19 control: A mathematical modelling study. Lancet Infect. Dis. 2020, 20, 1381–1389. [Google Scholar] [CrossRef]
- Kucharski, A.J.; Klepac, P.; Conlan, A.J.K.; Kissler, S.M.; Tang, M.L.; Fry, H.; Gog, J.R.; Edmunds, W.J.; Emery, J.C.; Medley, G.; et al. Effectiveness of isolation, testing, contact tracing, and physical distancing on reducing transmission of SARS-CoV-2 in different settings: A mathematical modelling study. Lancet Infect. Dis. 2020, 20, 1151–1160. [Google Scholar] [CrossRef]
- Qiu, X.; Miller, J.C.; MacFadden, D.R.; Hanage, W.P. Evaluating the contributions of strategies to prevent SARS-CoV-2 transmission in the healthcare setting: A modelling study. BMJ Open 2021, 11, e044644. [Google Scholar] [CrossRef] [PubMed]
- Razzak, J.A.; Bhatti, J.A.; Tahir, M.R.; Pasha-Razzak, O. Initial estimates of COVID-19 infections in hospital workers in the United States during the first wave of pandemic. PLoS ONE 2020, 15, e0242589. [Google Scholar] [CrossRef] [PubMed]
- McDermott, J.H.; Stoddard, D.; Woolf, P.J.; Ellingford, J.M.; Gokhale, D.; Taylor, A.; Black, G. A Non-Adaptive Combinatorial Group Testing Strategy to Facilitate Healthcare Worker Screening During the Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) Outbreak. medRxiv 2020, 7677, Preprint. [Google Scholar] [CrossRef]
- Chin, E.T.; Huynh, B.Q.; Chapman, L.A.C.; Murrill, M.; Basu, S.; Lo, N.C. Frequency of routine testing for COVID-19 in high-risk healthcare environments to reduce outbreaks. medRxiv 2020, 7015, Preprint. [Google Scholar] [CrossRef]
- See, I.; Paul, P.; Slayton, R.; Steele, M.; Stuckey, M.; Duca, L.; Reddy, S. Modeling effectiveness of testing strategies to prevent COVID-19 in nursing homes—United States, 2020. medRxiv 2021, 8255, Preprint. [Google Scholar] [CrossRef]
- Agius, R.M.; Robertson, J.F.R.; Kendrick, D.; Sewell, H.F.; Stewart, M.; McKee, M. Covid-19 in the workplace. BMJ 2020, 370, 3577. [Google Scholar] [CrossRef]
- CDC. COVID-19: Ventilation in Buildings, Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/coronavirus/2019-ncov/community/ventilation.html (accessed on 7 July 2021).
- Juneau, C.-E.; Pueyo, T.; Bell, M.; Gee, G.; Collazzo, P.; Potvin, L. Evidence-Based, Cost-Effective Interventions to Suppress The COVID-19 Pandemic: A Systematic Review. medRxiv 2020, 4726, Preprint. [Google Scholar] [CrossRef] [Green Version]
- Iannone, P.; Castellini, G.; Coclite, D.; Napoletano, A.; Fauci, A.J.; Iacorossi, L.; D’Angelo, D.; Renzi, C.; La Torre, G.; Mastroianni, C.M.; et al. The need of health policy perspective to protect Healthcare Workers during COVID-19 pandemic. A GRADE rapid review on the N95 respirators effectiveness. PLoS ONE 2020, 15, e0234025. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Strategies to Reduce COVID-19 Transmission at the Smithfield Sioux Falls Pork Plant. Available online: https://covid.sd.gov/docs/smithfield_recs.pdf (accessed on 5 May 2021).
- Megnin-Viggars, O.; Carter, P.; Melendez-Torres, G.J.; Weston, D.; Rubin, G.J. Facilitators and barriers to engagement with contact tracing during infectious disease outbreaks: A rapid review of the evidence. PLoS ONE 2020, 15, e0241473. [Google Scholar] [CrossRef] [PubMed]
- Lewis, D. Why indoor spaces are still prime COVID hotspots. Nat. Cell Biol. 2021, 592, 22–25. [Google Scholar] [CrossRef]
- Belgian Government. Summer Plan: Four Steps towards a More Normal Life. Coronavirus COVID-19. Available online: https://www.info-coronavirus.be/en/news/occ1105/ (accessed on 7 July 2021).
- Eykelbosh, A. Indoor CO2 Sensors for COVID-19 Risk Mitigation: Current Guidance and Limitations, Vanc. BC Natl. Collab. Cent. Environ. Health 2021. Available online: https://ncceh.ca/sites/default/files/FINAL%20-%20Using%20Indoor%20CO2%20Sensors%20for%20COVID%20MAY%2017%202021.pdf (accessed on 7 July 2021).
| Scheme | Design | Country | Setting | Population | Quality Score/4 a | Infection Prevention and Control Measures |
|---|---|---|---|---|---|---|
| [32] | Prospective Cohort Study | India | Hospital | Residents (n = 5), lab technicians (n = 10), nursing orderlies (n = 3) | 2 | In-house, homemade tools for standard operating procedures: face masks, OT gowns |
| [29] | Prospective Cohort Study | Canada | Hospital and nursing home residences | HCWs+ (n = 30) | 3 | Home-based 7-day control strategy for exposed HCWs, asymptomatic RT-PCR testing |
| [57] | Retrospective Cohort Study | Italy | Teaching hospital | Patients and HCWs (n = 103) | 2 | Nasal swab qPCR and IgG/IgM antibodies testing |
| [62] | Prospective Observational Study | Italy | Hospital | HCWs (n = 7595) | 3 | Separated and dedicated COVID areas, multiple hand hygiene installations, PPE, training protocols, implementation of surveillance system |
| [30] | Prospective Cohort Study | Korea | Hospital | HCWs and patients (n = 142) | 3 | Universal screening programme |
| [80] | Modelling Study | USA | Healthcare facilities | Residents and HCWs (n = 100) | LRB b | Routine asymptomatic PCR testing |
| [31] | Prospective Cohort Study | Korea | Hospital setting | HCWs (n = 317) | 2 | Contact tracing |
| [63] | Prospective Observational study | Canada | Tertiary care centre | HCWs (cohort 1: n = 1669, cohort 2: 4107, cohort 3: n = 1597) | 3 | Symptomatic/asymptomatic nasopharyngeal swab PCR testing |
| [65] | Prospective Observational Study | UK | Hospital setting | Staff (n = 10,034) | 3 | Naso-/oropharyngeal swab and/or immunoassay IgG testing; contact tracing |
| [75] | Mathematical Modelling | UK | n/A | Not reported | LRB b | Estimate of PCR test sensitivity, sensitivity and specificity of IgG antibody test, positive predictive value of a positive antibody test |
| [73] | Surveillance Study | Taiwan | Hospital | HCWs (n = 374) | 3 | Online body temperature surveillance, outbreak investigation and management, advising HCWs not to travel |
| [34] | Prospective Descriptive Study | Vietnam | Hospital laboratory Setting | Staff members (n = 32) | 2 | Risk assessment and management, laboratory training program, self-reporting and electronic reporting of COVID-19 symptoms, PPE stock monitoring system |
| [33] | Prospective Seroprevalence Study | Germany | Hospital setting | Clinical and non-clinical MRI staffs (n = 6305), and medical students (n = 1699) | 3 | PPE; PCR testing for SARS-CoV-2, anti-SARS-CoV-2 IgG and IgM testing |
| [79] | Modelling | USA | Hospital | HCW’s (n = 1350) | LRB b | Nasopharyngeal samples |
| [35] | Prospective Cohort Study | UK | Teaching hospital | HCWs (n = 360) | 3 | Symptomatic/asymptomatic HCW screening |
| [67] | Outbreak Investigation Report | Germany | Maternity and Perinatal centre | Not reported | 2 | Extensive testing; universal face masks; central monitoring of sick leaves; measures to ensure social distancing; continuous on-site visits by hygiene experts and staff training |
| [36] | Prospective Cohort Study | California, USA | Skilled nursing facility | Hospital staff and residents (n = 725) | 2 | Targeted testing: point prevalence surveys. |
| [56] | Retrospective Cohort Study | Korea | Hospital setting | Patients and HCWs (n = 2924) | 2 | Nasopharyngeal and oropharyngeal swabs, surveillance of people with contact history with confirmed COVID-19 patients. |
| [68] | Outbreak Investigation Report | Germany | Tertiary university hospital | HCWs (n = 432) | 3 | Quarantine of positive HCWs, containment measures including surgical masks; physical distancing, and systematic testing. |
| [48] | Cohort Study | China | Tertiary hospital | Patients (n = 1860) | 2 | Hospital layout adjustments, specialized training, pre-testing and triage, environmental cleansing, PPE |
| [23] | Cross-Sectional | France | Hospital | HCW’s (n = 314) | 1 | Nasal swab testing, self-isolation, and masks |
| [39] | Prospective Cohort Study | Belgium | Hospital | HCWs (n = 699) | 2 | SARS-CoV-2 RNA and anti-SARS-CoV-2 IgG antibodies testing |
| [27] | Case-Control study | International | n/A (online survey) | HCWs (n = 1130) | 1 | Use of respirators for aerosol generating procedures (AGP); PPE use and training |
| [25] | Cross-Sectional | Finland | Tertiary hospitals | HCWs (n = 1072) | 1 | Social distance of 1 m |
| [44] | Prospective Cohort Study | Italy | Hospital | HCWs (n = 5750) | 2 | Contact tracing, reinforced hygiene practices, PPE, education, and signage |
| [70] | Post hoc Analysis of a Randomized Controlled Trial | Vietnam | Hospital | HCWs (n = 607) | 3 | Washing method for cloth masks |
| [37] | Prospective Cohort Study | Germany | Hospital | Hospital staff (n = 1185) | 3 | Low-threshold SARS-CoV-2 testing facility |
| [26] | Cross-Sectional Study | Spain | Hospital setting | Hospital Workers (n = 2963) | 2 | Use of PPE |
| [74] | Surveillance Study | Italy | Hospital setting | Staff and residents under contract working (n = 5942) | 3 | Mass screening (oropharyngeal and nasopharyngeal swabs) with/without contact tracing |
| [77] | Mathematical Modelling Studies | International | Healthcare setting | HCWs (n = 224) | LRB b | Surveillance |
| [78] | Modelling Study | USA | Hospital setting | Hospital Workers (n = 53,000, number of hospital workers in the US) | LRB b | Use of PPE in all healthcare workers. Use of PPE only in high-risk workers. Restricting age of workers < 60 y; restricting age of workers < 50 y |
| [40] | Prospective Cohort Study | UK | Teaching hospital | HCWs (n = 1032), symptomatic HCW’s (n = 169), symptomatic household contacts (n = 52) | 3 | Asymptomatic screening using real-time RT-PCR Symptomatic screening using real-time RT-PCR Symptomatic screening of household contacts |
| [38] | Prospective Cohort Study | USA | Electrophysiology unit | Staff (n = 912) and patients (n = 758) | 2 | Universal asymptomatic testing for patients, caregivers, staff, and emergency medical service staff |
| [41] | Prospective Cohort Study | USA | 2 Community hospitals | HCWs (n = 21,014) | 1 | Universal masking |
| [42] | Prospective Cohort Study | Italy | Hospital | HCWs (n-2611) | 1 | PPE and sanitation guidelines implemented, epidemiological investigation and contact tracing of high-risk HCWs, symptomatic swab testing |
| [60] | Prospective Observational Study | Italy | 2 Large hospitals | HCWs (6800) | 3 | Contact tracing and testing of close contacts; random testing |
| [43] | Prospective Cohort Study | Japan | Hospital setting | HCWs (n = 49) | 1 | PPE (90% compliance) |
| [71] | Short-Term Prospective Study | Germany | Tertiary care centre | Staff (n = 1253) | 3 | Multimodal infection control: strict barrier nursing of known COVID-19 patients, including full PPE, visitor restrictions, universal face masks, universal RT-PCR admission screening of patients |
| [54] | Retrospective Cohort Study | Korea | Healthcare setting | Hospital staff (n = 87) and patients (n = 224) | 3 | Outbreak investigation surveillance |
| [55] | Retrospective Cohort Study | USA | Hospitals | HCWs (n = 9850) | 3 | Universal masking for HCWs |
| [45] | Prospective Cohort Study | USA | Hospital | HCWs (n = 832) | 3 | Universal face mask policy |
| [47] | Prospective Cohort Study | Singapore | Hospital | HCWs (n = 1642) | 3 | Enforcing reporting of HCWs with acute respiratory illness (ARI) to staff clinic for monitoring; ongoing syndromic surveillance; outbreak investigation and management |
| [61] | Prospective Observational Study | Singapore | Hospital | HCWs (n = 13,066) | 3 | Multi-tiered infection control strategy: improved patient segregation and distancing, heightened infection prevention and control measures including universal masking, testing of all symptomatic patients |
| [64] | Prospective Observational Study | Singapore | Hospital | Staff (n = 253) | 3 | Contact tracing; 14-day phone surveillance and 28-day follow-up of close contacts; testing of symptomatic contacts |
| [46] | Prospective Cohort Study | Malaysia | Hospital | HCWs (n = 400) | 2 | Full PPE, which includes an N95 mask, an isolation gown, gloves, eye protection and a head cover when providing care to patients under investigation or confirmed COVID-19 patients, and anti-SARS-CoV-2 antibodies serological tests |
| Study Reference | Design | Country | Population | Quality Score/4 a | Infection Prevention and Control Measures |
|---|---|---|---|---|---|
| [66] | Retrospective Observational Study | France | Staff (n = 360) and residents (n = 930) | 2 | Nursing home has enough masks for all residents and staff |
| [58] | Retrospective Cohort Study | UK | Nurses, care workers and non-care workers | 2 | Increased PPE: face masks, eye protection |
| [21] | Cross-Sectional Study | Belgium | Staff (n = 93) and residents (n = 119) | 1 | Anti-SARS-CoV-2 antibody testing in addition to RT-PCR testing |
| [81] | Modelling | USA | Residents and staff (n = 215) | LRB b | Serial testing of asymptomatic persons in response to an outbreak; serial testing of asymptomatic healthcare personnel in the absence of known cases |
| [59] | Prospective Observational Study | Germany | Staff (n = 135) and Residents (n = 160) | 2 | General screening and cohort isolation |
| [53] | Retrospective Cohort Study | UK | Care home staff (n = 474) | 2 | Nasal swab testing; working in multiple vs. single care home |
| [51] | Longitudinal Cohort Study | Massachusetts, USA | Care homes (n = 360) | 2 | 6-part intervention: 28-item checklist, payment incentive, on-site and virtual infection control consultation, weekly webinars, continuous question and answer communication, PPE-staffing-testing resources |
| [72] | Short-Term Prospective Study | France | Long-term care facilities (n = 124) | 2 | Staff compartmentalization within zones; self-assessment scale of the quality of the “barrier” measures |
| [50] | Cohort Study | UK | Staff (n = 320); residents (n = 349) | 1 | Implementation of a negative pressure isolation space |
| [28] | Case-Series Study | Washington, USA | Staff (n = 62) and residents (n = 80) | 1 | Enhanced hygiene practices were implemented |
| [69] | Outbreak Investigation | USA | Nursing facilities (n = 26) | 3 | Universal asymptomatic testing for patients, caregivers, and staff |
| Study Reference | Design | Country | Setting | Population | Quality Score/4 a | Infection Prevention and Control Measures |
|---|---|---|---|---|---|---|
| [22] | Cross-Sectional Study | Italy | Manufacturing facility | Employees (n = 1267) | 1 | Social distancing, individual hygiene rules, PPE, cleaning and sanitizing of environments, information, and training of workers |
| [49] | Prospective Cohort Study | USA | Offices | Employees (n = 27), household (n = 27), students, and volunteers | 2 | Nasal swabs, RT-qPCR measuring antibodies concentration by ELISA |
| [76] | Modelling Study | UK | General population | (n = 40,162) | LRB b | Physical distancing, isolation, tracing, and testing |
| [24] | Cross-Sectional: Point Prevalence | Belgium, Spain, Italy, France, USA, UK | Offices and industrial buildings | Workplaces (n = 411 for 1st week/n = 424 for 2nd week) | 1 | Environmental monitoring |
| [52] | Retrospective Cohort Study | USA | Meatpacking facility | Employees (n = 1000) | 1 | PPE and physical barriers |
| Preventive Measures Category | Study Reference |
|---|---|
| Surveillance | |
| Asymptomatic PCR testing | [28,35,38,39,49,57,63,65] |
| Symptomatic PCR testing | [23,35,40,63] |
| Symptomatic PCR testing of household contacts | [35,40] |
| RT-PCR testing of staff after environmental monitoring | [24] |
| Asymptomatic IgG/IgM immunoassay testing | [57,65] |
| Asymptomatic IgG/IgM immunoassay testing following an outbreak | [21] |
| Asymptomatic RT-PCR testing following an outbreak | [21] |
| Point prevalence surveys | [36,69] |
| Low-threshold SARS-CoV-2 testing facility | [37] |
| Outbreak Investigations and Response | |
| Syndromic surveillance, outbreak investigations | [47,73] |
| Contact tracing | [31] |
| Mass screening, contact tracing | [74] |
| Contact tracing, testing of close contacts | [60] |
| Contact tracing, 14-day phone surveillance, 28-day follow-up of close contacts | [64] |
| Asymptomatic RT-PCR prior to patient surgery, contact tracing of exposed HCWS | [54] |
| PPE | |
| Cloth masks compared to medical masks | [70] |
| Universal masking | [41,45,55] |
| Homemade tools for standard operating procedures | [32] |
| High PPE compliance | [26,33,43] |
| Adequate PPE supply | [66] |
| Masks with and without physical barriers | [52] |
| Respirators used instead of surgical masks | [27] |
| Eye protection and face protection | [58] |
| Education | |
| On-site and virtual infection control consultations | [51] |
| Changes in work arrangements | |
| Staff compartmentalization within zones | [72] |
| Negative pressure isolation space | [50] |
| Restricted worker mobility between facilities | [53] |
| Social distancing compliance | [25] |
| Combined Preventive Measures. | Study Reference |
|---|---|
| Hospital layout adjustments, training, pre-testing and triage, environmental cleansing, PPE | [48] |
| Standard operating procedure, staff training, symptom reporting, enhanced cleaning, inventory monitoring protocols | [34] |
| Social distancing, universal masking, testing of all symptomatic patients | [61] |
| Home-based 7-day infection control strategy for exposed HCWs—symptomatic, asymptomatic RT-PCR testing | [29] |
| General screening and cohort isolation | [42] |
| PPE and sanitation guidelines implemented, epidemiological investigation and contact tracing of high-risk HCWs, symptomatic swab testing | [46] |
| Integrated infection control strategy: zoning, PPE, mass surveillance | [62] |
| PPE, visitor restrictions, universal face masks, universal RT-PCR patient admission screening | [71] |
| Hospital shut down, universal testing of all inpatients, medical staff, and employees | [56] |
| Systematic testing, social distancing, monitoring of sick leaves, on-site visits by hygiene experts, staff training, direct communication of all measures to personnel and patients | [67] |
| Social distancing, surgical masks, systematic testing | [68] |
| Regulation of access to the company, social distancing, hygiene and PPE, cleaning and sanitizing of environments, worker education | [22] |
| Contact tracing, reinforced hygiene practices, PPE, education, and signage | [44] |
| Modelled Preventive Measures. | Study Reference |
|---|---|
| Variations in employee testing frequency (daily, weekly, bi-weekly, monthly) | [80] |
| Variations in testing frequency; outbreak vs. non-outbreak testing | [81] |
| Testing and symptomatic isolation; regular screening of high-risk groups; close contact quarantine | [75] |
| Non-adaptive combinatorial matrices used for group testing | [79] |
| Self-isolation and variations in contact tracing methods; mass testing | [76] |
| Variations in PPE use; worker age restrictions | [78] |
| Variations in PPE efficacy and testing frequency | [77] |
| Intervention | No. of Studies | n | Pooled Positivity Rate (%) a,b | 95% CI | Q | I2 | T2 | p-Value | Egger’s Test c | Egger’s Test p-Value |
|---|---|---|---|---|---|---|---|---|---|---|
| Asymptomatic RT-PCR d | 10 | 25077 | 1.7 e | 0.9, 2.9 | 202.32 | 96% | 0.0025 | <0.01 | 0.09 | 0.93 |
| Surveillance and Contact tracing | 9 | 6599 | 3.5 e | 0.8, 7.9 | 391.59 | 98% | 0.0191 | <0.01 | n/A | n/A |
| Universal Masking of Employees | 2 | 11684 | 24.0 f | 3.4, 55.5 | 692.34 | 100% | 0.0559 | <0.01 | n/A | n/A |
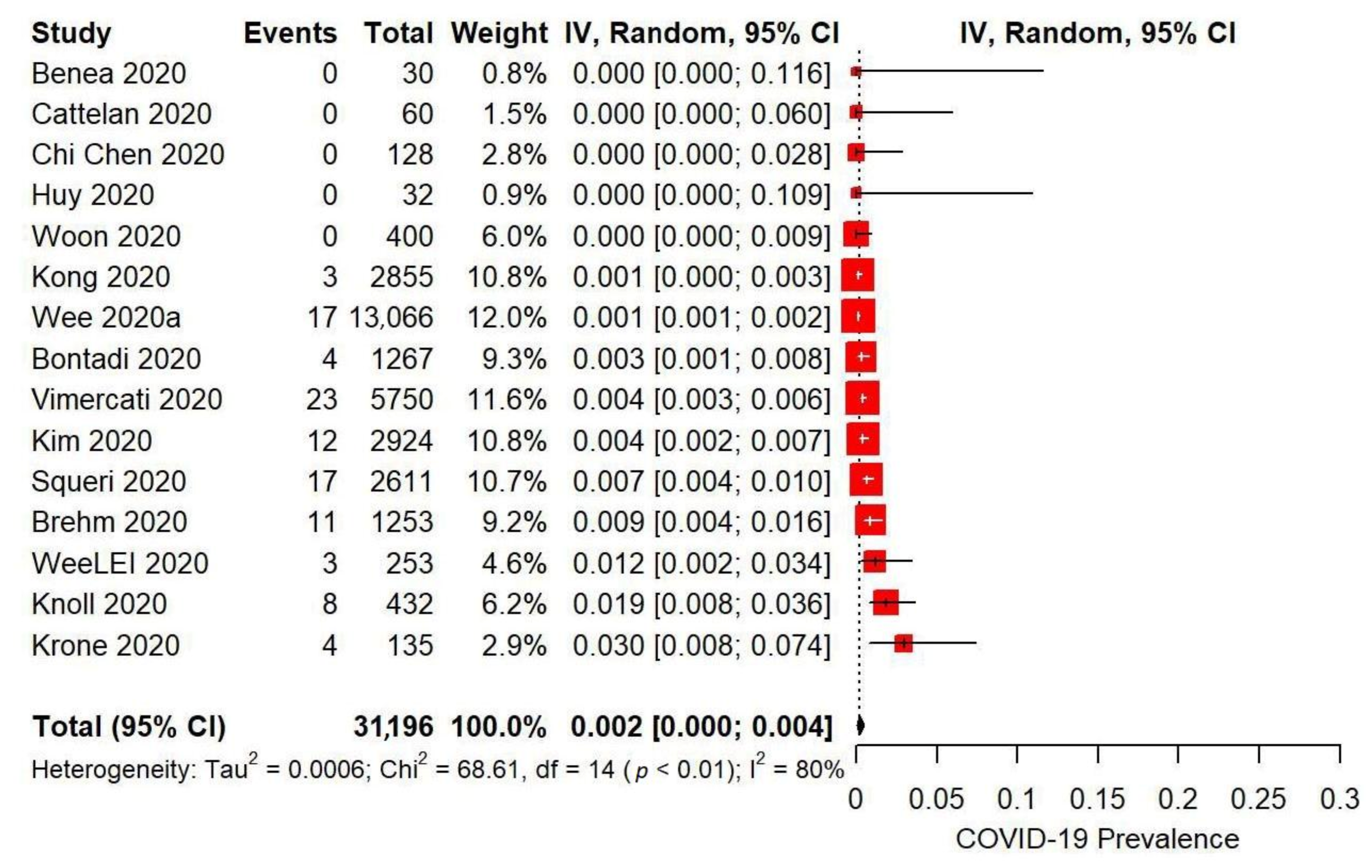
| Combined measures (>2 intervention categories) | 15 | 31196 | 0.2 f | 0.0, 0.4 | 68.61 | 80% | 0.0006 | <0.01 | 2.24 | 0.04 |
| Factor | QM (df) | R2 | Beta coefficient (99% CI) | Standard Error | p-Value a |
|---|---|---|---|---|---|
| Region | 7.137 (2) | 43% | |||
| Europe vs. Asia | 0.048 (0.0368, 0.0371) | 0.010 | 0.008 ** | ||
| North America vs. Asia | 0.041 (0.0402, 0.0425) | 0.093 | 0.223 | ||
| North America vs. Europe | 0.004 (0.003,0.006) | 0.093 | 0.963 | ||
| Intervention Duration (Days) | 43.96 (1) | 12% | −0.0002 (−0.0002, −0.0002) | 0.0001 | 0.214 |
| Community COVID-19 positivity (Under 5% vs. Over 5%) | 3.389 (1) | 0% | −0.036 (−0.0358, −0.0353) | 0.019 | 0.066 |
| Pandemic Wave Interval | 0.820 (2) | 0% | |||
| Deceleration vs. Acceleration | 0.028 (0.027,0.028) | 0.033 | 0.394 | ||
| Peak vs. Acceleration | 0.005 (0.004,0.005) | 0.023 | 0.848 | ||
| Specific Intervention Implemented (Yes vs. No) | |||||
| Asymptomatic RT-PCR testing | 4.961 (1) | 27% | 0.040 (0.0394,0.0399) | 0.018 | 0.023 * |
| Facility Zoning | 0.040 (1) | 0% | 0.004(0.0038,0.0044) | 0.021 | 0.842 |
| Employee Education | 1.610 (1) | 0% | −0.026 (−0.026, −0.026) | 0.020 | 0.205 |
| Environmental Cleaning | 3.733 (1) | 0% | −0.038 (−0.038, −0.038) | 0.020 | 0.053 |
| PPE | 2.133 (1) | 14% | −0.025 (−0.025, −0.025) | 0.017 | 0.144 |
| Syndromic Surveillance | 2.210 (1) | 16% | −0.026 (−0.027, −0.026) | 0.018 | 0.137 |
| Contact Tracing | 0.330 (1) | 0% | −0.012 (−0.012, −0.012) | 0.021 | 0.566 |
| Total Interventions Implemented | 6.102 (1) | 22% | −0.0109 (−0.0110, −0.0108) | 0.004 | 0.014 * |
| Study | Risk of Bias | Key Findings |
|---|---|---|
| [80] | Low |
|
| [81] | Low |
|
| [75] | Low |
|
| [79] | Low |
|
| [76] | Low |
|
| [78] | Low |
|
| [77] | Low |
|
| Study | Intervention Category/Setting | Findings | Conclusions |
|---|---|---|---|
| [24] | Surveillance/Hospital | OR calculation for locations with PCR or antibody positives (2400 environmental swabs) vs. locations without positives (3000 environmental swabs) reveals that locations with coronavirus-positive environmental surfaces had 10 times greater odds (p ≤ 0.05) of having positive employees compared to locations with no positive surfaces. |
|
| [70] | PPE/Hospital | The risk of COVID-19 infection was more than double among HCWs self-washing their masks compared with the hospital laundry (HR 2.04 (95% CI 1.03 to 4.00); p = 0.04). There was no significant difference in infection between HCWs who wore cloth masks washed in the hospital laundry compared with medical masks (p = 0.5). |
|
| [52] | PPE/Meatpacking | After initiating both universal masking and physical barrier interventions, 8/11 facilities showed a statistically significant reduction in COVID-19 incidence in <10 days. Facilities that only initiated a universal mask policy showed no significant difference before and after the intervention. |
|
| [51] | Education/Nursing home | Special focus facilities (n = 123) started with higher infection rates than control facilities (n = 237) but rapidly declined to the same level as the other facilities within a week of starting on-site and virtual infection control consultations. Compliance with cohorting and PPE were associated with large reductions in the weekly infection rate (−50%; p = .004; −23%; p = .0379) and increased odds of a zero-infection rate ([OR] = 3.0; p = .0076; OR = 2.16; p = .0003). |
|
| [72] | Changes in work arrangements/Nursing home | Long-term care facilities (LTCF) that organized staff compartmentalization within zones were significantly more likely to avoid a COVID-19 outbreak (OR = 0.19 (0.07–0.48)) as were LTCFs whose staff perceived high-quality implementation of preventive measures (OR = 0.65 (0.43–0.98)). |
|
| [53] | Changes in work arrangements/Nursing home | Staff working across different care homes (14/27, 52%) had a 3.0-fold (95% CI, 1.9–4.8; p < 0.001) higher risk of SARS-CoV-2 positivity than staff working in single care homes (39/227, 17%). Whole-genome sequencing identified distinct clusters of SARS-CoV-2 infection between staff only, including those with minimal resident contact. |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ingram, C.; Downey, V.; Roe, M.; Chen, Y.; Archibald, M.; Kallas, K.-A.; Kumar, J.; Naughton, P.; Uteh, C.O.; Rojas-Chaves, A.; et al. COVID-19 Prevention and Control Measures in Workplace Settings: A Rapid Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2021, 18, 7847. https://doi.org/10.3390/ijerph18157847
Ingram C, Downey V, Roe M, Chen Y, Archibald M, Kallas K-A, Kumar J, Naughton P, Uteh CO, Rojas-Chaves A, et al. COVID-19 Prevention and Control Measures in Workplace Settings: A Rapid Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2021; 18(15):7847. https://doi.org/10.3390/ijerph18157847
Chicago/Turabian StyleIngram, Carolyn, Vicky Downey, Mark Roe, Yanbing Chen, Mary Archibald, Kadri-Ann Kallas, Jaspal Kumar, Peter Naughton, Cyril Onwuelazu Uteh, Alejandro Rojas-Chaves, and et al. 2021. "COVID-19 Prevention and Control Measures in Workplace Settings: A Rapid Review and Meta-Analysis" International Journal of Environmental Research and Public Health 18, no. 15: 7847. https://doi.org/10.3390/ijerph18157847
APA StyleIngram, C., Downey, V., Roe, M., Chen, Y., Archibald, M., Kallas, K.-A., Kumar, J., Naughton, P., Uteh, C. O., Rojas-Chaves, A., Shrestha, S., Syed, S., Cléirigh Büttner, F., Buggy, C., & Perrotta, C. (2021). COVID-19 Prevention and Control Measures in Workplace Settings: A Rapid Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 18(15), 7847. https://doi.org/10.3390/ijerph18157847







